Abstract
Several studies have indicated the harmful effect of flare-up periods in pregnant women with inflammatory bowel disease (IBD) on their newborns. Therefore, an effective and safe medical treatment during pregnancy is of great concern in IBD patients. The aim of this study was to perform a meta-analysis on the outcomes of thiopurines use and a systematic review of antitumor necrosis factor (anti-TNF) drugs used during pregnancy in women with IBD. The results of cohorts evaluating the safety of anti-TNF drugs during pregnancy up to July 2013 were collected and analyzed. In the meta-analysis, a total of 312 pregnant women with IBD who used thiopurines were compared with 1149 controls (women with IBD who were not treated with any medication and women who were exposed to drugs other than thiopurines) to evaluate the drug effect on different pregnancy outcomes, including prematurity, low birth weight, congenital abnormalities, spontaneous abortion, and neonatal adverse outcomes. Results of statistical analysis demonstrated that congenital abnormalities were increased significantly in thiopurine-exposed group in comparison with control group who did not receive any medicine for IBD treatment. The summary odds ratio was 2.95 with 95% confidence interval = 1.03–8.43 (p = 0.04). We observed no significant differences in occurrence of other adverse pregnancy outcomes between compared groups. The results of cohorts evaluated the safety of anti-TNF drugs during pregnancy demonstrated no increase in occurrence of adverse pregnancy outcomes in comparison with controls except for the significant decrease in gestational age of newborns of drug-exposed mothers in one trial. In conclusion, a benefit–risk ratio should be considered in prescribing or continuing medicinal therapy during pregnancy of IBD patients.
Keywords
Introduction
Inflammatory bowel disease (IBD) in pregnant women may influence their offspring due to observed increase in prevalence of prematurity, low birth weight, Cesarean section, and congenital malformations. 1 Therefore, it seems necessary for women with IBD to receive medical counseling to prevent possible risks of their underlying disease on their newborns. The interaction between IBD and pregnancy is very important due to the possibility of coincidence of the onset of disease with peak reproductive years. 2,3 Several studies have demonstrated significant higher prevalence of Crohn’s disease (CD) among women. 4
IBD, including ulcerative colitis (UC) and CD, is a chronic relapsing and remitting disease, 5 characterized by gastrointestinal inflammation and some extraintestinal complications. 6
Till date, several drugs have been introduced for treatment of both UC and CD, including 5-amino salicylate (5-ASA), 7,8 corticosteroids, 9 immunosuppressive agents, antitumor necrosis factor (anti-TNF) drugs, 10 –12 antibiotics, 13,14 antioxidants, 15,16 probiotics, 17 –19 and some herbal medicines as supplemental therapy. 20 –22
Thiopurines and anti-TNF drugs such as infliximab are used widely to induce and maintain remission in IBD patients. Infliximab has been approved by US Food and Drug Administration (FDA) for treatment of CD. However, information on its safety in humans during pregnancy is scarce yet. 23
Thiopurines include 6-mercaptopurine and its prodrug azathioprine. It is reported that both drugs have the potential to induce spontaneous abortion. 24,25 After analyzing for the results from several studies, the European Crohn’s and Colitis Organization guidelines consider thiopurines to be safe and well tolerated in pregnant women. 26 There are some controversial data about their safety during pregnancy. Several studies demonstrated their safety for pregnant women. 27 –30 However, increased preterm birth, low birth weight, and congenital abnormalities have been reported. Nevertheless, it has been mentioned that these results might be due to the underlying diseases rather than the pharmacologic treatment. 31 Therefore, assessing a benefit–risk ratio of thiopurines and anti-TNF drugs during pregnancy in IBD patients is an immediate important challenge. 32
Infliximab, adalimumab, certolizumab, and natalizumab are considered as the most common biological agents used in IBD treatment. 33 No adverse effect has been reported according to three large-scale studies using infliximab. 34 – 36 Adalimumab has the same placental transfer compared to infliximab. 36 Some studies demonstrated its safety during pregnancy. 36 – 38 In addition, certolizumab and natalizumab are two other anti-TNF drugs with currently insufficient data on their effect on pregnancy. 26
There is sufficient evidence suggesting that anti-inflammatory drugs can cause adverse effect of vasculitis and consequently abortion. 39,40 Therefore, assessing a benefit–risk ratio of thiopurines and anti-TNF drugs during pregnancy in IBD patients is an immediate and important challenge. In this meta-analysis, we aimed to highlight the safety of thiopurines and anti-TNF drugs during pregnancy in IBD patients.
