Abstract
Accumulation of reactive oxygen species, such as hydrogen peroxide (H2O2), generated by inflammatory cells or other pathological conditions, leads to oxidative stress, which may contribute to the neuronal degeneration observed in a wide variety of neurodegenerative disorders such as Alzheimer’s disease. Recent investigations have described effective properties of tropisetron, such as antiphlogistic action or protection against β-amyloid induced-neuroinflammation in rats. Our data revealed that H2O2-induced cell death in rat pheochromocytoma cell line (PC12) can be inhibited by tropisetron, as defined by 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide assay, caspase 3 and caspase 12 levels. We further showed that tropisetron exerts its protective effects by upregulation of heme oxygenase-1, glutathione, catalase activity, and nuclear factor-erythroid 2 p45-related factor 2 level. Moreover, tropisetron was recently found to be a partial agonist of α7 nicotinic acetylcholine receptor (α7nAChR). The activation of α7nAChR could inhibit inflammatory and apoptotic signaling pathways in the oxidative stress conditions. In this study, selective α7nAChR antagonists (methyllycaconitine) reversed the effects of tropisetron on caspase 3 level. Our findings indicated that tropisetron can protect PC12 cells against H2O2-induced neurotoxicity through α7nAChR in vitro.
Introduction
5-Hydroxytryptamine 3 (5-HT3) receptor is the ligand-gated ion channel that increases intracellular cations such as calcium (Ca2+), sodium (Na+), and potassium (K+). Stimulation of this receptor can induce rapid and transient depolarization and excitation of neurons. The 5-HT3 receptor antagonist directly decreases depolarization and elevation of [Ca2+]i. 1 Recently, some studies have reported neuroprotective properties for 5-HT3 receptor antagonists both in vitro and in vivo, when administered in amyloid β (Aβ)-challenged cortical neurons of rats. 5-HT3 receptor antagonists substantially abated apoptosis, elevation of cytosolic Ca2+, glutamate release, reactive oxygen species (ROS) generation, and caspase 3 activation. These observed neuroprotective properties were all mediated through 5-HT3 receptor antagonism since they were completely blocked by concurrent treatment with a 5-HT3 receptor agonist. 1
In vivo studies have shown that 5-HT3 receptor antagonists, alongside their antiemetic effects possess notable immunomodulatory properties in the central and peripheral nervous system. 2 –6 In a previous study, we found that pretreatment with tropisetron significantly improved neurological deficits and diminished leukocyte transmigration into the brain, tumor necrosis factor α (TNF-α) level, and brain infarction in a murine model of embolic stroke. 7 Our recent investigation have revealed that tropisetron protects against Aβ-induced neurotoxicity through both 5-HT3 receptor-dependent and independent pathways in vivo. Seven days following Aβ inoculation, tropisetron significantly diminished the elevated levels of TNF-α, cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), nuclear factor kappa B (NF-κB), active caspase 3, cytochrome c release, and calcineurin phosphatase activity in the hippocampus. 8 In addition, in vitro studies have suggested that tropisetron can be an efficacious inhibitor of the signaling pathway leading to the activation of pro-inflammatory NF-κB, 9 a transcription factor that is pivotal for the upregulation of several neuroinflammatory mediators in Alzheimer’s disease (AD)-affected brain. 10
The aim of this study was to investigate the effects of tropisetron on hydrogen peroxide (H2O2)-induced apoptotic cell death in neuron-like rat pheochromocytoma cell line (PC12). Furthermore, we aimed to explore whether tropisetron could affect the anti-inflammatory NF-E2-related factor (Nrf2)/heme oxygenase-1 (HO-1) signaling pathway since activation of this pathway leads to the synthesis of a distinct set of antioxidant proteins that can efficiently protect cells from oxidative stress. 11
Moreover, as tropisetron was recently found to be a partial agonist of α7 nicotinic acetylcholine receptor (α7nAChR), the activation of which is shown to inhibit inflammatory and apoptotic signaling pathways in oxidative stress conditions, 12 we also investigated the involvements of α7nAChR in neuroprotective potential of tropisetron.
