Abstract
This study was designed to investigate the effect of agmatine sulfate (AG, CAS2482-00-0) in nicotine (NIC)-induced vascular endothelial dysfunction (VED) in rabbits. NIC was administered to produce VED in rabbits with or without AG for 6 weeks. Serum lipid profile, serum thiobarbituric acid reactive substances, reduced glutathione, superoxide dismutase generation, serum nitrite/nitrate, serum vascular cellular adhesion molecule-1 (VCAM-1), and aortic nuclear factor κB (NF-κB) levels were analyzed.Treatment with AG markedly improves lipid profile and prevented NIC-induced VED and oxidative stress. The mechanism of AG in improving NIC-induced VED may be due to the significant reduction in serum VCAM-1 levels and aortic NF-κB. Thus, it may be concluded that AG reduces the oxidative stress, nitric oxide production, VCAM-1 levels, and aortic NF-κB expression, thereby consequently improving the integrity of vascular endothelium.
Introduction
Cigarette smoking is an important risk factor for cardiovascular disease and premature death in the world today. 1 Smoking facilitates atherosclerosis, myocardial infarction, angina pectoris, peripheral vascular disease, and stroke. 2 Nicotine (NIC) is the main cigarette component known to exert proatherosclerotic, prothrombotic, and proangiogenic effects on vascular endothelial cells due to excessive lipid metabolism, vascular cell damage, hypertension, and thrombosis. 3 NIC, as demonstrated by previous studies, is responsible for the structural and functional alterations in blood vessels such as increased intimal thickening, impaired endothelium-dependent vasodilation, hyperplasia, atherosclerotic plaques, and aneurysmal dilation. 4
Chronic NIC exposure has been implicated in pathogenesis of cardiovascular disorders by inducing vascular endothelial dysfunction (VED) due to downregulating the expression of endothelial nitric oxide synthase (eNOS) and increasing the generation of reactive oxygen species (ROS). 5 Further, exposure to NIC induces oxidative stress that causes endothelial cell damage and disrupts the endogenous antioxidant defense mechanisms by downregulating catalase and superoxide dismutase (SOD) activities. 6 Concomitantly, different pro-inflammatory mediators such as cellular adhesion molecules including intercellular adhesion molecule 1 (ICAM-1) and vascular cellular adhesion molecule 1 (VCAM-1) 7,8 are also produced.
Agmatine (AG, CAS2482-00-0, 4-(aminobutyl)guanidine), an amine dication at physiological pH, is synthesized by decarboxylation of
The present study was undertaken to examine the effect of daily administration of AG on NIC-induced VED and to investigate its possible mechanisms.
Materials and methods
Animal care and experimental treatments
Age-matched 18 male New Zealand White rabbits weighing about 1.5–2 kg were employed in the present study. Animals were placed in a room with constant temperature of 22°C and an alternating 12-h light/12-h dark cycle. They were fed on standard chow diet and water ad libitum. Protocols were approved by the committee on Animal Experimentation of Faculty of Pharmacy, Mansoura University. Rabbits were divided into 4 groups (each of 6 animals) and treated for 6 weeks. Group I included control animals that received distilled water; group II was treated with AG alone (AG was dissolved in water at the dose of 20 mg/kg taken orally); group III was treated with NIC alone (administrated by intraperitoneal injection at the dose of 7.5 mg/kg in distilled water); and group IV was treated with NIC and AG.
Measurement of blood parameters
Prior to experimentation at the end of 6 weeks, the rabbits were fasted overnight. Then, the animals were anesthetized, and blood samples were collected from the marginal ear vein, followed by euthanization by an overdose of sodium pentobarbital and exsanguinated for aortic assessment.
Collected blood samples were allowed to clot, centrifuged (10,000g), and the sera were collected and stored at −80°C until analyzing the biochemical parameters.
Assessment of lipid profile in serum
Determination of serum concentrations of total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG) were carried out using commercially available kits (Randox Laboratories Ltd, UK). The assay was performed according to the manufacturer’s instruction. All samples were measured in duplicate. The concentration of low-density lipoprotein cholesterol (LDL-C) was calculated by the following equation: LDL − C = TC − (HDL − C + 0:2 TG).
