Abstract
Introduction:
Chemoprevention may involve perturbation of a variety of steps in tumor initiation, promotion, and progression.
Objective:
To investigate the antiproliferative and anti-inflammatory potential effects of diindolylmethane (DIM) and lupeol on experimental bladder carcinogenesis.
Methods:
Sixty healthy male Wistar rats were selected and randomly divided into six groups, with 10 rats in each group. Group I: control; group II: N-butyl-N-(4-hydroxybutyl) nitrosamine (BBN; 150 mg/gavage/twice a week) for 8 weeks, and then they were given 100 ppm concentrations of dimethylarsenic acid (DMA) in the drinking water for 28 weeks; group III: BBN + DMA + DIM (5 mg/kg body weight (b.w.)/day) treatment was started after BBN treatment, and it was orally administered for 28 weeks); group IV: BBN + DMA + lupeol (50 mg/kg b.w./day) treatment was started after BBN treatment, and it was orally administered for 28 weeks); and groups V and VI: DIM and lupeol treatment alone for 36 weeks. Bladder tissues were collected after 36th week study protocol for further analysis.
Results:
Our results revealed that DIM and lupeol treatment showed inhibition of tumor growth in the bladder by histopathological confirmations as well as significantly (p < 0.001) increased the expression of phosphotensin (PTEN) and significantly (p < 0.001) decreased the expression of tumor necrosis factor α, nuclear factor κβ (p65) were quantified using Western blot analysis. DIM and lupeol treatment significantly (p < 0.001) decreased the levels of Cox-2 in bladder tissue samples and NMP 22 in urine samples were quantified using enzyme-linked immunosorbent assay method.
Conclusion:
Preventive DIM and lupeol administration act as potent Cox-2 inhibitors, which activates the tumor suppressor protein PTEN against experimental bladder carcinogenesis by antiproliferative and anti-inflammatory properties.
Introduction
Urothelial carcinoma of the bladder is a common malignancy ranked 9th with an estimated 356,600 new cases diagnosed and approximately 150,000 deaths annually worldwide. It is the common cancer in developed countries, 1 predominantly manifests as a non-muscle invasive tumor characterized by an overall good prognosis following transurethral resection in patients with low-grade and intravesical chemotherapy and/or Bacillus Calmette–Guerin instillation in patients with high-grade bladder cancer. 2 It is the fourth most prevalent cancer in men and eighth in the women, with transitional cell carcinoma comprising up to 90% of bladder tumors are of epithelial origin. 3 Approximately 50% of the patients suffer from high-grade bladder cancer, which is the precursor for the muscle invasive cancer, and of these, 50% have distant metastasis within 2 years and approximately 50–60% of these patients will die within 5 years despite treatment at initial diagnosis. 4 The rat bladder cancer model of multistage carcinogenesis has been a useful experimental framework to study basic mechanisms associated with the initiation, promotion, and progression stages of carcinogenesis and defining newer chemopreventive agents. The intervention of cancer at the promotion stage appears to be most appropriate and practical. 5 N-Butyl-N-(4-hydroxybutyl) nitrosamine (BBN) is an environmental carcinogen of tobacco origin that reproducibly causes high-grade, invasive cancers in the urinary bladder. 6 The metabolism of BBN into N-butyl-N-(3-carboxypropyl) nitrosamine occurs in liver by alcohol dehydrogenase enzyme, which further initiates the carcinogenesis in the urinary bladder. 7 Dimethylarsenic acid (DMA) has the potential to promote rat urinary bladder carcinogenesis initiated with BBN, which is involved in stimulating the cell proliferation in the epithelium of urinary bladder. 8
Nuclear matrix protein 22 (NMP22) has been identified as a specific tumor marker for bladder cancer, and it is released from the nuclei of bladder tumor cells. NMP22 levels were increased (approximately 25-folds) in urine of the bladder carcinoma patients. 9,10 COX-2 plays major roles in tumor initiation/promotion, cell proliferation, and differentiation in urinary bladder carcinoma. 11 It is expressed in both transitional cell and squamous cell carcinomas of human urinary bladder. 12,13 The nuclear factor κB (NF-κB) promotes tumorigenesis by triggering the expression of genes involved in cell proliferation, survival, tumor promotion, immortalization, angiogenesis, and metastasis. 14 Tumor necrosis factor α is a multifunctional cytokine involved in apoptosis, cell survival, inflammation, and immunity. An inhibition of the NF-κB pathway by COX-2 inhibitors is reported to be successful in the treatment of tumor cells. 15 Phosphotensin (PTEN) is the tumor suppressor that regulates growth, survival, and proliferation. Inactivation of PTEN promotes tumorigenesis in urinary bladder of the humans. 16
3,3′-Diindolylmethane (DIM), an active metabolite of indole-3-carbinol, is present in cruciferous vegetables such as cabbage, broccoli, cauliflower, and Brussels sprouts that have been shown to prevent carcinogenesis in animals and humans. 17 It has chemopreventive effects against chemically induced tumors such as ovarian, pancreatic, prostate, and breast in rats and mice. 18 Lupeol, a triterpene found in fruits and vegetables such as white cabbage, green pepper, strawberry, olive, mangoes, and grapes, was reported to possess beneficial effects as a therapeutic and preventive agent. 19 Lupeol possesses pharmacological properties including anticancer, anti-inflammatory, antidiabetic, cardioprotective, and hepatoprotective activities. 20 –22 It shows inhibitory effect on the in vivo oral cancer model. 23 Hence the aim of the study was to determine the modulating effects of DIM and lupeol on BBN-induced experimental bladder carcinogenesis.
