Abstract
Methoxyflurane is a volatile, halogenated analgesic, self-administered in a controlled low dose from the Penthrox® inhaler for short-term pain relief. It was formerly used in significantly higher doses to produce anaesthesia, when it caused a specific type of dose-related renal tubular damage. The pathogenesis of the renal damage and clinical use of methoxyflurane are discussed here with evidence that a low but effective analgesic dose is not associated with the risk of renal adverse effects. The maximum dose employed to produce analgesia is limited to methoxyflurane 6 mL/day and 15 mL/week, producing a minimum alveolar concentration (MAC) of 0.59 MAC-hours. Renal damage is due to the metabolism of methoxyflurane and release of fluoride ions. Exposure of humans to methoxyflurane ≤2.0 MAC-hours, resulting in serum fluoride ≤40 µmol/L, has not been associated with renal tubular toxicity. The safety margin of analgesic use of methoxyflurane in the Penthrox ® inhaler is at least 2.7- to 8-fold, based on methoxyflurane MAC-hours or serum fluoride level, with clinical experience suggesting it is higher. It is concluded from clinical experience in emergency medicine, surgical procedures and various experimental and laboratory investigations that the analgesic use of methoxyflurane in subanaesthetic doses in the Penthrox inhaler does not carry a risk of nephrotoxicity.
Introduction
Methoxyflurane (CAS 76-38-0), 2,2-dichloro-1,1-difluoro-1-methoxyethane, is a volatile fluorinated hydrocarbon (Figure 1).

Structure of methoxyflurane, a fluorinated hydrocarbon.
It was first introduced as an inhalation anaesthetic for human use in the early 1960s. 1 After a few years of general availability, reports of dose-related renal tubular damage 2 –4 and the availability of newer anaesthetic agents led to its reduced use and disappearance from anaesthetic practice by the late 1970s and ultimately its discontinuation in the United States and Canada in 1999. In September 2005, the US Food and Drug Administration determined that Penthrane (Abbott Laboratories’ methoxyflurane) should be withdrawn from the market for safety reasons.
However, in Australia and New Zealand, methoxyflurane has never been withdrawn. It remains widely available and is much used, via a handheld inhaler, Penthrox®, as a self-administered, rapid-acting analgesic for short-term pain relief in civilian and military emergencies and in brief surgical procedures. Methoxyflurane has been available as an analgesic for prehospital use in Australia since 1975. It is 99.9% pure, and the impurities are of no toxicological significance based on chemical structure, in silico prediction and basic mutagenicity testing.
Whilst the clinical and anecdotal evidence indicate that methoxyflurane is a useful and effective analgesic agent, any potential for renal toxicity arising from its past use in significantly higher anaesthetic doses has continued to raise concerns about its safety. The aim of this article is to review all the laboratory and clinical data relevant to nephrotoxicity and methoxyflurane. and to evaluate the risk of nephrotoxicity due to its analgesic use.
Methods
This review concentrates on renal toxicity and it does not cover other safety considerations of methoxyflurane.
It is based on searches of Medline, Embase, Toxline and their subfolders, such as the Hazardous Substances Data Bank, and the Centre for Agriculture and Biosciences International and Scirus databases, employing the search terms listed in Table 1. Other information sources included standard textbooks and monographs on anaesthesia, toxicology and adverse events in humans, such as Davies’ Textbook of Adverse Drug Reactions, 5 Meyler’s Side Effects of Drugs, 6 and Side Effects of Drugs Annual. 7
Database search terms.

CABI: Centre for Agriculture and Biosciences International; HSDB: Hazardous Substances Data Bank.
‘Human’ and ‘animal’ references were accepted in any European language as well as Chinese and Japanese. The searches covered the period from 1949 to May 2013. Data, published and unpublished, were also obtained from the manufacturer. A follow-up PubMed search was conducted on 26 September 2014 to capture any additional articles relating to methoxyflurane and nephrotoxicity and none were identified.
