Abstract
Endosulfan, a well-known organochlorine pesticide, induces apoptosis and depletion of reduced glutathione (GSH) in human peripheral blood mononuclear cells (PBMC). Thus, for the amelioration of its effect, antioxidant and antiapoptotic potential of curcumin was evaluated. For ascertaining the attenuating effect of curcumin, various biochemical indices of cell damage such as cytotoxicity, oxidative stress, apoptosis (phosphatidylserine externalization, DNA fragmentation, and cytochrome
Introduction
Curcumin is the major active component of turmeric, a yellow compound originally isolated from the plant
Certain chemicals such as cadmium, acrolein, dibutyltin dichloride, acetaldehyde, dexamethasone, or 2,3,7,8-tetrachlorodibenzo-
Studies from our laboratory have shown that endosulfan is present in breast milk, adipose tissue, placental tissue, and umbilical cord blood. We have reported significantly higher levels (4.80 ng/ml blood) of endosulfan in women with adverse reproductive outcomes in comparison with healthy women population (3.85 ng/ml blood) of North India. 11,12 Subacute and chronic toxicity studies of enosulfan on human and animals have suggested that the vital organs including liver, kidney, testes, spleen, thymus, and lymph nodes are the main target organs bearing the major impact of their toxicity. 13,14 A dose-dependent effect of endosulfan on humoral and cell-mediated immune responses in rats was reported. 15 Endosulfan at 8 and 16 mg/kg, that is, 20% and 40% of median lethal doses significantly induced reduction in the anti-sheep red blood cell (anti-SRBC) antibody responses and suppressed the interferon γ and interleukin 4 cytokines levels. 15 Several in vitro studies have demonstrated that endosulfan (0–100 µM) induces the cytotoxicity/apoptosis in primary culture and cell lines. 16,17 Apoptosis of neuronal cells has been one of the causes of neurotoxicity as a result of endosulfan exposure. 16 Endosulfan induces oxidative stress and apoptosis in adult rat testes as a possible mechanism of action of endosulfan-induced toxicity. 18 However, the consequence of prolonged exposure to subchronic doses of endosulfan at cellular level still needs elucidation.
Cells may employ several mechanisms to protect themselves from the effects of toxicants. The cellular response to stress is represented at the molecular level by the increased synthesis of heat shock proteins (HSPs). Mammalian HSP27 belongs to the family of small heat shock proteins (sHSPs) that is rapidly phosphorylated at Ser78 and Ser82 in response to heat shock and other stressors, suggesting a role in the regulation of signal transduction pathways. 19 The stress proteins have also been important models in various mechanistic studies like regulation of transcription, embryogenesis, aging, and apoptosis. 20,21
Earlier reports show that curcumin (0–30 µM) stimulates the stress-induced expression of genes for HSP27, αβ-crystalline and HSP70 in cultured cells and in rats tissues in vivo when injected with 20 mg/kg body weight. 22 In our previous studies, we have reported that subchronic doses of pesticide induce apoptosis and also the role HSP27 in modulation of apoptosis following pesticide exposure. 23,24 This study is in line with our previous studies to demonstrate the linkage between cellular events and cell death and to evaluate the signaling mechanism revealing the specific role of HSP in endosulfan-mediated cytotoxicity.
Materials and methods
Chemicals
Endosulfan, (technical grade) was obtained from AccuStandard (New Haven, Connecticut, USA). Roswell Park Memorial Institute 1640 (RPMI-1640) media, Histopaque 1077, curcumin, and all other chemicals used in the present study were of analytical grade and purchased from Sigma–Aldrich Limited (St Louis, Missouri, USA). Stock solutions of endosulfan (99.9% purity) and curcumin (99% purity) were prepared using dimethyl sulfoxide (DMSO) and serially diluted with incomplete media (RPMI-1640 without fetal bovine serum) to prepare the 1 mM working solution. The final concentration of DMSO in working solution was up to 0.5%.