Materials and methods
Antibodies directed against caspase 3 and β-actin were obtained from Cell Signaling Technology (Beverly, Massachusetts, USA). Nrf2 antibody was purchased from Santa Cruz Biotechnology (Dallas, Texas, USA). HO-1 antibody was obtained from ABCAM (Cambridge, UK). H2O2 was obtained from Merck (Darmstadt, Germany). Electrochemiluminescence (ECL) kit was obtained from Amersham Bioscience (Piscataway, New Jersey, USA). Polyvinylidene fluoride (PVDF) was from Millipore (Billerica, Massachusetts, USA). Tropisetron and methyllycaconitine citrate (MLA) were purchased from Tocris Bioscience (Bristol, BS11 0QL, UK). All the other reagents, unless otherwise stated, were from Sigma Aldrich (St Louis, Missouri, USA).
Cell culture and treatment conditions
The PC12 cells used in this experiment were provided by Pasteur Institute (Tehran, Iran). Cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% horse serum, 5% fetal bovine serum, and 1% penicillin/streptomycin and were maintained at 37°C in a humidified atmosphere of 5% carbon dioxide in air atmosphere. Medium was changed three times a week. The cells were differentiated using nerve growth factor (NGF; 50 ng/ml) every other day for six days.
Treatment conditions
All PC12 cells were treated with escalating concentrations (10 nM, 100 nM, and 1 μM) of tropisetron in the presence or absence of MLA (10 nM), an α7nAChR antagonist, for 6 h, followed by 4 h exposure to H2O2 (250 μM).
Assessments of cell death and apoptosis
Conventional 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide (MTT) reduction assay was used to test the cell viability. The absorbance of the formed dark blue formazan crystals when intact cells were solubilized in dimethyl sulfoxide was measured at 630 nm. The percentages of reduced MTT were reported, assuming the absorbance of control cells as 100%.
Western blot analysis
The cells were lysed in buffer containing complete protease inhibitor cocktail. Then, 20 μg of total protein of each sample were electrophoresed on 12.5% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and then transferred to PVDF membrane. Next, membrane was blocked using nonfat dry milk–Trsi-buffered saline Tween 20 solution (2%) and probed with specific primary antibodies overnight. Immunoreactive polypeptides were detected by chemiluminescence using ECL reagents. Quantification of results was performed by scan of films and densitometric analysis was carried out by ImageJ software (ver. 1.41o, NIH, USA; http://rsb.info.nih.gov/ij/).
Measurement of GSH levels
The principle of the method to measure glutathione (GSH) levels was first described by Ellman 13 and depends on the reduction of GSH. Whole cell lysate was added to Ellman’s reagent containing 19.8 mg of 5,5'-dithiobis-(2-nitrobenzoic acid in 100 ml of 0.1% sodium nitrate and phosphate buffer (0.2 M pH 8.0). The absorbance was measured at 412 nm.
CAT activity assay
Catalase (CAT) activity was measured spectrophotometrically at 240 nm using H2O2 (0.01 M) as substrate, which was added to the cuvette at the end of the process. 14
Statistical analysis
Data are presented as the mean ± SEM. Each experiment was repeated three times, giving essentially identical results. Statistical analysis between groups was performed using one-way analysis of variance, followed by Tukey’s post hoc test (*p < 0.05 or # p < 0.05, **p < 0.01 or ## p < 0.01, and ***p < 0.001 or ### p < 0.001).
Results
Tropisetron-protected PC12 cells against H2O2-induced cell death
In the present investigation, we aimed to explore the neuroprotective impact of tropisetron against H2O2-induced apoptosis in the differentiated PC12 cells. The MTT assay was conducted to monitor cytotoxicity of different concentrations of tropisetron. H2O2 (250 µM) significantly decreased viability of PC12 cells (64.2% compared with the control cells). To evaluate the neuroprotective impact of tropisetron, differentiated PC12 cells were treated with escalating doses of tropisetron 6 h before exposure to H2O2 and were assayed for cell viability 4 h after H2O2 insult. Over the concentration range used, tropisetron alone did not exert any cytotoxic effect in PC12 cells. As illustrated in Figure 1, pretreatment of cells with 10 nM, 100 nM, and 1 μM of tropisetron enhanced survival of H2O2-challenged cells about 1.7, 2.2, and 2-fold, respectively, compared with the untreated H2O2-exposed cells.