Clinical use of methoxyflurane as an analgesic with the Penthrox® inhaler
Methoxyflurane vapour is administered as a short-term analgesic inhaled by the patient from a simple, green, whistle-shaped device, the Penthrox inhaler (Figure 2). Analgesia develops rapidly on using this device and can be maintained for as long as required until the 3 mL (4.12 g) charge of methoxyflurane has been exhausted. The maximum recommended dose is 2 inhalers/day equivalent to methoxyflurane 6 mL/day, up to a maximum of 15 mL/week. 8,9

Penthrox inhaler.
The more important value is the absorbed dose of methoxyflurane, as nephrotoxicity is a consequence of systemic exposure. The complete use of 3 mL from the Penthrox device would lead to an exposure to methoxyflurane of 0.3 MAC-hours, where MAC is the minimum alveolar concentration to produce surgical anaesthesia in 50% of healthy patients.
At clinically useful levels of analgesia obtained by intermittent inhalation of methoxyflurane in various clinical circumstances, the blood concentrations of methoxyflurane have variously been reported to be between 0.6 and 2.6 mg/100 mL (Table 2); for comparison Table 3 below shows the relationship between the blood fluoride and methoxyflurane levels after higher doses for anaesthetic use.
Blood concentrations of methoxyflurane when used as an analgesic.

General relationship between MAC-hours, serum fluoride and blood methoxyflurane levels in humans reported in the literature.a
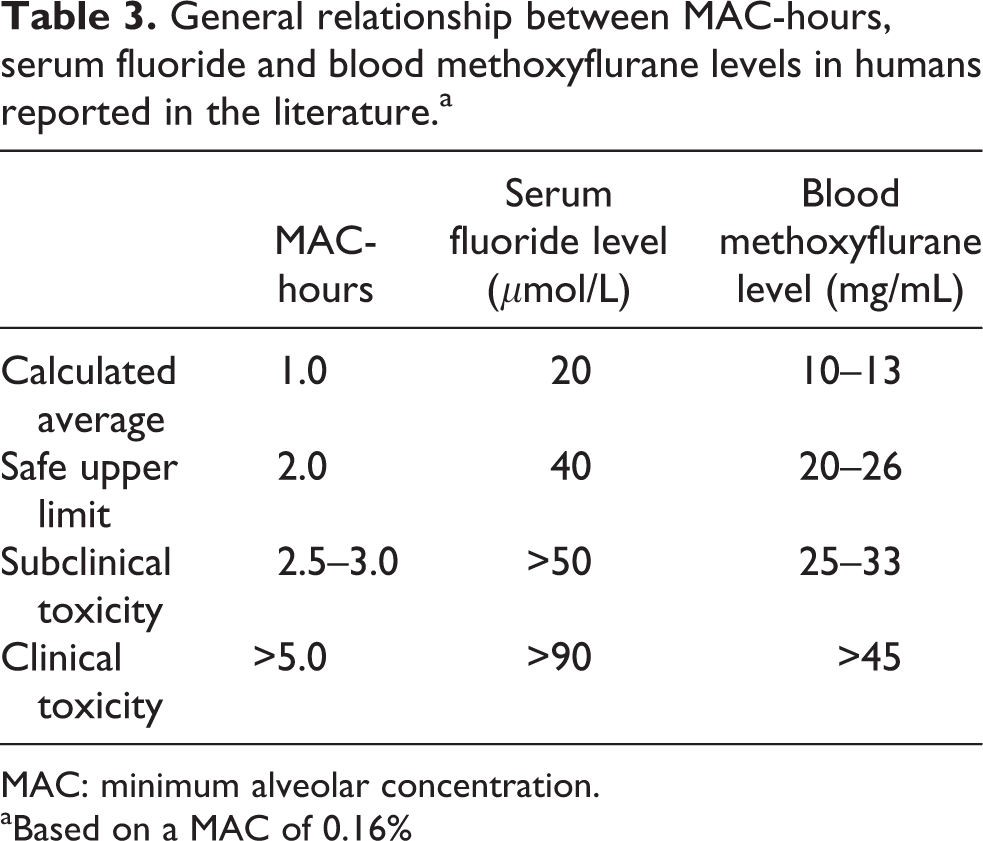
MAC: minimum alveolar concentration.
