Abstract
There are increasing concerns regarding the toxic effects of chlorpyrifos (CPF) on human health. Curcumin (CUR) is a yellow pigment isolated from turmeric ground rhizome of Curcuma longa Linn., which has been identified as an antioxidant agent. This study was designed to examine the protective effect of CUR and vitamin E (Vit E) on CPF-induced lung toxicity. Rats were divided into seven groups: control, CPF (13.5 mg/kg, orally), CPF + CUR (100 and 300 mg/kg, respectively, orally), CPF + α-tocopherol (Vit E, 150 mg/kg, intraperitoneally), CPF and CUR (100 and 300 mg/kg, respectively) in combination with α-tocopherol. The regimens were administered once daily for 28 days. At the end of the treatment period, lungs were collected for evaluation of oxidative factors and histopathological parameters. CUR and Vit E led to a decrease in lipid peroxidation in the lungs of the CPF-injected animals (48% and 51%, respectively). Glutathione peroxidase inhibited by CPF (91.9 nmol/min/mg protein) was induced again by CUR and Vit E (167.1 and 171.8 nmol/min/mg protein). CUR and Vit E caused a significant induction of superoxide dismutase (103.4 U/mg protein). Catalase activity almost returned to normalcy in CPF-intoxicated rats subjected to CUR + Vit E treatment (p < 0.001). Lung sections from CPF-treated rats displayed histopathological damages, while coadministration of CUR and Vit E resulted in apparently normal morphology with a significant decrease in injuries (p < 0.05). Our findings revealed that coadministration of Vit E and CUR to CPF-treated animals prevents the oxidative damages in the lung tissues.
Introduction
Annually, million tons of pesticides are employed to control a wide spectrum of insects and pests, but only less than 5% of these chemicals are estimated to reach the target organism. 1 Organophosphates (OPs) are among the most commonly used pesticides that have been employed for agricultural and military purposes. The extensive uses of OPs have been accompanied by a significant increase in the occurrence of poisoning with these agents. A variety of disorders including cancers, diabetes, birth defects, neurodegenerative diseases like Parkinson and Alzheimer, and cardiovascular diseases have been reported to be associated with chronic exposure to OPs. 2
Chlorpyrifos (CPF; O,O-diethyl O-(3,5,6-tricloro-2-pyridinol) phosphorothionate) is a well-known chlorinated OP which is widely used in agriculture industry. 3 –5 The toxic effects of OPs are due to the inhibition of acetylcholinesterase enzyme. 6 In addition to the role of CPF in controlling of insects, a number of additional effects such as hematological and immunological abnormalities, genotoxicity, teratogenicity, neurotoxicity, and hepatic dysfunction have been induced following CPF applicaction. 7,8 Animal studies showed that CPF also induces oxidative stress resulting in generation of lipid peroxidation products in different organs. 9,10 Evidences suggest that the lipophilic character of OPs such as CPF facilitates their penetration through the cell membrane to induce oxidative stress. 11
Antioxidant therapy can be considered as a good strategy for alleviating oxidative stress. Nowadays, there is a great tendency toward the consumption of herbal products as sources of antioxidants. This trend necessitates the evaluation of medicinal properties of natural products. 12
Curcumin (CUR; 1,7-bis [4-hydroxy-3-methoxyphenyl]-1,6-heptadiene-3,5-dione) is a yellow pigment originally isolated from turmeric ground rhizome of Curcuma longa Linn., which is widely used as a drug, cosmetic, and coloring agent. 13 Turmeric possesses a wide range of pharmacological properties including antitumor, anti-inflammatory, antiinfectious, antidiabetic, and antiangiogenic activities. 14 Various studies have demonstrated that CUR exhibits free radicals scavenging activities, inhibits lipid peroxidation, and enhances the activities of antioxidant enzymes such as glutathione-S-transferase (GST). 15,16
Vitamin E (Vit E) is a natural component of the membrane lipid bilayer and a fat-soluble antioxidant that prevents the propagation of free radical reactions. 17 The molecular and cellular mechanism of Vit E involve either scavenging of reactive oxygen species (ROS) or influencing cellular structure such as membrane and lipid domain and regulating specific enzymes. 18,19 It has been also demonstrated that Vit E by limiting the propagation of chain reaction of lipid peroxidation may inhibit the production of lipid peroxidation. 20 Several in vitro and in vivo studies have reported that the combination of vitamins with other antioxidants produces synergistic effects. 21,22
In light of above knowledge, this study was designed to evaluate the protective activity of two potent antioxidants, Vit E and CUR, against pulmonary oxidative damage induced by chlorpyrifos in rats.