Assessment of oxidative stress
Estimation of serum TBARS level
Thiobarbituric acid reactive substance (TBARS) serum level was estimated, and the results were expressed as micromoles per milliliter. A standard graph using 1,1,3,3tetramethoxypropane (1–50 μM) was plotted to calculate the concentration of malondialdehyde (MDA) equivalents to TBARS. 20
Estimation of serum SOD activity
Serum activity of SOD was determined according to the method followed by Marklund 21 by computing the difference between autoxidation of pyrogallol alone and in the presence of SOD enzyme. The enzyme activity of 1 unit is defined as 50% inhibition of autoxidation of pyrogallol under the assay conditions and was expressed as units per milliliter.
Estimation of serum GSH level
Serum reduced glutathione (GSH) activity was determined following the method described by Ellman, 22 which is based on the reduction of Ellman’s reagent (5,5′-dithiobis(2-nitrobenzoic acid)) by thiol (SH) groups to form 1 mole of 2-nitro mercapto-benzoic acid per mole of SH. An intense yellow color of nitromercaptobenzoic acid was produced that can be measured, and the concentration of GSH in the sample was estimated in reference to a calibration curve of GSH prepared from serial dilutions of a 240 μM GSH stock solution that were treated in identical manner as the lung homogenate samples. The result was reported as micromoles per milliliter.
Assessment of serum NOx concentration
Assessment of serum nitrite/nitrate (NOx) concentration was taken as an index for NO level. A 0.5% solution of 100 µl zinc sulfate was prepared and incubated for 5 min at room temperature with 100 µl serum samples. The supernatant resulted after centrifugation of the previous mixture at 3700g for 20 min at 4C was used for the measurement of NOx concentration using the Greiss method. 23
Assessment of serum VCAM-1 level in serum
Serum VCAM-1 was determined using capture enzyme-linked immunosorbent assay (ELISA) kits according to the instructions provided by the manufacturers (Uscn Life Science, Inc., Houston, Texas, USA).
Histopathological examination
After collecting blood samples, thoracic aorta was carefully removed and part of it was fixed in 4% paraformaldehyde and embedded in paraffin wax according to standard procedures for the other analyses. The slices were serially sectioned at 5 m and then stained with hematoxylin–eosin (H&E) or Oil Red O as previously described. 24
IHC analysis for NF-κB expression
A part of the aortic tissue was fixed in 4% paraformaldehyde and embedded in paraffin wax for immunohistochemical (IHC) analysis. Paraffin-embedded slices were serially sectioned at 5 μm. Immunostaining was performed using avidin–biotin complex method. 25 The immunostained sections were examined by an observer blinded to the treatments using a light microscope (Olympus, Japan). The intensity of staining was graded semiquantitatively, and each specimen was assigned a score on a scale from 0 to 3, designated as 0 (negative), 1 (weak), 2 (moderate), and 3 (strong). The immunoreactive score was designated by the staining intensity as previously described. 26
Statistical analysis
The results are presented as mean ± standard error of mean. Statistical analysis was carried out using one-way analysis of variance (ANOVA), followed by Tukey–Kramer multiple comparisons test as post hoc test. Statistical analysis for nonparametric scoring data was carried out using Kruskal–Wallis test, followed by Dunn’s test for comparing all pairs. Statistical significance was considered when p < 0.05. Nonlinear regression analysis was carried out using Graphpad Prism software (Graphpad Software Inc., San Diego, California, USA). Statistical analysis was carried out using Instat-3 computer program (GraphPad Software Inc., V3.05).
Results
Animals treated with AG alone showed no significant differences when compared with the control group (data not shown). Therefore, the results of this group were excluded from tables and figures in order to facilitate data comparison and interpretation.