Methods
Data sources
Electronic databases, including PubMed, Google Scholar, Web of Science, Scopus, and Cochrane Central Register of Controlled trials were searched for all cohort studies that evaluated the pregnancy outcomes of thiopurines and/or anti-TNF drugs during pregnancy in IBD women up to July 2013. The primarily applied Medical Subject Headings were azathioprine, mercaptopurine, infliximab, adalimumab, anti-TNF and pregnancy outcomes, adverse events, safety, ulcerative colitis, Crohn’s disease, and congenital abnormalities. All published studies as well as abstracts presented at the meetings were evaluated without any language limitations. The reference lists of searched articles were also reviewed for additional relevant studies.
Study selection
Three reviewers independently examined the title and abstract of each article to eliminate duplicates, reviews, case studies, and pilot studies. All studies investigating the influence of medical treatment during pregnancy in IBD women were considered. We excluded the studies in which the effects of paternal drug exposure were evaluated in newborns. Trials that evaluated any medical treatment other than thiopurines and anti-TNF drugs were excluded. We excluded the studies in which pregnant women used thiopurines and anti-TNF drugs for an underlying disease other than IBD. Any trials in which there was no comparison (unexposed group including both women with IBD who were not treated with drugs and women who were exposed to drugs other than thiopurines and anti-TNF drugs) were excluded from the meta-analysis. The studies that assessed pregnancy outcomes such as prematurity, birth weight, gestational age, and congenital malformations were included. Data were extracted as characteristics of included pregnant women, type of IBD, sample size, treatment type, and pregnancy outcomes. Finally, three reviewers checked for validity of the final eligible studies. There were no disagreements between the reviewers (Figure 1).
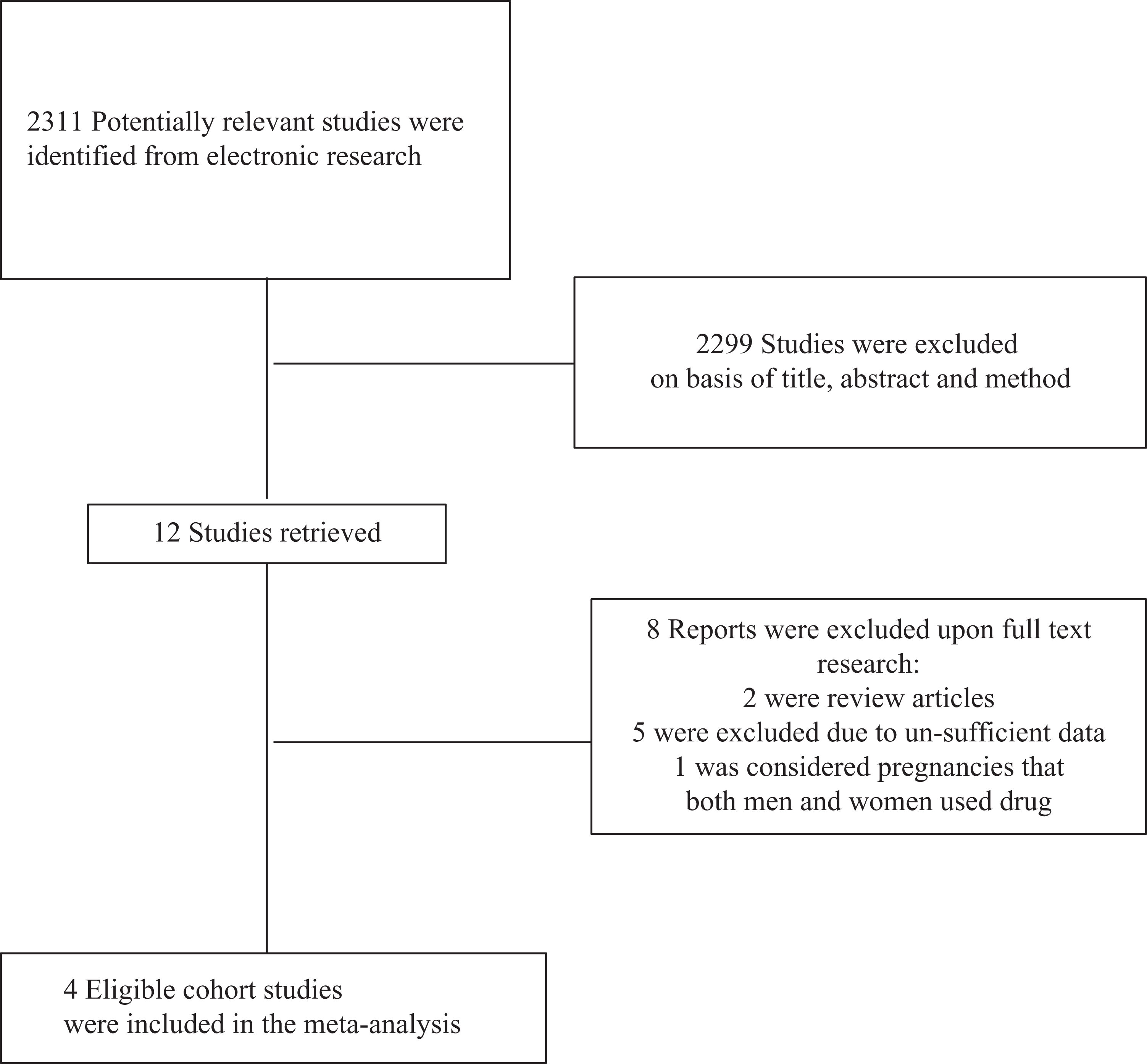
Flow diagram of the study selection process.
Statistical analysis for meta-analysis
Data from selected studies were extracted in the form of 2 × 2 tables. Included studies were weighted and pooled. The data were analyzed using Statsdirect software version 3.0.97. Odds ratio (OR) and 95% confidence intervals (95% CIs) were calculated using Der Simonian–Laird methods. The Cochran Q test and I 2 inconsistency were used to test heterogeneity. In case of heterogeneity or probability of few included studies in the meta-analysis, the random effect for individual and summary of effect size for OR was applied.
