Abstract
Objective:
Diabetes mellitus (DM) and hypercholesterolemia (HC) when poorly controlled lead to debilitating central nervous system complications including cognitive deficits and memory impairment. In the present study, we investigated the mechanisms that may be responsible for such deficits.
Methods:
Both diabetes and HC were induced in two groups of rats independently using alloxan and high cholesterol diet, respectively.
Results:
Acetyl cholinesterase was significantly increased in brain of diabetic rats. Also, brain malondialdehyde level was extremely elevated in both diabetic and hypercholesterolemic groups. Meanwhile, brain albumin was markedly decreased in both of them. However, the brain iron level was significantly increased in DM with concomitant increase in total antioxidant capacity in the same group as compared to the normal control. The concentration of brain calcium was noticeably increased in HC group. Our results were confirmed by the increased activity of lactate dehydrogenase in both DM and HC groups, indicating major brain cytotoxicity.
Conclusions:
Overall, our results suggested that both DM and HC have deleterious effects on the brain which may be attributed to oxidative stress and dysregulation of both cholinergic function and calcium level. Administration of antioxidant is recommended in both cases.
Introduction
A wealth of epidemiological and experimental evidence now suggests diabetes mellitus (DM) and hypercholesterolemia (HC) as two major risk factors for the development of various central nervous system (CNS) diseases including Alzheimer’s disease (AD). 1,2
DM is a major global health problem currently affecting more than 180 million people worldwide. Diabetes is associated with long-term complications such as retinopathy, nephropathy, and cardiomyopathy. Besides, the complications of the peripheral nervous system in diabetic patients, recent evidences demonstrated that diabetes may also have negative impacts on the CNS. 3,4 The hippocampus, a key area for learning and memory is considered a special target for alterations in diabetes. 5 This area is most vulnerable to uncontrolled hyperglycemia. Neuropathological and neurobehavioral effects manifest as cognitive dysfunction, decline in memory, and hippocampal atrophy. 6 The cognitive decline in diabetes is strongly correlated with a number of functional and structural abnormalities in the nervous system. 7
In vitro and in vivo studies have shown that HC results in enhanced levels of β-amyloid. 8 In addition, tau pathology has recently been associated with membrane cholesterol and β-amyloid-induced neurotoxicity. 9 Furthermore, oxygenated cholesterol derivatives, oxysterols, upregulate the expression of various inflammatory cytokines and chemokines that trigger neurodegenerative disease. 10
In the brain, DM and HC-associated vascular inflammation may contribute to early breakdown of the functional integrity of the blood–brain barrier (BBB). 1 The BBB helps to maintain brain homeostasis by preventing the entry of blood components into the brain tissue that could disturb the normal functioning of neurons. 11 Moreover, both DM and HC are associated with increased oxidative stress. 12,13 Oxidative stress has been implicated in cognitive decline. The excess formation of reactive oxygen species (ROS) can damage cellular proteins, lipids, or DNA, then inhibit their normal function and disturb homeostasis within the neuron, and, ultimately, results in cell death. 14 However, the multifactorial pathogenesis of cognitive decline associated with DM and/or HC is not completely understood. Therefore, the aim of our present study was to explore whether HC or diabetes in rats can affect (1) the cholinergic system, via evaluation of brain acetylcholine esterase and (2) the oxidative status through the determination of brain oxidant/antioxidant biomarkers.
Materials and methods
Animals
Thirty-two Sprague Dawley male rats weighing 110 ± 10 g were included in this study. Animals were purchased from Animal House of the National Research Centre (NRC), Cairo, Egypt. They were kept individually in stainless steel wire-bottomed cages at room temperature (25 ± 2°C) under 12-h dark/12-h light cycle. Animals were fed standard pellet diets, had free access to tap water, and acclimatized for 1 week. Then, the rats were divided into two independent experimental groups with 16 rats in each group. Again, each group was subdivided into two subgroups (each including eight rats). Animal experiments were conducted strictly according to the approved guidelines of Animal Care and Ethics Committee of the NRC.