Materials and methods
Animals and chemicals
Male albino Wistar rats were purchased from Central Animal House, Indian Institute of Science (Bengaluru, Karnataka, India). All animals were housed in polypropylene cages. The animals were kept in a room lighted for 12 h each day and maintained at 20°C, standard pellet diet (Hindustan lever Ltd) and water was provided ad libitum. Animal experiments were followed under the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (New Delhi, India). The study was approved by the Institutional Animal Ethical Committee, SRM University. OH-BBN and DMA were purchased from Tokyo Chemical Industry (Japan). Lupeol (Cat no. L5632) was purchased from Sigma Aldrich (St Louis, Missouri, USA). Horseradish peroxidase (HRP)-conjugated goat anti-rabbit immunoglobulin G were purchased from Santa Cruz Biotechnology, (Santa Cruz, California, USA). Rabbit monoclonal TNF-α antibody was purchased from Thermo Fisher Scientific (Waltham, Massachusetts, USA). Rabbit monoclonal phospho-NFκ β p65, PTEN, and β-actin monoclonal antibodies were purchased from Cell Signaling Technology (Danvers, Massachusetts, USA).
Experimental design
Sixty rats were procured and divided into six groups of 10 animals each. Group I rats were served as healthy control. Group II rats were treated with BBN (150 mg/gavage/twice a week) for 8 weeks, and the rats were given 100 ppm concentrations of DMA in the drinking water for 28 weeks. 24,8 Group III rats were treated with BBN, and after cessation of BBN treatment, rats were given 100 ppm DMA in the drinking water with oral coadministration of 5 mg/kg body weight (b.w.)/day DIM for 28 weeks. 25 Group IV rats were treated with BBN, and after cessation of BBN treatment, rats were given 100 ppm DMA in the drinking water with oral coadministration of 50 mg/kg b.w./day lupeol for 28 weeks. 23 Group V rats were treated with DIM alone (5 mg/kg b.w./day) for 36 weeks. Group VI rats were treated with lupeol alone (50 mg/kg b.w./day) for 36 weeks. Urine samples were collected a week before the experimental period. The experiment was terminated at the end of 36th week, and all the experimental rats were euthanized by cervical dislocation. Bladder tissue samples were collected for subsequent analysis.
Sample collection and preparation
The collected urine samples were centrifuged at 3000 r/min for 10 min at 4°C. The clear supernatants were immediately frozen at −80°C and kept until further analysis. The bladder tissues were excised and rinsed with cold 0.1 M potassium chloride, dried, and weighed. Bladder homogenate was prepared in isotonic phosphate buffer (pH 7.4) using a homogenizer for a minute. Bladder tissue cell lysates were prepared by incubation on ice with lysis buffer (50 mM Tris–hydrochloric acid (HCl; pH 7.5), 20 mM sodium chloride (NaCl), 5 mM ethylenediaminetetraacetic acid, 1% Triton X-100, 0.1% sodium dodecyl sulfate (SDS), and 5% glycerol + protease inhibitors), and centrifuged at 20,000g for 20 min.
