Abstract
Synthetic cannabinoid use has become widespread, leading to increased burdens on health care providers. Symptoms range from agitation and psychosis to seizures and acute kidney injury. We report a case where a patient was assessed and treated twice within 12 h for seizures following synthetic cannabinoid intoxication. Blood sample determinations showed low concentrations of analogues not previously reported, some of which are legal. Clinicians should be aware that synthetic cannabinoids may cause an array of severe health consequences. Given the ever evolving structure of available analogues, clinicians must also be prepared for other unexpected adverse effects.
Introduction
The use of synthetic cannabinoids has become widespread throughout North America, Europe, and Australasia. The term synthetic cannabinoids describes a large number of chemicals with unrelated structures, which bind to cannabinoid receptors (CB1 and CB2) and mimic the effects of delta-9-tetrahydrocannabinol (THC), the major active constituent of marijuana. However, they have been found to possess a toxicology profile different to that of THC; reported adverse effects have included vomiting and tachycardia, 1 drowsiness, anxiety and agitation, 1 psychosis 2 including aggressive, violent, and self-injurious behaviors, 3 hallucinations, 1 dependence and withdrawal, 4 tremors and seizures, 5,6 myocardial infarction, 7 ischemic stroke, 8 and acute kidney injury 9,10 Anticipation of clinical effects has been complicated by the wide range of different analogues being used recreationally.
In New Zealand, particularly due to legislation put in place to halt the widespread use of these drugs, rapid government identification and prohibition of individual analogues has led to them being quickly replaced by other cannabinoids; these new products were then remarketed, typically with the original brand name. Consequently, distributors and retailers are selling synthetic cannabinoids not previously reported in the peer-reviewed literature. This case report highlights several recently identified new analogues, some of which are still legally available for sale (the analogues PB-22, 5F-PB-22) and describes a patient presenting to an emergency department with a seizure followed by a second seizure several hours later, after smoking synthetic cannabinoids.
Case report
A 23-year-old male recreationally smoked a synthetic cannabinoid cigarette (trade name K2) at home and 6 h later he suffered what his wife described as generalized tonic–clonic seizure activity, including urinary incontinence and tongue lacerations. He did not seek medical assessment at this stage. Later, after smoking a further smaller amount 3 h after his first seizure, he started vomiting and presented at a local emergency department with nausea, dry mouth, and vomiting. On the initial evaluation, he had a normal neurologic and cardiopulmonary examination. The patient admitted to smoking a synthetic cannabinoid product and to a history of smoking other synthetic cannabinoid-containing products, including K2, more or less daily for the previous several years but denied use of any other drugs or medications. The patient had no previous history of seizures. There were no further seizures, and he was discharged home.
Three hours after discharge (4 h after last smoking the product), the patient experienced a second episode of seizure activity. He represented to the emergency department under his own volition and a computed tomography scan of the brain was obtained, which was read as normal. Blood biochemistries were normal except for a potassium of 3.3 mmol L−1 (normal range 3.5 to 5.2), bicarbonate of 20.9 mmol L−1 (normal range 22–32), creatine kinase of 338 U L−1 (normal range 60–220), and a lactate of 5.2 mmol L−1 (normal range 0.5–2.2). These abnormal parameters all resolved on repeat evaluation prior to discharge. In addition, the white blood cell count was 18.9 × 109 L−1 (normal range 4–11) with a left shift. Standard toxicology screening for THC and ethanol was negative. The patient was administered 1500 mL of intravenous normal saline and oral diazepam and discharged from the emergency department.
Two temporally separate blood samples were collected in ethylenediaminetetraacetic acid tubes from the patient on the second visit (5.5 and 8.3 h after the last exposure); the separated plasma was stored at −30°C for later analysis. Samples were prepared for analysis using a liquid/liquid extraction technique. The analysis was performed by liquid chromatography/mass spectroscopy (LC-MS) using a ThermoFisher Q Exactive high-resolution accurate mass LC-MS (HFL Sport Science, Cambridgeshire, UK). Identified synthetic cannabinoid analogues and their plasma concentrations are summarized in Table 1 and figures of the respective analogues are presented in Figure 1.
