Abstract
Introduction:
Cyclophosphamide (CPA) chemotherapy leads to ovarian failure and infertility. Tocotrienol (T3) is an antioxidant and anti-inflammatory agent. The role of T3 in ovarian protection throughout chemotherapy remains unclear.
Aim:
To investigate the role of T3 in the preservation of female fertility in CPA treatment.
Method:
Sixty female mice were divided into five treatment groups, namely, normal saline, corn oil only, T3 only, CPA and CPA + T3. The treatment was given for 30 days, followed by administration of gonadotrophin to induce ovulation. After killing, both ovaries were collected and examined histologically.
Results:
There was significant reduction in ovarian size in the CPA group compared with the normal group (CPA versus normal, mean area ± SD; 0.118 ± 0.018 vs. 0.423 ± 0.024 cm2; p ≤ 0.005), whilst concurrent administration of T3 with CPA leads to conservation of ovarian size (CPA + T3 vs. CPA, mean area ± SD; 0.285 ± 0.032 vs. 0.118 ± 0.018 cm2; p ≤ 0.005). Ovaries in CPA group showed abnormal folliculogenesis with accompanied reduced ovulation rate, follicular oedema, increased vascularity and inflammatory cell infiltration. These changes were reversed by concurrent T3 administration.
Conclusion:
Co-administration of T3 with CPA confers protection of ovarian morphology and function in vivo. These findings contribute to the further elucidation of CPA effect on ovary and suggest the potential of T3 use in preserving fertility in chemotherapy.
Introduction
Infertility is increasing worldwide, with approximately 15% incidence in both genders of reproductive age. Long-term anticancer drug treatment markedly affects non-target tissue such as the ovaries 1 causing iatrogenic infertility in young patients. 2,3 Cyclophosphamide (CPA) is an anticancer drug with a wide spectrum of usage. It is a cytotoxic agent acting to inhibit cell division. This drug affects the ovary in time- and dose-dependent fashion. 4,5 It has been reported to decrease follicle numbers, cause disturbances in oestrogen levels and deplete the follicles of their antrum. 6 Consequently, several strategies have been established to preserve fertility in chemotherapy administration. 2 This includes cryopreservation of mature ovaries and administration of gonadotrophin-releasing hormone (GnRH) antagonist to prevent ovulation during treatment. 7
Tocotrienol (T3) is a less investigated form of vitamin E compared with tocopherol. It has an unsaturated isoprenoid side chain with three double bonds compared with tocopherol, which has a saturated phytyl side chain. 8 T3 exists in four isomers, namely, α-, β-, δ- and γ-T3; these isomers occur naturally in plants. T3 has been reported to have potent antioxidant, anti-inflammatory, anticancer, anti-ageing, anti-thrombotic and anti-angiogenic properties. 9 The effect of T3 as a chemoprotective agent to sustain fertility following chemotherapy remains to be elucidated.
The aim of this study is to further characterize morphologic and functional changes in the ovary following long-term, low-dose treatment with CPA and the effect of T3 on the said ovarian changes when administered in combination with CPA.
Methods
This is a prospective, randomized, controlled, interventional animal study using mice as the subject. This study was reviewed and approved by the Laboratory Animal Care Unit Committee, Universiti Teknologi MARA (UiTM), Malaysia, in concurrence with Malaysian Guidelines for Good Clinical Practices, Ministry of Health Malaysia.
Animals
Sixty imprinting control region female mice aged between 8 weeks and 10 weeks and an average weight of 29.5 g were used. They were housed in plastic cages in an air-conditioned room (19 ± 2°C) with 12-h alternate light and dark period. They were fed pellet diet and given water ad libitum.
Experimental groups and procedures
The mice were randomly divided into five experimental groups (n = 12/group). The first group was given 0.15 ml normal saline intraperitoneally (i.p.; normal control), the second group with corn oil only (0.15 ml, oral gavage), the third group with T3 only (given 60 mg/kg body weight T3, oral gavage), the fourth group with 10 mg/kg body weight CPA injected i.p. 4 and the fifth group was administered the same dose of CPA, in addition to 60 mg/kg body weight T3 by oral needle gavages. 10 The T3 mixture (Malaysian Palm Oil Board, Malaysia) was composed of 27.45% α-T3, 57.99% γ-T3 and 14.56% δ-T3, diluted in corn oil to the desired concentration. All treatments were given for 30 days.
