Abstract
The second-generation selective 5-HT2 receptor antagonists and reuptake inhibitors (SARIs) class antidepressants are known to have fewer cardiovascular side effects than the older ones. However, several case reports showed that trazodone, one of the second-generation SARIs, induces QT prolongation, cardiac arrhythmia, and ventricular tachycardia. Although these clinical cases suggested trazodone-induced cardiotoxicity, the toxicological actions of trazodone on cardiac action potentials (APs) beyond the human ether-a-go-go related gene (hERG) remain unclear. To elucidate the cellular mechanism for the adverse cardiac effects of trazodone, we investigated its effects on cardiac APs and ion channels using whole-cell patch clamp techniques in human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) and transiently transfected human embryonic kidney cells (HEK293) with cardiac ion channel complementary DNA. Trazodone dose-dependently decreased the maximum upstroke velocity (
Introduction
Depressive disorders acting on the autonomic neurocardiac system may occur in patients with heart disease and are associated with cardiovascular mortality. 1 –4 First-generation antidepressants, such as tricyclic antidepressants (TCAs) were demonstrated to have fewer cardiovascular side effects; however, their broad mechanisms of action led to unwanted ventricular arrhythmias and sudden cardiac death, resulting in their removal from the market. 5,6 To overcome the cardiotoxicity of first-generation antidepressants, second-generation antidepressants, such as selective serotonin reuptake inhibitors 7 –9 and the selective 5-HT2 receptor antagonists and reuptake inhibitors (SARIs), which simultaneously act as postsynaptic serotonin receptor antagonists and presynaptic serotonin reuptake inhibitors, 10 –12 were introduced. These drugs are as effective as TCAs, but have higher safety and tolerability profiles. Thus, they are preferred over first-generation antidepressants.
Although trazodone, a tetracyclic SARI, is known to have fewer cardiovascular side effects than TCAs,
13
an increasing number of trazodone studies have raised concerns regarding its cardiac safety. Several previous studies showed that trazodone inhibits human ether-a-go-go related gene (hERG) channels by interacting at the aromatic binding site of Y652 and F656 in the S6 domain of hERG.
14,15
Trazodone also inhibits outward potassium ion (K+) channels, which includes transient outward K currents and transiently, rapidly inactivating (voltage-gated potassium) Kv channels .
16,17
In addition, trazodone was reported to cause QT prolongation and torsade de pointes at both therapeutic doses
18,19
and overdose levels.
20,21
Although the likely mechanism for drug-induced long QT syndrome (LQTS) is the interaction of trazodone with KCNH2-encoded hERG K channels,
15,22
additional genes associated with LQTS can also cause life-threatening cardiac arrhythmias.
23
Mutations in genes encoding cardiac ionic channels can disrupt the fine balance of ionic currents that shape the action potential (AP) and may lead to life-threatening arrhythmias. For example, mutations to the KCNQ1/KCNE1-encoded slow component of the delayed rectifying K current (
Cardiac electrophysiological assessment of the SARI trazodone is necessary to identify the mechanisms underlying its cardiovascular side effects; however, the effect of trazodone on cardiac APs, mediated by depolarization and repolarization via ion channels, remains unclear. To elucidate the cellular mechanism of trazodone-induced cardiac adverse events, we investigated the effects of trazodone on electrophysiological cardiac characteristics using the whole-cell patch clamp technique. We evaluated the effect of trazodone on APs in human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs), which express the major cardiac ion channels and recapitulate spontaneous mechanical and electrical activity similar to adult cardiomyocytes.