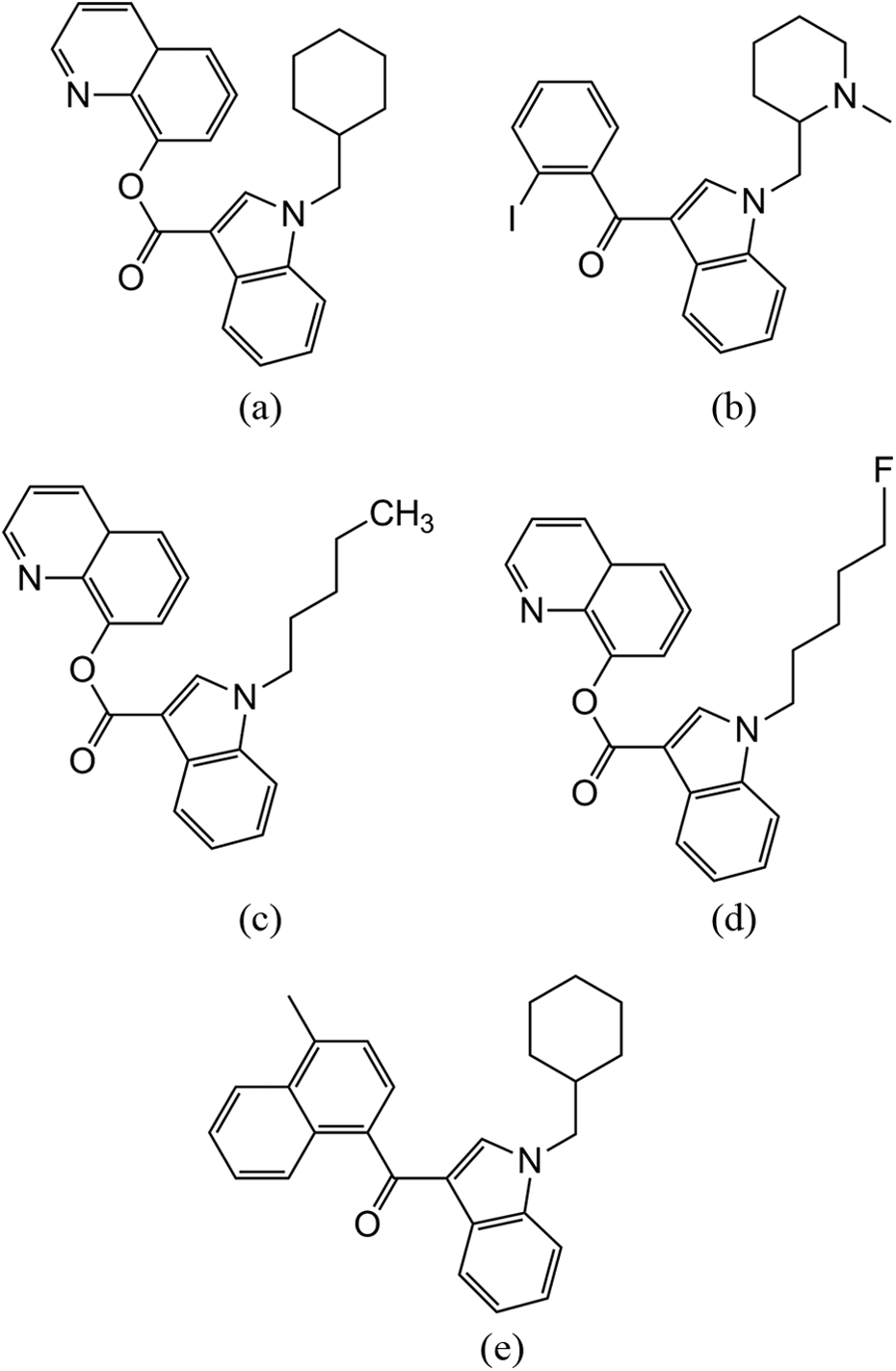
Synthetic cannabinoids identified in the plasma of the patient suffering seizures: (a) BB-22, (b) AM-2233, (c) PB-22, (d) 5F-PB-22, and (e) JWH-122 (N-methylcyclohexyl).
