Abstract
Here we evaluated the ability of
Introduction
Cirrhosis is one of the most frequent and serious liver diseases and is the end stage of progressive fibrosis, which causes major disruptions to metabolic function and blood circulation. 1 Cirrhosis is characterized by the accumulation of extracellular matrix (ECM) proteins, and it causes the perturbation of liver homeostasis, leading to the intracellular release of cytokines. In particular, the fibrogenic cytokine, transforming growth factor β (TGF-β), is a key factor in cirrhosis development as it stimulates the generation of ECM and inhibits matrix protein removal.
Excessive activation of cellular signalling pathways by nuclear factor κB (NF-κB) has important roles in innate immunity, liver inflammation, fibrosis and the prevention of apoptosis. NF-κB constitutes a critical target for manipulating pathophysiological processes in different hepatic diseases. The activation of NF-κB is related to oxidative stress-induced cell death.

Chemical structure of
These observations prompted us to investigate whether
Materials and methods
Chemicals
Treatment of animals
In this work, male Wistar rats (3 weeks old at the beginning of treatments) were used and were maintained on a standard rat chow diet with free access to drinking water. Eight animals were housed per polycarbonate cage and kept under controlled conditions at 22 ± 2°C with 50–60% relative humidity and 12-h light/12-h dark cycles. All animals received humane care, and the study complied with the institution’s guidelines, official Mexican regulations (NOM-062-ZOO-1999) and the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Sciences and published by the National Institutes of Health (NIH publication 86-23, revised 1985).
Cirrhosis induction
Rats initially weighing 100–110 g were divided into four groups of eight rats each. Animals in the control group were given mineral oil (0.25 mL, intraperitoneally (i.p.)) three times per week for 8 weeks. Cirrhosis was induced in the CCl4 group by the administration of CCl4 (0.4 g/kg, i.p.) three times per week for 8 weeks.
8,9
The CCl4-
Biochemical assays
Blood sample was collected by cardiac puncture, and the liver was rapidly removed. Serum was obtained to assess liver damage by measuring the enzymatic activities of alanine aminotransferase (ALT) 10 and γ-glutamyl transpeptidase (γ-GTP). 11
Assessment of lipid peroxidation
The extent of lipid peroxidation in liver homogenates was determined by quantification of malonyldialdehyde (MDA) 12 formation by the thiobarbituric acid method. Total protein contents were determined by the Bradford method, using bovine serum albumin as standard. 13
Determinations of liver GSH and GSSG
Glutathione (GSH) and glutathione disulfide (GSSG) levels were quantified as described by Hissin and Hilf. 14 GSH and GSSG assays were performed, on the same day when the animals were killed, in 0.1 M sodium phosphate, 0.005 M ethylenediaminetetraacetic acid buffer (pH 8.0) and kept on ice until used.
Collagen quantification
Collagen concentrations were determined by measuring the hydroxyproline contents of fresh liver samples digested with hydrochloric acid as previously described. 15,16
Histology
Liver samples were taken from all animals and fixed with 10% formaldehyde in phosphate-buffered saline for 24 h. Tissue pieces were then washed with tap water, dehydrated in alcohol and embedded in paraffin. Five micrometre-thick sections were mounted on glass slides and covered with silane. Staining was performed using haematoxylin and eosin and Masson’s trichromic stain.
Molecular biology assays
Total protein isolation
Trizol® reagent (Invitrogen™, Carlsbad, California, USA) was used to isolate the total protein content of liver tissue, and the total protein levels were determined by the bicinchoninic acid method.
Western blot assays
Samples containing 50 μg total protein were separated on 10% polyacrylamide gels, and the proteins were then transferred onto Immuno-Blot™ polyvinylidene difluoride membranes (Bio-Rad, Hercules, California, USA). 17 The blots were subsequently blocked with 5% skim milk and 0.05% Tween-20 for 1 h at room temperature and independently incubated at 4°C overnight with antibodies (Abs) selective against each protein: NF-κB (p65), TGF-β, Metalloproteinase 13 (MMP-13) and interleukin (IL)-1β Abs (MAB3026, MAB1032, MAB13426 and AB1832, respectively) were from Millipore Corp. (Billerica, Massachusetts, USA). IL-6, IL-10 and connective tissue growth factor (CTGF) Abs (SC-57315, SC-57245 and SC14939, respectively) were from Santa Cruz Biotechnology Inc. (Santa Cruz, California, USA). Membranes were then washed and exposed to a secondary peroxidase-labelled Ab (Zymed, San Francisco, California, USA) diluted to 1:1500 in blocking solution for 1 h at room temperature.
Blots were then washed and developed using the Western Lightning™ Plus-ECL enhanced chemiluminescence detection system (NEN Life Sciences Products, Elmer LAS Inc., Boston, Massachusetts, USA). After stripping, the blots were incubated with a monoclonal Ab directed against β-actin, 18 as a control for protein loading, and developed using the Western Lightning Plus-ECL kit. Images were digitally captured using a BioDoc-It imaging system (UVP, Upland, California, USA). 19
RT-PCR analysis
The messenger RNA (mRNA) levels of the cytokine TGF-β in liver samples were measured using reverse transcriptase polymerase chain reaction (RT-PCR). Total RNA was isolated from rat liver tissue using the Trizol reagent (Invitrogen), and the concentration and purity of RNA samples were determined by measuring the A 260–A 280 nm absorbance ratios. Complementary DNA (cDNA) was obtained using poly dT primed RT (Invitrogen), and Taq polymerase (Invitrogen) was used for the PCR reactions. 20
The primer sequences and PCR conditions used to amplify TGF-β and CTGF are described in Table 1. As a positive control, β-actin cDNA was amplified as previously described. Images were digitally acquired using a BioDoc-It Imaging System (UVP).
Primers used for RT-PCR.a