Results
Thiopurines
The electronic searches yielded 2311 items. Of these, four cohort studies used thiopurines in pregnant women with IBD were considered eligible (Figure 1). These four trials included a total of 1461 pregnant women with IBD who received thiopurines, other types of treatment, or no medicines as controls. Of these, 1127 women had CD and 329 and 5 had UC and unclassified IBD, respectively. Characteristics of included studies such as the number, mean age, and type of IBD in pregnant women and pregnancy outcomes are summarized in Tables 1 and 2, respectively.
Characteristics of included studies on the use of thiopurines during pregnancy.

AZA: azathioprine; CD: Crohn’s disease; IBD: inflammatory bowel disease; MP: mercaptopurine; ND: not determined;UC: ulcerative colitis.
Pregnancy outcomes (proportion of adverse outcome to number of exposed pregnancies) of included studies on the use of thiopurines.

A: exposed to thiopurines; B: receiving a drug other than thiopurine; C: not receiving any medication.
Prematurity due to exposure to thiopurines in comparison with other IBD medications
The summary OR of prematurity for all included data for thiopurines in comparison with other IBD medications 30,41,42 was 1.07 (95% CI = 0.42–2.7; p = 0.89; Figure 2(a)). The value of p for the Cochrane Q was 0.04 (Figure 2(b)) with I 2 (inconsistency) of 68.9% (95% CI = 0–88.8%).

(a) Individual and summary odds ratio for the outcome of “prematurity” for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (b) Heterogeneity indicators for the outcome of prematurity for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (c) Individual and summary odds ratio for the outcome of prematurity for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients. (d) Heterogeneity indicators for the outcome of prematurity for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients.
Prematurity due to exposure to thiopurines in comparison with no medications in pregnant IBD patients
The summary OR of prematurity for all included data for thiopurines in comparison with no medications in IBD patients in two studies 30,43 was 2.65 (95% CI = 0.85–8.3; p = 0.1; Figure 2(c)). The value of p for the Cochrane Q was 0.11 (Figure 2(d)).