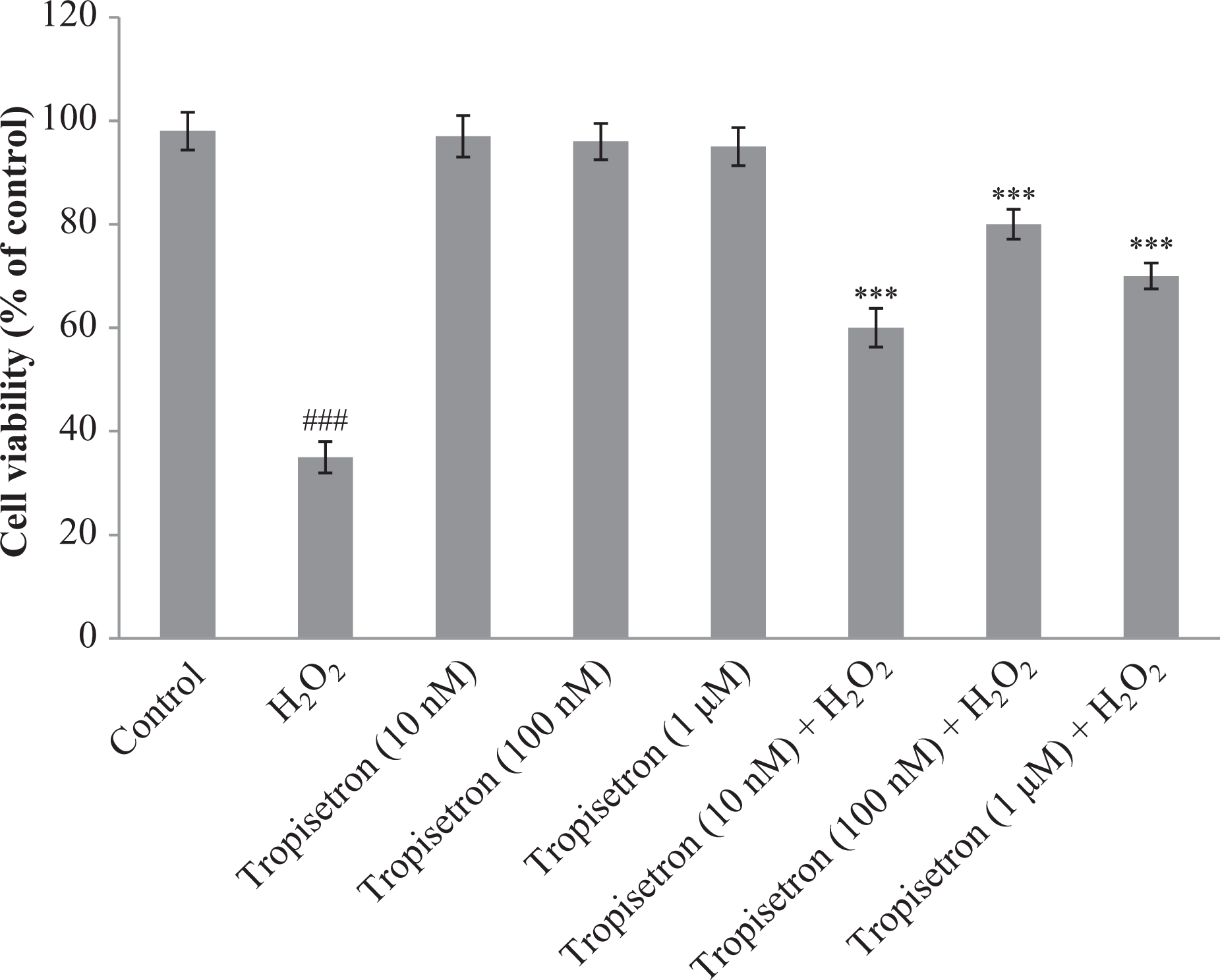
Neuroprotective effect of tropisetron against H2O2-induced cell death in PC12 cells. Differentiated PC12 cells were treated with different concentrations of tropisetron in the presence and/or absence of H2O2 (250 µM). Cell viability was determined by MTT assay and calculated as the percentage of living cells in treated cultures compared with those in control cultures. Each value represents the mean ± SEM (n = 3). ### p < 0.001: significantly different from control cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line; MTT: 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide.
Tropisetron decreased caspase 3 content of PC12 cells
Previous studies have identified that caspases are important mediators of apoptosis induced by various apoptotic stimuli. Caspase 3 is known as a downstream executioner in the caspase-dependent apoptotic cascade. 15 Therefore, we examined the impact of tropisetron treatment on caspase 3 activation. As we can see in Figure 2, the amount of cleaved (active) caspase 3 increased in the presence of H2O2 (2.8-fold); however, in cells pretreated with increasing concentration of tropisetron (10 nM, 100 nM, and 1 μM), cleaved caspase 3 protein level had diminished expression by 23.6%, 65.1%, and 63.7%, respectively, in comparison with H2O2-treated cells.