TGF-β: transforming growth factor β; CTGF: connective tissue growth factor; RT-PCR: reverse transcriptase polymerase chain reaction.
aPrimer sequences specific for the indicated rat genes and the sizes of the expected PCR products are shown.
Statistical analysis
Data are expressed as the mean values ± SE. Comparisons were performed using one-way analysis of variance (ANOVA), followed by Tukey’s test, using the GraphPad Prism 5.00 software. Differences were considered to be statistically significant when p < 0.05.
Results
The enzymatic activities of ALT (a marker of necrosis) and γ-GTP were increased approximately 1.5-fold in the group treated with CCl4 for 8 weeks compared with the control group, and administration of
Enzyme activities determined in serum in groups of six rats each.a

CCl4: carbon tetrachloride; γ-GTP: γ-glutamyl transpeptidase; ALT: alanine aminotransferase.
aEffect of the chronic administration of CCl4 on the activities of ALT and γ-GTP in control group, CCl4-treated rats (CCl4), CCl4-treated rats plus
bSignificantly different from control at p < 0.05.
cSignificantly different from CCl4 group at p < 0.05.
Chronic exposure to CCl4 increased fibrosis nearly sixfold when compared with the control. This effect was partially, but significantly, inhibited by

Liver collagen as measured by hepatic hydroxyproline content in livers from rats following chronic administration of CCl4 for 8 weeks. Control (CONTROL), CCl4-treated (CCl4), CCl4 plus
Sections of rat livers from the different treatment groups were stained with haematoxylin and eosin and are presented in Figure 3. The control group showed a normal appearance (Figure 3(a)), whilst samples from CCl4-treated group showed pronounced nodular fibrosis of the parenchyma with hypertrophic hepatocytes, steatosis and neoformed acidophilic cells (Figure 3(b)). Liver sections from the CCl4 +