aBased on a MAC of 0.16%
In contrast, the arterial blood concentration of methoxyflurane in anaesthesia has been reported to be about 6–13 mg/100mL. 16 –18
Pharmacokinetics and metabolism of methoxyflurane
Methoxyflurane is rapidly metabolized in humans by oxidative demethylation and defluorination (Figure 3). 16,19 –23

Metabolism of methoxyflurane.
In humans, including patients given methoxyflurane as an anaesthetic or as an analgesic, healthy women in labour and healthy volunteers, the pharmacokinetics and metabolism of methoxyflurane have been studied mainly by following blood and urinary concentrations of the parent compound, its principal metabolites (fluoride and dichloroacetic acid (DCAA)) and their relationship to the inhaled concentration, as well as the total inhaled dose of methoxyflurane given either in higher doses to produce anaesthesia or in smaller doses to produce analgesia. 16,19,21 –27
Absorption is rapid, hence the fast onset of the analgesic action, due to the physicochemical characteristics of methoxyflurane, and blood levels plateau after less than 1 h of constant exposure. The blood levels may fall on prolonged exposure over several hours. 13,16 The latter phase has been attributed mainly to changes in the respiratory pattern.
A conventional elimination half-life (t ½) of methoxyflurane from blood cannot be calculated because of the nature of the exposure. An apparent t ½ of the order of 15-20 min after surgical anaesthesia with methoxyflurane can be extrapolated from the shape of the disappearance curves after inhalation has ceased. 10,12,16 However, that is not a conventional clearance value because of the slow and limiting rate of diffusion of methoxyflurane from adipose tissue into the bloodstream and its subsequent clearance by metabolism and exhalation.
In humans, 50-75% of absorbed methoxyflurane is considered to be metabolized. 28 The metabolites formed (fluoride, DCAA as the dichloroacetate anion at physiological pH and 2,2-difluoro-2-methoxyacetic acid) are excreted in the urine. 19,24,27 In human liver microsomes, methoxyflurane is metabolized by several cytochrome P450 (CYP450) isozymes including 2E, 2C and 3A. 26
Although the metabolism of methoxyflurane by several different CYP450 isoenzymes does not entirely exclude the possibility of clinically important genotypic and phenotypic interactions, it is unlikely that they would influence the occurrence of renal toxicity due to methoxyflurane.
Pathogenesis of the nephrotoxic action of methoxyflurane
There are two principal hypotheses, both based on the production of toxic metabolites of methoxyflurane in the body.