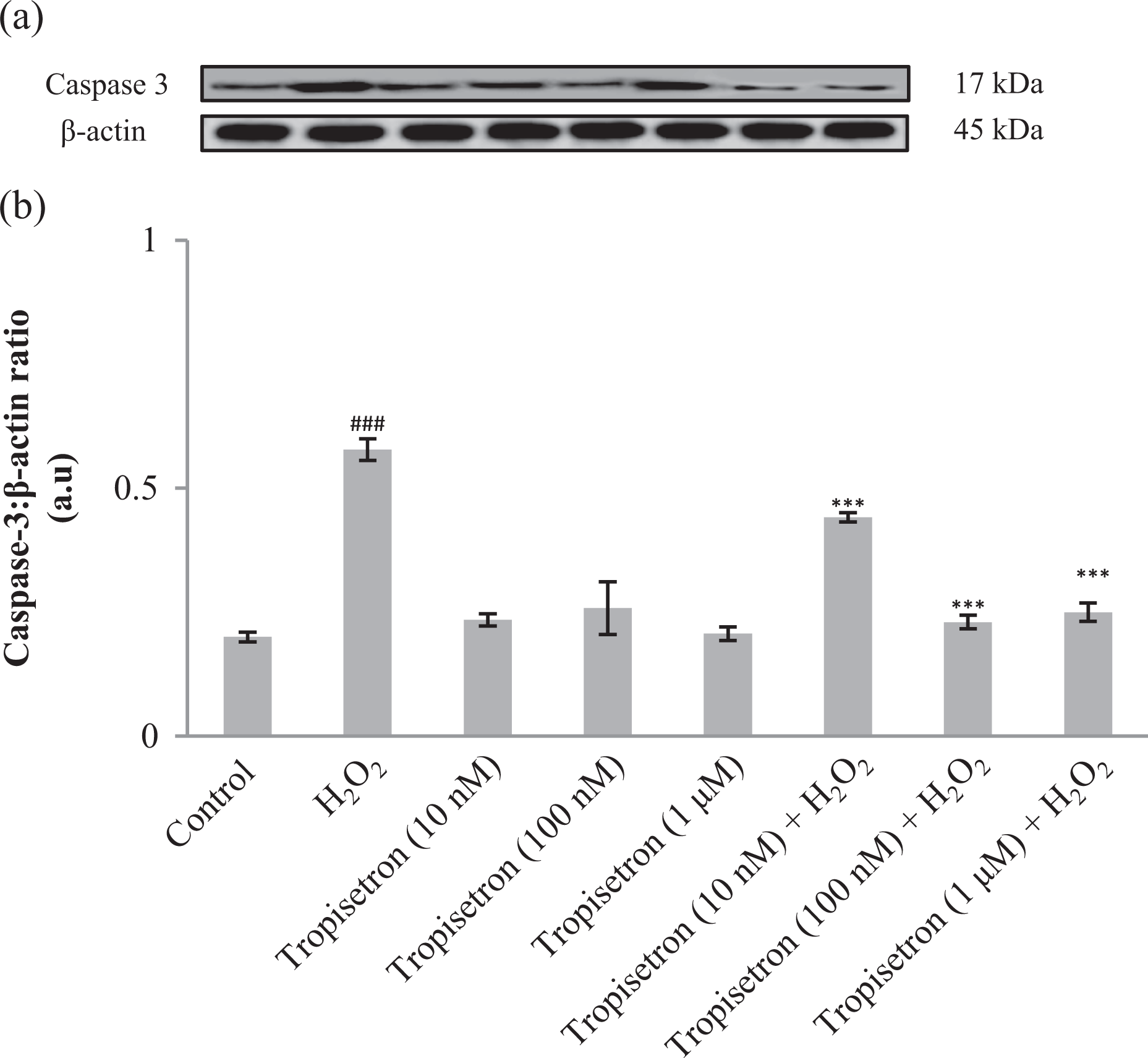
Caspase 3 levels in PC12 cells pretreated with tropisetron. PC12 cells were pretreated with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM) for 6 h and then exposed to H2O2 for 4 h. Cells were lysed with lysis buffer, and protein content was determined according to Bradford’s method. Twenty microgram proteins were separated on SDS-PAGE and subjected to anti-caspase 3 antibody (1:1000) and reprobed with anti-β-actin antibody (1:1000). One representative Western blot was shown (n = 3). The densities of corresponding bands were measured and the ratio was calculated. The median of three independent experiments is shown. ### p < 0.001: significantly different from untreated cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line; SDS-PAGE: sodium dodecyl sulfate–polyacrylamide gel electrophoresis.