Haematoxylin and eosin staining of rat liver samples. (a) Untreated control, (b) CCl4 treated, (c) CCL4 +
Moreover, Masson’s trichromic staining (Figure 4) revealed that small collagen fibres appeared only in portal areas within the livers of the control group (Figure 4(a)). Liver cirrhotic sections as well as nodular fibrosis surrounding hypertrophic hepatocytes were found in tissue samples from the CCl4-treated group (Figure 4(b)). The CCl4 +

Masson’s trichromic stain of rat liver samples. (a) Untreated control, (b) CCl4 treated, (c) CCL4 +
Membrane oxidative stress was evaluated by determining liver MDA levels. Lipid peroxidation was significantly increased by CCl4 treatment, and this increase was partially prevented by
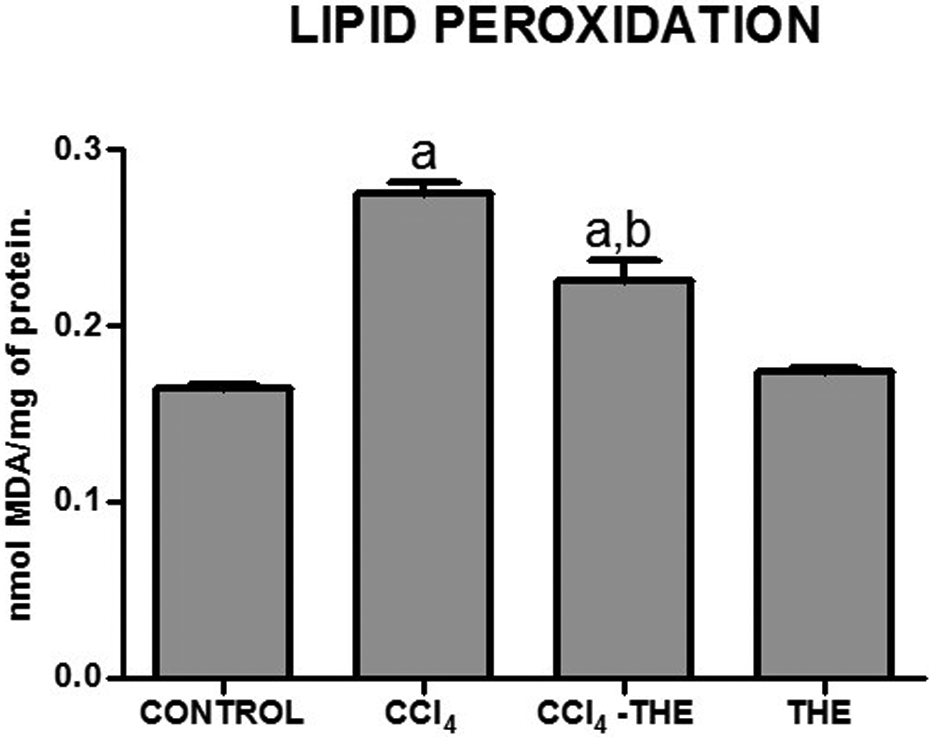
Liver lipid peroxidation as determined by MDA contents of samples from control (CONTROL), CCl4-treated (CCl4), CCl4 plus
Cytosolic oxidative stress was evaluated by the quantification of liver GSH levels (Figure 6). Decreases in the GSH levels of the CCl4-treated group were observed, and GSSG levels tended to increase in the CCl4-treated group compared with the control group.

Reduced GSH and oxidized GSSG, GSH/GSSG ratio and total glutathione (GSH + GSSG) determined in livers from control (CONTROL), CCl4-treated (CCl4), CCl4 plus
The administration of CCl4 increased p65 levels compared with the control group and

Expression of NF-κB-p65 as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus
Concomitant increases in IL-1β and IL-6 protein levels in liver samples obtained from CCl4-treated rats compared with the control group are shown in Figures 8 and 9, respectively; furthermore, the levels of these cytokines were reduced by co-administration of

Expression of IL-1β as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus

Expression of IL-6 as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus

Expression of IL-10 as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus
Following the administration of CCl4 over 8 weeks, significant increases in the expression of TGF-β (Figure 11) and CTGF (Figure 12) were detected when compared with the control group, and

