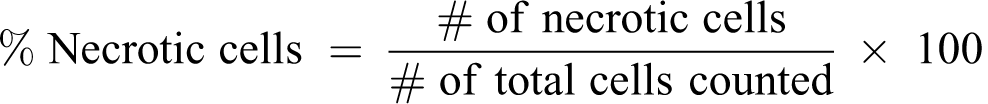Abstract
The aim of this study was to investigate genotoxic and cytotoxic effects of doxorubicin, silymarin, or in combination on HepG2 cells for 24 and 48 h. Both doxorubicin and silymarin caused dose-dependent inhibition of cell proliferation. After 48 h of treatment, doxorubicin application caused dramatically increased ratio of apoptotic cells. Both 24 and 48 h of silymarin and doxorubicin–silymarin combination caused significant increases in the rate of apoptotic cells. Applications of doxorubicin and silymarin separately for 24 h led to deoxyribonucleic acid (DNA) damages. After 48 h of incubation, doxorubicin-induced genotoxic damage was 2-fold higher than the silymarin-induced damage. After 24 and 48 h, DNA damage in response to combined applications of doxorubicin and silymarin was indifferent from silymarin- and doxorubicin-induced damage respectively. There was not any difference in genotoxicity levels between incubation periods in combined applications of doxorubicin and silymarin. Lipid peroxidation levels increased in all applications. Biopharmacotherapy with chemotherapeutic agents are of interest in the issue of adjuvant therapy. Here, we demonstrate in vitro potential genotoxic and cytotoxic antitumor effect of silymarin on HepG2 cells at achievable plasma level concentrations.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most prevalent solid cancer type worldwide, and the fourth leading cause of cancer-related deaths. 1 Liver transplantation and surgical resection considerably decrease mortality and increase survival rate. However, these therapies are still effective in a restricted number of patients. 2 Moreover, many patients are not operated because of advanced cirrhosis, large primary lesions, multifocal disease, thrombosis of major blood vessels, and extra hepatic metastases. Since 70% of the patients having resection are refractory to the disease, development of new therapy strategies rather than surgery is very important in the case of HCC. 3 HCCs are also highly resistant to conventional systemic chemotherapy. Among the chemotherapeutics used, only partial sensitivity is obtained with doxorubicin. Doxorubicin has multiple action mechanisms on cells. First, it intercalates and interferes with deoxyribonucleic acid (DNA) and ribonucleic acid syntheses. Second, it inhibits topoisomerase II leading to DNA strand breakages. 4 Third, it can cause the formation of reactive oxygen species, and free radical damage on cells. 5 Collectively, the effects are inhibition of cell proliferation, induction of G2-M cell arrest, or apoptosis. 6,7 Despite its multifactorial antitumor activity, only 20% of the HCC patients respond to doxorubicin treatment, according to recent studies. 8 New systemic combinational chemotherapy strategies are required. However, such studies have demonstrated that the results obtained were not better than that of doxorubicin. 9
Epidemiological studies demonstrate that herbal-based supplements might be effective in the prevention of cancer. Among these supplements, silymarin potency on the issue has raised great attention. 10 Silymarin is a bioactive component found in the extracts of the milk thistle (Silybum marianum) seeds. It is composed of four flavonoid isomers: silibinin, isosilibinin, silydianin, and silychristin. It has antioxidant and anticarcinogenic effects and has been used as a complementary and an alternative agent by chemotherapy patients. 11 On a voluntary basis, silymarin has been reported to be one of the most frequently used herbal preparations by cancer patients. 12 Furthermore, it is widely used for its antihepatotoxic effects in liver diseases. In the past 30 years, it is clinically used in Europe and Asia in the commercial form of Legalon™ and Silipide IdB1016. 10
Many compounds are widely used as complementary medication in patients taking chemotherapy. Effects of these compounds alone or in combination with chemotherapeutics should be well defined in tumors at the cellular basis. In addition, cell-based studies with natural compounds would enable the development of new systemic combinational chemotherapy strategies. The aim of this study was to investigate genotoxic and cytotoxic effects of mostly effective chemotherapeutic agent doxorubicin and widely used supplement silymarin alone or in combination at achievable plasma concentrations on HepG2 HCC cell line.