Diabetic experiment
Two groups of rats, namely, normal control (n = 8) and diabetic rats (n = 8) were fed balanced diet 15 for 6 weeks (Table 1). Diabetes was induced by 5% alloxan solution in normal saline intraperitoneally (125 mg/kg body weight) for fasted rats. 16,17 Diabetes was identified by polydypsia, polyuria, and by measuring blood glucose levels. After 2 weeks, rats with moderate hyperglycemia having blood glucose more than 116 mg/dl were included in the experiment.
Composition of experimental diets.

Hypercholesterolemic experiment
HC was induced in eight rats by feeding high cholesterol diet containing 1% cholesterol and 0.25% bile salts and saturated fat 18 for 4 weeks (Table 1). Then, the experiment begun by feeding the control rats with normal balanced diet and the experimental group with high cholesterol diet for 6 weeks. At the end of the experimental periods, rats in the two experiments were fasted overnight. Brains were dissected, washed with isotonic saline, weighed and relative brain weight was calculated (weight of brain/final body weight × 100). Then, they were immediately homogenized to give 10% (w/v) homogenate in ice-cold medium (pH 7.4) containing 50 mM tris(hydroxymethyl)aminomethane–hydrochloric acid and 300 mM sucrose. 19 The homogenate was centrifuged at 3000 r/min for 10 min in a cooling centrifuge. The supernatant was used for the determination of malondialdehyde (MDA) as a marker for brain lipid peroxidation using colorimetric method described by Sato et al. 20 Total antioxidant capacity (TAC) was estimated using commercial Biodiagnostic kit (Cairo, Egypt). 21 Brain calcium concentration was determined colorimetrically. 22 A cholinergic marker, acetylcholinesterase (AChE) activity was estimated using Centronic GmbH kit (Germany) according to the method described by Henry. 23 Brain iron was evaluated in the homogenate using the principle of Burits and Ashford. 24 Moreover, kinetic assay was applied to estimate lactate dehydrogenase (LDH) activity in brain homogenate according to Kachmar and Moss. 25 Finally, brain albumin was evaluated using kits provided from Biosystems (Barcelona, Spain). 26
Results
The effects of diabetes and HC on brain weights and relative brain weights are presented in Table 2. Diabetes significantly decreased rat brain weights (p < 0.05) as compared to their corresponding normal controls. Also, their relative weights showed slight decrease as compared to those of normal. On the other hand, rats fed with high fat diet showed slightly increased brain weights with negligible changes for relative brain weights as compared to their respective controls.
Brain weight and relative brain weight for various groups.a

aData are expressed as mean ± SE.
bp < 0.05.
Both diabetes and HC induced marked oxidative stress in the brain tissues. The level of MDA in diabetic rats was significantly increased (348 ± 34.4 nmol/g) versus (253 ± 22 nmol/g) for normal rats. Also, for hyperchrolesterolemic rats, it was significantly increased (225 ± 17.8 nmol/g) compared to their respective control (186 ± 12.9 nmol/g). The brain iron level was markedly increased (p < 0.0005; 7.05 ± 0.50 µg/g) in diabetic rats as compared to their normal (4.75 ± 0.17 µg/g). Meanwhile, iron level showed insignificant changes in hypercholesterolemic rats. TAC was significantly (p < 0.005) elevated (10.1 ± 0.22 µmol/g) in diabetic rats compared to their respective controls (8.50 ± 0.47 µmol/g), but it didn’t show any marked change in hypercholesterolemic rats. Brain albumin was significantly reduced in diabetic and hypercholesterolemic rats (63.1 ± 4.64 mg/g, p < 0.025 and 45.6 ± 2.32 mg/g, p < 0.0005, respectively) versus (82 ± 6.14 and 62.6 ± 1.63 mg/g, respectively) for their respective control (Table 3).
Changes of MDA, iron, TAC, and albumin parameters in diabetes and HC rat brain tissues.a

MDA: malondialdehyde; TAC: total antioxidant capacity; HC: hypercholesterolemia.
aData are expressed as mean ± SE.
bp < 0.005.
cp < 0.025.
dp < 0.05.
ep < 0.0005.
