Abstract
The aim of this study was focused on investigating the possible protective effect of erdosteine against gamma radiation-induced renal lesions in male albino rats. Twenty-eight albino rats were divided into four equal groups as follows: control group, irradiated group (animals subjected to whole-body gamma irradiation at a dose of 5 Gy), treated group (each rat received 100 mg/kg body weight once daily, orally by gastric tube, erdosteine for 1 week), and treated irradiated group (each rat received 100 mg/kg body weight once daily, orally by gastric tube, erdosteine for 1 week, then exposed to whole-body gamma irradiation at a dose of 5 Gy). The results revealed that the administration of erdosteine to rats before irradiation significantly ameliorated the changes occurred in kidney function (creatinine and urea) compared with irradiated group. Also the changes in serum tumor necrosis factor α, interleukin 1β, and interleukin 6 activities were markedly improved compared with the corresponding values of irradiated group. Kidney catalase and glutathione peroxidase (GPx) activities and reduced glutathione concentration showed approximately normal level when compared with the irradiated group. The histopathological results showed distinctive pattern of renal lesions in irradiated group, while in treated irradiated group the renal tissues showed relatively well-preserved architecture. Erdosteine acts in the kidney as a potent scavenger of free radicals to prevent or ameliorate the toxic effects of gamma irradiation as shown in the biochemical and histopathological changes and might provide substantial protection against radiation-induced inflammatory damage.
Introduction
Ionizing radiation, such as X-ray or gamma ray, damages tissue by creating an unbalanced free radical, disrupts molecules, such as DNA, producing mutations that can lead to cancer. Furthermore, the free radicals while interacting chemically with other molecules form toxic reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) that create further damage in cells and tissues. 1 The production of free radicals following exposure to ionizing radiation takes place in less than a second and their damage is instant. Our body, therefore, must have radiation protective means to prevent free radical damage and its later consequences. 2 Moulder and Cohen reported that irradiation can produce chronic renal damage in many species of animals at relatively low doses. 3 Also, Hanafy and Kandil reported that gamma radiation with a dose of 6 Gy in rats induced significant renal lesions that included atrophied glomeruli, widened Bowman’s capsule, high cellularity in the visceral layer of the Bowman’s capsule, and highly affected cytoplasm and nuclei of the convoluted tubules. 4 Agostino et al. found that the whole irradiated animals had severe renal damage involving the glomeruli, the tubules, the interstitial tissue, and the blood vessels. 5 Moreover, Kucuktulu reported that gamma radiation at a dose of 20 Gy are capable of inducing chronic nephritis. 6
Maasilta et al. reported that N-acetyl cysteine may inhibit free oxygen radicals and may have a radioprotective effect on patients receiving radiotherapy. 7 Another homocysteine derivative erdosteine is known to have protective role on the release of free oxygen radicals beside its mucolytic and mucomodulator properties. 8,9 Erden et al. reported that erdosteine may prevent the acute lung inflammation and fibrosis by suppressing the accumulation of neutrophils, inhibition of lipid peroxydation, and chemokine production and release. 10 Zeki et al. studied the effect of oral erdosteine on tissue malondialdehyde (MDA), nitric oxide (NO) levels, catalase (CAT), delayed glutathione peroxidase (GPX), and superoxide dismutase (SOD) activities in the cisplatin model of acute renal failure in rats. 11 A single dose of cisplatin caused kidney damage manifested by kidney histology as well as increases in plasma creatinine and blood urea nitrogen (BUN) levels. Treatment with free radical scavenger erdosteine attenuated increases in plasma creatinine and BUN and tissue MDA and NO levels, and provided a histologically proven protection against cisplatin-induced acute renal failure. Erdosteine also reduced depletion in the tissue CAT, GPx, and SOD activities. These results show that erdosteine may be a promising drug for protection in cisplatin-induced nephrotoxicity. Dal Negro et al. stated that the part of the beneficial anti-inflammatory protective effects of erdosteine may be mediated through the inhibition of proinflammatory cytokines tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), IFN γ, and interleukin 6 (IL-6) and this in turn would lead to reduced production of free radicals and subsequent damage. 12 However, further studies with different doses of erdosteine are warranted for clarifying the issue. The aim of this study was oriented to evaluate the protective effect of erdosteine in irradiation-induced experimental renal lesions in male albino rats.
Materials and methods
Animals
Adult male Swiss Albino rats (180–200 g) were obtained from the Egyptian Organization for Biological Product and Vaccines, Giza, Egypt. Animals were kept under good ventilation and illumination condition and received standard diet and water.