After completing treatments, all animals were subjected to induction of ovulation by injecting 5 IU human chorionic gonadotropin (hCG, Sigma, St Louis, Missouri, USA) 48 h after administration of 5 IU pregnant mare serum gonadotropin (Sigma). 11 The mice were killed by cervical dislocation 14–16 h after hCG administration.
Tissue collection, processing and evaluation
After killing, ovulated oocytes were immediately collected from the oviduct ampulla into M2 medium (Sigma) and counted under a dissecting microscope. Both ovaries were measured and then fixed in 10% formalin, subsequently embedded in paraffin, serially sectioned at 3 µm thickness and stained with haematoxylin and eosin. They were viewed under the light microscope. The types of follicles seen were classified into healthy and unhealthy follicles. The healthy follicles were further divided into the following five classes according to Erickson and Schriber 12 : (i) primordial follicles (PMFs), consisting of oocytes surrounded by a single layer of flat cuboidal cells; (ii) primary follicles, consisting of oocyte, zona pellucida and cuboidal granulosa cells; (iii) secondary follicles, recognized by oocyte surrounded by two to eight layers of granulosa cells with no initial antrum; (iv) early tertiary follicles, identified by a well-defined antrum surrounded by multiple layers of granulosa cells; and (v) Graafian follicles characterized with large antrum occupying most of the total follicle volume. Tertiary follicle and Graafian follicles are similar, except for the size of the follicle and the antrum. 12 The distinction is made so as to allow a more detailed analysis of the morphological differences between the groups.
The unhealthy follicles were defined as follicles with atretic and cystic changes. The number and diameter of each follicle, the number of corpus luteum, number of follicles with antrum formation and the number of oocytes were analysed using an image analysis software (NIS-Elements, Nikon, Japan), and the results were recorded. Degree of vascularity, follicular oedema and number of inflammatory cells were also quantitatively analysed using the same method.
Statistical analysis
All data were subjected to statistical analysis by one-way analysis of variance using Statistical Package for the Social Sciences version 19. Significant differences were analyzed by Duncan’s triplicates range test. 13 Results were presented as mean ± standard deviation; statistical significance was taken significant if the difference is at p value ≤0.05 and 0.01.
Results
Ovarian size
The ovary in CPA-treated group was significantly smaller than normal control (CPA vs. normal (mean ± SD); 0.118 ± 0.018 vs. 0.423 ± 0.024 cm2; p < 0.05). Co-administration of T3 with CPA leads to significant preservation of ovarian size, with the size being comparable with the normal group (Figure 1). The groups treated with T3 only and corn oil only do not show any significant difference in ovarian size compared with the normal control (Figure 1).

Comparison between ovarian sizes in all study groups. There was a significant increase in ovarian size of CPA + T3 group compared with CPA only group (*p < 0.05); the size of ovaries in CPA-treated group is significantly smaller than the NS control, T3 and corn oil groups (**p > 0.05); no differences were observed between the NS control, T3, corn oil and CPA + T3 groups. Data are represented as mean area. NS: normal saline; CPA: cyclophosphamide; T3: tocotrienol.
Ovulation rate
The CPA-treated group had an ovulation rate of 46% compared with the normal control. T3 + CPA co-administration increased the ovulation rate to approximately 78% of normal, p < 0.01. The ovulation rate in corn oil and T3 groups was not significantly different from the normal control (Figure 2).