Low birth weight due to exposure to thiopurines in comparison with other IBD medications
The summary OR of low birth weight for all included data for thiopurines in comparison with other IBD medications 30,41,42 was 0.82 (95% CI = 0.32–2.13; p = 0.69; Figure 3(a)).

(a) Individual and summary odds ratio for the outcome of “low birth weight” for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (b) Heterogeneity indicators for the outcome of low birth weight for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (c) Individual and summary odds ratio for the outcome of low birth weight for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients. (d) Heterogeneity indicators for the outcome of low birth weight for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients.
The value of p for the Cochrane Q was 0.12 (Figure 3(b)) with I 2 (inconsistency) of 52.5% (95% CI = 0–85.1%).
Low birth weight due to exposure to thiopurines in comparison with no medications in pregnant IBD patients
The summary OR of low birth weight for all included data for thiopurines in comparison with no medications in IBD patients (two studies) 30,43 was 1.95 (95% CI = 0.66–5.78; p = 0.23; Figure 3(c)).
The value of p for the Cochrane Q was 0.91 (Figure 3(d)).
Congenital abnormality due to exposure to thiopurines in comparison with other IBD medications
The summary OR of congenital abnormality for all included data for thiopurines in comparison with other IBD medications 30,41,42 was 0.99 (95% CI = 0.27–3.61; p = 0.98; Figure 4(a)).
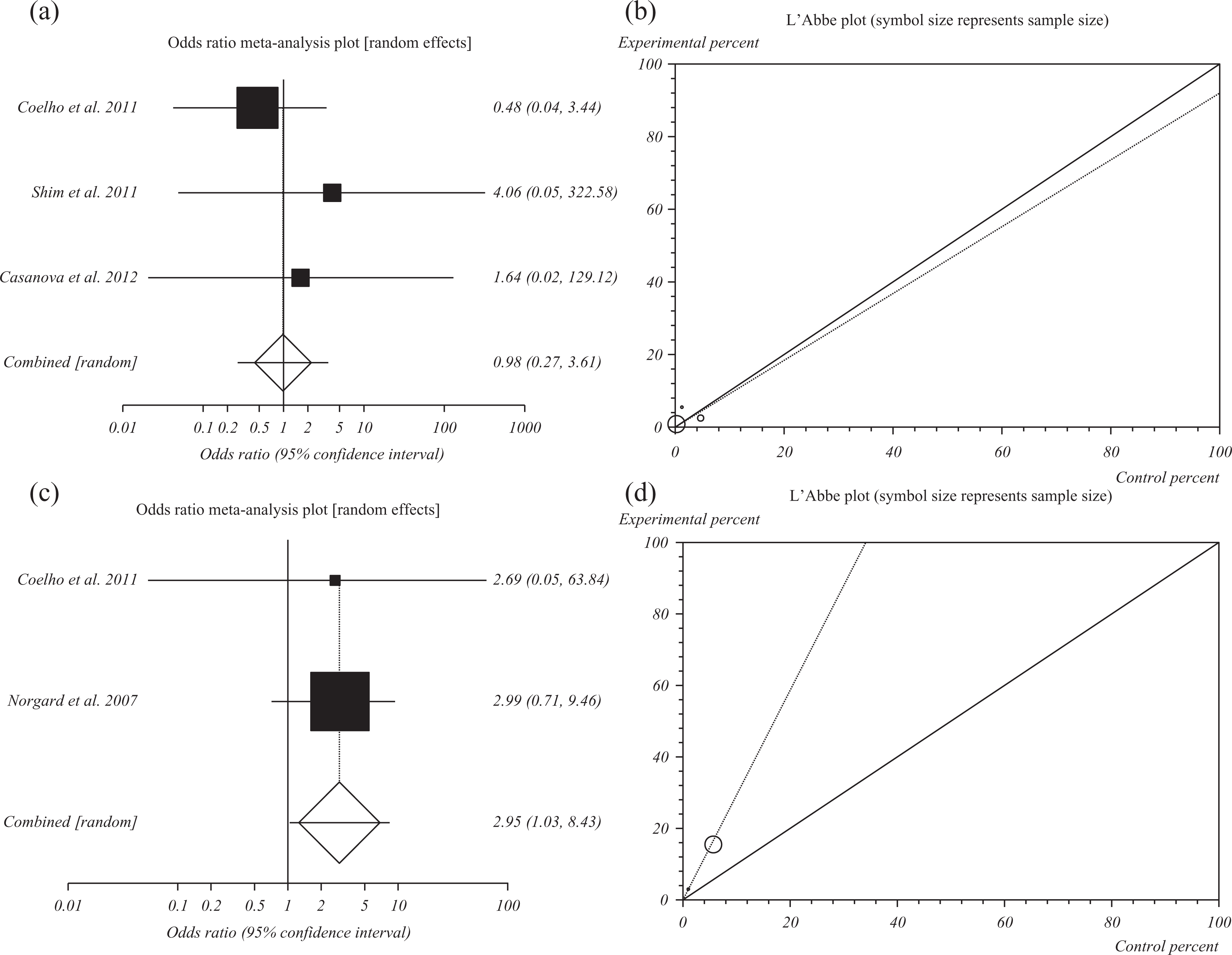
(a) Individual and summary odds ratio for the outcome of “congenital abnormality” for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (b) Heterogeneity indicators for the outcome of congenital abnormality for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (c) Individual and summary odds ratio for the outcome of congenital abnormality for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients. (d) Heterogeneity indicators for the outcome of congenital abnormality for studies including exposure to thiopurines in comparison with no medications in pregnant IBD patients.