Materials and methods
Chemicals
CUR, α-tocopherol (Vit E), CPF (96.5% purity (technical grade)), tris(hydroxymethyl)aminomethane) (Tris)–hydrochloric acid (HCl), nitroblue tetrazolium (NBT), bovine serum albumin (BSA), buffer, Coomassie blue, potassium cyanide, nicotinamide adenine dinucleotide phosphate reduced form (NADPH), ethylenediaminetetraacetic acid (EDTA), 5-50-dithio-bis-2-nitrobenzoic acid, and thiobarbituric acid (TBA) were purchased from Sigma Chemicals (St Louis, Missouri, USA). All other chemicals were of analytical grade and purchased from standard commercial suppliers.
Animals
Male 8-week-old specific pathogen-free Sprague Dawley rats, weighing between 180 g and 220 g, were obtained from the Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Islamic Republic of Iran. Rats were kept in a temperature-controlled room at 25°C and on a standard diet and water. All experimental procedures were approved by the Medical Ethics Committee for the use and care of laboratory animals of Tehran Medical University, Tehran, Islamic Republic of Iran (357; November 8, 2000).
Animal treatment schedule
CPF was reconstituted in corn oil to prepare a mixture of 1% concentration and was administered orally (p.o.) at the dose of 13.5 mg/kg (approximately 0.1 median lethal dose (LD50)) by a stomach tube. 23 Animals were randomly divided into seven groups (n = 8). In group I, rats received corn oil (p.o.) and saline intraperitoneally (i.p.) (negative control); in group II, CPF was administered (13.5 mg/kg, p.o.) to the animals for a period of 28 days (untreated CPF toxicity); in groups III and IV, CPF was administered as mentioned above and the rats were treated concomitantly once daily with CUR 100 and 300 mg/kg, respectively, for 28 consecutive days; in group V, CPF (150 mg/kg) + α-tocopherol (Vit E, i.p.) for 28 days; in groups VI and VII, in addition to CPF administration, the animals were treated concomitantly once daily with CUR (100 and 300 mg/kg) + 150 mg/kg α-tocopherol (Vit E, i.p.) for 28 days. At the end of the experimental period, rats were anesthetized and killed by cervical dislocation. Lung tissues were collected, washed with saline solution, and stored at −70°C for the biochemical studies. Also a small portion of tissue was fixed in 10% neutral formalin for histopathological examination.
Measurement of TBARS
Formation of TBA reactive substances (TBARS) was measured in lung homogenates (20% w/v) according to a previously described method. 24 Briefly, the tissue was homogenized in 100 mM potassium phosphate buffer (pH 7.4), and the homogenate was centrifuged for 5 min at 3000g. Then, 1 ml of supernatant was mixed with 2 ml of the TBA-trichloroacetic acid (TCA)-HCl reagent (TBA 0.37%, 0.25 N HCl, and 15% TCA) and vortexed thoroughly. The samples were kept in boiling water bath for 15 min and then allowed to cool at room temperature. The samples were then centrifuged at 3000g for 10 min, and the absorbance of supernatant was measured at 535 nm by spectrophotometer. The malondialdehyde (MDA) levels was determined from the extinction coefficient at 535 nm (ε = 1.56 × 105 M−1 cm−1).
Measurement of GPx activity
For GPx activity assay, lung samples were added to a solution containing 50 mM Tris (pH 7.5), 2 mM glutathione (GSH), NADPH, 0.15 U/ml glutathione reductase (GR), 0.4 mM azide, 5 mM EDTA, and 1 mM dithiothreitol buffer. The reaction was initiated by adding hydrogen peroxide (H2O2) and change in absorbance was monitored spectrophotometrically at 340 nm/min over 10 min.
Measurement of SOD activity
The basis of the enzyme assessment is the reaction of superoxide dismutase (SOD) with the indicator molecule NBT. Increase in absorbance at 560 nm over a 5-min period indicates the reduction of NBT by superoxide. Total enzyme activity is defined as the amount of protein needed for half-maximal suppression of the NBT.