The best established hypothesis about the causation of renal tubular lesion, and the one that is directly supported by extensive experimental evidence, attributes it to fluoride ions generated by the metabolism of methoxyflurane in the liver and kidney. 16,24 An alternative hypothesis invokes fluoride released in that way and a co-pathogenetic role for DCAA also produced by the metabolism of methoxyflurane in those organs. 21,27 A comprehensive review of the toxicity of DCAA has not revealed any direct link to nephrotoxicity. 29
Regardless of that hypothesis, the blood level of the major metabolite fluoride appears to be an appropriate indicator of exposure and effect. 16 Cousins and Mazze concluded from their clinical studies that exposure to methoxyflurane resulting in serum fluoride <40 µmol/L was not associated with clinical or laboratory evidence of renal damage and that inhalation of methoxyflurane ≤2.0 MAC-hours by patients did not result in serum fluoride levels exceeding the 40 µmol/L limit. 16
Nephrotoxicity of methoxyflurane in animals
Murray and Fleming showed notable increases in plasma and urinary fluoride in human, dog, rabbit, guinea pig, calf, rhesus monkey, mouse and rat after surgical anaesthesia with methoxyflurane. 30
Mazze et al. were able to reproduce the characteristic type of methoxyflurane nephrotoxicity in a dose-related manner in the F344 strain of rat and demonstrate the cardinal importance of fluoride in the pathogenesis of the renal lesion. 28,31,32 The blood fluoride level was shown to be a good marker of exposure and of the risk of nephrotoxicity. Pathological changes in the kidney resembled those in affected patients. 33
Dogs that inhaled a full anaesthetic dose of methoxyflurane for 5 h on two occasions at an interval of 9 days developed renal dysfunction typical of tubular damage and a notable increase in serum fluoride levels, in a similar fashion to the rat and humans. 34
A considerable and prolonged rise in serum and intra-renal fluoride was described in rabbits anaesthetized with methoxyflurane, and the renal tubular damage was attributed to the fluoride ion. 35
Clinical (human) nephrotoxicity and methoxyflurane
Exposure of humans to methoxyflurane as an anaesthetic
The pathogenesis, clinical and pathological features of methoxyflurane-induced nephrotoxicity after its use as an anaesthetic have been widely published. 4,25,36 –40
The key features are that the renal failure is of the ‘high output’ type, that is, there is polyuria with passage of a large volume of dilute urine, loss of the normal concentrating response to vasopressin and biochemical and histopathological evidence of damage, mainly to proximal renal tubules. This unusual type of renal dysfunction, due to proximal tubular damage, is characteristic of fluoride toxicity. 41 –45 Recovery has usually occurred after 10 days at the most, but prolonged damage has occasionally been described with associated renal interstitial fibrosis. 46,47 Crystal deposition in the renal tubules has been an unusual finding in renal biopsies.
It is important that in no instance has there been any feature of an autoimmune process affecting the kidney, nor do the histopathological findings resemble those occurring in proven autoimmune reactions. 48
The incidence of renal damage induced in patients by prolonged high-dose inhalation of methoxyflurane to produce full surgical anaesthesia has not been well studied, but it may have occurred to some extent in more than 10% of patients at a clinically apparent level, and perhaps an even higher proportion may have had some abnormal renal function markers for a limited period after such anaesthetic exposure. 16,17,38
There is substantial clinical and experimental evidence from humans to show that if exposure to methoxyflurane is limited, nephrotoxicity will not develop – neither as polyuria, excretion of hypo-osmolar urine, nor as lesser changes in markers of renal function and tubular cell integrity. Cousins and Mazze 16 in a controlled trial in surgical anaesthesia described how exposure of adult patients to methoxyflurane 2.5-5.0 MAC-hours was associated with subclinical renal damage (detectable by standard biochemical laboratory tests), exposure to >5 MAC-hours resulted in clinical renal failure and that ≤2.0 MAC-hours should be regarded as the upper limit of safe exposure not resulting in detectable renal dysfunction. One MAC-hour was equivalent to a blood methoxyflurane level of 10-13 mg/mL. 16
The limit on exposure of ≤ 2.0 MAC-hours advanced by Cousins and Mazze was based both on direct observations in patients and on the results of animal experiments. 16 It has been related to the serum fluoride ion concentration as the principal and quite possibly the sole cause of the renal tubular damage. The relationship between MAC-hours, serum fluoride and blood methoxyflurane levels reported in humans is shown in Table 3.
