Abstract
Objective:
Self-poisoning with (4-chloro-2-methylphenoxy) acetic acid (MCPA) is a common reason for presentation to hospitals, especially in some Asian countries. We encountered a case of a 76-year-old woman who experienced unconsciousness, shock and respiratory failure after ingesting 100 mL MCPA herbicide. We determined whether the surfactant in the formulation was the chemical responsible for the toxic symptom in this patient.
Design:
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell viability and lactate dehydrogenase (LDH) cytotoxicity assays were performed on human brain neuroblastoma SK-N-SH cells. The expressions of 84 genes in 9 categories that are implicated in cellular damage pathways were quantified using an RT2 Profiler™ PCR array on a human neuronal cell line challenged with polyoxyethylene tridecyl ether (PTE).
Setting:
Pesticide intoxication institute in university hospital.
Interventions:
Extracorporeal elimination with intravenous lipid emulsion.
Measurements:
Cell viability and gene expression.
Main Results:
In the MTT assay, MCPA only minimally decreased cell viability even at concentrations as high as 1 mM. Cells treated with 1-methoxy-2-propanol, dimethylamine and polypropylene glycol exhibited minimal decreases in viability, whilst the viability of cells challenged with PTE decreased dramatically; only 15.5% of cells survived after exposure to 1 µM PTE. Similarly, the results of the LDH cytotoxicity assay showed that MCPA had very low cytotoxicity, whilst cells treated with PTE showed incomparably higher LDH levels (p < 0.0001). PTE up-regulated the expressions of genes implicated in various cell damage pathways, particularly genes involved in the inflammatory pathway.
Conclusions:
The surfactant PTE was likely the chemical responsible for the toxic symptom in our patient.
Keywords
Introduction
(4-Chloro-2-methylphenoxy) acetic acid (MCPA), a chlorophenoxy herbicide, is a synthetic auxin that acts like indole-3-acetic acid, a naturally occurring plant hormone. 1 MCPA is one of the most commonly used herbicides worldwide, and acute self-poisoning with MCPA is a common reason for presentation to hospitals, especially in some Asian countries.2,3 In Korea, the manufacturing output of MCPA was 4.3 tons of active ingredient in 2013; Annual report of Korean Crop Protection Association, Seoul Korea.
In mammals, MCPA generally has low toxicity and is rapidly excreted. The oral median lethal dose (LD50) in rats is 765 mg kg−1 (Environmental Protection Agency, Registration Eligibility Decision for MCPA September 2004, http://www.epa.gov/oppsrrd1/REDs/mcpa_red.pdf).
However, the clinical features after oral exposure to a large amount of undiluted MCPA herbicide include gastrointestinal symptoms (i.e. vomiting and abdominal pain), central nervous system symptoms (i.e. confusion, coma, convulsion, ataxia and fasciculation), cardiovascular symptoms (i.e. hypotension and arrhythmia), respiratory failure, renal failure and electrolyte imbalance. Approximately 5% of patients die, typically 24–48 h post-ingestion. 3
There have been several studies on MCPA, including its effect on the blood–brain barrier,4,5 toxicokinetics, 6 and a clinical trial to determine the association between plasma levels of MPCA at a given time and clinical outcome. 7 However, the results do not explain the precise mechanism underlying MPCA herbicide toxicity, and doubt has been cast on the role of additives in various herbicide formulations.
Pesticide formulations contain both active and inert ingredients, that is, active ingredients kill the pest and inert ingredients help the active ingredients work more effectively. However, the safety of inert ingredients may not be tested as thoroughly as active ingredients. Furthermore, they are seldom disclosed on product labels. Accordingly, rather than active ingredients, physicians must be aware of inert ingredients when treating patients with acute pesticide intoxication.8,9
We recently encountered a 76-year-old female patient who ingested 100 mL undiluted MCPA stock solution. She experienced unconsciousness, shock and respiratory failure after ingesting the herbicide. We found that the formulation included various additives including surfactant, solvent and antifreeze. To investigate the implications of these additives in the patient’s toxic symptoms, we scrutinized her clinical features and performed in vitro experiments analyzing the cytotoxicity of each chemical in the herbicide.