Radiation processing
It was performed by using Gammacell-40 (Cesium-137) located at the National Centre for Radiation Research and Technology, Cairo, Egypt. Animals were irradiated with a single dose level of 5 Gy delivered at a dose rate of 0.46 Gy/min at the time of experimentation. Animals were not anesthetized before irradiation.
Chemicals and drugs
Erdosteine tablets (300 mg) were purchased from Sigma Company, Cairo, Egypt.
Experimental design
Twenty-eight rats were divided into four groups (n = 7). In group I (control group), rats were neither treated nor irradiated. Rats of this group received orally an equivalent volume of distilled water (vehicle of erdosteine) during the period of erdosteine administration. Rats in group II (irradiated group) were exposed to 5 Gy to induce biochemical and pathological changes before whole-body gamma radiations and like control group received orally an equivalent volume of distilled water during the period of erdosteine administration. In group III (treated group), rats were administrated 100 mg/kg body weight erdosteine, once daily for 1 week by gastric tube according to Fadilliogiu and Erdogan. 13 Group IV (treated irradiated group) rats were administrated 100 mg/kg body weight erdosteine, once daily for 1 week by gastric tube before exposure to whole-body gamma irradiation at a dose of 5 Gy. Rats were killed on the 5th day because pathological lesions were to be well observed postradiation exposure or erdosteine administration.
Sample collection
After an overnight fast, rats were anesthetized with ether and then killed. Blood samples from each rat were collected by retro-orbital puncture using blood capillary tubes. Serum was obtained immediately by centrifugation of blood samples at 3000 r/min for 10 min. Kidney was directly separated after the killing of rats, washed in ice-cold saline, then the kidney samples were homogenized in distilled water (10% w/v) using a homogenizer, and then the cell debris was removed by centrifugation at 3000 r/min for 10 min. The supernatant of homogenates was subjected to biochemical analysis. Tissue specimens from kidney were collected and fixed in 10%-buffered formalin solution followed by dehydration, clearing, and embedding in paraffin. Paraffin sections of 5-μ thickness were prepared and stained routinely with hematoxylin and eosin according to Bancroft and Stevens 14 and examined microscopically.
Estimation of biochemical parameters
The GSH content was determined photometrically according to the method described by Ellman. 15 GPx activity was assayed according to the method of Gross et al. 16 CAT activity was assayed using the method of Sinha. 17 Serum urea was estimated according to the method of Fawcett and Scott 18 and creatinine was assayed according to the method of Chasson et al. 19 Detection of serum TNF-α, IL-1β, and IL-6 was performed by enzyme-linked immunosorbent assay technique (BioSource International, Camarillo, California, USA) according to the manufacturer’s instructions.
Statistical analysis
Data were analyzed using one-way analysis of variance followed by least significant difference as is post hoc test. The results obtained were expressed by mean ± standard error of the mean. Differences were considered significant at p ≤ 0.05. 20
Results
As presented in Table 1, whole-body gamma irradiation induced a significant increase in the concentration of serum urea and creatinine compared with the control group. Pretreatment with erdosteine prior to gamma irradiation significantly abolish these radiation-induced changes.
The concentration of urea and creatinine in the different animal groups.a

aAll values are expressed as mean ± SE.
b p ≤ 0.05: significant when compared with the control group.
c p ≤ 0.05: significant when compared with the irradiated group.
The effects of gamma radiation on endogenous antioxidant status are shown in Table 2. Gamma irradiation induced significant decrease in the kidney CAT, GPx activities, and GSH content compared with control group. Administration of erdostine for seven consecutive days prior to gamma irradiation resulted in a significant increase of kidney CAT, GPx activities, and GSH content compared with the irradiated group.