Factors that triggered and confirmed neuropathy such as brain AChE, calcium, and LDH were estimated and presented in Table 4. It shows that brain AChE activity was significantly elevated (35.5 ± 3.05 U/g, p < 0.025) in diabetes as compared to their respective control (26.2 ± 2.20 U/g), while this enzyme showed insignificant changes in hypercholesterolemic rats. High fat diet extremely increased brain calcium level (1095 ± 71.5 µg/g, p < 0.05) compared to corresponding control group (958 ± 9.51 µg/g). Diabetic rats exerted insignificant changes concerning brain calcium level. However, both diabetic and hyperlipidemic rats showed marked leakage of LDH from brain cells (31.2 ± 1.39 U/g, p < 0.0005 and 26.6 ± 2.39 U/g, p < 0.01, respectively) compared to their coordinate controls (15.9 ± 0.98 and ± 18.6 ± 1.27 U/g, respectively). LDH activity was higher in diabetic rats than hypercholesterolemic rats.
Effect of diabetes or HC on brain tissue biomarkers that triggered neuropathy.a
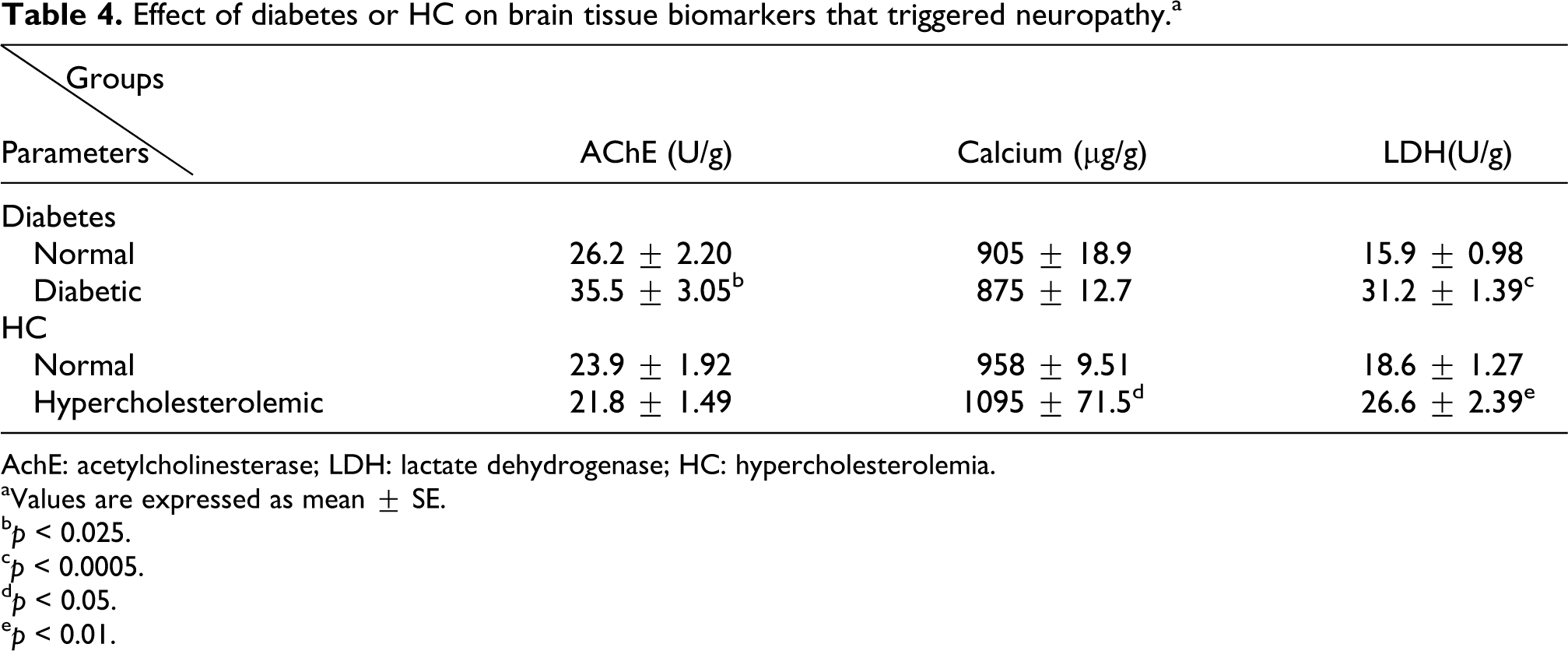
AchE: acetylcholinesterase; LDH: lactate dehydrogenase; HC: hypercholesterolemia.