Tropisetrin treatment increased Nrf2 and HO-1 levels
As depicted in Figure 3(a) and (b), a low level of Nrf2 was detected in H2O2-treated cells (decrease by 31.8% as compared to the control cells), while pretreatment with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM) 6 h prior to H2O2 exposure caused a significant increase in Nrf2 protein levels (about 1.1, 1.32, and 1.31-fold, respectively, compared to H2O2-treated cells). HO-1 levels also displayed a dramatic decrease (63.9%) in H2O2-exposed cells compared with the control cells. On the contrary, in cells incubated with 10 nM, 100 nM, and 1 µM of tropisetron, HO-1 levels rose about 1.3, 2.4, and 2.2-fold, respectively, compared to H2O2-treated cells receiving no prior treatment (Figure 3(c)).

Nrf2 and HO-1 levels in PC12 cells pretreated with tropisetron. PC12 cells were pretreated with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM) for 6 h and then exposed to H2O2 for 4 h. The protein level was determined with Western blotting (n = 3). ### p < 0.001: significantly different from untreated cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line; Nrf2: nuclear factor-E2-related factor; HO-1: heme oxygenase 1.
Tropisetron affects caspase 12 levels in PC12 cells
Caspase 12 level increased 2.8-fold in the presence of H2O2 as compared to the control; However, in cells pretreated with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM), a significant reduction was observed compared to H2O2-challenged cells (27.5%, 65.1%, and 63.3% respectively; Figure 4).

Caspase 12 levels in PC12 cells pretreated with tropisetron. PC12 cells were pretreated with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM) for 6 h and then exposed to H2O2 for 4 h (n = 3). ### p < 0.001: significantly different from untreated cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line.
Tropisetron augmented GSH level and CAT activity in PC12 cells
As shown in Table 1, H2O2 decreased GSH level about 62% compared with the control cells; however, in cells pretreated with different concentrations of tropisetron (10 nM, 100 nM, and 1 μM), GSH content was enhanced 2.32, 2.36, and 2.32-fold, respectively, compared to untreated H2O2-challenged cells.
Effects of tropisetron on GSH level and CAT activity in H2O2-treated PC12 cells.a
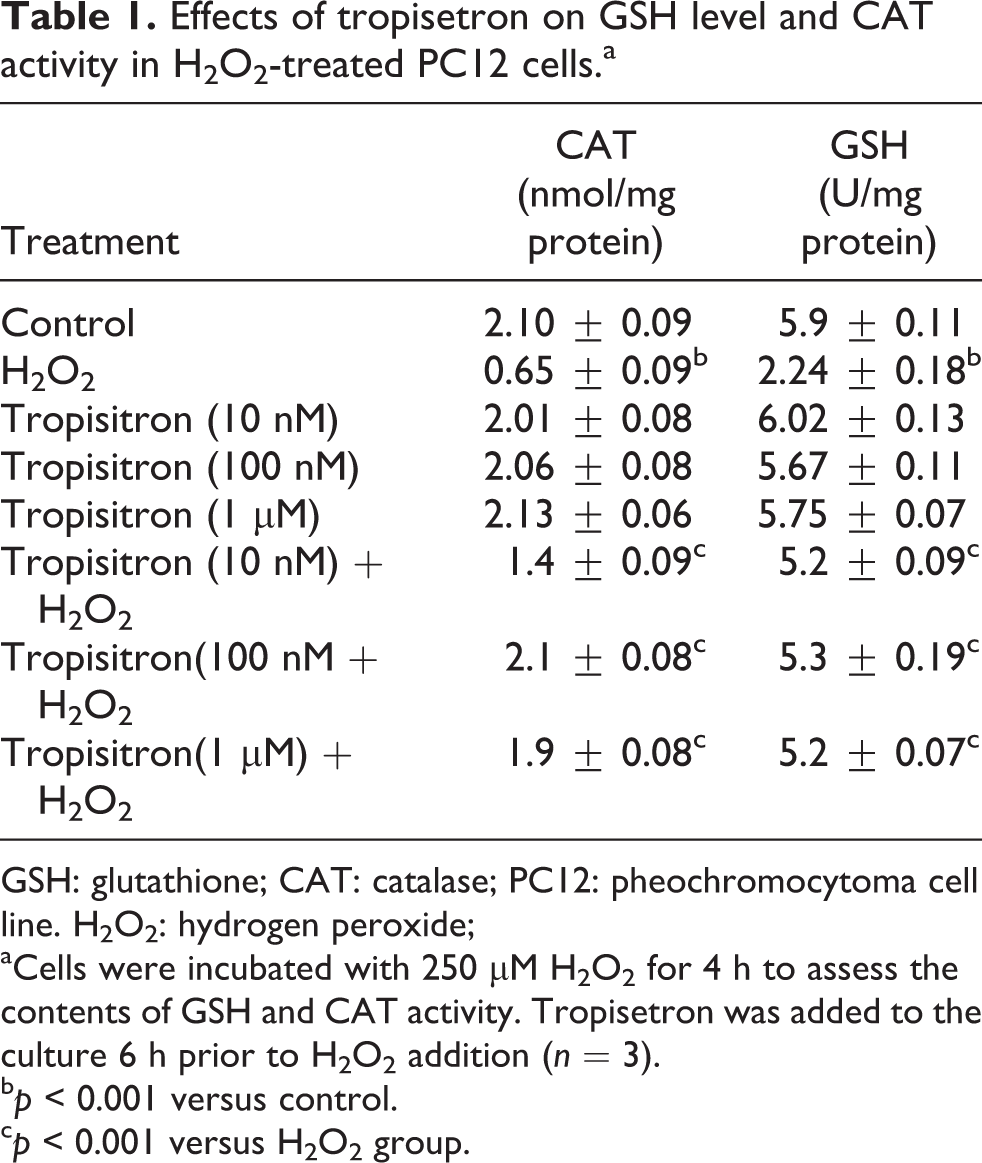
GSH: glutathione; CAT: catalase; PC12: pheochromocytoma cell line. H2O2: hydrogen peroxide;