27
–30
We further evaluated the effect of trazodone on major ion channels, including
Methods
Cell culture
hiPSC-CMs (iCell Cardiomyocytes; Cellular Dynamics International, Madison, Wisconsin, USA) were cultured for single-cell electrophysiology recordings. Frozen vials of hiPSC-CM were thawed in a water bath maintained at 37°C and mixed with an ice-cold plating medium (iCell Cardiomyocyte Plating Medium). The cells were transferred to four-well culture plates containing 0.1% gelatin-coated glass coverslips. The cells were then maintained in a culture incubator at 37°C in an atmosphere of 93% air and 7% carbon dioxide. After 2 days of culture, the plating medium was replaced with the culture medium (iCell Cardiomyocyte Maintenance Medium). This medium was changed every 48 h. HEK293 cells were transiently transfected with cardiac major ionic channel cDNA using lipofectamine. The transfected cDNAs, including
Whole-cell patch clamp recordings in hiPSC-CMs
Whole cell hiPSC-CM recordings were performed at 37°C using an external solution containing (in mM): sodium chloride (NaCl), 145; potassium chloride (KCl), 5.4; (4-2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 10; magnesium chloride (MgCl2), 3.5; glucose, 5; calcium chloride (CaCl2), 1.8; 4-aminopyridine (4-AP), 2; and tetrodotoxin (TTX), 0.01. The internal solution contained (in mM): Cs-Asp (cesium aspartate), 120; cesium chloride, 20; NaCl, 5; tetraethylammonium chloride, 10; HEPES, 10; ethylene glycol tetraacetic acid (EGTA), 10; and magnesium adenosine triphosphate (Mg-ATP), 5. We recorded typical APs of the hiPSC-CMs in the current-clamp mode. Spontaneous beating activity of single hiPSC-CMs were recorded and only hiPSC-CMs that could beat stably were included in the analysis. Following stabilization of the AP waveforms, the average from five recorded APs was analyzed for each test concentration. In the voltage-clamp mode, a standardized step protocol was used to elicit the major cardiac calcium ion (Ca2+) currents (
Whole-cell voltage-clamp recordings in HEK293 cells
Statistical analysis of data
pCLAMP (Axon Instruments, Foster City, California, USA), Origin 8 (OriginLab Corp, Northampton, Massachusetts, USA), and Excel (Microsoft, Redmond, Washington, USA) software were used for data acquisition and analysis. The concentration–response relationships for drug-induced blockage were calculated using SigmaPlot (Systat Software, San Jose, California, USA). The half-maximal inhibitory concentration (IC50) value, defined as the drug concentration that reduced the ionic currents by 50%, was obtained using the sigmoidal Hill equation:
Results
Effects of trazodone on AP waveforms in hiPSC-CMs
To assess whether the changes in cardiac AP properties were due to the effect of trazodone, we obtained current-clamp recordings in hiPSC-CMs (Figure 1). A spontaneous beating activity was recorded and AP-related parameters were quantified as the control condition in hiPSC-CMs (
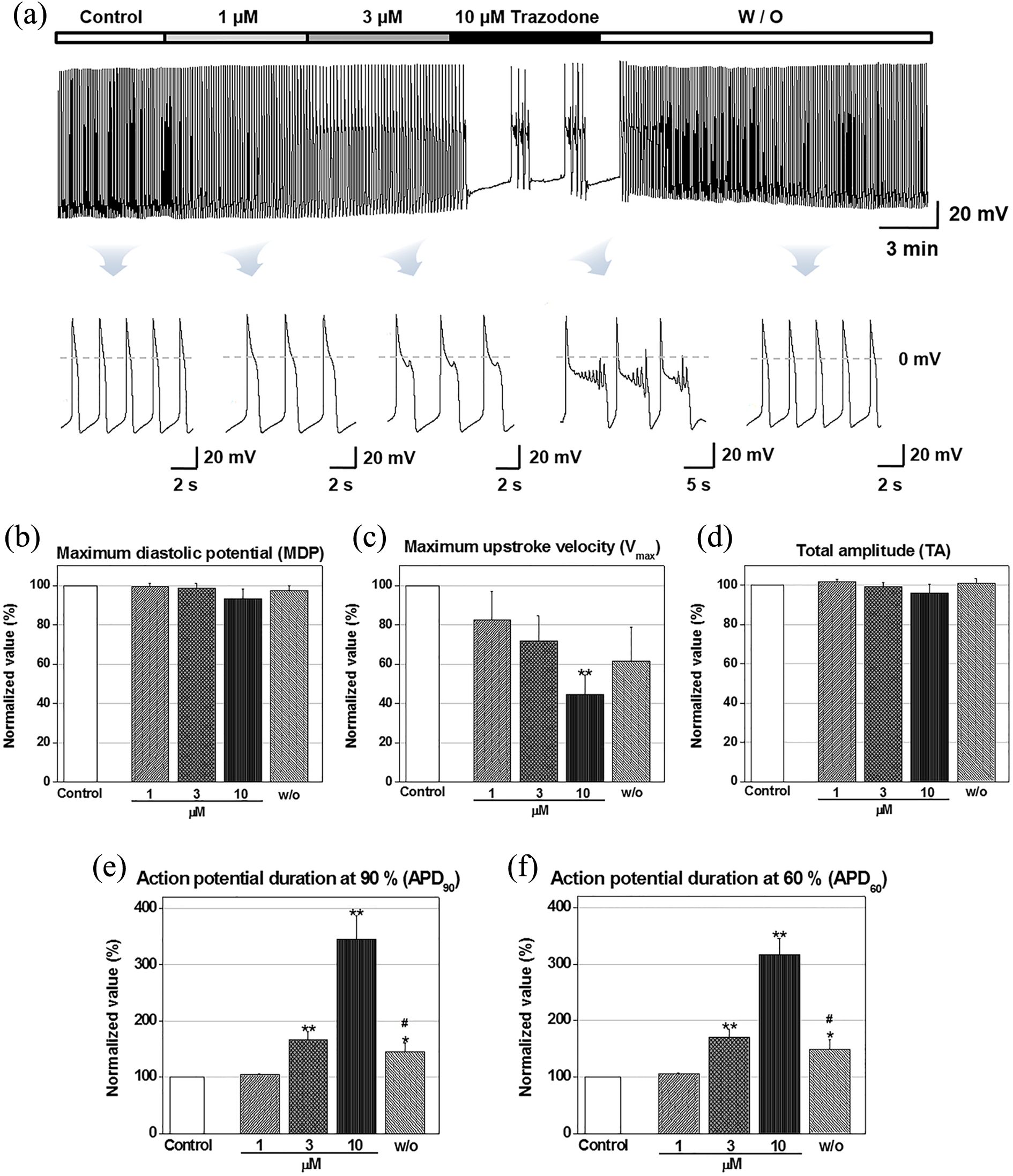
Cardiac actions of the SARI trazodone on action potential waveforms in hiPSC-CMs. (a) Representative traces induced by 1 μM trazodone. Representative traces show the typical waveform of EAD induced by 3 and 10 μM trazodone. (b) to (f), Normalized AP parameters in control and in the presence of trazodone. MDP: maximum diastolic potential;
Effects of trazodone on cardiac repolarization-related currents: I Kr and I Ks in HEK293
To investigate the cellular mechanism underlying the effects of trazodone on repolarization-related currents, KCNH2-encoded fast components of the delayed

Effect of the SARI trazodone on

Effect of the SARI trazodone on
Effects of trazodone on cardiac depolarization-related currents: I Na in HEK293 and I Ca in hiPSC-CMs
To investigate the effect of trazodone on the depolarization-related currents, the SCN5A-encoded inward Na current (

Effect of the SARI trazodone on
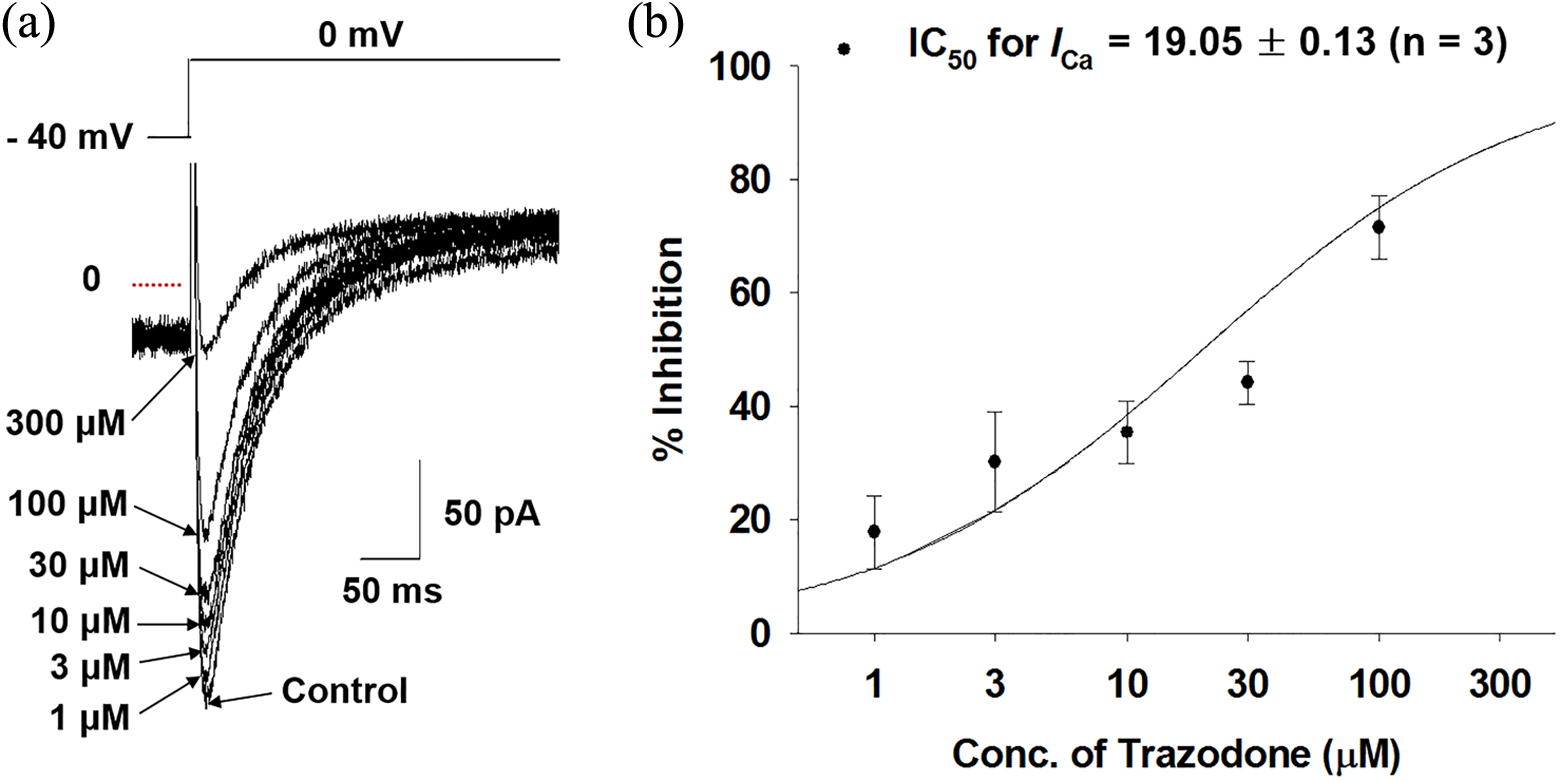
Cardiac actions of the SARI trazodone on
Discussion
Most clinical studies using SARIs suggest that trazodone has significant advantages over first-generation antidepressants; however, recent studies indicate that trazodone may have cardiovascular side effects.
18
–21
Several clinical studies suggest that cardiotoxicity is induced via trazodone acting on hERG channels, which are involved in cardiac repolarization, thereby inducing QT prolongation and ventricular arrhythmias.
15,22
Although it is widely accepted that ventricular arrhythmias are primarily caused by
In this study, we hypothesized that trazodone modulates cardiac APs, which is controlled by interactions between depolarizing (
One potential mechanism by which trazodone dose dependently prolongs APD90 (Figure 1(e)) is inhibition of repolarization-related K+ currents. Trazodone inhibited
Cardiotoxicity testing, including in vitro assays and in vivo animal models, is an important part of preclinical drug evaluation prior to first-in-human clinical trials; however, many drugs that have passed animal tests have unanticipated cardiotoxicity during clinical phases.
32
–34
