Abstract
Researchers have been evaluating several biodosimetric/screening approaches to assess acute radiation injury, related to mass causality. Keeping in mind this background, we hypothesized that effect of whole-body irradiation in single fraction in graded doses can affect the secretion of various salivary components that could be used as acute radiation injury/toxicity marker, which can be used in screening of large population at the time of nuclear accidents/disaster. Thirty Sprague Dawley rats treated with whole-body cobalt-60 gamma irradiation of dose 1–5 Gy (dose rate: 0.95 Gy/min) were included in this study. Whole mixed saliva was collected from all animals before and after radiation up to 72 h postradiation. Saliva was analyzed for electrolytes, total protein, urea, and amylase. Intragroup comparison of salivary parameters at different radiation doses showed significant differences. Potassium was significantly increased as the dose increased from 1 Gy to 5 Gy (p < 0.01) with effect size of difference (r > 0.5). Sodium was significantly altered after 3–5 Gy (p < 0.01, r > 0.5), except 1 and 2 Gy, whereas changes in sodium level were nonsignificant (p > 0.5). Urea, total protein, and amylase levels were also significantly increased as the radiation dose increased (p < 0.01) with large effect size of difference (r > 0.5). This study suggests that salivary parameters were sensitive toward radiation even at low radiation dose which can be used as a predictor of radiation injury.
Introduction
Although radiological events occur infrequently, they have the high potential of causing a mass casualty, such as nuclear accidents of Chernobyl and Fukushima. Dose estimation and screening is an early assessment tool to classify the severity of acute radiation injury, which is imperative to minimize morbidity and mortality through rationally directed medical intervention after a radiological or nuclear disaster. 1
For the mass screening of radiation-exposed victims, chromosomal aberration-based assays are impractical due to time-consuming process (24–72 h), despite their high specificity and sensitivity. 2 At the time of radiological disaster, time is the most precious determinant of morbidity and mortality. Incapacitating psychosocial manifestations with symptoms like nausea, vomiting, headache, delirium, and unconsciousness are very frequent immediately after such incidence necessitating immediate recognition of truly exposed victims. 3 That is why rapid response from laboratories is required for medical team to manage triage and treatment in case of a mass casualty scenario. 4,5 The treatment should be initiated within 24 h after exposure and geared toward those individuals in the moderate and severe exposure categories 6 (with total or near-total body exposure greater than 2 Gy), which is not possible with conventional biodosimetry approaches mentioned above.
In the present study, saliva was taken as a noninvasive test sample due to its advantages over blood collection. Drawing blood is impractical in emergency scenario, making saliva a better diagnostic medium, where repeated saliva samples can be collected easily. 7 Although many studies have reported the effect of gamma radiation on salivary gland output and its biochemical parameters, there is no wide literature available which shows the periodic study of the salivary constituents as toxicity biomarkers of acute radiation injury. The aim of the present study is to observe the effect of whole-body radiation on salivary parameters that can be used during the screening of mass casualty caused by nuclear accidents or occupational exposure.
Materials and methods
Animals selected
Thirty male Sprague Dawley rats (8–9 weeks old) with an initial body weight of 200–250 g were obtained from animal house. Rats were given free access to standard laboratory animal feed (Hindustan Lever Ltd, Mumbai, Maharashtra, India) and water ad libitum. The temperature of the animal room was maintained at 22 ± 3°C and relative humidity at 30–70% throughout the experiment. The rats were housed in polypropylene cages on paper cutting bedding. Animals were maintained on a 12-h light/12-h dark cycle in accordance with the protocols approved by the Institutional Animal Ethics Committee (IAEC). Rats were divided randomly into the following five groups:
Group I: irradiated with a single dose of 1 Gy (n = 6); Group II: irradiated with a single dose of 2 Gy (n = 6); Group III: irradiated with a single dose of 3 Gy (n = 6); Group IV: irradiated with a single dose of 4 Gy (n = 6); and Group V: irradiated with a single dose of 5 Gy (n = 6).
Preradiation exposure saliva sample of all group animals were considered as control sample for the study.