Materials and methods
Case presentation
A 76-year-old female patient presented to the emergency room of our hospital 3 h after ingesting 100 mL undiluted MCPA herbicide. On arrival, respiratory arrest developed with semi-comatose consciousness. Blood pressure was 90/60 mm Hg. Immediate first-aid treatment was applied, including tracheal intubation and mechanical ventilation. Urine output fell below 10 mL h−1, and metabolic acidosis developed. Continuous renal replacement therapy (CRRT) was initiated with the aid of norepinephrine. In addition, 500 mL 20% lipid emulsion product was injected intravenously over 2 h, followed by a maintenance dose of 1000 mL over the next 24 h.
On the third hospital day, the patient’s blood pressure was 120/80 mm Hg without inotropic agent and self-respiration had recovered. Tracheal extubation was also performed on the third hospital day, and consciousness returned to near normal by the fourth hospital day. Laboratory findings including complete blood count, blood chemistry, and arterial blood gas analysis are summarized in Table 1 and Figure 1.
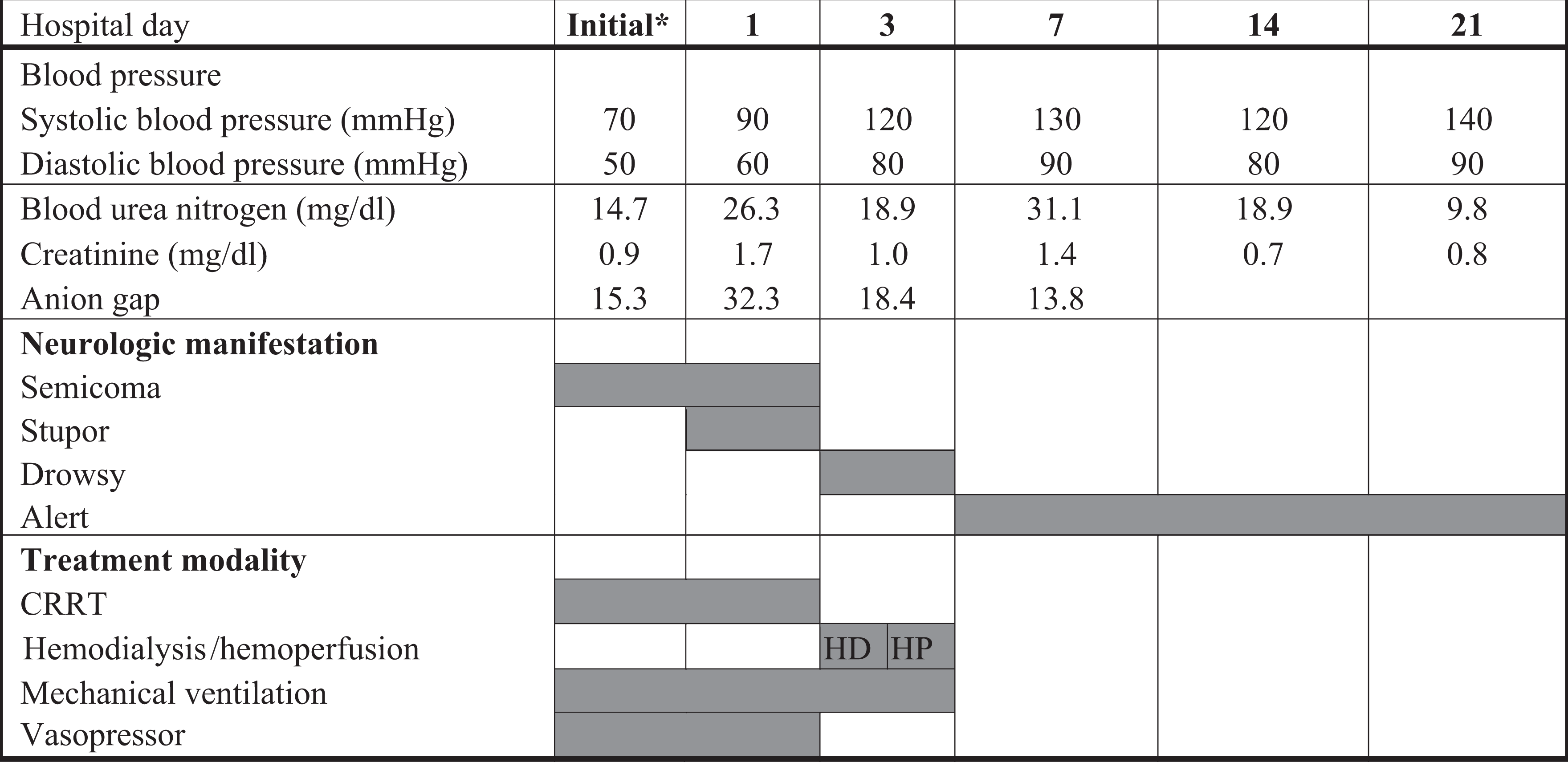
Toxic symptoms and treatment modalities of the patient HP and HD were carried out through a jugular venous catheter for 4–5 h at a blood flow rate of 250–300 mL min−1. The HP membrane was an Adsorba 300 C, Gambro (Gambro Dialysatoren GmbH Co., KG Hechingen, Germany) with polypropylene housing material, activated charcoal adsorbent, a 300-m2 surface area and cellulose coating material for the adsorbent. The HD dialyzer was a polyflux 170H dialyzer (Polyamix membrane; Gambro Dialysatoren). HD was initially performed and then changed to CVVHDF due to unstable vital signs. CVVHDF using the Gambro Prismaflex® system was performed on the third and fourth hospital day (48 h). The CVVHDF membrane was an AN69 membrane (acrylonitrile and sodium methyl sulphonate copolymer; Gambro Dialysatoren). The total removed plasma volume by CVVHDF was 12,784 mL over 48 h. On the fourth hospital day, the patient’s vital signs became stable, and CVVHDF was changed to HD, which was maintained for 2 weeks to treat the acute renal failure. The patient recuperated from the shock state within 7 hospital days. *Initial: first day in the ER. HD: haemodialysis; HP: haemoperfusion; CVVHDF: continuous venovenous haemodiafiltration; ER: emergency room.
Sequential measurements of blood chemistry, complete blood cell counts and arterial blood gases analysis.
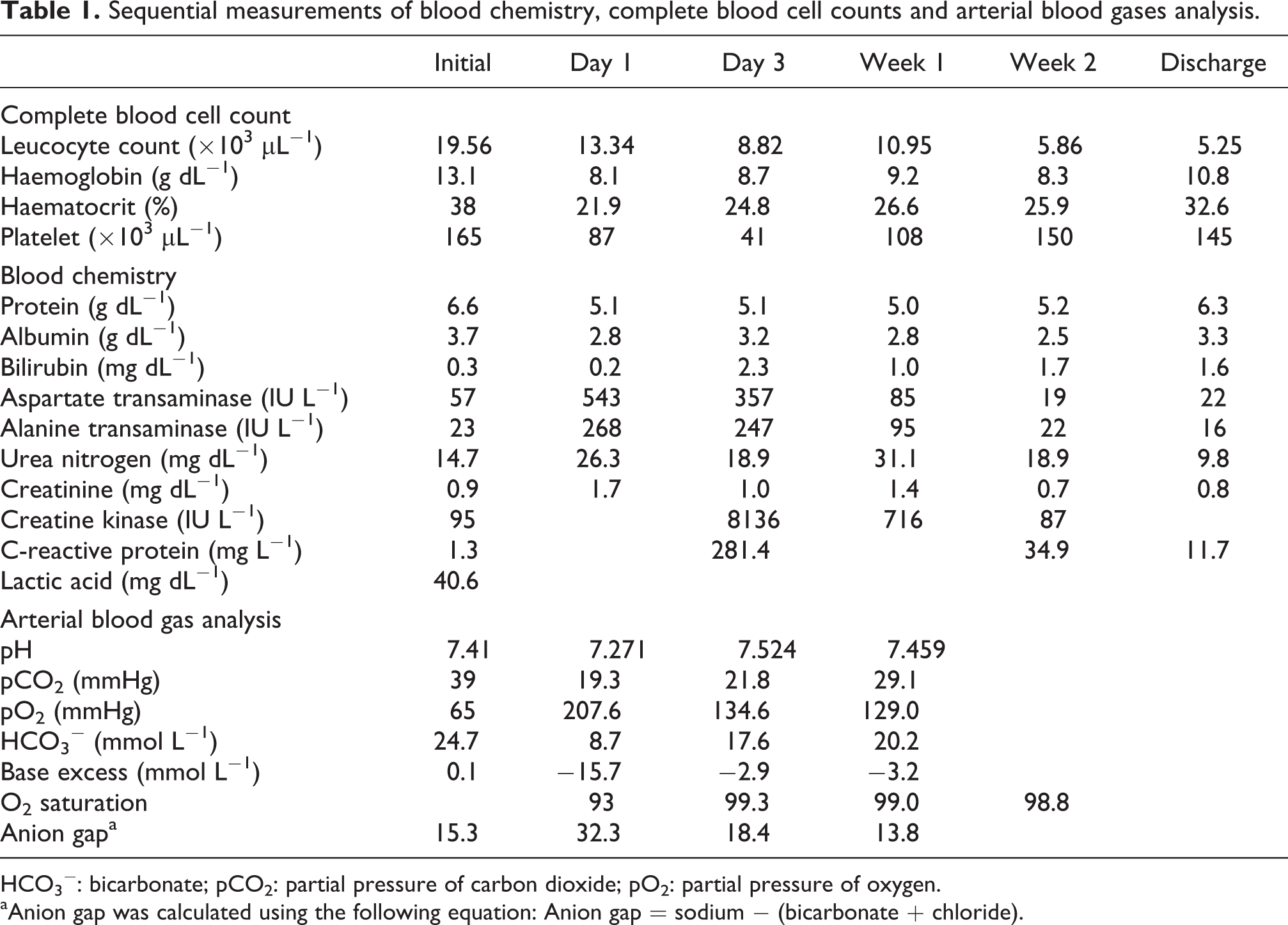
HCO3 −: bicarbonate; pCO2: partial pressure of carbon dioxide; pO2: partial pressure of oxygen.
aAnion gap was calculated using the following equation: Anion gap = sodium − (bicarbonate + chloride).
In vitro cell viability and cytotoxicity of active and inert ingredients
Chemicals
MCPA was purchased from Santa Cruz Biotechnology Inc. (Dallas, Texas, USA). The chemicals that were purchased from Sigma-Aldrich (St Louis, Missouri, USA) were dimethylamine, polyoxyethylene tridecyl ether (PTE), polypropylene glycol (PPG), and 1-methoxy-2-propanol (Table 2). The chemicals that were purchased for the cell viability assay were 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide reagent (MTT, Sigma-Aldrich), culture medium (Roswell Park Memorial Institute medium (RPMI)-1640; HyClone, Logan, Utah, USA), penicillin–streptomycin (Life Technologies, Paisley, UK) and dimethyl sulfoxide (DMSO; Sigma-Aldrich).
Formulation of the MCPA herbicide.

MCPA: (4-chloro-2-methylphenoxy) acetic acid; PTE: polyoxyethylene tridecyl ether; PPG: polypropylene glycol; CAS: chemical abstracts service.
aInsert rate is usually presented as the ‘weight%’ or ‘weight by volume’ of additives in a formulation and is not disclosed by the manufacturer. Therefore, we are only able to present estimated values.