Isolation and treatment of human PBMCs
The blood samples from healthy volunteers attending blood bank of University College of Medical Sciences and GTB Hospital, Delhi, India, for blood donation were collected in heparinized tubes after obtaining informed consent per institutional ethical committee guidelines.
PBMCs were separated from peripheral blood by the density gradient centrifugation method. 5 Briefly, 5 ml blood was layered carefully over equal volume of Histopaque 1077 and subjected to centrifugation for 30 min at 2000 r/min. PBMCs were collected from the buffy layer formed at the plasma–Histopaque 1077 interface and diluted in Hank’s balance salt solution. Cells were counted in Neubauer chamber using trypan blue and diluted with media to get a final concentration of 2 × 106 cells/ml of media and incubated in 5% carbondioxide (CO2)–95% humid atmosphere at 37°C in CO2 incubator (MCO-15 AC, Sanyo, Japan). Cells were treated with increasing concentrations of endosulfan (0–100 μM) and along with curcumin (25 µM) for 24 h. The doses of endosulfan were based on earlier reports that endosulfan at concentration ranging from 10 µM to 100 µM induces apoptosis in Jurkat cells and in SH-SY5Y cells. 16,25 The dose of curcumin used was based on previous reports that the protective efficacy of curcumin toward HSP27 synthesis is in the range of 10–30 µM. 21 Control cells were exposed to 0.5% DMSO.
Cytotoxicity assay
Cell viability, as an indicator of cytotoxicity, was determined by measuring the capacity of the PBMC to reduce 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma Chemical Co.) to formazan. Briefly, a 5 mg/ml solution of MTT was prepared in incomplete RPMI media. MTT solution (20 µl) was added to each well of pesticide-treated PBMC and control cells in 96-well plates, followed by incubation at 37°C for 4 h in a 5% CO2, 95% humid atmosphere. Following incubation, intracellular formazan dissolved in DMSO gave a purple color and was quantified at 540 nm using a microplate reader (Bio-Rad-680, Hercules, California, USA) with DMSO as the blank. 23 The results are presented as percentage of the control cells.
Detection of apoptosis
Annexin V-Cy3.18 binding assay
This was carried out using annexin V–Cy3.18 (AnnCy3) and 6-carboxy fluorecein diacetate (CFDA) according to the manufacturer’s protocol. Briefly, induced cells were washed twice with phosphate-buffered saline (PBS) and were suspended at a concentration approximately 1 × 106 cells/ml. A circle of 1 cm diameter was drawn on a polyprep poly-
DNA fragmentation assay
Cultured cells were collected by centrifugation at 1500 r/min for 5 min at 4°C. The cell pellets were washed twice with PBS, immediately lysed with 400 μl of lysis buffer (1% Triton X, 50 mM Tris–hydrochloric acid (HCl), pH 7.5, 20 mM ethylenediaminetetraacetic acid (EDTA)) and centrifuged at 4500 r/min for 5 min at 4°C in a microcentrifuge (Eppendorff). The supernatant-containing DNA was removed and incubated at 50°C for 3 h after mixing well with 20 μl of 10% sodium dodecyl sulfate (SDS) and 5 μl RNase A (10 mg/ml). After incubation, 5 μl proteinase K (15.6 mg/ml) was added, and the mixture was further incubated at 37°C for 3 h. DNA was precipitated by ethanol, separated by centrifugation and dissolved in Tris-EDTA (TE) buffer. Aliquots of DNA from different groups along with 100 bp marker were electrophoresed using 1% agarose gel containing 0.01% ethidium bromide at 80 V/18 A for 3 h. The bands were visualized and photographed using a gel documentation system (UVP DigiDoc It LS, UVP-Analytik Jena AG, Jena, Germany).