Measurement of CAT activity
Catalase (CAT) activity in lung tissue was measured spectrophotometrically according to the method of Luck. 25 The enzyme activity was assayed in samples by measuring the absorbance at 240 nm in a reaction medium containing 10 mM H2O2 and 50 mM sodium phosphate buffer (pH 7.0). The amount of enzyme activity was calculated using the molar extinction coefficient of H2O2. The results were expressed as micromoles of H2O2 decomposed/min/mg protein.
Protein estimation
The protein content was quantified by the method described by Bradford. 26 Briefly, concentrated Coomassie blue (G250) was diluted in 250 µl distilled water. Then, 750 µl of the dye was added to 50 µl of sample, and the mixture was incubated at room temperature for 10 min. An absorbance measurement was carried out at 595 nm by a spectrophotometer (CE 7250, Cecil, UK). A standard curve was prepared using different concentrations of BSA ranging between 0.25 and 1 mg/ml.
Histopathological studies
Lung tissues of rats in all experimental groups were collected to be used for histopathological observations. Samples were fixed in 10% buffered formaldehyde solution, embedded in paraffin, sectioned at 5 µM, fixed on slides, and stained with hematoxylin and eosin. The tissue sections were evaluated under light microscopy and photographed with a digital camera (Olympus DP12, U-TVO.5XC-2, Japan).
Statistical analysis
The experimental results are presented as mean ± standard error of mean. Following the assurance of normal distribution of data, one-way analysis of variance and Newman Keuls’ post hoc test were carried out using GraphPad prism Version 5.04 (San Diego, California, USA) software, and the value of p < 0.05 was considered significant.
Results
Effects of CUR and Vit E treatments on lung lipid peroxidation
Determination of TBARS in lung homogenates revealed that the levels of TBARS increased significantly in CPF-exposed animals (approximately 47%). Treatment of CPF-intoxicated rats with CUR (100 and 300 mg/kg, p.o.) and Vit E (i.p.) significantly decreased TBARS levels (Figure 1). Compared with CUR alone, coadministration of CUR and α-tocopherol in rats exposed to CPF caused a significant reduction in TBARS levels (p < 0.05).
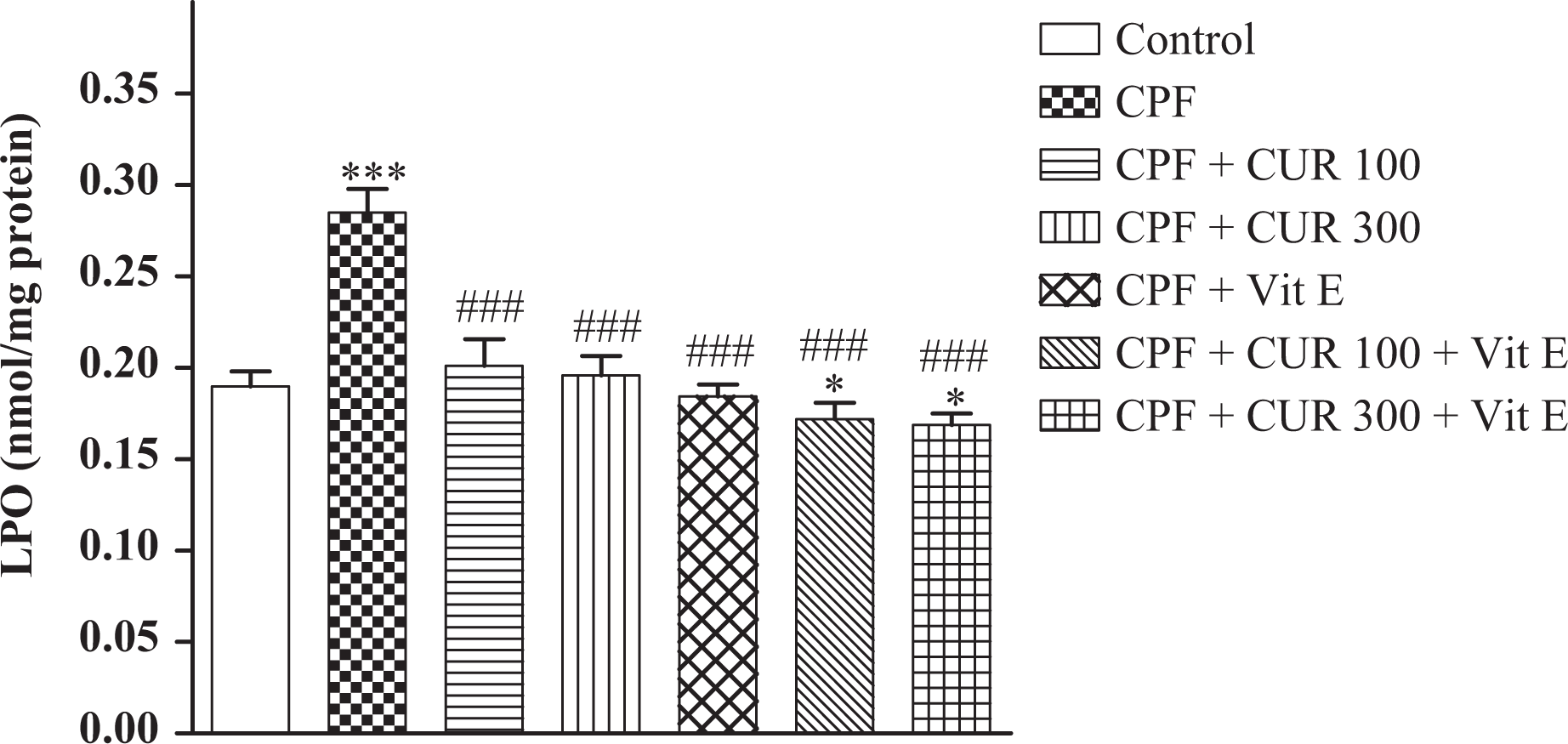