Effect on serum lipid concentration
Upon treatment with NIC, there were significant elevation in serum TC, TGs, and LDL-C levels and a significant reduction in serum HDL-C level compared with the normal group. Oral treatment with AG for 6 weeks concomitant with NIC administration significantly decreased serum TC, TGs, and LDL-C levels and increased serum HDL-C level when compared with NIC-treated rabbits (Table 1).
Effect of AG on serum changes of TC, TGs, LDL-C, and HDL-C levels induced by NIC in rabbits.a

AG: agmatine (7.5 mg/kg/day, oral); NIC: nicotine (7.5mg/kg/day, SC); TC: total cholesterol; TG: triglycerides; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; ANOVA: analysis of variance; SC: subcutaneous.
aValues are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using one-way ANOVA, followed by Tukey–Kramer multiple comparisons test.
bp < 0.05: significantly different from normal group.
cp < 0.05: significantly different from NIC-treated group.
Effect on serum oxidative stress
As shown in Table 2, subcutaneous (SC) injection of NIC for 6 weeks significantly increased serum MDA level when compared with the normal group and significantly decreased the serum SOD and GSH levels when compared with normal group. Chronic AG treatment in rabbits administered with NIC resulted in a significant decrease in serum MDA level compared with the NIC-treated group and resulted in significant increase in serum SOD and GSH levels when compared with the NIC-treated group.
Effect of AG on serum level changes of MDA, SOD, and GSH induced by NIC in rabbits.a
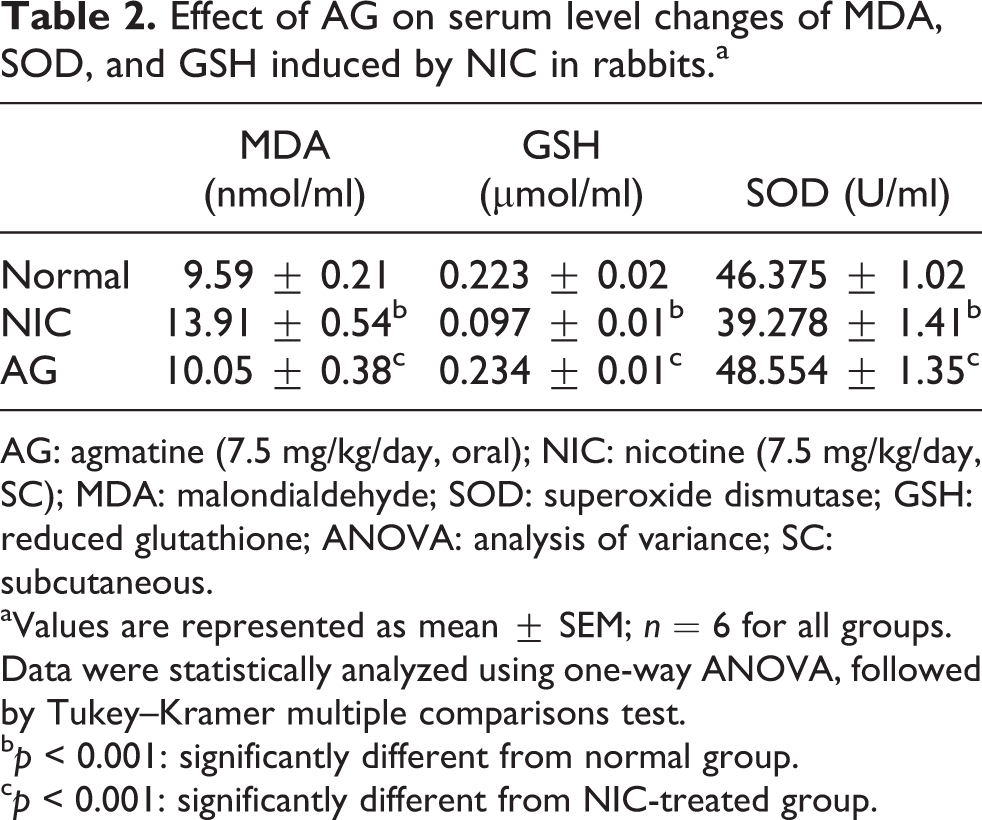
AG: agmatine (7.5 mg/kg/day, oral); NIC: nicotine (7.5 mg/kg/day, SC); MDA: malondialdehyde; SOD: superoxide dismutase; GSH: reduced glutathione; ANOVA: analysis of variance; SC: subcutaneous.
aValues are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using one-way ANOVA, followed by Tukey–Kramer multiple comparisons test.
bp < 0.001: significantly different from normal group.
cp < 0.001: significantly different from NIC-treated group.