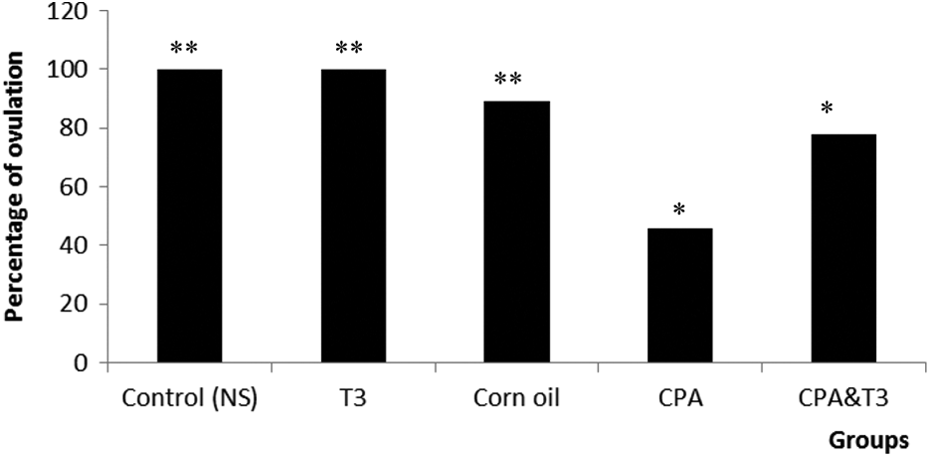
Comparison of ovulation rate in between the control, corn oil, T3, CPA and CPA + T3 groups. Significant decrease in ovulation was seen in the CPA group compared to all control groups (**p < 0.001). An increase in ovulation was observed in CPA + T3 compared with the CPA group (*p < 0.05). There is no significant difference between the control groups and the CPA + T3 group (p = 0.07). Data are represented as percentage. CPA: cyclophosphamide; T3: tocotrienol.
Folliculogenesis assessment
CPA-exposed ovaries have significantly reduced the number of follicles compared with the control (p < 0.05; Table 1). Ovaries exposed to CPA showed reduced follicle maturation at all stages, but the decrease was more marked in the secondary, tertiary and Graafian follicle stages compared to earlier stages. In addition, more unhealthy (cystic and atretic) follicles were seen in this group (Figure 3). Administration of CPA + T3 restored the follicle number to near normal, with significant increase compared with the CPA group (Table 1; p < 0.05). Furthermore, the number of unhealthy follicles was reduced and the population size of healthy follicles was re-established (Figure 3). The diameter of the follicles and antrum was significantly decreased in CPA-treated group compared with the control. Co-administration of T3 with CPA restored their diameter (Figure 4(a) to (c)). Moreover, there is correlation between the diameter of follicles and development of antrum and oocytes in this group, as was also seen in the normal, T3 only and corn oil groups. In the CPA group, the unhealthy follicles showed no correlation between the above parameters, indicating disordered folliculogenesis.
Number of follicles according to their maturation in all groups.a

aThere was significant reduction in all follicle types in CPA-treated group; co-administration of T3 leads to marked improvement in all categories; data are represented as mean ± SD. Comparison between control (NS) versus T3; corn oil and CPA + T3 yielded insignificant p values (p ≥ 0.05).
b p < 0.001: control vs. CPA.
c p = 0.01: CPA vs. CPA + T3.

The numbers of unhealthy follicles in different stages in follicullogenesis in CPA and CPA + T3 groups. There is overall decrease in the number of unhealthy follicles in all maturation level of follicles (p < 0.05). CPA: cyclophosphamide; T3: tocotrienol.
Further histopathological observations
In addition to the above changes, CPA-treated ovary has significantly more degree of vascular channels (Figure 4(c)), follicular oedema (Figure 4(d)) and inflammatory cell infiltration (Figure 4(e)) than the control groups (p < 0.01; Figure 4). Co-administration of T3 with CPA reduces the changes (p < 0.05; Figures 4(f) and 5).