Effect of CPF, CUR, and Vit E on MDA level in lung tissue of experimental rats. Values are expressed as mean ± SEM of eight animals in each group. *p < 0.05: values differ significantly from CPF + CUR; ***p < 0.001: values differ significantly from negative control; ###p < 0.001: values differ significantly from CPF group. CPF: chlorpyrifos; CUR: curcumin; Vit E: vitamin E; MDA: malondialdehyde; SEM: standard error of mean.
Effects of CUR and Vit E treatments on lung GPx activity
As shown in Figure 2, lung GPx activity was suppressed due to CPF administration (approximately 54%), while the enzyme activity was again induced following CUR or Vit E treatment. Co-treatment of CPF-treated rats with CUR and α-tocopherol also led to a significant increase in GPx activity (p < 0.001).

Effect of CPF, CUR, and Vit E on pulmonary GPx level in rats. Values are expressed as mean ± SEM of eight animals in each group. ***p < 0.001: values differ significantly from negative control; ###p < 0.001: values differ significantly from CPF group. CPF: chlorpyrifos; CUR: curcumin; Vit E: vitamin E; GPx: glutathione peroxidase; SEM: standard error of mean.
Effects of CUR and Vit E treatments on lung SOD activity
Changes in lung SOD activity showed that the enzyme activity that was initially suppressed due to CPF administration (approximately 58%) was induced again following CUR and Vit E administration (Figure 3). Coadministration of CUR (300 mg/kg) + α-tocopherol to CPF-exposed rats also improved the SOD activity compared with those received only CUR (p < 0.05).

Effect of CPF, CUR, and Vit E on SOD activity in lung tissue of experimental rats. Values are expressed as mean ± SEM of eight animals in each group. *p < 0.05: values differ significantly from CPF + CUR; ***p < 0.001: values differ significantly from negative control; ###p < 0.001: values differ significantly from CPF group. CPF: chlorpyrifos; CUR: curcumin; Vit E: vitamin E; SOD: superoxide dismutase; SEM: standard error of mean.
Effects of CUR and Vit E treatments on lung CAT activity
The pulmonary CAT activity of all experimental animals is shown in Figure 4. CPF intoxication decreased the CAT activity, whereas CUR treatment (100 and 300 mg/kg, p.o.) caused an increase in the enzyme activity (p < 0.001). Our results showed that CAT almost returned to normalcy in CPF-intoxicated rats treated with CUR + α-tocopherol.