Methods
Cell culture and treatments
In the present study, HepG2 human HCC cells, which are capable of metabolizing many nutrients, metabolites, and xenobiotics, were used. In addition to the secretion of plasma proteins and lipoproteins, these cells also have inductive P450 cytochromes.
1
Cells were purchased from the American Type Culture Collection (ATCC) and cultured according to the protocol provided by the ATCC as an attached type monolayer culture. Cells were maintained in Dulbecco’s minimal essential medium (Biochrom AG, Berlin, Germany) supplemented with 10% heat-inactivated fetal bovine serum (Biochrom AG),
Assay for cell proliferation
The effects of doxorubicin, silymarin, and their combination on the proliferation of HepG2 cells were colorimetrically tested by biochemical reduction of 3-(4,5-dimethyl-2-thiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTTSigma-Aldrich). The compounds were diluted from high to low concentrations horizontally in 96-well microtiter plates. Cells were seeded to each well (1 × 105 cells) with the exception of medium control wells. The plates were incubated for 24 or 48 h and then 20 µL of MTT solution (5 mg/mL) was added to each well. After incubation for 4 h, 100 µL sodium dodecyl sulfate (Sigma-Aldrich) solution (10% w/v) was added to each well. The plates were further incubated overnight to allow the dissolution of formazan crystals that were produced by mitochondrial activity of the viable cells. The inhibition of cell proliferation was determined by measuring the optical density of the chromogenic product at 540 nm with an enzyme-linked immunosorbent assay reader (Biotek Instrument ELx800, Winooski, Vermont, USA). Inhibition of cell proliferation and inhibitory concentration of 50 (IC50), which are drug concentrations at which 50% of cells are viable, was calculated from the logarithmic trend lines of the proliferation graphs.
Assessment of apoptosis and necrosis by acridine orange/ethidium bromide (AO/EtBr) staining
Apoptosis, necrosis, and viable cells were differentiated after staining with a modified AO/EtBr double staining.
16
Trypsinized cells were resuspended in 0.5 mL phosphate-buffered saline (PBS), and cell suspension was mixed with AO (4 µg/mL) and EtBr (0.5 µg/mL). The suspension was immediately examined by fluorescence microscopy (Nikon, Eclipse 600, Japan) at 400× magnification. A minimum of 300 cells were considered for every treatment. Frequencies of apoptotic and necrotic cells, which were identified according to the previous study,
16
were calculated as:
Alkaline comet assay (alkaline single-cell gel electrophoresis; SCGE)
For the determination of genotoxic effects of doxorubicin, silymarin, and doxorubicin–silymarin treatments on HepG2 cells, alkaline SCGE were performed as previously described. 17 In brief, trypsinized cells were resuspended in 0.5 mL PBS and 5 µL of cell suspension were mixed with 35 µL of 1% (w/v) low melting point agarose (LMPA; Sigma-Aldrich) and added on to the slides coated with 0.5% (w/v) normal melting point agarose (NMPA; Sigma-Aldrich). Coverslips were placed and slides were incubated on ice packs until the solidification of agarose. Coverslips were removed and 40 µL of 1% (w/v) LMPA was added on to the slides. Slides were incubated in lysis solution (2.5 M sodium chloride, 100 mM ethylenediaminetetraacetic acid (EDTA) disodium salt, and 10 mM Tris; pH 10) at 4°C (dark) for 2 h. Slides were incubated in electrophoresis buffer (300 mM sodium hydroxide, 1 mM EDTA disodium salt; pH > 13) for 20 min at dark and electrophoresis was performed at 24 V (300 mA) for 30 min. After neutralization (0.4 M Tris; pH 7.5), the slides were stained with 2 µg/mL EtBr and observed under fluorescence microscope (Nikon, Eclipse 600, Japan). A computerized image analysis system (comet assay IV; Perceptive Instruments, Bury St Edmunds, UK) was employed. Olive tail moment (TM), which is a function of tail and head length, and fraction of total DNA in the tail, was used as the end point measure of DNA damage. Tail intensity (TI) values were also given. A minimum of three SCGE slides were prepared for each treatment and in total 150 nuclei were analyzed per treatment.