Effect on serum NOx level
SC injection of NIC daily to rabbits for 6 weeks showed a significant decrease in serum NOx level compared to normal rabbits. Chronic AG treatment concomitant with NIC resulted in a significant increase in serum NOx level compared with NIC-treated group (Figure 1).

Effect of AG treatment on serum NOx level induced by NIC in rabbits. Values are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using one-way ANOVA, followed by Tukey–Kramer multiple comparisons test. **p < 0.01: significantly different from normal group; ###p < 0.001: significantly different from NIC-treated group; AG: agmatine; NIC: nicotine; NOx: nitrite/nitrate; ANOVA: analysis of variance.
Effect on serum VCAM-1 level
SC injection of NIC in rabbits for 6 weeks showed a significant increase in serum VCAM-1 level compared to normal rabbits. Chronic oral treatment for 6 weeks with AG and NIC resulted in a significant decrease in serum VCAM-1 level compared with NIC-treated group (Figure 2).

Effect of AG treatment on serum VCAM-1 level changes induced by NIC in rabbits. Values are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using one-way ANOVA, followed by Tukey–Kramer multiple comparisons test. ***p < 0.001: significantly different from normal group; ###p < 0.001: significantly different from NIC-treated group. AG: agmatine; VCAM-1: vascular cellular adhesion molecule 1; NIC: nicotine; ANOVA: analysis of variance.
Effect on histopathological results
Light microscopic examination of sections prepared from rabbit aorta after staining with H&E revealed that NIC-treated aortas demonstrated endothelial damage with moderate elevation of the endothelium and subendothelial accumulation of fat cells compared with normal aortas that showed none of the above pathological changes. Regarding AG-treated aortas, examination of their H&E-stained slides showed no or slight elevation of endothelium with no or slight accumulation of fat cells compared with normal aortas (Figure 3(a) to (c)). Confirmation of accumulation of fat cells subendothelially was done by staining with Oil Red O stain as a frozen section. NIC-treated aortas showed moderate-to-severe intense red staining compared with the normal group that showed no red staining. Aortas treated with AG showed no or mild staining (Figure 4(a) to (c), Table 3).

Histopathological examination of aortas (H&E; ×20). (a) Normal, intact endothelium with no subendothelial lipid collection. (b) NIC, moderately elevated endothelium with subendothelial lipid collection. (c) AG, intact endothelium with no subendothelial lipid collection. H&E: hematoxylin–eosin; AG: agmatine; NIC: nicotine.

Histopathological examination of aortas (Oil Red O; ×20). (a) Normal, no subendothelial red staining. (b) NIC, moderate-to-severe subendothelial red staining denoting presence of fat cells. (c) AG, no subendothelial red staining. AG: agmatine; NIC: nicotine.
Effect of AG treatment on subendothelial fat cell accumulation induced by NIC in rabbits.a

AG: agmatine; NIC: nicotine.
aValues are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using Kruskal–Wallis test, followed by Dunn’s multiple comparisons test.
bp < 0.01: significantly different from normal group.
cp < 0.05: significantly different from NIC-treated group.
Effect on aortic NF-κB expression
Scoring of IHC staining using monoclonal antibodies against NF-κB showed that chronic SC injection of NIC led to significant increase in NF-κB expression in aortic endothelial cells compared with the normal group. Chronic oral treatment with AG and NIC resulted in a significant decrease of NF-κB expression in aortic endothelial cells compared with NIC-treated group (Figure 5(a) to (c), Table 4).