The value of p for the Cochrane Q was 0.41 (Figure 4(b)) with I 2 (inconsistency) of 0% (95% CI = 0–72.9%).
Congenital abnormality due to exposure to thiopurines in comparison with no medications in pregnant IBD patients
The summary OR of congenital abnormality for all included data for thiopurines in comparison with no medications in IBD patients (two studies) 30,43 was 2.95 (95% CI = 1.03–8.43; p = 0.04; Figure 4(c)).
The value of p for the Cochrane Q was 0.95 (Figure 4(d)).
Spontaneous abortion due to exposure to thiopurines in comparison with other IBD medications
The summary OR of spontaneous abortion for all included data for thiopurines in comparison with other IBD medications 41,42 was 0.87 (95% CI = 0.29–2.68; p = 0.81; Figure 5(a)).

(a) Individual and summary odds ratio for the outcome of “spontaneous abortion” for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (b) Heterogeneity indicators for the outcome of spontaneous abortion for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy.
The value of p for the Cochrane Q was 0.26 (Figure 5(b)).
Neonatal adverse outcomes due to exposure to thiopurines in comparison with other IBD medications
The summary OR of neonatal adverse outcomes for all included data for thiopurines in comparison with other IBD medications 41,42 was 0.63 (95% CI = 0.2–1.95; p = 0.42; Figure 6(a)).
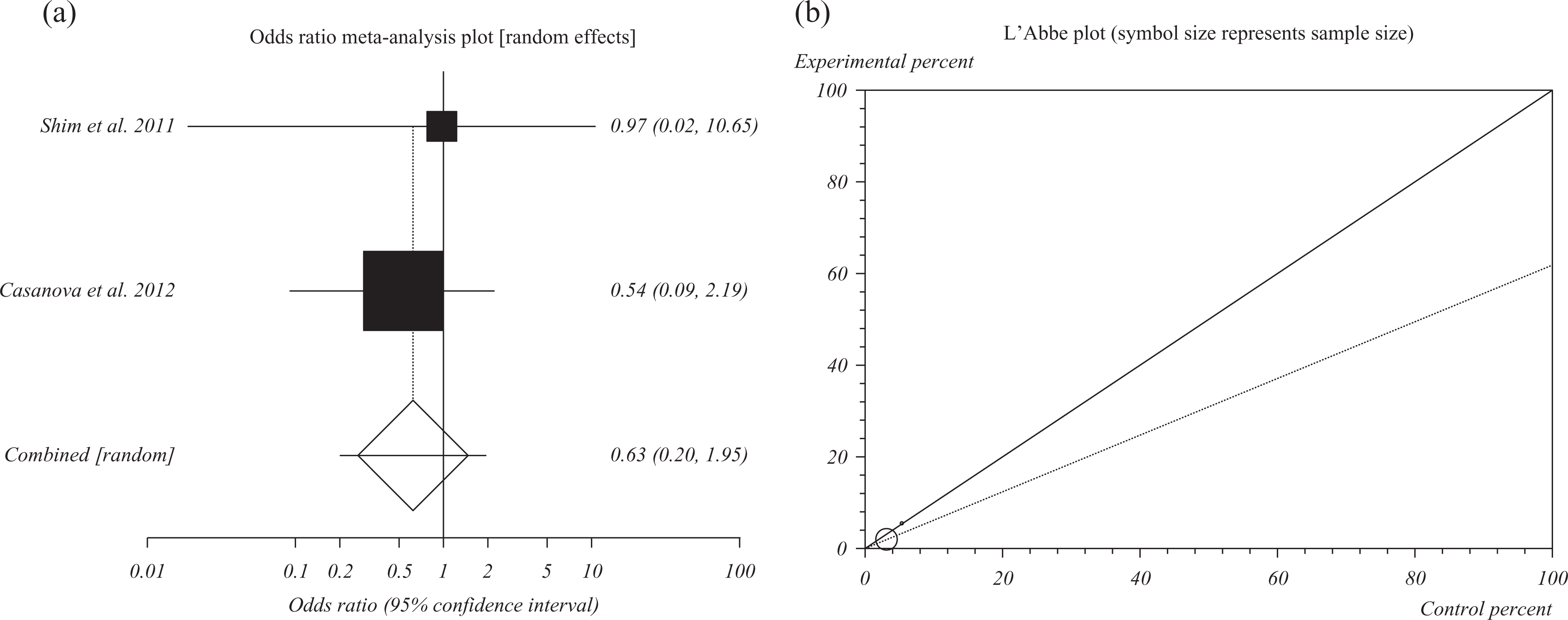
(a) Individual and summary odds ratio for the outcome of “neonatal adverse outcomes” for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy. (b) Heterogeneity indicators for the outcome of neonatal adverse outcomes for studies including exposure to thiopurines in comparison with other IBD medications during pregnancy.