Lipid peroxidation
Lipid oxidation was determined by the modified method of Ahn et al. 18 Cells were seeded to 25 cm2 culture flasks and treated with doxorubicin, silymarin, and doxorubicin and silymarin for 24 and 48 h. Trypsinized cells were resuspended in 1 mL PBS and lysed with repetitive freezing and thawing at −20°C with vigorous vortexing in between freezing and thawing. Protein amount of lysates was determined according to Bradford. 19 Twenty-five microliters of 0.04 M butylated hydroxytoluene (Sigma-Aldrich) in ethanol were added to prevent the artificial increase in malondialdehyde during the experiment. After addition of 0.5 mL of 30% (w/v) trichloroacetic acid (TCA; Sigma-Aldrich), the suspension was vortexed. Three milliliters of thiobarbituric acid (TBA; Sigma-Aldrich)/TCA (20 mM TBA in 15% (w/v) TCA) solution were added and the samples were vortex mixed. Samples were incubated in boiling water for 1 h for the development of color. Samples were cooled to room temperature on ice and centrifuged at 4000 rpm for 5 min. Absorbance of the supernatants were measured at 532 nm and TBA reactive substance (TBARS) concentrations were determined using extinction coefficient of 155 mM−1 cm−1. Lipid peroxidation was expressed as nanomole of TBARS per milligram protein.
Statistical analysis
All data are expressed as mean ± standard error of the means. All statistical analyses were performed using SPSS (Statistical Package for the Social Sciences; SPSS Inc., Chicago, Illinois, USA) software. Shapiro–Wilk test was used to test normality of distributions, and the homogeneity of variances was tested by Levene’s test. In case of normal distribution and homogeneity of variances, treatments were statistically evaluated by one-way analysis of variance at 0.05 levels, and post hoc Tukey analysis was carried out to find groups whose mean differences were significant. Differences between 24 and 48 h in treatments were statistically evaluated by independent samples t-test at the 0.05 level. If data were not normally distributed and variances were not homogenous, Mann–Whitney U or Kruskal–Wallis with post hoc Dunn’s test was used at 0.05 levels for the comparison of incubation periods and treatments, respectively.
Results
Antiproliferative effects of doxorubicin and silymarin on HepG2 cells
The effects of doxorubicin and silymarin on the proliferation of HepG2 cells for 24 and 48 h were examined by MTT assay. According to Figure 1, both doxorubicin and silymarin significantly (p < 0.05) inhibited the growth of HepG2 cells in a concentration- and time-dependent manner. For doxorubicin, IC50 for 24 h was 6.94 ± 0.04 µM where it was 2 ± 0.1 µM for 48 h of treatment. In the case of silymarin, IC50 values were 396.5 ± 83.5 ng/mL and 208 ± 12 ng/mL for 24 and 48 h of treatment, respectively. At the relevant concentrations of plasma level, that is, 4 µM doxorubicin and 300 ng/mL silymarin, cell proliferation of HepG2 cells decreased to 52.02% and 53%, respectively, after 24 h of treatment. Although 48 h of treatment with both doxorubicin and silymarin caused higher decrease in cell proliferation (28.02% and 40.72%, respectively), doxorubicin was more effective in longer incubation period than was silymarin.

Effects of (a) doxorubicin and (b) silymarin on the proliferation of HepG2 cells for 24 and 48 h. All the differences in cell proliferation of treatment and control groups were significant at 0.05 levels according to one-way ANOVA except for 1 µM for 24 h in doxorubicin applied group. ANOVA: analysis of variance.
Doxorubicin- and silymarin-induced apoptosis in HepG2 cells
Table 1 summarizes the results of AO/EtBr staining. Doxorubicin treatments at both 24 and 48 h significantly induced apoptosis when compared to control HepG2 cells. Frequency of apoptotic and necrotic cells increased significantly when the incubation duration of doxorubicin was increased. Silymarin treatments significantly increased the percentage of necrotic and apoptotic cells when compared to the control group, and the percentage of necrotic cells increased significantly after 48 h of silymarin treatment when compared to 24 h of treatment. Combined application of doxorubicin and silymarin to HepG2 cells for 24 and 48 h significantly increased apoptosis and necrosis when compared to control cells. In addition, 24 h of combined application increased apoptosis and necrosis when compared to single applications of both doxorubicin and silymarin. After 48 h of combination treatment, the percentage of apoptotic cells increased when compared to silymarin treatment alone. Moreover, increased incubation period in the combined application caused significant increases in the percentage of apoptotic and necrotic cells.
Frequencies of apoptotic and necrotic cells in doxorubicin- and silymarin-treated HepG2 cells.