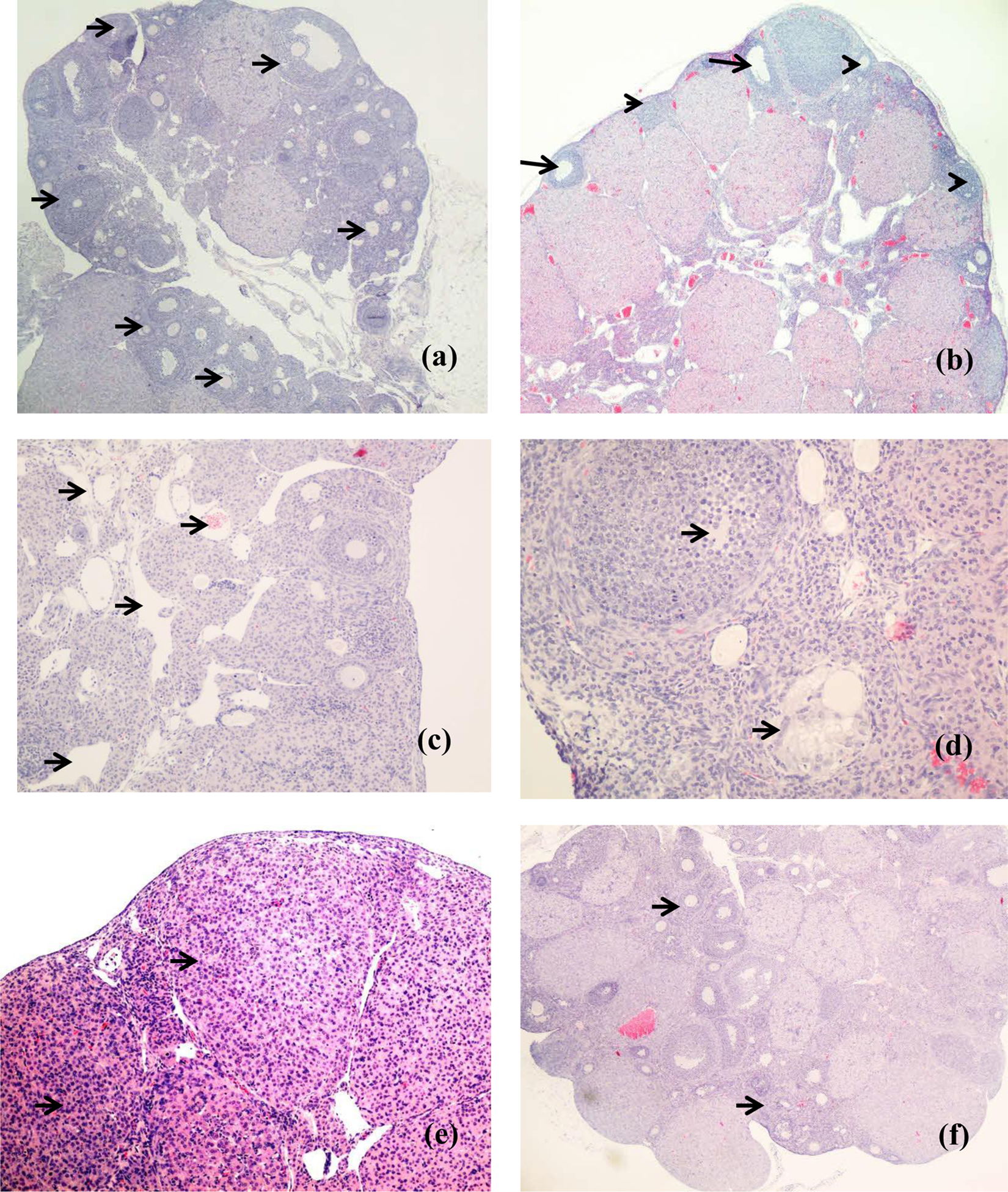
Photomicrographs of representative sections from the ovaries of the various groups. (a) Ovary from the normal saline control showing numerous follicles of differing maturity (arrows) present almost circumferentially within the cortex of the ovary; ×4 magnification. (b) CPA-exposed ovary showing marked decrease in follicles (arrows) and increased number of atretic follicles (arrow head; ×10). (c) Higher magnification (×20) of the area of the ovary with increase in vascular channels (arrows), especially within the medulla. (d) Section showing oedematous follicles (arrows). (e) Section showing areas with increase in cellularity due to inflammatory cell infiltration (arrow; ×20). (f) Section from ovary from CPA + T3 group showing near-normal follicle (arrow) numbers and morphology (×4 magnification). CPA: cyclophosphamide; T3: tocotrienol.

Image analysis and quantitative assessment was performed on (a) oedema of the follicles; (b) combined area of increased vascularity and (c) number of inflammatory cells. There were significant differences in CPA-treated group compared with the normal control in all parameters measured (*p < 0.001). In addition, T3 treatment led to reduction of oedema, vascularity and inflammatory cell infiltration (#p < 0.05). Oedema, vascularity and inflammatory cell infiltration of the normal saline, T3, corn oil and CPA + T3 groups are comparable (p > 0.05). CPA: cyclophosphamide; T3: tocotrienol.
Co-administration of T3 with CPA was found to restore the ovarian histology to near normal (Figure 4(f)). This includes the follicles, corpus luteum and the medullary cells. As for the other control groups (T3 only and corn oil), they were of similar histology to normal control group (Figure 4(a)).
