Abstract
We studied the effect of silymarin and dimercaptosuccinic acid (DMSA), a chelating agent that was administered individually or in combination against lead (Pb) toxicity in rats. Wistar rats (200 ± 20) were randomly divided into five groups. Group A served as a control. Groups B–E were exposed to 2000 ppm of lead acetate in drinking water for 8 weeks. Group B served as a positive control. Group C received silymarin (100 mg kg−1 orally) for 8 weeks. Group D received DMSA (75 mg kg−1 orally) once daily for the last 5 days of treatment. Group E received DMSA and silymarin as groups C and D, respectively. The effect of Pb was evaluated and accordingly the treatments on blood lead levels (BLLs), renal system, and genotoxic effects were calculated using comet assay. The BLLs were significantly increased following the exposition of lead acetate. The administration of silymarin and DMSA provided reduction in BLLs. Silymarin and DMSA provided significant protection on the genotoxic effect of Pb. The toxic effect of Pb on kidneys was also studied. Our data suggest that silymarin and DMSA improve the renal histopathological lesions.
Introduction
Lead (Pb) is a very toxic heavy metal. Exposure to lead is impossible to avoid due to it being ubiquitous in the environment. Pb exposure can induce a broad range of physiological dysfunctions including central nervous system, hematopoietic system, reproductive system, liver, and kidneys. Consequently, Pb toxicity is a significant clinical entity especially in the health of humans. 1 –3 Chelating agents are used to reduce the Pb toxicity; however, some of them have undesirable side effects. Dimercaptosuccinic acid (DMSA) is a dithiol compound and has been used to treat Pb toxicity. 4 This effective Pb chelator is considered as an alternative to sodium calcium edetate, particularly when an oral antidote is preferred. 5 Considering that Pb causes the generation of reactive oxygen species (ROS), oxidative stress has been proposed as a possible mechanism involved in Pb toxicity. This suggests that some antioxidants may play a role in the treatment of Pb poisoning. 6 –8 Silymarin, a flavonolignan, is considered as a strong antioxidant and has been widely used as a natural medicine for liver diseases. 9 –11 The review of Dashti-Khavidaki et al. 12 provided much evidence that silymarin has a potential renoprotective effect against nephrotoxic drugs. The new medicinal properties indicate that silymarin plays protective roles in several pathologies such as Alzheimer’s disease, Parkinson’s disease, sepsis, burns, osteoporosis, diabetes, and hypercholesterolemia. These pharmacological effects are attributed due to its strong antioxidant activity. 13 –17 In this study, we evaluated the toxic effect of lead acetate on the kidneys and blood of rats based on histopathological alterations and DNA damage. We also evaluated the protective effect of silymarin and DMSA either individually or in combination against Pb toxicity.
Methods
Lead acetate, silymarin, DMSA, and all other compounds were purchased from Sigma-Aldrich (St Louis, Missouri, USA).
Animals and experimental design
Male albino Wistar rats aged 2 months and weighing 200 ± 20 g were obtained from the animal house of Division of Natural Sciences, University of Guanajuato, Mexico. All experiments were carried out according to the institutional ethical guidelines and the Mexican guidelines NOM-062-Z00-1999 for the care of experimental animals. 18 The rats were randomly housed (five rats per cage) and maintained at standard conditions with a 12-h light/12-h dark cycle and temperature of 22 ± 3°C. The animal experiments were conducted for 8 weeks.
Group A, the negative control, was given only standard rat chow and water. Groups B, C, D, and E were exposed to 2000 ppm of lead acetate in their drinking water ad libitum for 8 weeks. Group B was the positive control. Group C was given silymarin at doses of 100 mg kg−1 orally for 8 weeks. Group D was given DMSA at doses of 75 mg kg−1 orally for 5 days in the last week of treatment. Group E received DMSA and silymarin as groups C and D, respectively.
Silymarin and DMSA solutions were freshly prepared and were administered by gavage. Control animals were administered water in the same way on the same schedule. One day after the last treatment, rats were anesthetized with sodium pentobarbital, and blood samples were collected with Pb-free needles via intracardiac puncture. Kidneys were resected, rinsed in cold saline, and used for histopathological observations.