Cytochrome c assay
Cytochrome
Estimation of GSH
Cellular GSH was estimated spectrophotometrically in a reaction mixture containing cell lysate, nicotinamide adenine dinucleotide phosphate (NADPH) and 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB). 5 Cell lysate was prepared by repeated freeze-thaw method using deionized water. The reaction mixture (1 ml) contained 25 μl of reduced GSH in HCl, 50 μl of NADPH (0.2 μmol/ml in 0.01M/0.005 M phosphate EDTA buffer), 10 μl of glutathione reductase (1 unit), and 800 μl of phosphate EDTA buffer (pH 7.5). The test sample was prepared by adding 25 μl of cell lysate instead of reduced GSH added in standard. The chromophoric product resulting from reaction of reagent DTNB (0.6 μmol) with GSH, that is, 2-nitro-5-thiobenzoic acid was measured spectrophotometrically at 412 nm immediately after addition of DTNB for 0–5 min time intervals. GSH concentration is expressed as percentage of control cells.
Estimation of HSP27 mRNA expression
Isolation of RNA and estimation of HSP27 expression by RT-PCR
To isolate RNA, at least 0.5 × 106 cells were resuspended in Perfect RNA lysis buffer (Eppendorf, Hamburg, Germany), total RNA was extracted by Perfect RNA Eukaryotic Mini kit (Eppendorf), and the amount of extracted RNA was estimated by spectrophotometry at 260 nm. Total RNA was reverse transcribed using the high-capacity complementary DNA (cDNA) Archive Kit (Applied Biosystems, Foster City, California, USA), according to the manufacturer’s instructions. Real-time polymerasde chain reaction (PCR) was performed with an ABI prism 7000 apparatus (Applied Biosystems) using the assay on demand kits for the gene expression of HSP27. The forward and reverse-specific primer sequences of HSP27 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) used in this study were selected from previous published data.
26,27
The thermal profile for each gene was stage 1, 2 min at 50°C; stage 2, 10 min at 95°C; stage 3, 40 cycles including 15 s at 95°C and 1 min at 60°C linearity of real-time PCR assays were tested by constructing standard curves by use of serial two fold dilutions of cDNA and regression coefficients (
HSP27 level estimation
Estimation of HSP27 levels was carried out using commercially available phospho-HSP27 [pSer82] ELISA kit (Sigma-Aldrich, Inc.) per manufacturer’s protocol. Briefly, washed cells were lysed in cell extraction buffer. In appropriate wells of microtiter plates, standard, sample, or controls along with anti-phospho-HSP27 [pSer82] detection antibody were added (except chromogen blank) and incubated for 2 h at room temperature. Anti-rabbit immunoglobulin G-HRP was added to each well, except chromogen blanks, and incubated for 30 min at room temperature. Chromogen (100 μl) was added into the wells and incubated approximately for 30 min at room temperature. The reaction was terminated by the addition of 100 μl of stop solution, and absorbance was measured at 450 nm on microtiter plate reader (Bio
Statistical analysis
All assays were repeated at least five times, and data are presented as means ± SD. Data were analyzed by repeated-measure analysis of variance using the SPSS version 17 statistical program and comparison between the various treated groups were carried out by the Tukey’s multiple comparison test. Only data with
Results
Effects of curcumin on survival of cells
To find out the protective effect of curcumin on endosulfan-induced cytotoxicity, PBMCs were incubated for 24 h with selected doses of endosulfan (0–100 µM), along with curcumin (25 µM) in culture media and were subsequently estimated for cell survival by MTT assay. Endosulfan showed its effects on the percentage of survival cells in dose-dependent manner. Above 10 μM of endosulfan, 10%–30% reduction in the number of surviving cells was noted. When the control cells were cotreated with curcumin, no appreciable changes were noted in cell survival. Following cotreatment with curcumin on endosulfan-treated groups, a significant improvement in the percentage of surviving cells was observed, however, no significant increase was depicted in cell survival at higher doses of pesticide (50 and 100 μM; Figure 1).