Effect of CPF, CUR, and Vit E on the CAT in lung tissue of experimental rats. Values are expressed as mean ± SEM of eight animals in each group. ***p < 0.001: values differ significantly from negative control; ###p < 0.001: values differ significantly from CPF group. CPF: chlorpyrifos; CUR: curcumin; Vit E: vitamin E; CAT: catalase; SEM: standard error of mean.
Histopathological examinations
On day 28, the biopsies of the lungs from all groups were processed for histopathological assessment. Based on the histopathological results, the appearance of the lung in control animals was normal (Figure 5(a)). Lung tissues of CPF-treated rats showed interalveolar septa thickening, distortion of alveoli, infiltration of inflammatory cells, and interstitial pneumonia with pulmonary edema (Figure 5(b)). However, when CPF was administered with CUR or CUR + α-tocopherol, a significant decrease in distortion of alveoli, infiltration of inflammatory cells, interstitial pneumonia, edema, and distortion of alveoli were observed (Figure 5(c) and (d)).

Histopathological observations. Light microscopy showed lung histologic sections of rats from different groups. (a) Negative control; (b) CPF (8.5 mg/kg); (c) CPF + CUR at the dose of 300 μg/kg b.w; (d) CPF + co-treatment of CUR with α-tocopherol (150 mg/kg b.w.). H&E-stained sections of lung in control group showed normal pathology. In CPF group, thickening of pulmonary interalveolar septa, distortion of alveoli, infiltration of inflammatory cells, and interstitial pneumonia with pulmonary edema were observed. Section of liver from CPF-intoxicated animals which were treated with CUR and Vit E showed only moderate accumulation of inflammatory cells. Arrow in (b) indicates infiltration of inflammatory cells. CPF: chlorpyrifos; CUR: curcumin; Vit E: vitamin E; H&E: hematoxylin and eosin; b.w.: body weight.
Discussion
The lung is the first organ to come in contact with inhaled toxic chemicals. OP insecticides can cause adverse effects in various organs such as the lungs. OP compounds lead to a wide range of pulmonary injuries such as cellular aggregation in the vascular walls or air spaces, neutrophil infiltration, hemorrhage, alveolar congestion, and emphysematous changes. 27,28 In addition to inhibition of cholinesterase enzyme, oxidative stress is proposed as a major toxicity mechanism for OP compounds. For example, diazinon, a nonsystemic OP, induces changes in cellular antioxidants such as GSH, GR (i.e. GSSG-Rd), CAT, SOD, GPx, and GST, which are traditional biomarkers of oxidative stress. 29 CPF is a lipophilic OP that can easily pass through the cell membrane and induce a high level of damage. 30 In previous investigations, it was demonstrated that CPF induces oxidative stress as well as histopathologic changes in different tissues. 31,32
This study was designed to evaluate the protective effects of CUR and Vit E on pulmonary oxidative damages induced by CPF. The results of this investigation clearly suggest that CPF induces pulmonary oxidative stress mainly by modulating antioxidant defense. The oral LD50 of CPF for male rats is 135 mg/kg body weight. 33 Although in this study the dose of CPF was much lower than oral LD50 (1/10 LD50) and none of the rats died during the experimental period, changes in lungs oxidative status as well as histopathological parameters clearly suggest that CPF induces pulmonary damages.
Considering the active role of ROS and its metabolites in cellular physiology and pathogenesis of a number of diseases, they are subject of intense research. 8 The measurement of tissue lipid peroxidation as a biomarker for oxidative stress has been of predictive importance. 34 MDA is the most important oxidation by-product of peroxidized polyunsaturated fatty acids, has a major cause in damaging cell membrane thereby acting as an important indicator of lipid peroxidation occurrence. 35 Different studies have shown that pesticides increase MDA levels in various tissues. 36,37 Data in this study also show that MDA content significantly increases in the lung tissue of rats treated with CPF. These findings are closely similar to those obtained by Uzun and coworkers. 38,39 The increase in pulmonary MDA level probably ascribed to the excessive production of ROS resulting from the oxidative stress induction.