Cell culture
Human brain neuroblastoma SK-N-SH cells were purchased from the Korean Cell Line Bank (Seoul, South Korea). Cells were maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum and 1% penicillin–streptomycin antibiotic. Cells were expanded into a humidified carbon dioxide (CO2) incubator (5%) at 37°C using T-75 flasks after trypsinization and subsequent washing with phosphate-buffered saline.
MTT cell viability assay
The volume of vehicle for the test chemicals in the plates was always ≤1.0% of the medium to minimize vehicle effect on the cells. Test chemicals were subsequently exposed to media with different concentration gradients from 0.01 µM to 1 mM. A chemosensitive and colorimetric MTT assay was used to measure cell viability and performed as described by Mickisch et al. 10
LDH cytotoxicity assay
Lactate dehydrogenase (LDH) is a stable cytoplasm enzyme present in all cells; it is rapidly released into the cell culture supernatant if cells are lysed or experience membrane damage. Colorimetric LDH release was measured at 490 nm using an enzyme-linked immunoassay reader (Roche Inc., Pleasanton, California, USA).
Gene expression study for stress- and toxicity-related pathways
PTE was the only subject for further gene expression experiments. To investigate the diverse pathways of cytotoxicity at the gene level, the expressions of 84 genes responsible for human stress and toxicity pathways were quantified. They included the following nine categories of cell damage pathways such as oxidative/metabolic stress, hypoxia, apoptosis signalling, autophagy signalling, necrosis signalling, inflammatory response, cell cycle checkpoint/arrest, DNA damage signalling (other responses) and heat shock proteins/unfolded protein response.