Effect of curcumin on the viability of endosulfan-exposed PBMCs. PBMC culture treated with endosulfan (5–100 μM) and along with curcumin (25 μM) for 24 h. Cell viability was determined by measuring the capacity of cells to reduce MTT to intracellular purple formazan, which was quantified at 540 nm. The results are presented as percentage of survival cells. a
Curcumin attenuates apoptosis in endosulfan exposed human PBMC
PBMCs were exposed to 0–100 µM endosulfan for 24 h and simultaneously curcumin (25 µM) was administrated. Pesticide-induced apoptosis was examined using AnnCy3 binding assay, DNA fragmentation assay, and the release of cytochrome
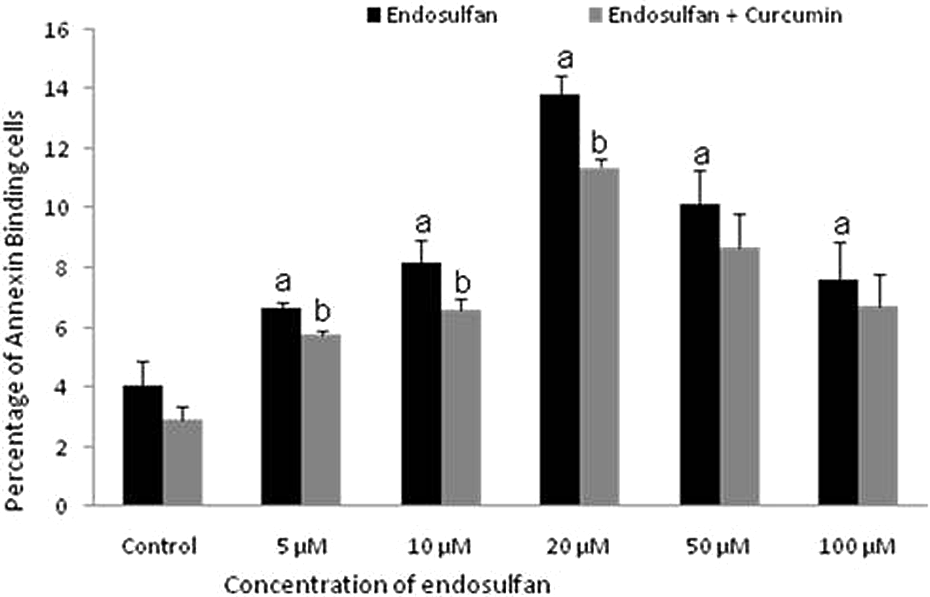
Effect of curcumin on the apoptotic effect of endosulfan-exposed PBMCs. Values depict the percentage of cells that bind to annexin V undergoing apoptosis observed under fluorescent microscope after 24 h. a

Effects of curcumin on DNA Fragmentation of PBMC after endosulfan treatment (24 h). Lane 1:100 bp marker, lane 2: 10 µM endosulfan-treated cells, lane 3: 20 µM endosulfan-treated cells, lane 4: 50 µM-treated endosulfan-treated cells, lane 5: control cells, lane 6: 20 µM endosulfan-treated cells along with 25 µM curcumin. PBMCs: peripheral blood mononuclear cells.

Effect of curcumin on endosulfan-induced cytochrome
Curcumin: An inducer of HSP
To evaluate the role of curcumin as an inducer of HSP27, the expression of HSP27 was studied by real-time PCR and sandwich ELISA after exposing PBMCs to endosulfan and along with curcumin for 24 h. The expression of HSP27 messenger RNA (mRNA) was significantly increased at subchronic doses of endosulfan up to 20 μM in a dose-dependent manner. However, no significant increase in the expression of HSP27 mRNA was observed at higher concentration of endosulfan. Two and half fold increase in HSP27 mRNA expression was observed following treatment with curcumin at 20 µM endosulfan-treated group (Figure 5). The levels of HSP27 was estimated by sandwich ELISA using anti-HSP27 antibody. The similar pattern of dose-dependent effect was observed on the levels of phospho-HSP27 in these treated PBMCs (Figure 6). HSP27 expression was further enhanced significantly by the addition of curcumin (Figures 5 and 6).