Quantification of rat NMP22
Urine samples were used to quantify the levels of urinary bladder cancer marker enzyme NMP22 using rat NMP 22 enzyme-linked immunosorbent assay (ELISA) kit (My Biosource, San Diego, California, USA)
Detection of cell proliferating marker COX-2
Bladder tissue cell supernatant were used to measure the levels of cell proliferating marker COX-2 using rat COX-2 ELISA kit (CusaBiotech; Wuhan Hi-tech Medical Devices Park, Wuhan, China).
Western blotting
The bladder tissue cell supernatant was collected and protein concentration was determined using the Piece BCA assay kit (Thermo) with bovine serum albumin as a standard control. Equal amounts of sample were loaded on SDS-polyacrylamide gel electrophoresis and transferred onto nitrocellulose membranes at 250 mA for 1 h. Membranes were stained with 0.5% Ponceau in 1% acetic acid for confirmation and were blocked for 2 h in TBST (10 mM Tris-HCl, pH 7.4, 150 mM NaCl, and 0.1% Tween 20) containing 5% fat-free dried milk and then incubated with primary antibodies overnight. Membranes were incubated with corresponding HRP-labeled secondary antibodies, and β-actin was selected as an internal control. Protein bands were visualized by chemiluminescence with the enhanced chemiluminescence detection kit according to the manufacturer’s instructions.
Histopathology examination
After formalin fixation (10% phosphate-buffered (pH 7.4)) and dehydration process, paraffin-embedded urinary bladder sections (5 μm) were stained by hematoxylin and eosin. Histopathology for urinary bladder sections was evaluated at 400× magnifications.
Statistical analysis
Data were analyzed using analysis of variance, and the group mean ± SD was compared with Tukey multiple range tests using SPSS 19 software version. p Values of 0.05 were considered to be statistically significant.
Results
DIM and lupeol inhibit the tumor development by its anticarcinogenic properties
DIM and lupeol treatment inhibits the tumor formation in the urinary bladder, and the tissues were subjected to histopathological evaluation. Control healthy urinary bladder tissue section showed normal architecture (Figure 1(a)), BBN-treated urinary bladder tissue section showed the carcinoma as indicated (Figure 1(b)), preventive DIM restores the bladder to normal architecture (Figure 1(c)), preventive lupeol completely prevented the tumor incidence (Figure 1(d)), and DIM and lupeol-alone treated urinary bladder tissue section showed no abnormality (Figure 1(e) and (f)).

DIM and lupeol treatment inhibits the bladder cancer in experimental rats. Histopathological changes in the bladder of control and experimental rats. ((a), (e) and (f)) Photomicrographs showing normal architecture of bladder in healthy control, DIM (5 mg/kg b.w.) and lupeol (50 mg/kg b.w.) alone-treated experimental rats. (H&E, ×400). (b) Photomicrograph showing carcinoma in rats treated with BBN + DMA (H&E, ×400). (c and d) Photomicrograph showing inhibition of tumor formation in BBN + DMA + DIM (5 mg/kg b.w.) and BBN + DMA + lupeol (50 mg/kg b.w.) treatment groups (H&E, ×400). DIM: diindolylmethane; BBN: N-butyl-N-(4-hydroxybutyl) nitrosamine; DMA: diindolylmethane; H&E: hematoxylin and eosin; b.w.: body weight.
NMP22 as a bladder cancer detection marker
The levels of bladder cancer-specific urinary marker NMP22 was quantified in control and experimental rats. NMP 22 (approximately 17-folds) levels were significantly (**p < 0.001) increased in BBN-treated rats when compared with control. NMP22 levels were significantly (##p < 0.001) decreased in preventive DIM and lupeol-treated rats when compared with BBN-treated experimental rats. DIM and lupeol-alone treated rats showed no significant (NS) changes in the levels of NMP 22 when compared with control (Figure 2).