aCells were incubated with 250 µM H2O2 for 4 h to assess the contents of GSH and CAT activity. Tropisetron was added to the culture 6 h prior to H2O2 addition (n = 3).
b p < 0.001 versus control.
c p < 0.001 versus H2O2 group.
H2O2 exposure decreased CAT activity by 69.04% in comparison with the control cells. Prior treatment with tropisetron (10 nM, 100 nM, and 1 μM) augmented CAT activity by 2.1, 3.2, and 2.9-fold, respectively, when compared with H2O2-insulted cells.
MLA offsets tropisetron effects on PC12 cell survival
In order to explore the possible involvement of α7nACh receptors in the neuroprotective action of tropisetron, a noneffective dose of MLA was defined and tested to investigate the interaction of tropisetron; for this purpose, cells were exposed to both tropisetron (100 nM) and MLA (10 nM) in the presence or absence of H2O2. As Figure 5 illustrates, MLA nullified the protective effect of tropisetron and showed 43% decrease compared to tropisetron (100 nM) and H2O2-treated group. This result proposed that MLA could abolish the protective effect of tropisetron on caspase-3 level.

Differentiated PC12 cells were treated with effective concentrations of tropisetron (100 nM) in the presence of H2O2 (250 µM) and noneffective concentration of MLA (10 nM). Cell viability was determined by MTT assay (n = 3). ### p < 0.001: significantly different from untreated cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line; MLA: methyllycaconitine citrate; MTT: 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide.
MLA altered antiapoptotic action of tropisetron
As depicted in Figure 6, no significant difference was observed in caspase 3 levels between H2O2-insulted cells and cells that were treated by H2O2, tropisetron, and MLA, indicating that α7nAChR blockade abrogated the antiapoptotic impact of tropisetron in this context.

Caspase 3 levels in PC12 cells pretreated with tropisetron (100 nM) and MLA (10 nM). PC12 cells were pretreated with effective concentrations of tropisetron (100 nM) and noneffective concentration of MLA (10 nM) for 6 h and then exposed to H2O2 for 4 h. Protein levels were determined by Western blot analysis as described in Materials and Methods section (n = 3). # significantly different from untreated cells. ### p < 0.001: significantly different from untreated cells; ***p < 0.001: significantly different from H2O2-treated cells. H2O2: hydrogen peroxide; PC12: pheochromocytoma cell line. H2O2: hydrogen peroxide; MLA: methyllycaconitine citrate.