Total RNA was isolated from SK-N-SH cells treated with 0.01 μM PTE for 24 h or untreated cells using the RNeasy mini kit (Qiagen, Valencia, California, USA). Complementary DNA was synthesized using the Maxime RT premix kit (Intron, Seongnam, Korea) according to the manufacturer’s instructions. Gene expression was quantified using the RT 2 Profiler™ PCR Array (Cat. no. PAHS-003ZD-12; SABiosciences Corporation, Frederick, Maryland, USA). SYBR Green PCR Master Mix (Applied Biosystems, Grand Island, New York, USA) was added, and the arrays were run on a CFX96 Real-Time system (Bio-Rad, Hercules, California, USA) with cycling programmes recommended by the manufacturer. Gene expression levels were determined by using the data analyzer template provided by SuperArray (http://pcrdataanalysis.sabiosciences.com/pcr/arrayanalysis.php) using glyceraldehyde 3-phosphate dehydrogenase, β-actin and ribosomal protein L13a as references. The non-stimulated condition was set to 1.
Statistics
Four replicates of each in vitro experiment were performed on three separate occasions (n = 12 per chemical). Data were first normalized to the control and plotted in a graph at 1 mM concentrations. Inhibitory concentration at 50% (IC50) and 5% (IC05) values were calculated using GraphPad Prism (GraphPad Software Inc., La Jolla, California, USA) where appropriate. Values in the figures are expressed as mean ± standard deviations. Student’s t test was used to perform pairwise comparisons between the chemically treated and untreated groups. The level of statistical significance was set at p < 0.05. All statistical analyses were performed using GraphPad Prism (GraphPad Software Inc.).
Results
Effects of MCPA and additives on human neuronal cells
In the MTT assay, MCPA only mildly decreased cell viability (61.9 ± 1.8%), even at concentration as high as 1 mM. Cells treated with 1-methoxy-2-propanol, dimethylamine and PPG exhibited minimal decreases in cell viability. However, the viability of cells challenged with PTE decreased dramatically to 5.4 ± 0.3% (Figure 2(a)). However, only 15.5% of cells survived after exposure to 1 μM PTE; the IC05 and IC50 values were calculated to be 0.01 and 0.15 μM, respectively (data not shown).

Cell viability and cytotoxicity following exposure to MCPA and additives. SK-N-SH cells were cultured for 24 h in the presence of indicated chemicals at 1 mM. (a) Shown are the percentages of viable cells in each group in comparison with control group (untreated), which were calculated using MTT assay. Error bars indicate standard deviation of each group. Astericks above each bar represent statistical significance compared to control group or other group. Note that the cell viability was preserved even at very high concentration of all the chemicals except PTE. (b) Shown are the percentages of released LDH in each group in comparison with Triton X-treated group. Error bars indicate standard deviation of each group. Asterix or letters above each bar represent statistical significance compared to control group or other group. **p < 0.001: Student’s t test, n = 12 for each chemical; ***p < 0.0001: Student’s t test, n = 12 for each chemical. MCPA: (4-choloro-2-methylphenoxy) acetic acid; PPG: polypropylene glycol; PTE: polyoxyethylene tridecyl ether; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; LDH: lactate dehydrogenase.
Similarly, the results of direct cytotoxicity measured by measuring LDH showed that MCPA had very low cytotoxicity against SK-N-SH cells (4.3 ± 1.6%, Figure 2(b)); meanwhile, 1-methoxy-2-propanol, dimethylamine and PPG had minimal or no cytotoxicity. On the contrary, cells treated with PTE exhibited incomparably greater LDH release (37.8 ± 2.8%) than cells exposed to any other additive (MCPA versus PTE; p < 0.001, Figure 2(b)). Taken together, the results demonstrate that PTE is a unique toxic chemical in the formulation of the MCPA herbicide.