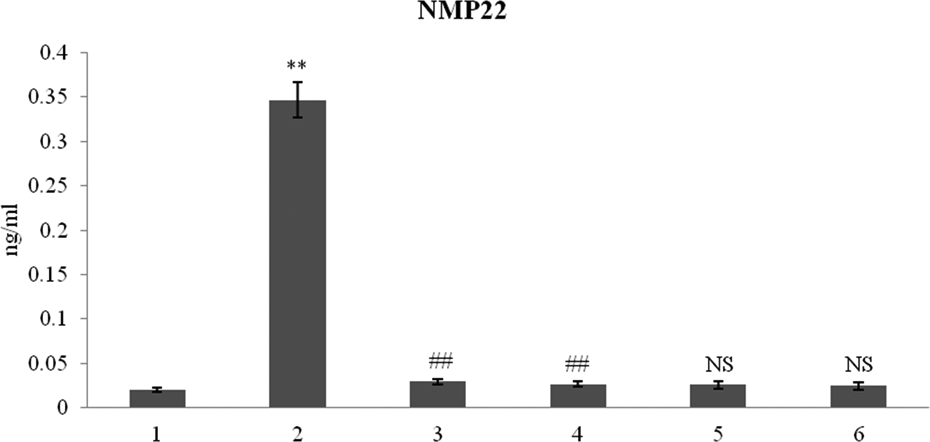
Nuclear matrix protein 22 as a bladder cancer detection marker levels in urine samples of control and experimental rats. Values are expressed as mean ± SD. **p < 0.001: versus control, ##p < 0.001: versus BBN + DMA, and NS denotes no significant changes in DIM and lupeol alone-treated groups compared with control. Units expressed as nanogram per milliliter. 1: control (untreated rat); 2: BBN + DMA; 3: BBN + DMA + DIM (5 mg/kg b.w.); 4: BBN + DMA + lupeol (50 mg/kg b.w.); 5: DIM (5 mg/kg b.w.); 6: lupeol (50 mg/kg b.w.). BBN: N-butyl-N-(4-hydroxybutyl) nitrosamine; DMA: diindolylmethane; NMP22: nuclear matrix protein 22; b.w.: body weight.
DIM and lupeol inhibit tumor cell proliferation in experimental bladder carcinogenesis
As shown in Figure 3, the levels of cell proliferation marker COX-2 in control and experimental rats were quantified using ELISA kit. COX-2 (approximately threefold) levels were significantly (**p < 0.001) increased in BBN-treated rats when compared with control. COX-2 levels were significantly (##p < 0.001) decreased in DIM and lupeol-treated (preventive) rats when compared with BBN-treated experimental rats. DIM and lupeol alone-treated rats showed NS changes in the levels of COX-2 when compared with control (Figure 3).

The antiproliferative effects of DIM and lupeol in in vivo bladder cancer models. The levels of Cox-2 were quantified in control and experimental rats. Values are expressed as mean ± SD. **p < 0.001: versus control, ##p < 0.001: versus BBN + DMA, and NS denotes no significant changes in DIM and lupeol alone-treated groups compared with control. Units expressed as nanogram per milligram tissue. 1: control (untreated rat); 2: BBN + DMA; 3: BBN + DMA + DIM (5 mg/kg b.w.); 4: BBN + DMA + lupeol (50 mg/kg b.w.); 5: DIM (5 mg/kg b.w.); 6: lupeol (50 mg/kg b.w.). DIM: diindolylmethane; BBN: N-butyl-N-(4-hydroxybutyl) nitrosamine; DIM: diindolylmethane; BBN: N-butyl-N-(4-hydroxybutyl) nitrosamine; DMA: diindolylmethane; b.w.: body weight.
DIM and lupeol inhibit cell survival, cell immortalization by promoting the action of apoptosis
Our Western blot data showed the quantitative expression of NF-κβ, TNF-α, and PTEN in control and experimental rats (Figure 4(a)). We investigated the expression of cell survival proteins. NF-κβ (approximately 2.2-folds) and TNF-α (approximately 0.6-folds) were significantly (**p < 0.001) increased and tumor suppressor protein PTEN (approximately 10-folds) significantly (**p < 0.001) decreased in BBN-treated rats when compared with control. Preventive (DIM and lupeol) treatment significantly (##p < 0.001) decreased the protein expression of NF-κβ and TNF-α, and significantly (##p < 0.001) increased the expression of PTEN (Figure 4(b), (c) and (d), respectively). DIM and lupeol-alone treated rats showed NS changes in the expression of NF-κβ and TNF α when compared with control. DIM and lupeol alone-treated rats showed mild significant changes (*p < 0.05 vs. control) in the expression of PTEN.