IHC staining of aortas (×20). (a) Normal, no brown staining. (b) NIC, moderate-to-severe brown staining denoting presence of positive cytoplasmic staining of NF-κB by IHC. (c) AG, no brown staining. IHC: immunohistochemical; NIC: nicotine; NF-κB: nuclear factor κB.
Effect of AG treatment on aortic NF-κB expression induced by NIC in rabbits.a

AG: agmatine; NF-κB: nuclear factor-κB; NIC: nicotine.
aValues are represented as mean ± SEM; n = 6 for all groups. Data were statistically analyzed using Kruskal–Wallis test, followed by Dunn’s multiple comparisons test.
bp < 0.01: significantly different from normal group.
cp < 0.05: significantly different from NIC-treated group.
Discussion
The vascular endothelium integrity impairment and serum NOx level reduction had been documented to be an indicator for experimental VED. 27,28 The present study examined the effect of AG, an NO modulator, on NIC-induced VED. The results demonstrated the beneficial therapeutic effect of AG in attenuating NIC-induced VED. Initially, serum MDA, SOD, and GSH levels were measured to evaluate the damage induced by NIC administration for 6 weeks. Oxidative stress has an important role in the development of VED by decreasing eNOS activity and reducing the generation and bioavailability of NO. 29 NIC exposure was proved to generate ROS in large amounts by activating nicotinamide adenine dinucleotide phosphate oxidase 30 and decreasing the antioxidant defense mechanisms via downregulation of catalase and SOD 31 and a resultant inhibition of NO activity. In addition, NIC reduced the production and bioavailability of NO via downregulation of eNOS and upregulation of asymmetric dimethylarginine (ADMA). 30,32 Thus, NIC-induced VED is suggested to be mediated through the development of oxidative stress. This is strongly proven by the data obtained from the present study which showed that NIC-administered rabbits exhibited high level of serum TBARs and decreased the activity of SOD enzyme and GSH with marked decrease in serum NOx concentration. This was also supported by the results previously reported by Zaghloul et al. 33 Treatment with AG markedly overcame NIC-induced generation of ROS in rabbits by significantly decreasing TBARS and increasing the activity of SOD and the level of GSH in serum that explain the antioxidant mechanism involved in the vascular protecting potential of AG. This is consistent with the previous reports that have shown the ability of AG to scavenge free radicals and resultant decrease in oxygen derivative, such as hydrogen peroxide. 34,35 The mechanism of the antioxidant effects of AG is still unknown, however, it has been attributed previously to its effect in suppressing NF-κB activation in hyperglycemic mesangial cells. 36 The protective effect of AG against oxidative stress would also be associated with the nucleophilic structure of AG. 36 In this regard, the antioxidant property of AG may be similar to that previously demonstrated for other polyamines, such as spermine, which act as scavengers against ROS. 37 Other proposal for the scavenging effect of AG is that AG reacts with hydroxyl radical to form dihydroxyaminobutyl guanidine, and guanidobutyric aldehyde is then formed by spontaneous dehydration and subsequent hydrolysis. 38
Therefore, the present study showed that AG effectively enhance the antioxidant defense mechanism against NIC-induced VED.