Blood Pb determination
Blood lead levels (BLLs) were determined following the procedure standardized in the laboratory according to the NOM-199-SSA1-2000 using an atomic absorption spectrophotometer. 19
Comet assay
The comet assay used was based on a previously described protocol. 20 In brief, lymphocytes were mixed with 500 µL of 0.7% agarose, and 250 µL of the mixture was transferred onto a frosted slide precoated with normal melting agarose (1%). A coverslip was added, and the slide was cooled on ice to allow the agarose to harden. Then, the slides were treated for 1 h with cold lysis buffer (100 mM tris(hydroxymethyl)aminomethane, pH 10; 5 M sodium chloride; 1 M ethylenediaminetetraacetic acid (EDTA); 1% (v/v) Triton X-100; 10% (v/v) dimethyl sulfoxide (DMSO)). After lysis, the slides were washed and immersed in cold alkaline unwinding electrophoresis solution (10 N sodium hydroxide and 1 M ethylenediaminetetraacetic acid disodium salt in deionized water, pH 13.5) for 30 min and subjected to electrophoresis for 30 min at a constant voltage of 25 V. All procedures were carried out in the dark. The slides were stained with 4′,6-diaminido-2-phenylindole and analyzed using fluorescence microscopy. Triplicate samples, each containing 50 cells, were quantified for each condition. The tail moment was used as a DNA damage parameter.
Histopathological examination
Immediately after removal, the kidney was fixed in 10% formalin for 5 days and then dehydrated and embedded in paraffin wax. After dehydration and clearing, tissue was embedding in paraffin wax for 1 h in each step. Specimens were cut into 5-μm-thick sections and stained with hematoxylin and eosin stain. Histopathological changes were observed using light microscopy.
Statistical analysis
The results are expressed as mean ± SD. The comparison of means was carried out using one-way analysis of variance test complemented with Tukey test. For comet assay, data were analyzed using Kruskal–Wallis test. The values of p < 0.05 were considered statistically significant. All statistical analyses were performed using JMP software (software 4.0.3).
Results
Pb exposure caused a significant increase in BLLs. (Table 1). BLLs decreased significantly in groups that received DMSA for 5 days (Table 1). Both silymarin and DMSA were effective in reducing the Pb concentrations in blood, though the efficiency of DMSA was better than silymarin.
Effects of silymarin, DMSA, or their combination on blood lead levels in lead-exposed rats.a

Pb: lead; DMSA: dimercaptosuccinic acid.
aLead acetate: 2000 ppm in drinking water; silymarin: 100 mg kg−1; DMSA: 75 mg kg−1; silymarin + DMSA: 100 + 75 mg kg−1. The results are expressed as mean ± SD (n = 5).
bp < 0.05: compared with positive control group.
Lymphocytes from rats treated with lead acetate showed significantly increased DNA damage compared with lymphocytes from the control group (this was determined based on the tail moment values determined using comet assay). DNA damage was reduced significantly in rats cotreated with silymarin and DMSA (Table 2). Lymphocytes from rats exposed to Pb and treated with silymarin and DMSA showed similar tail moment values with respect to the lymphocytes from negative control group.
Effects of silymarin, DMSA, or their combination on the tail moment value in peripheral blood lymphocytes of lead-exposed rats.a

Pb: Lead; DMSA: dimercaptosuccinic acid.
aLead acetate: 2000 ppm in drinking water; silymarin: 100 mg kg−1; DMSA: 75 mg kg−1; silymarin + DMSA: 100 + 75 mg kg−1. The results are expressed as mean ± SD (n = 5).
bp < 0.05: compared with positive control group.
Histopathological examinations indicate that control rats showed normal glomerular and tubular morphology with a well-preserved nucleus and cytoplasm (Figure 1). The degree of injury was significant in group exposed to Pb. Cells of renal tubules have loss of apical microvilli; in addition, it has minimal amounts of cytoplasm. Renal cells were irregular in shape and showed changes in nuclear and cell sizes (Figure 2). The treatment of Pb-exposed animals with silymarin and/or DMSA showed marked improvement in histopathological findings (Figures 3 and 4). Significant differences were not found between groups treated with silymarin and DMSA. Interestingly, the combined treatment of silymarin and DMSA showed more normal morphology than their individual administration (Figure 5). Finally, we did not find any differences between the control group and the group that received combined treatment.