Irradiation of animals
The animals were anesthetized with intravenous injection of 0.1 ml diazepam (10 mg/ml) prior to radiation treatment. Rats were then irradiated with a single dose of 1–5 Gy whole-body radiation by cobalt-60 teletherapy unit (model Bhabhatron II, Panacea Medical Technologies Pvt. Ltd, Bangalore, Karnataka, India), at dose rate of 0.95 Gy/min with a focus to skin distance of 120 cm. The total radiation field, in which rats were irradiated, was 20 × 20 cm2. The anesthetized rats were placed in plastic cages during exposure. The animals were maintained and treated in accordance with the protocols approved by the IAEC.
Saliva collection
Subcutaneous (S.C.) injection of 1 ml Ringer solution with 2.5% glucose was given to each animal to overcome the effect of dehydration, starting at day 1 of the irradiation period and lasting for 3 days. The rats were anesthetized with intramuscular injection of fentanyl (0.2 mg/ml) and diazepam (5 mg/ml) separately in thigh. Saliva secretion was stimulated by S.C. injection of pilocarpine (2.5 mg/kg body weight). Saliva secretion of pre- (T0) and postirradiation, stimulated whole mixed saliva sample was collected at different time points (T1 = 2 h, T2 = 24 h, T3 = 48 h, and T4 = 72 h) were collected. During saliva collection, the rats were positioned on a table inclined 10°, with the mouth placed over a polypropylene cup and collected for 20 min. Tubes-containing saliva samples were kept on chilled ice during the experiment. The saliva samples were stored at −80° until analysis. After saliva collection, 0.05 ml of atropine and 0.5 ml of antidote naloxone hydrochloride were given to all rats to stop the saliva secretion and for the recovery from anesthesia.
Sialochemical analysis
Stored saliva was thawed and centrifuged at 800 g for 10 min at 4°C to remove debris and food particles. In whole mixed saliva, salivary sodium and potassium concentrations were measured by the method described by Gowenlock 8 and Voda et al. 9 , respectively.
Total protein
Total protein in saliva was determined by the bicinchoninic acid method using an assay kit (Pierce Biotechnology, Rockford, Illinois, USA) with bovine serum albumin as a standard.
Urea estimation
The level of urea was quantified in saliva samples by diacetylmonoxime method described by Wootton. 10 Briefly, 1 ml diluted saliva was added in mixed color reagent (670 µg thiosemicarbazide and 2.68 mg diacetyl monoxime) and mixed acid reagent (sulfuric acid and 10 mg ferric chloride). Working urea solution (2 mg/ml) was used as urea standard. Then, the reaction mixture was kept in boiling water bath for 20 min and cooled immediately. Absorbance was measured at 520 nm against a blank using ultraviolet–visible spectrophotometer (T-20+, Labindia, Thane, Maharashtra, India). The concentration of urea was calculated and expressed in terms of milligram per deciliter of saliva sample.
Measurement of α-amylase
The amylase activity was determined by a colorimetric enzymatic method with commercially available kit (Salimetrics, State College, Pennsylvania, USA). The method used a chromagenic substrate, 2-chloro-p-nitrophenol linked with maltotriose. The enzymatic action of α-amylase on this substrate yielded 2-chloro-p-nitrophenol, which was spectrophotometrically measured at 405 nm. The amount of α-amylase activity present in the sample was directly proportional to the increase in absorbance at 405 nm.
Data and statistical analysis
Intergroup comparison of the levels of biochemical parameters was done using Kruskal–Wallis test, followed by Mann–Whitney test for post hoc comparisons. Intragroup comparison of the parameters was done using nonparametric Friedmann test, followed by Wilcoxon signed-rank test for post hoc comparisons. Median scores of all the groups were recorded. The values of p < 0.05 were taken as significant. Effect size (r) is a measure for quantifying the difference between two groups or we can say it measures the strength between two groups. The values of r < 0.3 were taken as small effect size, r value between 0.3 and 0.5 as medium effect, and r > 0.5 as large effect size. All analyses were performed by SPSS for Windows (version 20.0; SPSS Inc., Chicago, Illinois, USA).
Results
All rats remained alive during our study period of 72 h follow up. The amount of saliva collected was 0.5–1.0 ml for each time point. There were no significant changes in the amount of saliva collected upto 72 h. The results of various salivary parameters in all groups across the 72 h postradiation follow up are summarized for intragroup comparison (Table 1) and intergroup comparison (Table 2).