DIM and lupeol treatment inhibits cell survival, cell immortalization by promotes the action of apoptosis. (a) Western blot analysis examined the protein expression levels of p-NF-κβ, TNF-α, and PTEN. β-Actin was applied for equal loading. Lane 1: control (untreated rat); lane 2: BBN + DMA; lane 3: BBN + DMA + DIM (5 mg/kg b.w.); lane 4: BBN + DMA + lupeol (50 mg/kg b.w.); lane 5: DIM (5 mg/kg b.w.); lane 6: lupeol (50 mg/kg b.w.). (b to d) The gray analysis of p-NF-κβ, TNF-α, and PTEN. 1: control (untreated rat); 2: BBN + DMA; 3: BBN + DMA + DIM (5 mg/kg b.w.); 4: BBN + DMA + lupeol (50 mg/kg b.w.); 5: DIM (5 mg/kg b.w.); 6: lupeol (50 mg/kg b.w.). Values are expressed as mean ± SD. **p <0.001: versus control; ##p < 0.001: versus BBN + DMA; *p < 0.05: versus control, and NS denotes no significant changes in DIM and lupeol alone-treated groups compared with control. DIM: diindolylmethane; TNF-α: tumour necrosis factor-α; BBN: N-butyl-N-(4-hydroxybutyl) nitrosamine; DMA: diindolylmethane; PTEN: phosphotensin; b.w.: body weight.
Discussion
Cancer chemoprevention is increasingly being realized as an important area for cancer prevention, which, in addition to providing a practical approach of identifying potentially useful inhibitors of cancer development, also affords excellent opportunities to study the mechanisms of carcinogenesis. Zuo et al. reports that BBN chemical carcinogen-induced experimental bladder cancer showed the increased expression of the COX-2 protein, which was found in preneoplastic and neoplastic lesions of tumor tissues. 26 NF-κB is activated by various stimuli, including growth factors, carcinogens, and tumor promoters. 27 Decreased PTEN expression has been observed in a large number of invasive bladder cancers. 28 DIM is a potent inhibitor of NF-κB and COX-2 and is a well-known chemopreventive agent. It significantly decreased the Cox-2, TNF α, and NF-κB in lung metastasis of 4T1 murine mammary carcinoma cells in BALB/c mice. 29 DIM induced apoptosis in human breast cancer cells through inactivation of NF-κB pathways. 30,31 DIM work synergistically to inhibit cell proliferation and induce apoptosis in colorectal cancer and pancreatic cancer through modulating NF-κB activity. 32,33 DIM inhibits the development of cervical cancer by upregulating PTEN expression in the cervical epithelium of the transgenic mouse models. 34 Bioactive molecules such as DIM prevented cancer through inhibiting cellular oxidation and modulating drug metabolizing enzymes that may be useful adjuncts in the prevention of bladder cancer. 35 Lupeol was reported to modulate several molecules that directly or indirectly play a role in inflammatory process. 19 Lupeol treatment inhibited cell survival by inactivation of NF-κB in human epidermoid carcinoma cells. 36 Topical application of lupeol prior to 12-O-tetradecanoylphorbol-13-acetate (TPA) application to CD-1 mice showed significant decrease in skin hyperplasia and protein expression of COX-2, classical marker of inflammation and tumor promotion, and also resulted in the inhibition of NF-κB. 37 Lee et al. reports that increased PTEN protein expression in lupeol treatment was found in hepatocellular carcinoma cells as well as animal models. 38 Our data revealed that DIM inhibited oncogenesis and cancer cell growth, and induced apoptosis in experimental model, suggesting that DIM could serve as a potent agent for the prevention of tumor progression and/or treatment of cancers. As well as lupeol inhibits or abrogates the inflammatory processes under in vivo situations and such studies provided several mechanistic facets of anti-inflammatory action. In conclusion, we found that oral administration of DIM and lupeol decreased the expression of cell survival, inflammatory marker proteins such NF-κB and TNF α as well as increased the tumor suppressor protein PTEN. DIM and lupeol act as potent Cox-2 inhibitors by reducing the levels of Cox-2 in epithelium of the urinary bladder. Histopathology and Western blot results indicated that DIM and lupeol may be potential clinical candidates for the treatment of bladder cancer.
Footnotes
Acknowledgments
The infrastructure was kindly provided by SRM Medical College Hospital and Research Centre, SRM University, Tamil Nadu, India.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Department of Science and Technology, Government of India, under the Young Scientist Fast Track Scheme.