The antioxidant enzymes such as SOD, GPx, and CAT play central roles in cellular antioxidative defense. SOD enzyme catalyzes the dismutation of superoxide radicals to form H2O2. In this study, the activity of this enzyme was inhibited in rats exposed to CPF which may be due to the consumption of the enzyme in converting the superoxide radicals. 12 In addition to SOD, we observed that CAT and GPx were suppressed in CPF-intoxicated animals. These enzymes are also antioxidant key factors that convert H2O2 into water. 14 It has been proven that superoxide radicals are the potent inhibitors of CAT. 40
The main function of GPx, which uses GSH as a substrate, is to diminish soluble H2O2 and alkyl peroxides. 41 In our study, GPx activity was inhibited in lung tissues of rats treated with CPF. These results are consistent with a previous study in which both GPx and GST activities were decreased in rats exposed to CPF. 42 Thus, the suppression of antioxidant enzymes activities following CPF exposure could be attributed to depletion of the enzyme substrates such as GSH, overproduction of free radicals as evidenced by increased lipid peroxidation, and direct effect of CPF on the enzymes activity. These cellular oxidative damages following CPF exposure were confirmed by histopathological observations which revealed interalveolar septa thickening, distortion of alveoli, infiltration of inflammatory cells, and interstitial pneumonia with pulmonary edema (Figure 5(b)).
Scavenging ROS or their precursors, binding metal ions needed for the catalysis of ROS generation, and inhibiting ROS formation are possible mechanisms of action for antioxidants. 43 Numerous antioxidant compounds like Vit E, vitamin C, β-carotene, uric acid, ubiquinols, and flavonoids have been shown to play significant roles in the nonenzymatic protection against oxidative damages. 11,44 The protective effects of CUR have been demonstrated against some oxidative compounds such as carbon tetrachloride, ferric nitrilotriacetate, and malathion. 45,46 Due to the antioxidant potential and relatively low toxicity of CUR, there is a strong tendency toward investigation of its protective roles in oxidative damages. On the other hand, previous studies reported that antioxidants like Vit E and CUR had protective effects against oxidative stress induced by different toxic compounds. 47,48 Based on our results, we can assume that pulmonary toxic effects of CPF that are mainly attributed to induction of oxidative stress are reversed by administration of Vit E and/or CUR possibly via scavenging or neutralizing ROS.
CUR with and without Vit E decreased lung MDA, which is an end product of lipid peroxidation, in rats exposed to CPF. Phenolic and β-diketone functional groups as well as methoxy group of CUR possess considerable free radical scavenging properties. 49,50 Another proposed mechanism for CUR antioxidant properties is the inhibition of superoxide anion and hydroxyl radical generation via the preventing oxidation of ferrous ion to ferric ion through Fenton reaction. 14,49 Previous investigations have also shown the protective effect of Vit E against several xenobiotics. 51 –53 Vit E as a lipid-soluble antioxidant was reported to have protective function against oxidative stress and reduce the production of lipid peroxides through scavenging free radicals. 54,55
Besides, CUR and Vit E treatment induced the antioxidant enzymes activity which proves the antioxidant potential of these compounds. These data are in consistency with previous studies reporting that CUR improves the antioxidant enzyme activity. 56,57
It is interesting to note that coadministration of CUR and α-tocopherol significantly improved some oxidative parameters such as TBARS levels and SOD activity in comparison with CUR alone. These results suggest that CUR and α-tocopherol may have synergistic effects against CPF-induced pulmonary toxicity. In support of our results, earlier studies have shown that flavonoids can interact together which display distinct characteristics such as synergistic antioxidative properties. 58,59 Several other studies have also reported the combination of antioxidants with vitamins produces synergistic effects. 21,22 However, more investigations are needed to evaluate the synergistic effects of CUR and α-tocopherol. The protective role of Vit E and CUR was further confirmed by their effects on CPF-induced histopathological changes in the lungs (such as distortion of alveoli, infiltration of inflammatory cells, and interstitial pneumonia; Figure 5(c) and (d)).
In conclusion, the data of this study clearly indicate that CPF induces pulmonary oxidative stress mainly by modulating antioxidant defense. CUR and Vit E can attenuate CPF-induced pulmonary damage. This effect can be attributed to free radicals scavenging ability of CUR. Future studies should focus on molecular mechanisms involved in the modulatory action of CUR on CPF-induced pulmonary damage.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