Kidney CAT and GPx activities and reduced GSH level in the different animal groups.a
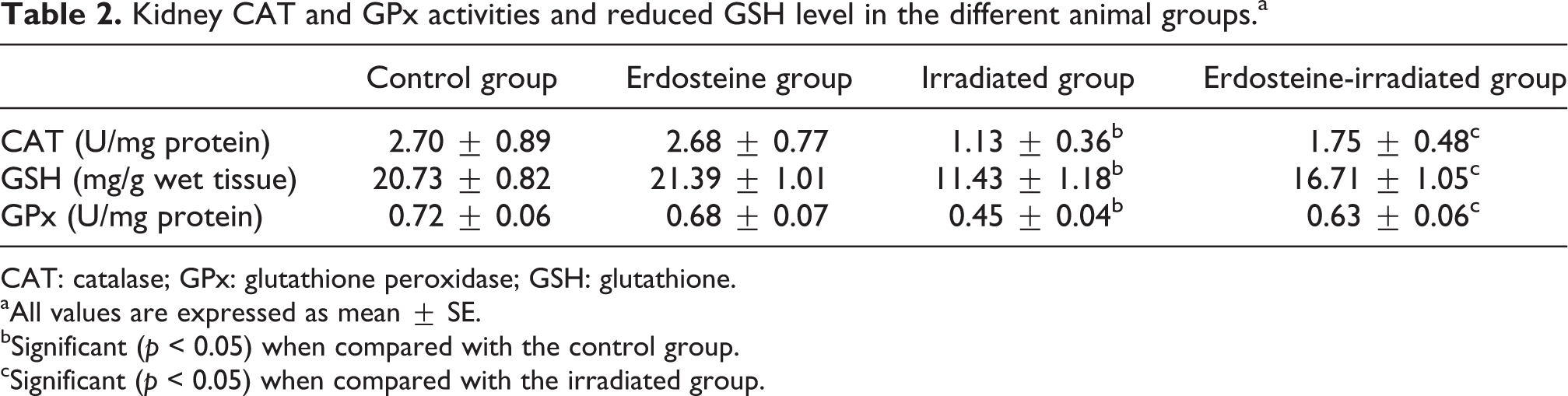
CAT: catalase; GPx: glutathione peroxidase; GSH: glutathione.
aAll values are expressed as mean ± SE.
bSignificant (p < 0.05) when compared with the control group.
cSignificant (p < 0.05) when compared with the irradiated group.
Also, as shown in Table 3, significant increases in some inflammatory markers represented in serum TNF-α, IL-1β, and IL-6 levels were observed in the irradiated group compared with the corresponding values of control rats. The administration of erdosteine before exposure to gamma irradiation significantly limits the elevation in serum TNF-α, IL-1β, and IL-6 levels compared with the irradiated group.
Serum TNF-α, IL-1β, and IL-6 levels in the different animal groups.a
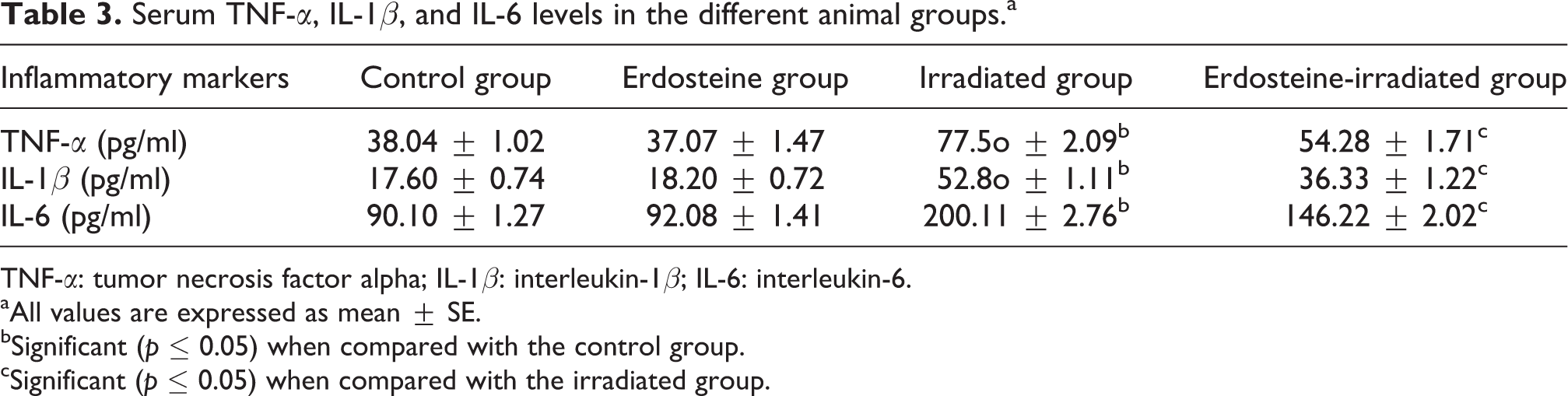
TNF-α: tumor necrosis factor alpha; IL-1β: interleukin-1β; IL-6: interleukin-6.
aAll values are expressed as mean ± SE.
bSignificant (p ≤ 0.05) when compared with the control group.
cSignificant (p ≤ 0.05) when compared with the irradiated group.