In a smaller, independent study, 17 full anaesthesia with methoxyflurane for more than 2 h, at a MAC of 0.19% or 0.35% for >1 h, was associated with some biochemical evidence of renal damage, that is, the data implied that exposure ≤2 MAC-hours was not associated with biochemical evidence of renal damage. No report has been found that the proposed limit of serum fluoride ≤40 µmol/L has not been ‘safe’ under normal circumstances, that is, exposures up to 2.0 MAC-hours have not been associated with nephrotoxicity. 22,23
Results from the recent clinical trial of Spruyt et al. in bone marrow biopsy patients 49 have demonstrated the overall safety in use of short-term methoxyflurane analgesia and have shown that although its use was associated with a rise in total fluoride in serum, the level did not reach even the ‘safe upper limit’ proposed by Cousins and Mazze 16 (see Figure 4 and Table 4). It is noteworthy that no cases of methoxyflurane-induced nephrotoxicity have been reported in children after full anaesthesia with this agent. 4
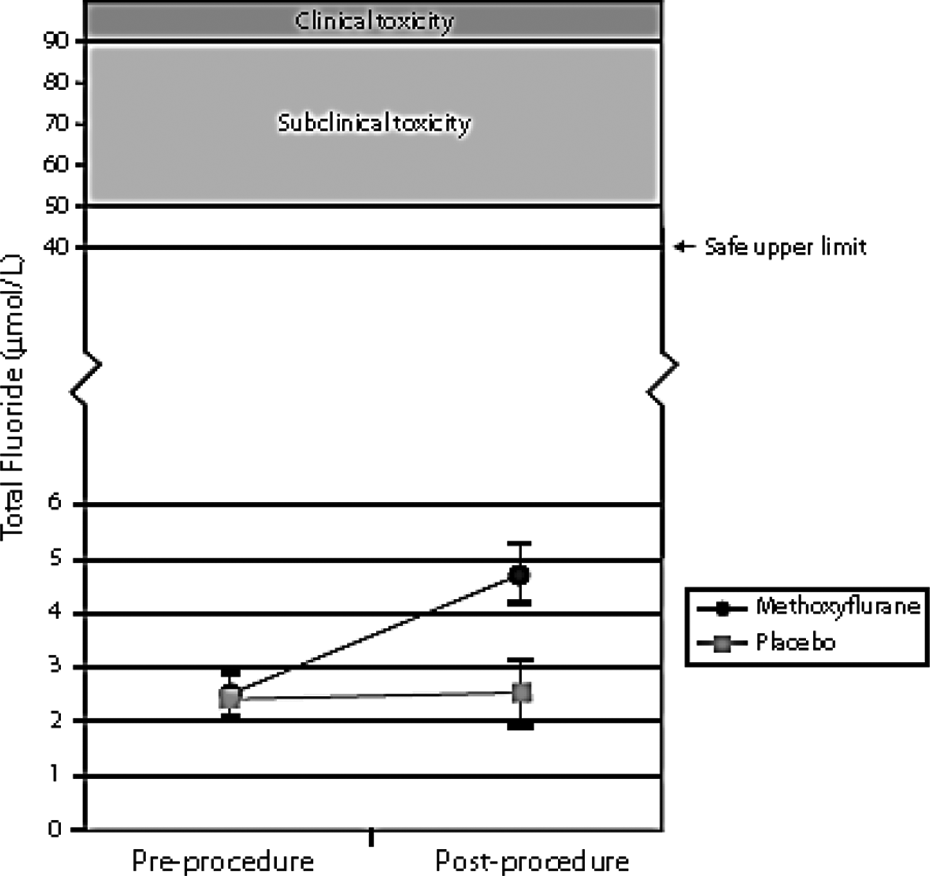
Total serum fluoride levels pre- and post-bone marrow biopsy. 49
Serum total fluoride levels (μmol/L) in patients before and after bone marrow biopsy under methoxyflurane analgesia.a

BMB: bone marrow biopsy; Max: maximum; Min: minimum; SD: standard deviation.
aData from Spruyt et al. 49
bTwo-sample t-test.