aValues are expressed as mean ± SE.
bp < 0.025.
cp < 0.0005.
dp < 0.05.
ep < 0.01.
Discussion
DM and HC are the most common disorders that are associated with long-term complications. Both affect eyes, kidneys, blood vessels, heart, and CNS. 27 The mechanism causing brain dysfunction in DM and HC appears to be a multifactorial process. A growing body of evidences suggests that DM- and HC-related cerebral dysfunction is largely a consequence of changes within CNS that are secondary to chronic hyperglycemia, 28 hyperlipidemia, 29 cholinergic function, 30 inflammation, 31 BBB permeability, 27 and oxidative stress. 32,33 The brain is especially sensitive to oxidative stress due to the high lipid composition of cell membranes and low levels of antioxidant enzymes. 34 Therefore, our study was conducted to evaluate oxidant/antioxidant status and cholinergic change and how far the BBB permeability affected in diabetic or hypercholesterolemic rat model.
Our results demonstrated that the weight of the brain of diabetic rats was significantly lower than that of control rats. This finding was in agreement with that of Zare et al. 35 The decrease in brain weight may be resulted from the reduction of body weight induced by diabetes. 36 On the contrary, HC increased the weight of rat brain which coincides with the finding of Charradi et al. 37 High cholesterol diet increased body weight gain 38 with concomitant increase in brain weight. However, both of the decrease (DM) and the increase (HC) in brain weight are indicative, to some extent, of brain pathological condition which was confirmed by the analysis of pivotal markers in our study.
Cholinergic neurotransmission plays a crucial process underlying memory and cognitive function. Acetylcholine (ACh) is an important neurotransmitter in the central cholinergic system through ACh receptor. It is synthesized by choline acetyltransferase (ChAT) and hydrolyzed by AChE and plays an important role in learning and memory capabilities. The dynamic balance of ACh is kept by both ChAT and AChE. The imbalance of ChAT and AChE is one of the important factors for cognitive dysfunction that causes ACh metabolic disorder. 7 Our emerged data showed that AChE activity was significantly increased in diabetic rats.
An increase in AChE activity has been found to inhibit cell proliferation and promote apoptosis. 39 Moreover, increased AChE activity led to fast ACh degradation and subsequent down stimulation of ACh receptors. This causes a reduction of both cholinergic (learning and memory) and non-cholinergic (cell proliferation and neurite outgrowth) function. A decrease in ACh levels in the synaptic cleft contributes to a progressive cognitive impairment and possibly other neurological dysfunction. 40 –42 Multiple studies showed that cholinergic dysfunction (AChE) contributes to cognitive deficits in diabetes. 7,43,44
However, AChE activity exhibited nonsignificant changes in our hypercholesterolemic rats. It is an unexpected finding contradicting with that of Charradi et al.’s finding. 37 This discrepancy may be attributed to the older age of rats and longer duration of the experiment in Charradi’s work. However, Li et al. 45 proved that feeding high cholesterol diet may or may not affect cognition.