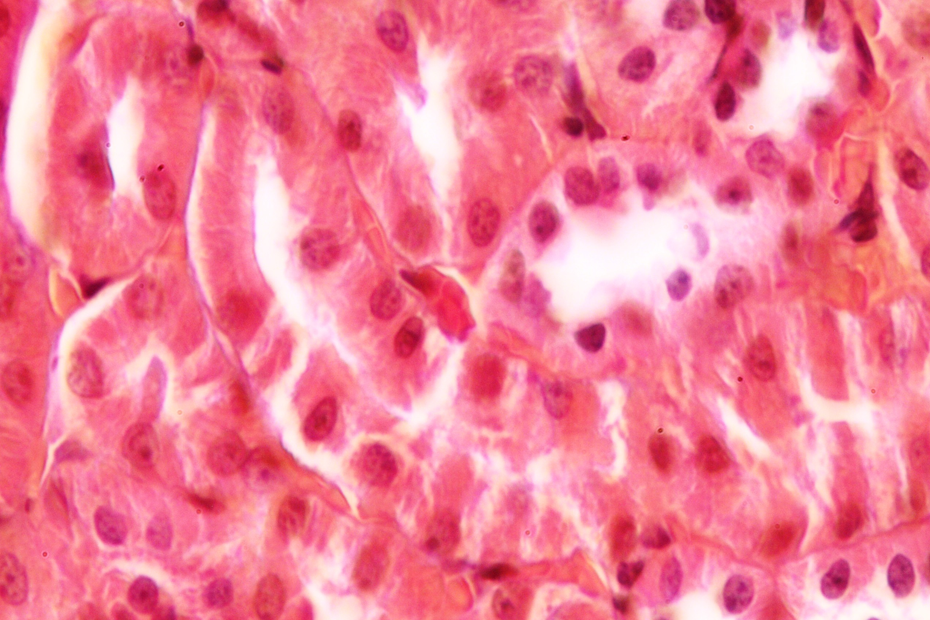
Control group showing normal renal architecture.

Lead acetate causes epithelial desquamation in the proximal tubules. This group has marked decrease in cytoplasmic volume, and some nuclei are observed practically naked.
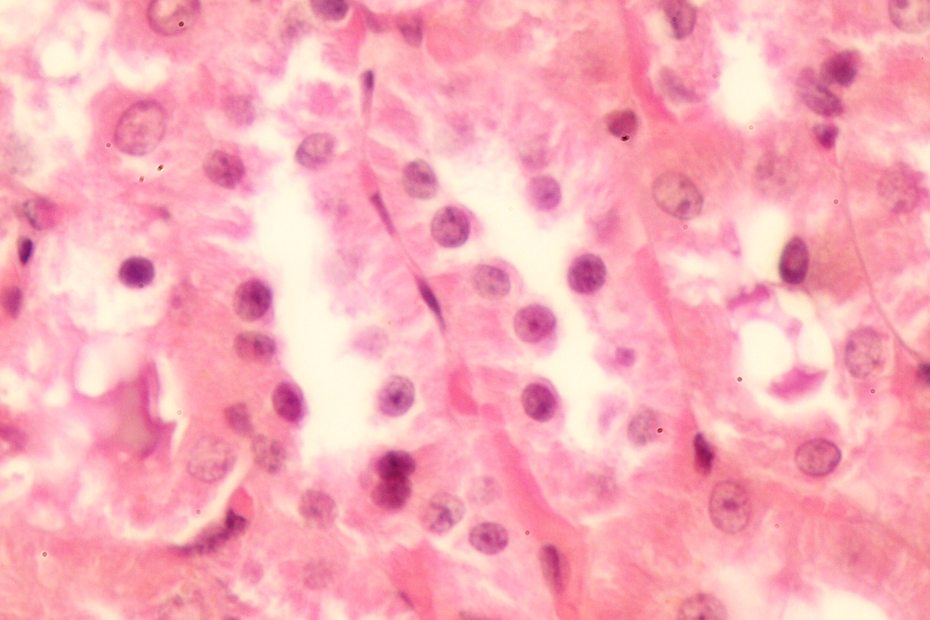
Effect of silymarin on lead acetate-treated group showing cytoplasmic preservation with more cytoplasmic volume.