Histopathological finding
The kidney of control rats (group I) showed greater details of the renal corpuscle and tubules. The corpuscle consists of glomerulus and Bowman’s capsule. The glomerulus is a tuft of capillaries formed from the afferent glomerular arterioles and supported by fine connective tissue. The Bowman’s capsule contains simple squamous epithelium, at the urinary pole; the squamous epithelium of the partial layer change to cuboidal epithelium of the proximal convoluted tubules. Numerous tubules (proximal and distal) that lie adjacent to renal corpuscles contain a single layer of cuboidal epithelium (Figure 1). In group II (irradiated group), rats’ renal cortex showed atrophied glomeruli. Widened Bowman’s space and thickened basement membrane were seen. The convoluted tubules showed distinctive pattern of ischemic renal injury represented by losing its details with partial or complete obstruction of its lumens, the cuboidal epithelium cells of proximal and distal renal tubules showed nuclear changes mainly pyknosis and karyolysis and infiltrated with leukocytes (Figure 2). In some cases, hyalinization of tubules were present (Figure 3). In most of the cases in group IV (treated irradiated group) rats, the histological structure of kidney showed relatively well-preserved architecture without degenerative changes. In few cases, slightly reversible changes in the convoluted tubules or focal degeneration were seen with distorted tubules lined by cuboidal epithelium; the cells have small round nuclei without hyaline casts in its lumina (Figure 4). The kidney of erdosteine group showed normal histological appearance as control group.

Kidney of control rat (group 1) showing normal structure (H&E 400×). H&E: hematoxylin and eosin.

Kidney of irradiated rat (group II) showing dilatation of some tubules with leukocytic infiltration around necrotic tubules (H&E 400×). H&E: hematoxylin and eosin.

Kidney of irradiated rat (group II) showing hyalinization of renal tubules (H&E 400×). H&E: hematoxylin and eosin.

Kidney of treated irradiated rat (group IV) showing normal renal tubules (H&E 400×). H&E: hematoxylin and eosin.
Discussion
Exposure to ionizing radiation increases the production of ROS and directs the irradiated cells into a state of oxidative stress that has been implicated in a variety of natural and pathological processes. 21 Antioxidants including erdosteine, which protect against oxidative damage, lower the risk of injury to vital molecules and to varying degrees may help or prevent the onset and progression of diseases. 22,23
Plasma creatinine concentration is a more potent indicator than the urea concentration in the first phases of kidney disease. Furthermore, urea concentration begins to increase only after parenchyma tissue injury. 24 In our study, plasma creatinine and urea levels were higher in the irradiated group when compared with the control group and there were significant decreases in plasma creatinine and urea levels in the treated irradiated groups when compared with the irradiated group (Table 1). The results support the findings of Hanafy and Kandile that whole-body gamma irradiation of rats (6 Gy) increases the level of plasma creatinine and urea. 4 Zeki et al. added that treatment with free radical scavenger erdosteine attenuated increases in plasma creatinine and urea and tissue MDA and NO levels, and provided a histologically proven protection against cisplatin-induced acute renal failure. Erdosteine also reduced depletion in the tissue CAT, GPx, and SOD activities. 11 These results show that erdosteine may be a promising drug for protection against cisplatin-induced nephrotoxicity. In the current study, the oral administration of erdosteine prevented gamma radiation-induced nephrotoxicity in rats as evidenced by significant reduction in serum urea and creatinine levels. Our results indicate that erdosteine has exerted protection effect against radiation-induced nephrotoxicity. CAT is an enzyme that catalyzes the decomposition of H2O2 to oxygen and water and is present in all aerobic cells. 25 The induction of CAT was expected in response to gamma radiation-induced H2O2 production, where CAT protects from oxidative stress by preventing the accumulation of H2O2, reducing it to water and oxygen. 26 However, the decrease in the activity of CAT recorded in this study (Table 2) might be due to the leakage of the enzyme from the oxidant injured cells. 27
Reduced glutathione (GSH) participate in the cellular system of defense against oxidative damage, 28 directly as a free radical scavenger or indirectly by repairing initial damage to macromolecules and could maintain protein and nonprotein sulfhydryl (SH) group in reduced form. 29 The significant reduction in kidney GSH demonstrated in this study (Table 2) after gamma radiation exposure could be due to an enhanced utilization in large amount to combat the radiation-induced free radical damage, as GSH is a major nonenzymatic antioxidant. 30
GPx plays an important role in the defense mechanisms of mammals against damage by catalyzing the reduction of H2O2 and a large variety of hydroperoxides into water and alcohols, respectively, consuming GSH as the hydrogen donor and its depletion leads to GPx inactivation. 29 The significant decrease in kidney GPx activity of irradiated rats (Table 2) could be attributed to its inactivation by lipid peroxidation byproducts. 31
The data of this study indicated that erdosteine ameliorated the disturbance in the antioxidant system that resulted from exposure to gamma radiation. The antioxidant activities of erdoseine suggested that erdosteine ingredients could have potent anti-inflammatory activities. Erdosteine contains two blocked sulfhydryl groups one of which, after hepatic metabolization and opening of the thiolactone ring, becomes available both for the mucolytic and free radical scavenging and antioxidant activity too. Moretti and Marchieni reported that there are several experimental evidences that support the protective effect of erdosteine in acute injury induced by a variety of pharmacological or noxious agents, mediated by products of oxidative stress. 32 Experimental data in animal exposed to a noxious agent revealed that co-treatment with erdosteine increases the tissue antioxidant enzyme activities such as SOD, CAT, and GPx compared with the toxic agent alone; meanwhile erdosteine decreases the tissue level of NO and xanthine oxidase, which catalyze oxygen-free radical production. They add that erdosteine prevents the accumulation of free oxygen radicals when their production is accelerated and increases antioxidant cellular protective mechanisms. The final result is a protective effect on tissues, which reduces lipid peroxidation, neutrophil infiltration, or cell apoptosis mediated by noxious agents.