Oxidative deterioration of cellular membranes consequently affects ion channels, receptors of various ligands, and also neurotransmission. Thus, oxidative stress may affect AChE. 37,46
Oxidative stress refers to the cytopathologic consequences of a mismatch between the production of free radicals and the ability of the cell to detoxify them. Growing data from experimental models and human brain studies suggest that oxidative stress may play an important role in neuronal degeneration in diseases such as AD. An increase in ROS can produce damage to vital molecules and induce necrosis or apoptosis for neurons, astrocytes, and microglia. 46,47 Our results demonstrated that both DM or HC extremely increased thiobarbituric acid reactive substances (TBARS) level (considered as end product of lipid peroxidation (MDA)), which coincides with Bhutada et al. 48 and Charradi et al., 37 respectively. A correlation occurs between HC and brain oxidative stress. Also, plasma lipid peroxidation levels correlated with brain tissue lipid peroxide. 29 Increased oxidative stress in the brain tissue induced the production of ROS that oxidized membrane unsaturated fatty acids with concomitant increase in TBARS as an end product of lipid peroxidation. Meanwhile, in diabetes, persistent hyperglycemia induces glycated hemoglobin (HbA1C) which generates free radicals and other advanced glycation end products (AGEs). AGEs via binding to its membrane receptors generate pro-oxidants and evoked both ROS and nitrogen species formation, 49 consequently, increasing lipid peroxidation.
Experimental evidence suggest that the initiation and promotion of neurodegenerative diseases are associated with accumulation of high concentrations of metals like copper, aluminum, and zinc. Moreover, iron in brain regions was associated with function loss and cell damage. 50 Metal homeostasis imbalance and neuronal loss are both present in neurodegenerative diseases. However, iron is necessary in many cellular functions, especially in the brain, where it participates in many neuronal processes. The current results demonstrated that diabetes increased brain iron level dramatically. Excess glucose induces HbA1C in majority that releases iron. 51 Iron overload and ROS increase BBB permeability. 52 On the other hand, HC didn’t change brain iron in our rats that was in accordance with Charradi et al. 37 . Albumin, the most abundant circulating protein in plasma not only has numerous physiological and pharmacological functions but also has a powerful antioxidant property. 53 Our results revealed that brain albumin level was extremely reduced in both DM and HC rats. This could be explained by the increased brain oxidative stress in both cases that decreased the level of brain albumin. 54 Besides, hypoalbuminemia induced by DM was confirmed by Park et al., 55 who found that albumin level was decreased in periferal blood cells of diabetic rats.
DM and HC increased cytotoxicity and decreased cell viability in the brain as revealed from the elevation of brain LDH activity. The deleterious effects of DM are more pronounced than that of HC. LDH is the enzyme catalyzing the interconversion of pyruvate and lactate and is essential for both production and utilization of lactate. Astrocytes are the predominant lactate-producing cells, while the neurons are predominantly those using lactate as an energy source. 56 It is formed through three distinct genes: LDH-A, LDH-B, and LDH-C. The LDH-A and LDH-B gene products are found in the CNS. 57 ROS resulted from the aberrant metabolism such as that in DM and HC has been shown to elicit cellular injury with concomitant release of LDH have particularly deleterious effects on the nervous system. 58 –60
Another deleterious effect of oxidative stress appeared on brain calcium level. As induction of oxidative stress is known to activate transient receptor potential (TRP) protein superfamily, a diverse group of voltage-independent calcium-permeable cation channel with concomitant calcium influx via these channels trigger apoptosis and cell death in brain regions. 61 Our emerged data demonstrated the finding that HC extremely elevated brain calcium level that confirms the above hypothesis and is also in accordance with those of Kamat et al. 62 and Prasad and Muralidhara. 63 Meanwhile, DM showed no significant changes in brain calcium concentration, which may be attributed to the marked elevation of brain TAC to the degree that opposes activation of TRP.
Conclusion
Our findings clearly demonstrated the adverse neurological effects of DM and HC on rats. Various mechanisms may be present, including increase of oxidative stress manifested by an increase of TBARS level and a decrease of albumin level in both DM and HC, and an increase of iron concentration in DM only. Moreover, an elevation of AChE activity and calcium level may also be causes for such neuropathy. Our results were confirmed by elevation of LDH activity that indicated cytotoxicity and major brain injury. It was seen that both DM and HC cause significant neuropathy. Administration of antioxidant is recommended.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