Therefore, the development of a sensitive in vitro model suitable for cardiac safety testing is extremely important. The most well-known electrophysiological studies use immortalized mammalian cell lines overexpressing single ion channel; however, these are not representative of native human cardiomyocytes. Although the IC50 value of trazodone for
Recently, hiPSC-CMs have been established as an attractive model for cardiac toxicity testing, as they express the major ion channels and recapitulate spontaneous mechanical and electrical activity, similar to adult cardiomyocytes. 27 –30 We evaluated the effect of trazodone on AP parameters in hiPSC-CMs, which is a physiologically relevant preclinical evaluation of drug-induced ventricular arrhythmias. Our findings indicated that hiPSC-CMs are sensitive and accurate predictors of trazodone-induced cardiotoxicity. Trazodone-induced prolonged APD may result from the complex interactions between Na+, Ca2+, and K+ currents, though its profound effect on ventricular APD may be related to its ability to regulate K+ channels. Although hiPSC-CMs have opened new horizons for drug screening and detecting undesired proarrhythmic side effects of preclinical drugs, 35 –37 they may still show gene expression profiles similar to immature human fetal cardiomyocytes. 38 –40 The electrophysiological properties of partially mature hiPSC-CMs have mixed subtypes and restricted functionality compared to human adult cardiomyocytes, which can limit their application in drug screening. 41 –43 To overcome this constraint, it will be crucial to develop a maturation protocol for hiPSC-CMs and verify and validate the obtained results in the initial screening steps.
Trazodone was developed as 150 mg and 300 mg caplets for once daily administration and known to be well absorbed after oral administration without selective localization in any tissue.
44
In a clinical setting, the plasma concentration of trazodone increases proportionally after administration of a single dose or under steady-state conditions.
45,46
Fagiolini et al. reported that plasma concentration (
Collectively, our in vitro studies indicated that trazodone dose-dependently inhibits all of the major cardiac ion channels, including hERG. The prolonged APD and decreased
Footnotes
Authors’ Note
These authors SL and HAL contributed equally to this work as first authors. The authors SJK and KSK contributed equally to this work as corresponding authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning (MSIP), Republic of Korea (No. NRF-2012M3A9C7050138).