The value of p for the Cochrane Q was 0.65 (Figure 6(b)).
Anti-TNF drugs
The birth outcomes following exposure to anti-TNF drugs in pregnant women with IBD have been reviewed including three eligible cohort studies in which anti-TNF drugs were used 3 months before conception and/or during pregnancy to treat IBD (Table 3). A total of 82 pregnant women with CD and 1 with UC used infliximab from 3 months before conception and during first trimester. Exposure to infliximab did not cause any congenital malformation, intrauterine growth retardation, or low birth weight, except for 11 miscarriages and 16 therapeutic terminations in comparison with 58 live births were reported. 35 In order to evaluate the correlation between use of anti-TNF drugs and pregnancy complications, 318 nonexposed pregnant women with IBD were compared to 66 patients who used anti-TNF drugs alone (n = 29) or in combination with other medicines (n = 37). Infliximab and adalimumab were not associated with any unfavorable pregnancy outcomes, including preterm delivery, low birth weight, congenital malformations, intrauterine growth restriction, intensive care unit admission, and elective abortion. 41 Schnitzler et al. have reported that exposure to infliximab and adalimumab in pregnant women with IBD was not associated with more adverse pregnancy outcomes than IBD. This cohort study involves 196 pregnant women with the mean age of 30 years affected by UC and CD. Gestational age of newborns was significantly decreased. Nevertheless, low birth weight, congenital malformations, and abortion rate remained nonsignificant. 36
Characteristics and outcomes of included studies on the use of anti-TNF-α drugs within 3 months before conception and/or during pregnancy until the second trimester.