Concentrations of synthetic cannabinoids identified in patient’s plasma at 5.5 and 8.3 h after last exposure.

Discussion
Recreational use of synthetic cannabinoids, like that of other psychoactive substances, places an additional burden on health care services; this is exacerbated by their legal status and ease of access from convenience stores and gas stations. Data from The American Association of Poison Control Centers suggest the majority of acute synthetic cannabinoid exposures do not produce major adverse outcomes 11 ; however, they may elicit adverse effects that are more serious than marijuana, and reports have demonstrated there is a risk of potentially life-threatening effects, likely depending on the dose or the analogue involved. 1 –3,5 –10
This patient had a long history of smoking cannabinoids without apparent adverse effect; however, marketed brands evolve over time as different analogues are prohibited and replaced with reformulated products containing analogues not subject to legal restrictions. Adverse clinical effects suffered by this patient may be explained by the analogues present in the formulation he smoked, and the dose, which can vary substantially between product batches not subjected to consistent quality control. Analogues identified in the patient’s blood, 5.5 and 8.3 h after his last exposure, showed low concentrations (varying from 9 to 148 pg mL−1) of the cannabinoid agonists BB-22 (QUCHIC), AM2233, 5F-PB-22, and PB-22 and a variation of JWH-122 (with a cyclohexylmethyl substitution; Figure 1). To the knowledge of the authors, there are no toxicology profiles for these analogues and although it cannot be conclusively proven that one or more of these analogues was directly responsible for his seizures, his lack of seizure history and temporal relationship to smoking is highly suggestive of a causal link. Both this report and recent investigations 12,13 have suggested concentrations evident immediately after exposure are possibly at and below parts per billion highlighting the potency of these drugs.
Synthetic cannabinoids will likely continue to be an important public health problem as new analogues are synthesized and marketed to replace those that have been prohibited. Clinicians in primary care, emergency care, and specialty care settings should be aware that synthetic cannabinoids may lead to an array of severe health consequences typically not associated with marijuana. The constellation of clinical effects may range from agitation and tachycardia to psychosis, seizures, acute kidney injury, and cerebral strokes. Furthermore, given the ever evolving structure of available analogues, clinicians must also be prepared for other adverse effects and be aware of complications with a delayed onset. There is no specific treatment or antidote for synthetic cannabinoid poisoning; agitation may respond to provision of a nonstimulating environment while more severe psychosis or seizures may require benzodiazepine administration. 14 Symptoms appear to be self-limited and typically resolve in 8–12 h. In individuals presenting with seizures of unknown etiology, inquiring about the use of novel psychoactive substances may yield important information. Patients should additionally be counseled on the possible severe adverse health consequences of using these potent drugs of abuse.
Limitations
While this patient appeared to have seizures, it was not witnessed by medical personnel and therefore cannot definitively be called a seizure. Additionally, while analytical confirmation of the analogues involved was undertaken in this case, we cannot exclude the possibility that another compound or other underlying condition may have contributed to the seizures displayed. However, this patient had no prior history of seizure activity.
Footnotes
Acknowledgment
The authors wish to acknowledge Craig Rodgers, Southern Community Health Laboratories, Invercargill, for performing plasma sample preparation.
Conflict of interest
The authors declared no conflicts of interest.
Funding
The study was funded, in part, by a grant from the University of Otago’s Dean’s Bequest Fund.