Discussion
The PC12 cell line is a well-characterized model for investigating neuronal differentiation and neurobiological events. 16 –18 H2O2 has been widely used as an inducer of oxidative stress in in vitro models. 19 The exposure of cultured cell to H2O2 leads to an imbalance in the energy metabolism and production of hydroxyl and peroxyl radicals that have adverse effects on important macromolecules. 20 Our results demonstrated that cell death induced by H2O2 was greatly decreased in the presence of tropisetron, a 5-HT3 receptor antagonist. This data indicated that tropisetron can significantly protect PC12 cells from H2O2-induced cytotoxicity. Results of this investigation confirmed our very recent study indicating that tropisetron is a neuroprotective agent in rat model of AD (Both Aβ and H2O2 increase oxidative stress approximately in the same ways. It has been shown that Aβ can cause oxidative stress via H2O2 as an intracellular messenger). Tropisetron attenuated Aβ-induced hippocampal neuroinflammation as verified by reduction in TNF-α, COX-2, inducible nitric oxide synthase as well as nuclear translocation of p65, NF-κB level, blunted hippocampal apoptotic markers, active caspase-3 levels, and cytochrome c release. 8
Many signals and metabolic events are important in the regulation of cell death, whereas the intracellular redox level, in particular, has been shown to play a critical role. Redox regulation is an interesting and important issue, which is tightly associated with oxidative stress. 21 HO-1, also known as heat shock protein 32 (Hsp-32), is a novel enzyme with potent anti-inflammatory, antioxidant, and antiproliferative effects. 22 –24 HO-1 is the rate-limiting enzyme in the conversion of heme into biliverdin, releasing free iron and carbon monoxide. Biliverdin is rapidly metabolized to bilirubin, which is a potent antioxidant. Activity of this factor is probably involved in the protection against oxidative stress. 25 HO-1 belongs to a family of cytoprotective and detoxification genes that possess antioxidant responsive elements (AREs) in their regulatory regions. 26 The Nrf family of transcription factors mediates adaptive responses to stress via binding to AREs. Previous studies have suggested that Nrf2 plays a key role in antioxidant-induced HO-1 expression. 27 Here we hypothesized that tropisetron could induce Nrf2 nuclear translocation and increase HO-1 expression. In addition, it has been shown that tropisetron can increase γ-glutamylcysteine synthetase level (the rate limiting step in GSH biosynthesis), one of the other Nrf2 target genes, suggesting that Nrf2 may play a pivotal role in tropisetron-induced cytoprotective effects in PC12 cells. 11,28
Effects of tropisetron on HO-1 level were paralleled with the recent interesting study of Liu et al. reporting that tropisetron administration after trauma–hemorrhage decreased pro-inflammatory mediator levels and likely attenuated liver injury through p38 mitogen activated kinase (MAPK) through upregulation of HO-1. 29
Apoptosis has been consistently linked to H2O2-induced neuronal damage in PC12 cell line. 30 At the molecular level, apoptosis occurs by activation of caspase cascade. Caspase 3 is the final effector responsible for the characteristic morphology of apoptosis. 31,32 Administration of H2O2 in PC12 cells caused a sharp rise in caspase 3 level, accompanied by increased level of cleaved caspase 12, demonstrating the involvement of endoplasmic reticulum (ER) apoptotic pathway. 33 Caspase 12 has been localized on the cytoplasmic side of the ER using immunofluorescence and subcellular fractionation techniques. 34,35 The observation in which caspase 12 activation occurs during ERs stress-induced apoptosis has favored the idea that caspase 12 could be the initiator caspase in ER stress-mediated apoptosis. 33,36 In this study, we showed that tropisetron can target and attenuate ER-mediated apoptosis and this effect was nullified by coadministration of MLA (selective α7 nicotinic receptor antagonist). 37 Effects of tropisetron on caspase 3 level is in parallel with our recent investigation indicating that tropisetron potently decreased hippocampal caspase 3 contents following Aβ administration. 8
The cholinergic vagal pathway appears to play an important role in modulating inflammatory responses, as evidenced by observing the increased lipopolysaccharides-induced TNF-α serum levels and hepatic TNF-α responses after vagotomy. On the other hand, electrical stimulation of the vagus nerve or treatment of vagotomized animals with acetylcholine (Ach) prevents TNF-α release. 38 The primary cholinergic target for these effects appears to be the α7nAChR. The key role played by α7nAChR in inflammatory processes is supported by reports that the anti-inflammatory effects of nicotine on macrophages can be counteracted by selective α7nAChR antagonists such MLA. 39