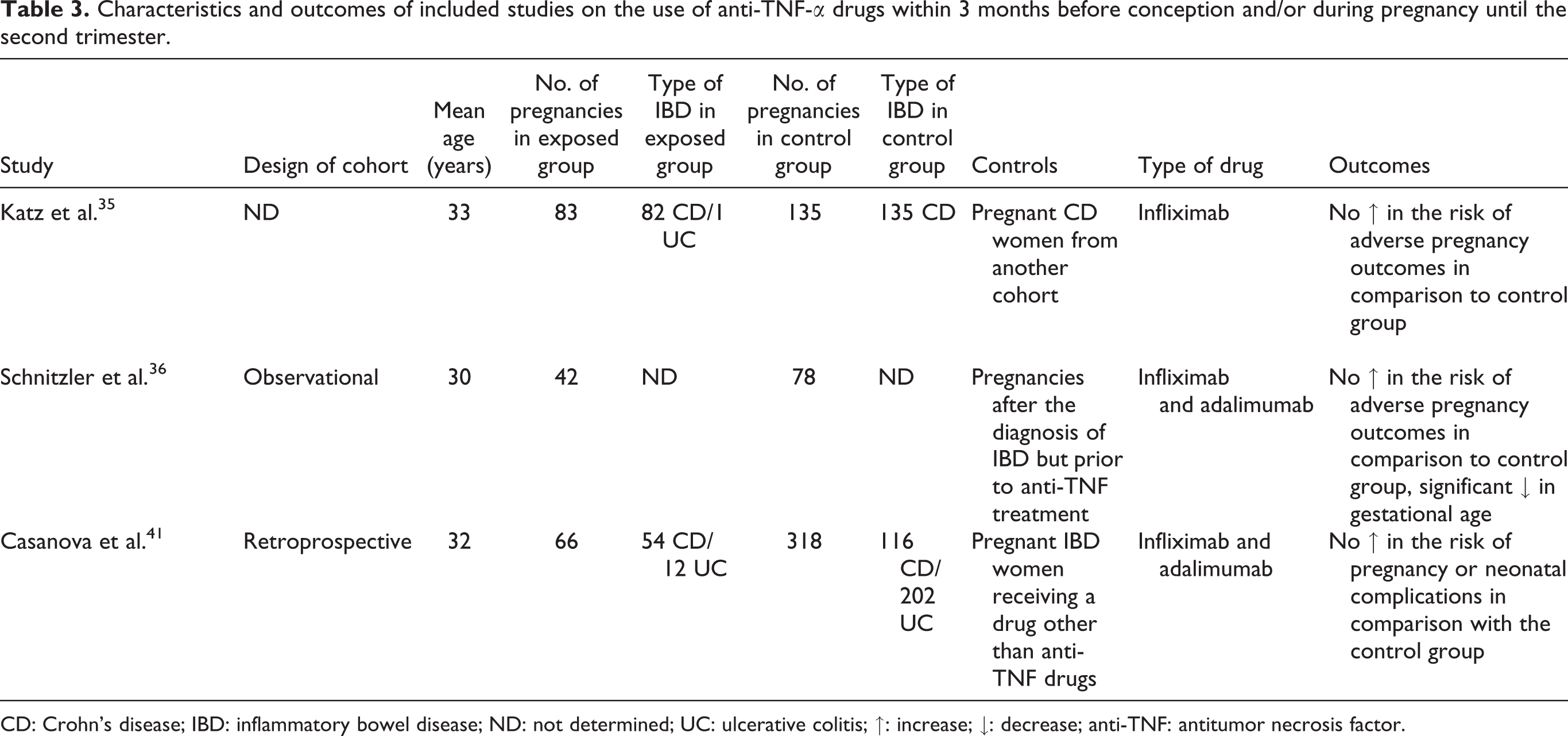
CD: Crohn’s disease; IBD: inflammatory bowel disease; ND: not determined; UC: ulcerative colitis; ↑: increase; ↓: decrease; anti-TNF: antitumor necrosis factor.