Superscript letters represent significant difference in frequency of apoptotic cells between treatment groups (i.e. acontrol 24 h vs. other 24 h of treatments; bcontrol 48 h vs. other 48 h of treatments; cDOX 24 h vs. DOX 48 h, SLY 24 h, and DOX + SLY 24 h of treatments; dDOX 48 h vs. SLY 48 h of treatments; eSLY 24 h vs. DOX + SLY 24 h; fSLY 48 h vs. DOX + SLY 48 h of treatments; gDOX + SLY 24 h vs. DOX + SLY 48 h of treatments). Superscript numbers represent significant difference in frequency of necrotic cells between treatment groups (i.e., 1control 24 h vs. other 24 h of treatments; 2control 48 h vs. other 48 h of treatments; 3DOX 24 h vs. DOX 48 h and DOX + SLY 24 h of treatments; 4 DOX 48 h vs. SLY 48 h of treatments; 5SLY 24 h vs. SLY 48 h, DOX + SLY 24 h of treatments; 6DOX + SLY 24 h vs. DOX + SLY 48 h of treatments). DOX: doxorubicin; SLY: silymarin.
Genotoxic effects of doxorubicin and silymarin on HepG2 cells
Results obtained with the comet assay are summarized in Figure 2. In Figure 2(a), representative micrographs of the DNA damage obtained from comet assay and evaluation are given. Doxorubicin and silymarin treatments induced time-dependent increase in average TMs (Figure 2(b)) as well as TI. Control groups had 1.73 ± 0.23 (TI; 7.3 ± 0.5) and 1.88 ± 0.08 (TI; 7.7 ± 0.6) µm TM for 24 and 48 h, respectively, whereas these values increased to 7.7 ± 0.74 (TI; 27.9 ± 2.2) and 12.83 ± 1.55 (TI; 50.2 ± 4.1) µm in doxorubicin applied cells for 24 and 48 h, respectively. Conversely, TM was higher in 24-h silymarin-treated cells than the 48-h silymarin-treated cells (11.13 ± 0.81 and 7.5 ± 0.74 µm TM, respectively, with 46.6 ± 3.4 and 25.2 ± 1.1 TI, respectively). In concordance, 24 and 48 h combination applications of doxorubicin and silymarin caused increases in TMs (11.50 ± 0.70 and 10.17 ± 0.18 µm, respectively) and TIs (45.5 ± 2.6 and 32.8 ± 1.9, respectively). Results obtained with 24-h doxorubicin treatment were significantly lower than the results obtained with 24 h of silymarin and combination treatments. Conversely, DNA damage was significantly higher in the 48-h doxorubicin treatment group than the 48-h silymarin and combination treatment groups. Nonetheless, DNA damage in the 48-h doxorubicin- and silymarin-treated groups was significantly higher than that in the 48-h silymarin-treated group.

(a) Representative micrographs for comet evaluation and (b) induction and frequency of DNA damage in doxorubicin- and silymarin-treated HepG2 cells. * represents significant difference between 24-h treatment groups (i.e., *control vs. treatments, **DOX vs. SLY and DOX + SLY treatments); × represents significant difference between 48 h treatment groups (i.e., ×control vs. treatments, ××DOX vs. SLY and DOX + SLY treatments, ×××SLY and DOX + SLY treatments); a and b values represent significant difference between 24 and 48 h of incubation periods. DNA: deoxyribonucleic acid; DOX: doxorubicin; SLY: silymarin.
Lipid peroxidation levels in doxorubicin- and silymarin-applied HepG2 cells
According to TBARS levels (Figure 3), all treatments caused significant increases in lipid peroxidation levels when compared to controls (0.33 ± 0.01 and 0.34 ± 0.01 nmol/mg protein for 24 and 48 h, respectively). Lipid peroxidation in doxorubicin-treated cells (1.91 ± 0.05 and 2.04 ± 0.05 nmol/mg protein for 24 and 48 h, respectively) was significantly higher than that of silymarin- and doxorubicin–silymarin-treated cells. The 24 h of combined application of doxorubicin and silymarin caused significantly lower lipid peroxidation (1.33 ± 0.05 nmol/mg protein) than that of 24-h silymarin alone application (1.61 ± 0.08 nmol/mg protein). Conversely, 48 h of combined application caused significantly increased lipid peroxidation (1.56 ± 0.12 nmol/mg protein) when compared to results obtained with the 48-h silymarin treatment (1.13 ± 0.03 nmol/mg protein). Twenty-four hours of silymarin treatment caused significantly higher lipid peroxidation than that of 48 h of silymarin treatment.