Radiation exposure of normal tissues inflicts an inflammatory response involving activation of stress-sensitive kinases, transcription factors, 33 and increased production of inflammatory cytokines. 34 The radiation-induced increases in the level of TNF-α, IL-1β, and IL-6 recorded in this study are presented in Table 3. In general, a cascade of inflammatory events is regulated through the production of proinflammatory mediators. Enhanced expression of proinflammatory cytokines, chemokines, and adhesion molecules, and their close interactions facilitate proinflammatory pathways by recruiting and transmigrating inflammatory cells from blood to tissues. 35,36 Several previous in vivo and in vitro studies have demonstrated that TNF-α and IL-1β are the most important proinflammatory cytokines that exert a central role in acute and chronic inflammation. It is well known that TNF-α and IL-1β strongly promote inflammatory responses in a wide spectrum of cell types, and overproduction of these cytokines has been implicated in a variety of human diseases including atherosclerosis, autoimmune disorders, and cancer. 37 In addition, increased levels of TNF-α and IL-1β gene expression were observed as an initial response of the mouse and rat brains to brain irradiation 38 and partial-body irradiation. 39 Upregulation of TNF-α and IL-1β expression was also found in lung and intestine after irradiation. 21,40 IL-6 is another multifunctional proinflammatory cytokine that plays a major role in the mediation of the inflammatory and immune responses initiated by infection or injury. 41 Recent studies have suggested that elevated levels of IL-6 messenger RNA and protein expression may be responsible for the radiation-induced inflammation in the intestine and whole brain. 39,40 Additionally, both total body and localized irradiation resulted in a small but significant increase in IL-6 levels in rats. 42 The amelioration of inflammatory factors in erdosteine-irradiated group (Table 3) suggested that erdosteine could have potent anti-inflammatory activities. The results corroborate the findings of Marabini et al. that erdosteine exert anti-inflammatory and anti-oxidant properties due to the presence of a pharmacologically active sulfhydryl group that has been found to have a free radical scavenging activity. 43
The histopathological results of this study indicated that the irradiated group revealed atrophied glomeruli. Widened Bowman’s space and thickened basement membrane were seen. The cuboidal epithelium cells of proximal and distal renal tubules showed nuclear changes mainly pyknosis and karyolysis and infiltrated with leukocytes. In some cases, hyalinization of tubules occurred. The results are in agreement with the findings of Hanafy and Kandile. 4 In group IV (treated irradiated group), the histological structure of kidney showed relatively well-preserved architecture without degenerative changes. In few cases, slightly reversible changes in the convoluted tubules or focal degeneration were seen with distorted tubules lined by cuboidal epithelium, the cells have a small round nuclei without hyaline casts in its lumina that corroborate the results obtained by Zeki et al. that erdosteine may be a promising drug for protection against nephrotoxicity. However, further studies with different doses of erdosteine are warranted for clarifying the issue. 11
In conclusion, this work documents that treatment with erdosteine offers protection from radiation-induced nephrotoxicity. The deterioration of proinflammatory cytokine and histological damage in kidney caused by gamma irradiation are markedly improved by erdosteine treatment. These observations may be attributed partly to the considerable antioxidant effect of erdosteine and suggest that it may be a valuable prophylactic agent against a variety of conditions and diseases.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