Intragroup comparison of salivary parameters over 72 h.a

T0: preradiation saliva sample, control; T1: postradiation 2 h sample; T2: postradiation 24 h sample; T3: postradiation 48 h sample; T4: postradiation 72 h sample.
aMedian value of potassium and sodium is expressed as millimoles per liter, total protein as milligrams per milliliter, urea as milligrams per deciliter, and amylase as units per milliliter.
Intergroup comparison of salivary parameters.
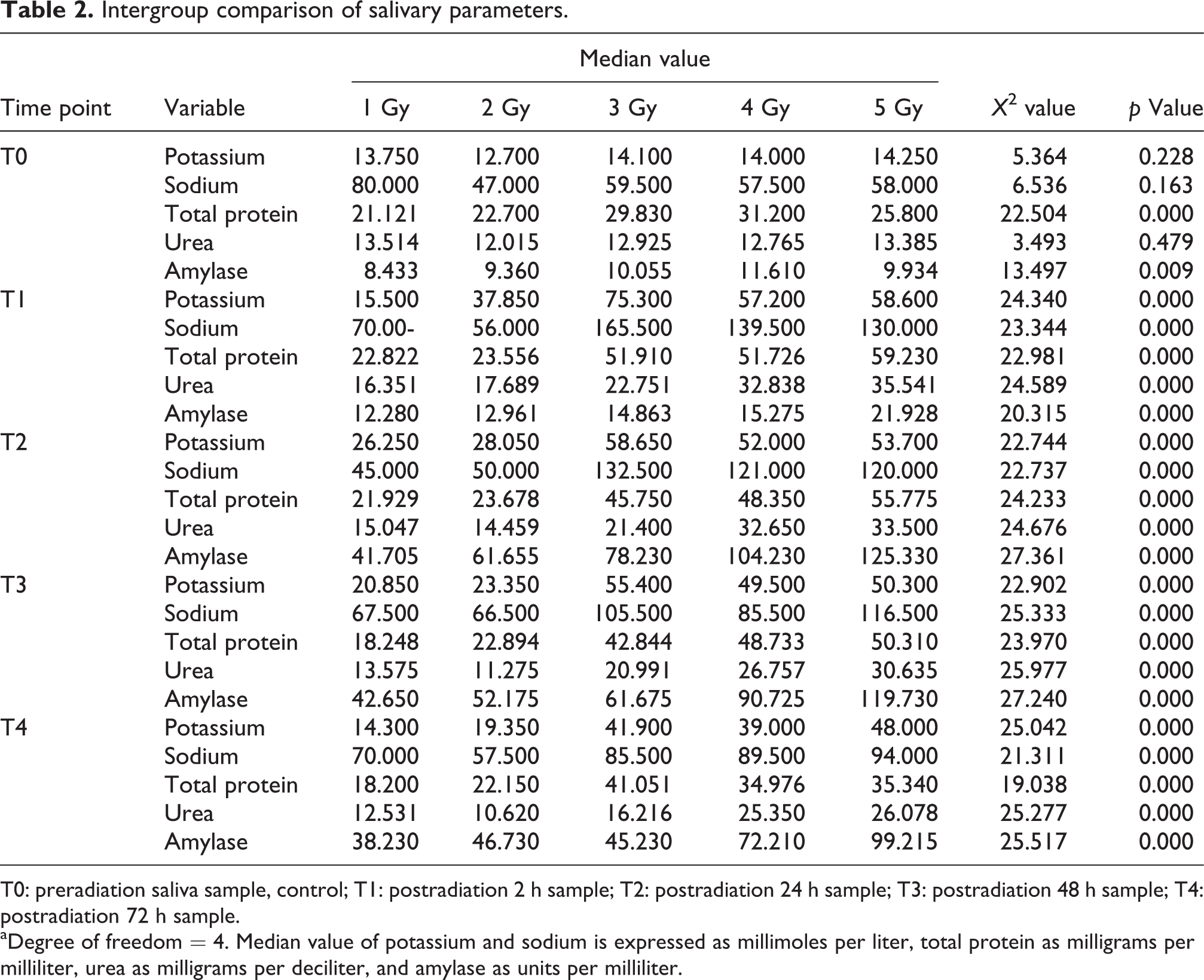
T0: preradiation saliva sample, control; T1: postradiation 2 h sample; T2: postradiation 24 h sample; T3: postradiation 48 h sample; T4: postradiation 72 h sample.
aDegree of freedom = 4. Median value of potassium and sodium is expressed as millimoles per liter, total protein as milligrams per milliliter, urea as milligrams per deciliter, and amylase as units per milliliter.