Lipid peroxidation (TBARS) levels of doxorubicin- and silymarin-treated HepG2 cells. * represents significant difference between 24-h treatment groups (i.e., *control vs. treatments, **DOX vs. SLY and DOX +SLY treatments, ***SLY vs. DOX + SLY treatments); × represents significant difference between 48 h of treatment groups (i.e., ×control vs. treatments, ××DOX vs. SLY and DOX + SLY treatments, ×××SLY and DOX + SLY treatments); a represents significant difference between 24 and 48 h of incubation periods in SLY treatment. TBARS: thiobarbituric acid reactive substance; DOX: doxorubicin; SLY: silymarin.
Discussion
Surgical therapies against HCC provide efficiency to some extent. However, patients who cannot be operated or have metastasis suffer from poor prognosis. In these cases, locoregional therapies for liver are more applicable. Such treatment strategies include intra-arterial infusion of combinational chemotherapy, chemoembolization, and selective internal radiation, which is still not applicable in all cases. Single-agent chemotherapy strategy including doxorubicin, on the other hand, has low response rate in HCC. 8
Silymarin has been used for over 2000 years in the herbal treatment of hepatitis and cirrhosis for its protective effect on the liver. Silymarin has antioxidant, anti-inflammatory, membrane stabilizing, and immunomodulatory functions. 11 It has been used in many countries as complementary and alternative therapy. 12 Previous in vivo studies demonstrated that silymarin had protective effect on doxorubicin-induced hepatotoxicity by (i) decreasing hydroxyl radical effects, (ii) reducing oxidative stress, (iii) protecting integrity of the genome, and (iv) its antagonist effect on apoptotic and necrotic pathways of normal liver cells 12,20,21
In the present study, doxorubicin and silymarin were applied alone to HepG2 human HCC cells or in combination for 24 and 48 h. Doxorubicin and silymarin concentrations were determined based on the previous studies reporting achievable peak plasma concentrations. 13,15 In the case of silymarin, peak plasma concentrations achieved with 600 mg of daily supplements, 300 ng/mL (0.62 μM), were applied to cells. According to MTT assay (Figure 1), both doxorubicin and silymarin significantly inhibited growth of HepG2 cells in a concentration- and time-dependent manner. The IC50 values of doxorubicin were 6.94 ± 0.04 µM and 2 ± 0.1 µM for 24 and 48 h of treatments, respectively. In the case of silymarin, the IC50 values were 396.5 ± 83.5 ng/mL and 208 ± 12 ng/mL for 24 and 48 h treatments, respectively. Results also demonstrated that both doxorubicin and silymarin caused apoptotic and necrotic cell death in 24 and 48 h (Table 1). Unlike silymarin applications, doxorubicin-induced cell death was considerably increased in the 48-h treatment group when compared to the 24-h doxorubicin treatment group. Silymarin induced apoptosis was higher than that of doxorubicin in the 24-h treatment period. Combined applications of doxorubicin and silymarin caused higher apoptosis and necrosis of cells in 24 h than single agent applications. However, when the incubation period was increased to 48 h, the results of the AO/EtBr staining obtained from combined applications were statistically in different from those obtained using single agent applications.
The comet assay or SCGE assay is a rapid, sensitive, and relatively simple method for detecting DNA damage. It is accepted as a standard method for assessing DNA damage in individual cells. 22 According to SCGE results (Figure 2), doxorubicin caused time-dependent DNA damage. On the contrary, DNA damage caused after 48 h of silymarin treatment was lower than that of 24 h of treatment. Results obtained from combinational applications did not change based on the incubation period.
Increase in TBARS is an indication of lipid peroxidation in cells. Both 24 and 48 h of doxorubicin treatment caused increase in TBARS. These values were higher than that of TBARS obtained from cells treated with silymarin only and a combination of silymarin and doxorubicin. Lipid peroxidation caused by silymarin decreased when the incubation period was prolonged to 48 h, whereas the values of doxorubicin treatment did not change significantly.