Effect of DMSA on lead acetate-treated group showing cytoplasmic preservation with more cytoplasmic volume. DMSA: dimercaptosuccinic acid.

Effect of silymarin and DMSA on lead acetate-treated group displaying remarkable improvement in the histological appearance of renal tubules. DMSA: dimercaptosuccinic acid.
Discussion
Pb has been used for centuries, and its chronic toxicity has caused various health problems. Oxidative stress has been proposed as a possible mechanism involved in Pb toxicity; therefore, the treatment of this toxicity is directed on one side to remove Pb from the body and on the other to scavenge generated ROS. In this regard, the treatment of Pb toxicity can include some therapeutic strategies such as chelation and antioxidant administration. Numerous studies have shown that antioxidants administered individually or combined with chelating therapies have a positive impact on Pb-induced toxicity. 21 –23 In the present study, the role of silymarin and DMSA on Pb toxicity was investigated in Wistar rats. The doses of silymarin and the course for 5 consecutive days for DMSA were based on the previous reports. 24,25 We found that Pb exposition caused a significant increase in the BLLs compared with the control group. DMSA administration caused a significant decrease in BLLs. Similar results were obtained by other researchers who found that DMSA is an effective chelator and BLLs can be diminished within cycles of 5 days of treatment. 26 Silymarin treatment causes only a moderate reduction in BLLs, and its combination with DMSA was not better than individual administration. Silymarin is a mixture of flavonolignan isomers and flavonoids and is well recognized as possessing a wide range of pharmacological properties. 27 Silibinin is a major compound of silymarin. This compound has been reported to have antioxidant efficacy as metal chelators. Muthumani and Prabu 28 suggest that silibinin has a protective effect on arsenic-induced toxicity in hepatic tissue in rats. Shalan et al. 29 studied the combined administration of silymarin and vitamin C on Pb toxicity, and they reported that the coadministration provided improvement in the biochemical, molecular, and histopathological findings; however, they did not report its effects on BLLs.
Many studies have shown that Pb can induce genotoxicity. 30 It has been suggested that this effect is associated with ROS produced by Pb exposure. 31 The present results show that lead acetate is genotoxic in Wistar rats. This damage is indicated by a significant increase in the tail moment values determined using comet assay. The treatment with silymarin during Pb exposition for 8 weeks diminished the DNA damage compared to that of the positive control. This might be due to the antioxidant effect of silymarin and its capacity to scavenge ROS. This finding agrees with other investigations, which showed that silymarin is a potent antioxidant and might act as an antigenotoxic agent. 32,33
The renal system is one most damaged by Pb exposition. This study also showed that Pb induced renal toxicity, as evidenced by histopathological changes that were consistent with the observations of other investigators. 34,35 Renal sections of rats exposed to lead acetate showed abnormal histology, including the loss of apical microvilli and minimal amounts of cytoplasm. Our results revealed that silymarin and DMSA ameliorated the kidney damage induced by lead acetate. Despite the fact that the silymarin was less effective in reduced BLLs, the histopathological examination of kidney tissues confirmed that silymarin improved the histological alterations. These results may be attributed to the protective antioxidant effect of silymarin. Furthermore, renal tissue was found similar to the control group when silymarin and DMSA were administered together. These results are consistent with those reported in the previous literature which suggested that coadministration of antioxidant and chelating agent is better than individual treatment. 6,36
In conclusion, the findings of the present work indicated that exposure to lead acetate induced severe DNA damage and histological changes in the kidney. These changes were supported by reduction of DNA damage in lymphocytes and histopathological observations of the kidney. Silymarin and DMSA had a protective effect against Pb toxicity. This effect may be due to its antioxidant and chelating effects. DMSA was found to be more effective than silymarin in reduced BLLs. These effects may be due to its attribution to eliminating Pb via chelating. These results suggest that combined administration of silymarin and DMSA has a potential protective effect in Pb-exposed rats.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Universidad de Guanajuato (DAIP-UG 2013).