Salivary potassium
Dose-dependent changes were observed in potassium concentration in all irradiated groups. After acute exposure of radiation, a significant increase in potassium concentration was recorded 2 h postradiation. Inspection of p values of post hoc comparison using Wilcoxon signed-rank test between control values and at each time point after irradiation revealed that there was a significant increase of potassium levels in all groups. Medium effect size was observed in group 1 (r = 0.2–0.6, p < 0.05), while large effect size was observed in groups 2 to 5 at each time point in comparison with the control group. Effect size of the differences was found to be in medium range in group 1 (r = 0.2–0.6, p < 0.05), while it was high (r > 0.5) at 2, 24, and 48 h postradiation in groups 2 to 5. This indicates the positive relation between radiation dose and time. It is observed from Table 2 that there was a significant change in potassium concentration in saliva at different radiation doses. It was also significant (p < 0.001) at each time period in all groups. Post hoc comparison using Mann–Whitney test between all radiation doses in control values and at each time point after irradiation revealed the increased level of potassium.
Salivary sodium
The change in salivary sodium in groups 1 and 2 was not significant as mentioned in Table 1, while significant (p < 0.001 in all groups) in groups 3 to 5. Post hoc comparison showed significant difference at all time points with p < 0.001 in groups 3 to 5. Effect size of the difference at each time period after 3, 4, and 5 Gy was more than 0.6 as compared to controls, indicating a large difference. It is clear from Table 2 that the difference of median values was significant in all radiation groups (p < 0.05). It is evident from post hoc comparison that effect size of the difference is increased (r = 0.5–0.8) as radiation dose increased (groups 1 to 5), except in group 1 where it was found <0.5 at 24 and 48 h.
Salivary urea
It is evident from Table 1 that difference of median was highly significant in all radiation groups (p < 0.01). Post hoc comparison test revealed that effect size was high at 2 h in group 1 (r = 0.6), while 24, 48, and 72 h showed small effect size (r < 0.5). Groups 2 to 5 showed large effect size (r > 0.5) after different time points, while 48 h in group 2 and 72 h in group 3 showed small effect size. It is clear from Table 2 that median value differences of control and postradiation sample showed high significance (p < 0.0001). All groups showed this trend. Post hoc comparison revealed that effect size of difference increased with r value more than 0.5 as radiation dose increased. Effect size was small at 2 and 24 h in group 1.
Salivary total protein
Table 1 shows that the difference of median values observed was significant (p < 0.01) in all groups, except for group 2 (p = 0.281). Post hoc tests revealed significant difference from groups 1 to 5 at each points with medium to high effect size (r = 0.2–0.63). The effect was more at higher radiation doses. It is observed from Table 2 that the difference of median values was found highly significant (p < 0.001). Post hoc tests revealed significant difference from groups 2 to 5 at each point with high effect size (r > 0.5). Effect size was observed <0.5 in group 1 at 2 and 24 h.
Salivary amylase
Table 1 shows that the change in salivary amylase was significant in all groups at each time point (p < 0.001). Post hoc comparison revealed that effect size of difference at each time point in all groups was more than 0.6 as compared to control, indicating a large difference. Table 2 showed that the differences of median value of salivary amylase were highly significant (p < 0.001) at each time point in groups 1 to 5. Post hoc comparison revealed that effect size of difference at each time point in all groups was more than 0.5, except at 2 h in group 1 (r < 0.5).