Expression of TGF-β as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus

Expression of CTGF as determined by Western blot analysis of hepatic extracts. (a) A representative Western blot is shown. (b) Control (CONTROL), CCl4-treated (CCl4), CCl4 plus
The effects of

Expression of the latent (a) and active (b) forms of MMP-13 as determined by Western blot analysis of hepatic extracts. A representative Western blot is shown (c). Control (CONTROL), CCl4-treated (CCl4), CCl4 plus
Chronic CCl4 intoxication increased both TGF-β and CTGF mRNA levels. Interestingly, co-treatment with

mRNA levels of TGF-β and CTGF. RT-PCR products were obtained using specific primers for each gene and β-actin as described in the text. Control (CONTROL), CCl4-treated (CCl4), CCl4-treated plus
Discussion
In the present study, we demonstrated that
We hypothesized that
Randle et al.
25
reported acute intoxication with xenobiotics including CCl4, increased glutamate cysteine ligase (GCL) activity, and the rate-limiting enzyme in the GSH biosynthetic pathway. However, free radicals produced by chronic CCl4 intoxication reduced hepatic GSH contents as a result of its metabolism (Figure 6); in fact, oxidative stress, including lipid peroxidation and GSH consumption, is one of the most common mechanisms leading to liver damage.
26
–29
Although inhibition of NF-κB could decrease GSH biosynthesis by regulating GCL expression,
30,31
the antioxidant properties of
Whole liver tissue was used to measure p65, the main subunit of NF-κB RelA.
33
As we used a specific Ab recognizing p65 that does not cross-react with NF-κB when bound to IκB, but does when activated, the p65 increase shown in Figure 7 represents the activation of this transcription factor. The CCl4-induced NF-κB activation is highly dependent on oxidative stress and liver inflammation is closely associated with the development of hepatic fibrosis and cirrhosis.
34,35
In this study, we showed that
Our results are supported by histopathological assays showing that following CCl4 intoxication, the hepatic parenchyma is distorted and fibrosis increased significantly as assessed by hydroxyproline content and visualized by staining with haematoxylin and eosin and trichromic stain. All of these effects were also partially prevented by
Cirrhosis is strongly associated with oxidative stress, chronic inflammation and increased TGF-β expression.
31,34
–39
The fibrogenic cytokine TGF-β plays a pivotal role in the activation of hepatic stellate cells to generate myofibroblasts, which in turn increase the production of ECM proteins leading to the progression of fibrosis;
37
is a prominent pro-fibrogenic cytokine with antiproliferative effects; and can up-regulate the deposition of ECM. In this study,
CTGF is a downstream mediator of TGF-β signalling in fibroblast cells and is widely thought to promote the development of fibrosis in collaboration with TGF-β. The crucial role of CTGF in fibrogenesis was evidenced by a significant up-regulation of ECM in fibrotic livers.
39
–41
In this study, we evaluated CTGF protein and mRNA levels, finding that the
Degradation of the interstitial matrix is regulated by inhibiting the synthesis of the metalloproteinases and increasing the production of the tissue inhibitor of metalloproteinases. Nevertheless, our results revealed a moderate presence of activated MMP-13 following 8 weeks of CCl4 treatment and a remarkable increase when CCl4-rats were treated with
Conclusions
The anti-fibrotic properties of
Footnotes
Acknowledgements
The authors express their gratitude to Biol. Mario Gil Moreno, Mr Ramón Hernández and QFB Silvia Galindo Gomez for their excellent technical assistance and to M.V.Z. Rafael Leyva Muñoz, MVZ Benjamin E Chavez and M.V.Z. Ricardo Gaxiola for animal handling and care. The authors also acknowledge the Animal Lab Facility, UPEAL-Cinvestav.. The authors also acknowledge the support from Conacyt-PNPC, 2015.
Funding
JEP-V was a Conacyt fellow (225157) and JS was partially supported by Conacyt grant 127357.