Expression of HSP27 mRNA in PBMCs after pesticide exposure. Total RNA was extracted from at least 0.5 × 106 cells treated PBMCs. Isolated total RNA was then reverse transcribed, resulting cDNA was used as a source for amplification. Relative expression of HSP27 mRNA compared with the GAPDH housekeeping gene following exposure to endosulfan and cotreatment with curcumin was examined using real-time PCR. Data were expressed as percentage of control cells and calculated from five individual measurements for each time point. *

Effect of curcumin on levels of HSP27 in endosulfan-treated cells. a
Effect of curcumin on GSH level
Intracellular GSH level was estimated in endosulfan-exposed cells spectrophotometrically. Intracellular GSH level was found to be depleted in a dose-dependent manner when the cells were incubated with these pesticides for 24 h (Figure 7). Coadministration of curcumin replenished the GSH levels significantly as compared to endosulfan-treated cells (Figure 7).
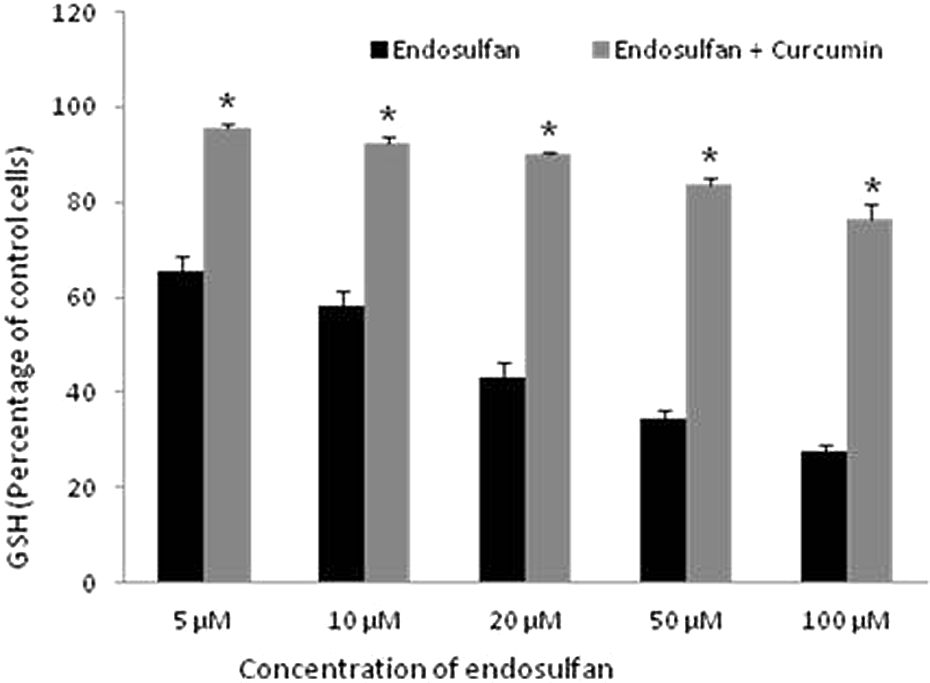
Effect of curcumin on GSH level in endosulfan-treated cells. *
Discussion
In this study, we found that curcumin attenuates cytotoxicity/apoptosis in human PBMCs induced by endosulfan (Figure 1), we also testified that curcumin causes the induction of HSP27. This study is in continuation of our previous study in which we had reported that endosulfan-induced apoptosis of PBMCs and depletion of reduced GSH was ascribed as an underlying mechanism.