Discussion
Ionizing radiation affects body physiology in different ways. To study and monitor the effect of radiation, one needs a series of analysis to explore the different damaging events that may occur. Early screening biomarkers may be useful in measurement of acute or chronic effect caused by total body irradiation or partial body irradiation. Numerous investigators have described the nature of salivary gland damage after irradiation in different animals such as rats, rabbits, 11,12 and monkeys. 13 The identification of radiation injury biomarkers is a necessary tool in mass screening at the time of nuclear accident or occupational exposure. 14,15 Most of the radiation biomarkers have been identified in blood cells and serum, including those of DNA damage, repair, 16,17 and chromosomal aberrations. 18 Although at present, cytogenetic techniques are widely used as biological dosimetry method in situation ranging from nuclear accidents 19 to space travel, 20 the accuracy is limited by many factors, particularly variation in background frequency of stable translocations apart from a processing time of 48–72 h. These techniques require high inputs in terms of man power, money, and infrastructure, which make them less practical mostly in developing countries.
Ionizing radiation has severe effect on salivary gland, including malfunctioning of gland, progressive cell death, and stable phase, followed by recovering phase. 21 The present study has demonstrated that salivary parameters, including sodium, potassium, urea, total protein, and amylase, can play a vital role in dose estimation and screening for acute radiation exposure. Ionizing radiation induces apoptosis in serous cells of salivary gland, which is facilitated by efflux of potassium and sodium ions. 22,23 The increased content of electrolytes in saliva is due to apoptosis and various other physiological changes occurring in salivary gland. There are various studies which reported that ionic strength of apoptotic cell is less than their nonshrunken counterparts. Similarly, we have investigated that salivary potassium was found sensitive toward low-dose radiation exposure. Partial recovery from radiation-induced damage was observed after 24–48 h postirradiation, suggesting onset of regeneration of salivary gland tissue (Figure 1(a)). Urea levels were also significantly increased at 2 h postradiation, but decreased at 24–72 h follow up. By passive diffusion, serum urea reaches to the salivary gland and increases the level of salivary urea content. Levels of urea were high at higher doses in comparison with low dose, but interesting trend was observed in this parameter. A plateau was observed after 2 h, and it did not show much deviation till 72 h, indicating the slower rate of recovery from radiation injury.
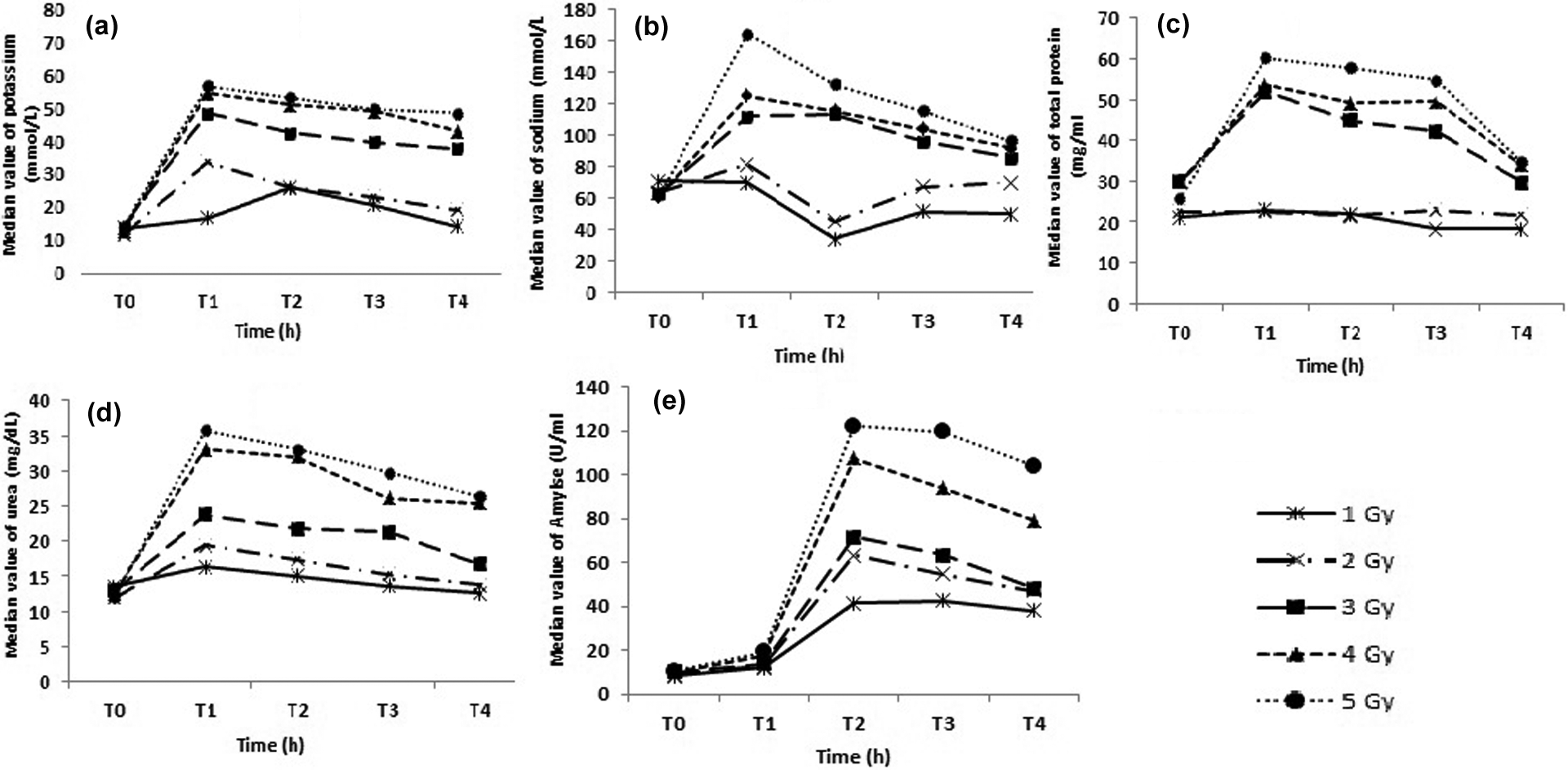