Discussion
Effect of CPA on the ovary
In this study, we found reduction in ovarian size, number of follicles and ovulation rate following long-term, low-dose CPA treatment. This is in agreement with previous reports on animal 14,15 and human studies 16 utilizing higher dose and shorter duration of CPA. Meirow et al. 5 reported depletion of PMFs with administration of a single dose of 20 mg/kg CPA; in addition, 50% reduction in PMF was seen with 75 mg/kg CPA treatment. They also reported significant reduction in pregnancy in animals treated with CPA 1 week before mating but not if mating occurred after that time point. This indicated that mature follicles are more affected by CPA. 5 In this experiment, we induced ovulation directly after CPA therapy, and therefore, the reduction in ovulation rate may be due to a similar reason as above, that is, the affected mature follicles fail to ovulate.
We observed that mature follicles were more significantly compromised compared to primary and PMF in chronic low-dose CPA treatment. The sign of folliculogenesis impairment was in the form of vacuolations in primary and secondary follicles and cystic changes in tertiary and Graafian follicles. This made these mature follicles appear larger than healthy follicles. In addition, the follicular antrum was sometimes absent or decreased in diameter due to the presence of degenerative granulosa cells and inflammatory cells within these unhealthy follicles. The infiltration of inflammatory cells leads to subsequent necrosis and produces large antrum in long-term CPA treatment. Co-treatment with T3 leads to reduction in this adverse effect, with lesser number of inflammatory cell infiltrations and better preservation of follicle morphology observed. Indeed, many studies had proved that T3 had anti-inflammatory effect. 17
There was also significant reduction in the number of corpus luteum following CPA exposure, reflecting the decrease in ovulating follicles in this group. Another study 4 also reported increase in medium- and large-sized follicles in addition to increased inflammatory cell infiltration in chronic, low-dose CPA treatment. However, this study did not further describe the follicular morphology that leads to the increase in follicle size. Our study provided further evidence on the deleterious effects of long-term CPA treatment on quality and quantity of folliculogenesis.
Effect of co-administration of T3 with CPA on the ovary
We found that co-administration of T3 with CPA significantly improved ovarian morphology and function to near normal, as evidenced by preservation of follicular size and ovulation rate to near-normal values. This indicates that T3 confers ovarian protection in chemotherapy. Published work on the effect of T3 administration with CPA on ovarian morphology and function is scarce. At the moment, ovarian function preservation was achieved with suppression of ovulation during CPA treatment by administration of combined oestrogen and progesterone preparation or a GnRH antagonist. 4,7
CPA caused ovarian toxicity by oxidative damage on the cells as evident from significant increase in malondialdehyde level and decrease in glutathione and superoxide dismutase levels found with its administration. This leads to increase in granulosa cell apoptosis, resulting in degeneration and depletion of ovarian follicles. Vitamin E plays an important physiological role as a free radical scavenger in the body. The presence of unsaturated isoprenoid side chain in T3 results in its increased fluidity and mobility between the membranes, 18 thus leading to an increase in the efficiency of T3 interaction with lipid radicals compared with tocopherols. 19 In addition, T3 is incorporated and transferred in between the cell membrane at a rapid rate 20 due to shorter side chain of its chromanols. 8 The protective effect of T3 on oxidative stress has also been reported in carbon tetrachloride-induced liver damage. 21 It was reported that T3 reduces the serum level of hepatic enzymes, a marker of hepatic cell injury due to the oxidative damage caused by carbon tetrachloride.
Limitations of the study
The limitations of this study are the lack of data on serum oxidative stress parameters and hormone levels. The data on oxidative markers would provide mechanistic explanation of the CPA-induced cell damage and changes seen with T3 supplementation. Measurement of hormone levels, specifically serum oestrogen and progesterone, would allow further correlation with other parameters on the effects of CPA and T3.
Conclusion
T3 confers protection in follicle number, morphology and function of the ovary when administered with CPA in vivo. T3 is a potential chemoprotective substance for the preservation of ovarian fertility following chemotherapy. Further investigation is needed to elucidate the mechanism of action of T3 when given in combination with CPA.
Footnotes
Acknowledgement
The authors thank Dr A Gapor from Malaysian Palm Oil Board for providing tocotrienol used in this study.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by Universiti Teknologi MARA (UiTM) through the UiTM Excellence fund (grant number: 600-RMI/ST/DANA 5/3/DST (131/2008)).