5
Using three different approaches in this study, namely, AnnCy3 binding assay, DNA fragmentation assay, and quantitative detection of cytochrome
The stress response is one mechanism that the cell could potentially employ to protect itself from cellular damage after exposure to xenobiotics. In doing so, they may influence the shape of the dose–response curve at low-level exposures to the environmental pollutants. The most obvious feature of the stress–response consists of acute alterations in cellular gene expression defined by a cessation of normally occurring protein synthesis and induction of the synthesis of HSPs after exposure of cells to stress. 29 Subsequently to withdrawal of stress, the heat shock response is downregulated, normal protein synthesis is reestablished, and the cell recovers from the stressful event. Alternatively, if the level of stress is too great to be attenuated by the stress response, cell death can ensure by necrosis and/or apoptosis. In recent years, HSPs have emerged as regulators of apoptosis. It has been suggested that HSP27 protects cells from apoptotic cell death, triggered by ionizing radiation, oxidative stress, and cytotoxic drugs. 30
Although widely studied in cell lines and other organ systems, expression of the stress proteins has not been studied till date in human PBMCs following endosulfan exposure. HSP27 mRNA expression in pesticide-treated PBMC revealed dose-dependent HSP synthesis at subchronic doses of endosulfan, however, there is no significant increase in the expression of HSP27 mRNA at higher concentration of endosulfan (Figure 5). The mRNA expression of HSP27 was consistent with protein levels of HSP27 (Figure 6). The results of the present study portray that HSP27 inhibits apoptosis in endosulfan-treated cells. These results are in agreement with our previous report of increased expression of HSP27 following malathion exposure.
24
The mechanisms by which HSPs inhibit apoptosis are still not clear. In addition to its basic function of promoting the refolding of damaged proteins and thus averting apoptosis, HSPs may directly interfere with execution of apoptosis by interacting with key components of apoptotic signaling pathway such as cytochrome
At cellular level, measurement of reduced GSH is a marker of cellular antioxidant defense. 33 In the present study, intracellular GSH levels were found to be depleted in a dose-dependent manner in human PBMCs following endosulfan exposure for 24 h (Figure 7). A decrease in GSH alone can act as a potent early activator of apoptotic signaling. Increased ROS production following mitochondrial GSH depletion has been reported to induce apoptosis in a B cell lymphoma cell line. 33 The result obtained by us clearly indicates an association between GSH depletion, endosulfan-induced oxidative stress, and increased apoptosis of PBMC.
Antioxidants have been proven to play an important role in the regulation of a vast array of physiological and pathological processes. They principally contribute to the protection of cells and tissues against deleterious effects of reactive oxygen species and other free radicals. Several antioxidants like vitamin C, vitamin E, and
An in vitro study has shown that curcumin protects smooth muscle cells against cell apoptosis evoked by oxidative stress. 43 It has been reported that curcumin is able to induce the synthesis of HSP70 in human leukemia cells. 44 The results obtained in this study indicate that curcumin protects PBMCs from endosulfan-induced apoptosis by enhancing synthesis of HSP27. These results are in concurrence with previous studies, demonstrating that high expression of HSPs was often related to resistance to cytotoxicity and promotes cell survival by inhibiting key steps of the apoptotic cascade normally leading to cell death. 45,46
Previously, Gao et al. reported that curcumin maintained the cellular redox state via upregulation of GSH levels.
47
In this case, the resistance caused by HSP27 against endosulfan-induced apoptosis seems to be mediated, at least in part, by modulation of GSH levels. A positive correlation was observed between HSP levels and GSH depletion in endosulfan-treated cells (
Hence, the present study demonstrates that PBMC undergoes dose-dependent apoptotic cell death following endosulfan exposure and also highlights the effectiveness of curcumin in counteracting xenobiotic-induced cytotoxicity. These findings further support the hypothesis that oxidative stress, as indicated by GSH depletion, results in the induction of apoptosis by the release of cytochrome
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