Renal function after methoxyflurane analgesia: Clinical experience
There have been various reports discussing renal function after methoxyflurane inhalation to produce analgesia in diverse clinical settings. For example, in a clinical experiment Artusio et al. described inhalation of methoxyflurane 0.3-0.6% for 6-8 min, two or three times at intervals of 4-6 days using Abbott’s handheld inhaler, the Analgizer® (and two more conventional inhalers), by 10 healthy subjects. There was no clinical or biochemical indication of any renal dysfunction over the subsequent 4 days. Serum fluoride was not measured. 12
Laird and Chrystal discussed a few markers of renal function after use of methoxyflurane as a short-term analgesic whilst changing dressings in 12 burns patients; after sufficient inhalation to produce analgesia for 35-40 min, there was only a brief fluctuation in serum uric acid level, but always within normal limits over the next 2-3 days and no change in blood urea nitrogen or creatinine. 50
A record linkage study was conducted in 135,770 patients, including 247 renal colic patients being transported to hospital by ambulance in Western Australia. The objective of the analysis was to assess long-term safety in patients who had received brief exposure to methoxyflurane to produce analgesia by comparing their health outcomes with a matched cohort of patients who had not received methoxyflurane. 51
Of the total group of 135,770 patients, 17,629 (13%) received at least one dose of methoxyflurane. Of those patients who had received methoxyflurane on at least one occasion, 14,410 (81.7%) patients received it only once, 1575 (8.9%) received it on two separate occasions and a further 341 (2.0%) were given it on three separate occasions. There was one patient who received methoxyflurane on 27 separate occasions. Recorded health event rates, when age standardized, were similar between the two groups. 51 There was no evidence of an increased risk of renal dysfunction in patients receiving methoxyflurane compared to patients who did not, the time taken to the first occurrence of disease was similar in the methoxyflurane- and non-methoxyflurane-treated patients, and no link between methoxyflurane analgesia and subsequent renal disorders was found. 51
When used for the treatment of acute pain in 300 patients, 90 of whom were aged 12-17 years, the new STOP! Study, which compared methoxyflurane-induced analgesia and a placebo, via a Penthrox inhaler, showed that methoxyflurane was not associated with nephrotoxicity based on blood biochemistry tests done −10 to +5 min from the start of inhalation of methoxyflurane as well as at the 14-day follow-up. 52
Similarly, following its use for short-term pain management during bone marrow biopsy in 97 patients, of whom half received methoxyflurane and the other half received placebo, blood samples for urea and electrolytes showed no difference between the two groups. Fluoride levels 30-45 min after the biopsy were significantly higher in the methoxyflurane group, although not in the known toxic range, and the highest individual blood fluoride level was 10 µmol/L. 51
The weakness of most of these studies is that there was only limited investigation of renal function, and there was no report of clinical or laboratory features of renal damage. The results represent valuable information but they are not conclusive. They have, however, been consistent with the earlier observations of Cousins and Mazze that showed exposure to methoxyflurane of ≤ 2.0 MAC-hours with a corresponding serum fluoride level ≤40 µmol/L was not associated with harmful effect on the kidney. 16
A quantitative summary was made of serum indicators of nephrotoxicity in eight obstetric studies in which methoxyflurane had been used as an analgesic. 15,53 –59 The investigation was conducted by Medical Developments International, Australia, the manufacturer of Penthrox. The weighted mean levels of serum inorganic fluoride, blood urea, serum creatinine and serum uric acid before and after the use of methoxyflurane were calculated, and these values were compared with the corresponding values in controls.
The analysis was performed using a random effects model in comparisons where heterogeneity between studies was significant (p < 0.05) and a fixed effect model for comparisons where heterogeneity was not significant (p > 0.05). There were no significant differences between controls and methoxyflurane-treated patients after analgesia in the levels of blood urea, serum creatinine or serum uric acid (all p > 0.05). As expected, there was a significant difference in levels of serum inorganic fluoride between controls and methoxyflurane-treated patients after analgesia (p < 0.0001). It was noted, however, that the mean value of serum inorganic fluoride after methoxyflurane analgesia from these studies did not exceed 20 µmol/L, – a level far below the 50 µmol/L previously linked to the occurrence of subclinical nephrotoxicity. 16 It should also be borne in mind that methoxyflurane administration in these studies was over a prolonged period during labour and delivery.