When doxorubicin is administered to patients, plasma concentration of doxorubicin rapidly decreased within few hours. But the drug showed a longer antitumor effect. 13 On the other hand, the plasma level of silymarin has previously been shown to decrease in parallel with its cytotoxicity. 15 Concordantly, the cytotoxicty of doxorubicin increased when incubated for a longer period of time, whereas the antiproliferative and genotoxic effects of silymarin decreased after 24 h of incubation period.
In a previous study, 25–100 µM silibinin was reported to increase the antitumor effects of conventional chemotherapeutics including doxorubicin on Michigan Cancer Foundation-7 mammary carcinoma cell line. In the same study, apoptotic effects of doxorubicin increased considerably. 23 Likewise, in prostate carcinoma cells, 10–100 μM silibinin increased the inhibitory effects of doxorubicin on cell cycle. 24 Considering HCC, in vitro growth inhibitory effects of 120 mM ≤ silibinin were reported by Lah et al on HuH7, HepG2, Hep3B, and PLC/PRF/5 cells. 25 Inhibition of HepG2 cell growth by 100 mg/mL application of silymarin was also reported. 26 Based on the previous findings, in the present study we tested the in vitro combinational effects of silymarin and doxorubicin at silymarin concentrations achieved in the plasma when reasonable daily supplements were taken. In fact, 0.62 μM (300 ng/mL) of silymarin concentration (rather than active constituent silibinin) is far less than the previously reported concentrations. In our study, combination of silymarin and doxorubicin increased the apoptotic cell rates when compared to single agent. However, similar pattern was shown neither for results of comet assay nor for TBARS. According to our results, 0.62 μM silymarin alone had antitumor potency caused by DNA damage, induction of apoptotic and necrotic cell death and lipid peroxidation. When silymarin was applied in combination with doxorubicin, lipid peroxidation did not seem to increase when compared to doxorubicin application. DNA damage in this case was higher and different than doxorubicin for 24 and 48 h of treatments, respectively, which is probably related to direct genotoxic effect of doxorubicin, which becomes more considerable after division of cells. Though increases in lipid peroxidation and DNA damage may point out silymarin-induced oxidative damage in HepG2 cells, there could also be other molecular mechanisms directly affecting apoptosis in these cells, since apoptosis in HepG2 cells increased when silymarin and doxorubicin were applied in combination.
Considering cytotoxic and genotoxic potential of silymarin, comparable results with doxorubicin were obtained: (1) silymarin inhibited growth of HepG2 cells in a concentration- and time-dependent manner, (2) in particular, at the plasma level concentrations the achievable cell proliferation of HepG2 cells decreased approximately to 50% after 24 h of doxorubicin or silymarin treatments, and after 48 h cytotoxicity of doxorubicin increased more drastically than that of silymarin, (3) both doxorubicin and silymarin caused apoptotic and necrotic cell death after 24 and 48 h, whereas combined applications of doxorubicin and silymarin caused higher apoptosis and necrosis of cells in 24 h than that of single agent applications, (4) doxorubicin caused time-dependent DNA damage, whereas DNA damage induced by silymarin was more prominent in 24 h of treatment, and (5) single agent and combined applications caused increments in lipid peroxidation, and lipid peroxidation caused by silymarin decreased when the incubation period was prolonged to 48 h. Here we report in vitro potential genotoxic and cytotoxic antitumor effect of silymarin on HepG2 human HCC cells at achievable plasma level concentrations with emphasis on time relation.
Silymarin is already in clinical use as an anti-hepatotoxic agent and consumed as a dietary supplement around the world, and currently, it is known to be devoid of any toxicity and side effects. Newer chemotherapeutic modulators and drugs target specific cancer cell signal transduction pathways. Potential of silymarin for targeting mitogenic and survival pathways in cancer cells as well as invasion/metastasis has been demonstrated in some cancers. Though, more studies at molecular level and probably with other HCC cell lines are required, this study gives a preliminary insight to further in vivo research on its potential effect on HCC. Research to develop and formulate silymarin or, probably, its natural and semisynthetic analogues as both preventive and therapeutic agents is ongoing. Furthermore, investigational clinical trials are needed to schedule its use against various cancers.
Footnotes
Funding
This study was approved by Baskent University Institutional Review Board (Project No.: DA 10/01) and supported by Baskent University Research Fund.