Effect of 1–5 Gy radiation dose on salivary parameter at different time intervals: (a) salivary potassium, (b) salivary sodium, (c) salivary total protein, (d) salivary urea, and (e) salivary amylase. T0: control; T1: postradiation 2 h sample; T2: postradiation 24 h sample; T3: postradiation 48 h sample, and T4: postradiation 72 h sample.
The assessment of total protein enables the examination of number of healthy functional cells along with secretary capacity of salivary gland. Quantitative change was observed in total protein level, which accord well by reduction in healthy acinar cells. At lower dose of 1 and 2 Gy, there was no significant change in total protein level when compared with the control, which makes it less impeccable at lower doses than higher doses. Radiation-induced damage to salivary gland involves interphase cell death of serous acinar cell component, which is sensitive for the low-dose radiation of 100 rad (1 Gy) inducing leakage of salivary amylase into circulation causing hyperamylasemia. 24 Concentration of salivary amylase showed (Figure 1(e)) an interesting response to irradiation, which was observed at all doses studied. In terms of sensitivity, salivary amylase did not show significant detectable changes as early as 2 h postirradiation. After 2 h postirradiation, amylase level did not change significantly in group 1 to 5, while showed maximum change at 24 h and subsequently remained at this level or decreased. At the end of the experimental period, the concentration of amylase was two to three times more than the control values, which suggest that salivary amylase activity must be measured for screening purposes after suspected radiation exposure to serve as biological indicator triage tool for identifying individuals with acute radiation injury.
The authors conclude with a preamble that further studies at larger scale are needed and current approach may help in accelerating such efforts, leading to a new focus in radiobiology and cancer research by opening avenues for identification of salivary biomarkers. Noninvasive nature of the study makes it more impeccable. The animal data collected in this study could be further studied to develop new methods of radiation dose assessment, allowing for identification of nonirradiated individuals. It could also be helpful in triage of exposed individuals requiring immediate medical intervention for survival versus those exposed to lethal doses of radiation who require palliative care only. The study has the potential for the screening of the large population exposed to ionizing radiation because of its cost-effectiveness, which is a crucial parameter for the developing countries where implementation and standardization of cytogenetic techniques are difficult.
Footnotes
Acknowledgment
The author SKS is thankful to Indian Council of Medical Research (ICMR), New Delhi, India, for providing senior research fellowship (3/2/2/163/2012/NCD-III).
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