Overall, therefore, a clear link has been established between higher anaesthetic exposure to methoxyflurane and kidney damage, and none between the much lower exposure when its use is restricted to short-term analgesia, even in field use in the general population of adults and children.
In summary, from the overwhelming majority of formal clinical studies and observational reports since the mid-1960s, analgesia produced by methoxyflurane was not associated with overt evidence of renal dysfunction in adults or children. The duration of exposure ranged from about 6-8 min to 40-60 min or more in prehospital and accident and emergency use as well as obstetrics, brief orthopaedic and dental procedures and wound dressings. The total number of healthy subjects and patients amounted to several thousand adults and children down to the age of 1 year.
Discussion
There is a large body of clinical and laboratory evidence to show the safety of low-dose use of methoxyflurane to produce analgesia, especially that it does not cause nephrotoxicity. Methoxyflurane-induced nephrotoxicity is related to total dose (time and concentration) and the rate of metabolism. The product of metabolism, inorganic fluoride, results in concentration-related nephrotoxicity. The dose employed to produce analgesia is limited to methoxyflurane 6 mL/day of 0.1-0.7%, and 15 mL/week, which results in exposure 0.59 MAC-hours, which is well below the reported level of risk of nephrotoxicity of 2 MAC-hours. 16 This is supported by emerging safety data on extensive clinical use of low dose methoxyflurane for analgesic use. Additionally, the manufacturer’s records show that more than 5 million doses of methoxyflurane have been sold for use as an analgesic with no pharmacovigilance-related trends suggesting nephrotoxicity.
Extensive clinical experience of the use of methoxyflurane as an anaesthetic has shown an association with a notable incidence of high output renal dysfunction and even dose-related, frank renal failure due to proximal tubular damage resulting from the release of fluoride ions after enzymatic metabolism of the agent.
Anaesthetic use had involved inhalation of a high dose of methoxyflurane, as the MAC was about 0.16% 16 or even 0.35% 17 and the period of administration had mainly been >60 min.
Because of the incidence of nephrotoxicity after its high-dose anaesthetic use, methoxyflurane disappeared from anaesthesia practice in the late 1970s. 4 In 1999, the principal manufacturer Abbott Laboratories stopped distributing Penthrane (methoxyflurane) in the United States and Canada. However, methoxyflurane for analgesic use remained widely available in Australia and has been widely employed there since 1975 for short-term emergency pain relief in ambulance, emergency departments, mines and sports fields as well as to alleviate brief pain associated with such procedures as changing burns dressings, in colonoscopy, 60 bone marrow biopsies 49 and prostate biopsies. 61
In addition to the low dose required to produce analgesia, further protection against the risk of nephrotoxicity comes from the limited exposure to methoxyflurane associated with the recommended, intermittent use of the Penthrox inhaler; the charge of methoxyflurane would last for no more than 30-60 min when used continuously, and the usual pattern of self-administration is for briefer periods only when the patient feels the need for pain relief. As a device for self-administration, it is very likely that a patient would cease to inhale from it once pain relief had been achieved.
Confirmation of the safety of blood fluoride levels in patients using the Penthrox inhaler to obtain analgesia has recently been obtained in a controlled clinical trial. 49 In a series of 49 patients undergoing various procedures with analgesia provided by the Penthrox ® inhaler, the highest individual blood fluoride level was 10 µmol/L, which is well below the upper safety limit of 40–50 µmol/L demonstrated by Cousins and Mazze. 16
In relation to potential interactions with other drugs, there is no evidence to indicate that prior treatment with drugs that might affect the metabolism of methoxyflurane will increase the risk of toxicity because it is metabolised by several CYP450 isozymes and exposure is quantitatively limited when it is used to produce analgesia.
It is concluded that the experimental and clinical evidence show that use of methoxyflurane in the low doses used for analgesia does not carry any particular risk of causing renal dysfunction or damage.
Footnotes
Acknowledgements
I am grateful to Medical Developments International for permission to access company study data and reports when undertaking this review and to Kim Magner for her help.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