Previous studies proved that increasing the level of circulating lipids impairs the vascular endothelium integrity and induces VED. 27,39 It has been shown that NIC administration alters the lipid profile by significant elevation in TC, TGs, and LDL-C and consequent reduction of HDL-C levels. 40 These data are also in parallel with that proved by many investigators reporting the increase in TC and TG levels in rats treated with NIC. 41 These results were further confirmed by the histopathological examination of the aorta of NIC-injected rabbits using H&E and Oil Red O staining, which revealed moderate elevation of the endothelium with subendothelial accumulation of fat cells compared with normal aorta. Lipid profile changes induced by NIC reported by Ashakumary and Vijayammal 42 may be attributed to increased synthesis and secretion of apolipoprotein B, the major apoprotein secreted as very low-density lipoprotein (VLDL) and LDL. Also elevated TC level may be caused by the decreased activity of plasma lecithin cholesterol acyl transferase enzyme, which is involved in the esterification of cholesterol. Increased levels of VLDL in rabbits injected with NIC results from decreased activity of LPL in extrahepatic tissues that result in decreased uptake of serum VLDL. 42 In addition, alteration in lipid profile by NIC demonstrated by Latha et al. 43 may also be due to increased tissue cholesterogenesis as proved by the increased activity of hydroxymethyl glutaryl coenzyme A reductase. Several reports evidence that high circulating lipids impair the integrity of vascular endothelium and induces vascular endothelial dysfunction. 27,39 Thus, it would be possible that, in the present study, increased serum levels of TC, TGs, and LDL-C and consequently decreased serum level of HDL-C may be involved in the induction of VED in NIC-injected rabbits. However, treatment with AG prevented NIC-induced alteration in lipid profile by reducing the circulating levels of TC and TGs and elevating HDL-C levels. In addition, histopathological examination of the aorta from AG-treated rabbits showed no or slight elevation of endothelium with no or slight accumulation of fat cells compared with aorta from NIC-treated rabbits.
Sabha et al.
44
have demonstrated that treatment of humans with NIC impairs the endothelium-dependent reactivity of large peripheral blood vessels. NIC impairs bioavailability of NO via downregulation of eNOS and upregulation of ADMA.
30,32
Excessive production of
To further elucidate the mechanism of AG improving NIC-induced vascular damage, the present study examined the effect of AG on NIC-induced increased expression of the pro-inflammatory NF-κB and the serum concentration of VCAM-1. These metabolic pathways are important for the development of NIC-induced atherosclerosis and hypertension. 49
Increased expression of VCAM-1, ICAM-1, and E-selectin and the production of chemokines, such as monocyte chemoattractant peptide-1 oxidants, activated ECs resulting in attraction and trans-endothelial migration of monocytes that are believed to be critical to the initiation and progression of atherosclerosis. 50 –52 Increase in serum or plasma level of soluble/surface ICAM-1 and VCAM-1 as the markers of endothelial activation have been reported for atherosclerosis. 53,54 Therefore, they were usually considered as valued markers in various vascular diseases. It is evident from numerous reports that inflammatory factors influence on adhesion molecules are mediated through the classic intracellular NF-κB signal way. 55,56 In the present study, IHC analysis for NF-κB showed a significant increase in its expression in aortic ECs in NIC-treated rabbits compared to normal rabbits. Also, ELISA revealed that NIC induced the expression of VCAM-1. These results are in agreement with those of Rodella et al. 57 who reported that significant increase in NF-κB and VCAM-1 expression was observed in rats treated with 100 µg/ml NIC in drinking water. In the present study, AG treatment for 6 weeks produced a significant decrease in serum VCAM-1 level and NF-κB expression in aortic ECs when compared with the NIC-treated rabbits. Alterations of intracellular signaling, including the NF-κB pathway accounts for the neuroprotective effects of AG. 11 Increased production of free derivatives activates NF-κB, 34,58 thus the antioxidant effects of AG in hyperglycemic mesangial cells were associated with suppressing NF-κB activation, whereas polyamines were not as much effective. 36 It is known that loss of NO in the microcirculation can result in the upregulation of proadhesive cell adhesion molecules. 59 Therefore, AG may inhibit the expression of NF-κB and subsequent inhibit the expression of VCAM-1 through its antioxidant effect and through activation of eNOS and normalizing NO production.
The present study showed that AG can protect against NIC-induced VED. The protective effect of AG may be related to its ability to nearly normalize lipid profile, decrease lipid peroxidation, enhance antioxidant status and normalize NO, and suppress the expression of the pro-inflammatory transcription factor NF-κB and the serum concentration of VCAM-1.
Footnotes
Acknowledgments
The author acknowledges Dr AA Shalabe, Department of Pathology, Faculty of Medicine, Mansoura University for providing assistance in completing the pathology section.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