Now it is well established that activation of α7nAChR results in augmentation of intracellular anti-inflammatory and survival signaling pathways. In important neurodegenerative diseases, such as AD, Aβ disrupts α7nAChR antioxidative stress signaling culminating in neuronal apoptosis. Indeed the Aβ-α7nAChR interaction activates the apoptotic enzyme caspase 3 and causes cleavage of the DNA repairing enzyme poly-(adenosine diphosphate ribose) polymerase resulting in neurotoxicity and cell death. On the other hand, nicotine inhibits membrane degradation and DNA fragmentation through α7nAChR. 40,41 Different mechanisms could take part in α7nAChR mediated antiapoptotic and antiphlogistic aspects that one of which is inducing HO-1 by Nrf2. 42,43 Intriguingly, tropisetron can act as a partial agonist of α7nAChR, 44 which is associated with neuroprotective properties and inflammatory inhibition responses. 45
Results of this study indicated that tropisetron can attenuate H2O2-induced neurotoxicity in PC12 cell lines by decreasing the contents of apoptotic proteins (caspase 3 and caspase 12) and increasing the levels of anti-inflammatory transcriptional factors like Nrf2 and HO-1. The most important finding of our research is that α7nAChR mediated some neuroroprotective actions of tropisetron in PC12 cells as MLA reversed it. It should be considered that PC12 cells also expressed 5-HT3 receptors and involvement of these receptors in neuroprotective aspects of tropisetron should not be excluded. 18 In support of this notion, a very recent study has shown that tropisetron suppresses collagen synthesis induced by transforming growth factor β1 (TGFβ1). This effect was independent of 5-HT3 receptor and was mediated by α7nAChR. 46
For the first time in an intriguing study, Swartz et al. showed that tropisetron could elicit prominent neuroprotective actions against glutamate-induced neurotoxicity in retinal ganglion cells (RGCs). Glutamate (500 µM) evoked excitotoxicity in RGCs and decreased RGC survival by an average of 62% compared with control conditions, while RGCs pretreated with tropisetron (100 nM) before glutamate survived to an average of 105% compared with the controls.
47
Inhibition studies using the α7nAChR antagonist, MLA (10 nM), supported the hypothesis that neuroprotective aspects of tropisetron against glutamate-induced excitotoxicity is mediated by α7nAChR activation. The most important finding of Swartz et al. is that internalization of N-methyl-
It is noteworthy that the activation of other pivotal inflammatory signaling pathways such as p38 MAPK can be affected by tropisetron. Stratz et al. have shown that in primary human monocytes, tropisetron prevented the phosphorylation and thus activation of the p38 MAPK, which is involved in posttranscriptional regulation of different cytokines such as TNF-α, thereby exerting anti-inflammatory properties. 48
The 5-HT3 receptor is a pentamer consisting of five monomers that form a centrally permeable cylindrical body which can be readily penetrated by small cations. The 5-HT3A–E subunits are different in the amino terminal and the transmembrane region. 49 Functional homomeric 5-HT3A receptors and heteromeric 5-HT3A/B receptors are expressed neuronally, which show a 40-fold difference between the single-channel conductances for agonist action, possibly due to structural differences at the interfaces between the extracellular domains of the subunits. Homomeric and heteromeric 5-HT3 receptors show clearly differing biophysical and pharmacological properties. 49 Coexpression of the 5-HT3B subunit alters receptor desensitization and deactivation and reduces the sensitivity to serotonin. 49 The diversity of homomeric and heteromeric subtypes of 5-HT3 receptors and their different expression patterns in tissues can lead to various and unpredictable responses elicited by 5-HT3 receptor agonists.
In conclusion, our investigation revealed that activation of Nrf2 signaling by tropisetron may protect neuron-like PC12 cells against apoptosis. These effects were completely or partially mediated by α7nAChR. Various mechanisms might contribute to the beneficial effects observed here with tropisetron: (i) Decrease in intracellular Ca+2 as a result of 5-HT3 ion channel blockade. This aspect was previously reported in primary cortical neural culture. (ii) Inhibition of calcineurin phosphatase activity, which was shown both in vivo and in vitro. 8 (3) Tropisetron acts as a partial agonist of α7nAChR, 44 which is associated with neuroprotective properties and inhibition of inflammatory responses. 45 (4) In its structure, tropisetron bears indole, an electron-rich aromatic ring and thus serves as an electron donor, thereby reducing and quenching electrophilic radicals, 3 and this property enables tropisetron to combat oxidative stress in an inflammatory context. The findings of this study altogether with low incidence of adverse effects reported for tropisetron in clinical researches suggest that tropisetron represents a therapeutic potential to halt the neuroinflammatory responses in neurodegenerative diseases.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by funds from the Tehran University of Medical Sciences.