Pathway-related gene expression following PTE treatment
Pathway-focused gene expression after PTE treatment showed that all nine pathways were up-regulated by 1.3 to 23.6-fold compared with the untreated group (data not shown). The genes in the inflammatory response pathway were the most profoundly affected among the tested pathways; the following eight (of nine) genes of proteins involved in inflammatory responses increased more than 10-fold after PTE treatment: chemokine (C–C motif) ligand 2, 11 CD40 ligand, 12 C-reactive protein, 13 interferon gamma, 14 interleukin-1 α, 15 interleukin-6, 16 toll-like receptor 4, 17 and tumour necrosis factor. 18 Among 11 hypoxia-related genes, the genes of 5 proteins – aryl hydrocarbon receptor nuclear translocator, 19 carbonic anhydrase IX, 20 erythropoietin, 21 haeme oxygenase (decycling) 1 22 and matrix metallopeptidase 9 23 – were up-regulated by more than 10-fold. In addition, unc-51-like kinase 1, 24 the gene encoding a protein for autophagy and ataxia telangiectasia mutated 25 and the gene for DNA damage were highly expressed following PTE exposure. Furthermore, the genes of five of six proteins involved in apoptosis signalling – apoptosis-related cysteine peptidase, 26 myeloid cell leukaemia sequence 1, 27 tumour necrosis factor receptor super family member 1a (TNFRSF1A), 28 TNFRSF10A 28 and FAS (TNFRSF6) 29 – were up-regulated by more than 10-fold. Finally, among the set of 13 genes for heat shock proteins, activating transcription factor 6, 30 BCL2-binding component 3, 31 BH3-interacting domain death agonist, 32 DNA-damage-inducible transcript 3 33 and heat shock 70-kDa protein 5 34 were up-regulated by 10.7- to 23.6-fold.
Discussion
The so-called ‘surfactant syndrome’ in acute pesticide intoxication is a symptom complex comprising hypotension, unconsciousness and respiratory failure, similar to the symptoms of the present case.35,36 Therefore, we hypothesized that the surfactant in the MCPA formulation ingested by the patient may have been the chemical responsible for her symptoms. Confirming our suspicions, the present results demonstrate that PTE was the most toxic among chemicals in the formulation. Assuming the in vitro tests using the neuronal cell line are biologically similar to the conditions of nerve cells in vivo, the observed toxicity in the nerve cells in vitro are relevant to the toxic symptoms in the central nerve system in the present patient.
We previously reported that surfactants can negatively affect cells in several ways, including breaking down the cell membrane, altering metabolic and mitochondrial activity, disrupting total protein synthesis and facilitating mitochondrial damage-induced apoptosis and necrosis.8,9 Pathway-focused gene expression after PTE treatment showed that the genes of all of nine pathways were up-regulated by 1.3- to 23.6-fold compared to the untreated group. Inflammatory responses were the most profoundly affected pathway among the tested pathways.
The appropriateness of the treatment modalities administered in the present case may be controversial among clinical toxicologists. Most if not all physicians would agree that early extracorporeal elimination is an essential treatment modality for patients with acute chemical intoxication; this can be achieved in many ways including haemodialysis (HD), haemoperfusion (HP) and CRRT according to the dialyzability of the target molecules and the patient’s condition. 36 On the first hospital day, there was no choice for extracorporeal elimination besides CRRT, because the patient’s blood pressure was too low to apply HD and/or HP (Table 2). On the second hospital day, when the patient’s blood pressure was normalized, we initially performed one HD session to correct acid–base unbalance, followed by one HP session for chemicals with low water solubility, such as MCPA (water solubility: 825 mg L−1). In addition, we recently reported the effect of intravenous lipid emulsion to treat surfactant toxicity in patients with acute glyphosate herbicide intoxication. 35 The leading theory for the therapeutic effect of lipid emulsions is that by creating an expanded intravascular lipid phase, the equilibrium established drives the offending drug from the target tissues into the newly formed ‘lipid sink’. 37
The present study has some limitations. First, because of the lack of toxicokinetics data, we were unable to calculate how much of each chemical was absorbed after the herbicide was ingested. Second, it is impossible to precisely replicate the patient’s physiologic state in vitro. Nevertheless, the present results demonstrate that the inert ingredient PTE is more toxic than the active ingredient MCPA. PTE up-regulated the expressions of genes implicated in several cell damage pathways, particularly the inflammatory response pathway.
In conclusion, the result of the present experiment suggests that the surfactant PTE is the chemical responsible for the toxic symptoms in our patient. Extracorporeal elimination with intravenous lipid emulsion was an effective treatment modality in this case.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was carried out with the support of the Soonchunhyang University Research Fund, Republic of Korea.