In a retrospective observational study in 12 women with IBD (4 UC and 8 CD cases), infliximab exposure during conception and pregnancy did not cause any congenital malformation, intrauterine growth retardation, and low birth weight, except for one case of premature infant. 44
In a retrospective chart review, infliximab was used to treat 10 pregnant women with CD. From 10 participants, 3 prematurity and 1 low-birth weight cases were observed. 45 In another retrospective case-control trial, pregnancy outcomes were investigated according to IBD activity and medical treatment in the same pregnant women before and after IBD diagnosis. Infliximab as one of the common medical treatments was suggested to have no effect on pregnancy outcomes and health status of newborns. 46 One recent study has confirmed the efficacy and safety of infliximab and adalimumab during pregnancy in women with IBD. Among 41 pregnancies, 34 ended in live births. Five spontaneous and two therapeutic abortions occurred. Except six preterm infants and one case of hip dysplasia, no congenital malformations and complications were observed. 47
Discussion
We conducted a meta-analysis and systematic review on pregnancy outcomes following exposure to thiopurines and anti-TNF drugs in women with IBD. A total of 312 pregnant women with CD (n = 236), UC (n = 74), or unclassified IBD (n = 2) who used thiopurines were compared to 1149 nonexposed IBD controls to evaluate the drug effect on different pregnancy outcomes including prematurity, low birth weight, congenital abnormalities, spontaneous abortion, and neonatal adverse outcomes. The thiopurine-exposed group was separately compared with the control group who received other types of drugs or pregnant women who received no drug treatment. The malformation categories and their incidences were also included. The latter reference group received no medication for IBD from 30 days before conception up to the end of third trimester. Results of statistical analysis demonstrated that congenital abnormalities were significantly increased in the thiopurine-exposed group in comparison with the control group. Although the overall incidence risk of congenital malformation was not significantly different in thiopurine-exposed and control groups, the differences in malformation categories or their impact should be taken into account. Table 4 shows the numbers and types of several reported congenital abnormalities in four cohorts of thiopurines treatment during pregnancy.
Types of observed congenital abnormalities in included studies.

A: exposed to thiopurines; B: receiving a drug other than thiopurine; C: not receiving any medication; ND: not determined.
We observed no statistically significant difference in the occurrence of other adverse pregnancy outcomes between compared groups. There was no difference between azathioprine and other drugs including 5-ASA, corticosteroids, and anti-TNF drugs when the pregnancy outcomes between two groups were compared. Comparison of pregnancy outcomes except for congenital abnormalities between thiopurines and control group showed no statistically significant difference. Of course, the incidence risk of prematurity and low birth weight in the thiopurine-treated group was clinically important. This may indicate that the study has kind of insufficient power meaning that increasing the sample size in the compared groups might change this clinical importance as statistically significant and thus could be reported as the adverse effect of thiopurines on pregnancy outcomes. For that reason, there should be a conservative decision on administration of these drugs due to their probable side effects during pregnancy as safety concern is always an issue. Furthermore, small number of participants can be considered as one of limitations in the present meta-analysis, though this is inevitable because conducting clinical trials to assess adverse effects in pregnancy is neither possible nor moral. Several factors such as concerns about safe treatments and self-pregnancy terminations could be considered. Therefore, further cohort studies should be performed to reach more convincing data and results.
We also reviewed the pregnancy outcomes following the use of anti-TNF drugs during pregnancy in women with IBD. The results demonstrated no increase in occurrence of adverse pregnancy outcomes in comparison with controls (Table 3) except for the significant decrease in gestational age of newborns in drug-exposed mothers in one cohort. 36
A previous meta-analysis evaluated pregnancy outcomes in pregnant women with IBD following exposure to 5-ASA drugs. 48 Among different types of medicines prescribed to induce and maintain the IBD patients in remission, thiopurines and anti-TNFs were identified more effective. Generally, some studies with limited sample sizes introduced use of thiopurines and anti-TNF drugs safe during pregnancy. However, it has been recommended not to continue infliximab after the gestational week 30. 41,49
In a meta-analysis performed by Cornish et al., the influence of IBD on pregnancy outcomes was evaluated. The results indicated no significant increase in the incidence of adverse pregnancy outcomes such as still birth, spontaneous abortion, and low birth weight in newborns following continuing drug therapy including thiopurines and anti-TNFs. They reported an increase in the number of congenital abnormalities and suggested the link of increased risk to IBD itself, not the drug. 1
The present meta-analysis shows an increase in the risk of congenital malformations in thiopurines-exposed group in comparison with healthy women. Conversely, this difference was not significant when compared with the IBD controls that were not exposed to thiopurines. Therefore, thiopurines may have a minimal risk to infants but might be critical for IBD management. 50
Akbari et al. have recently published a similar meta-analysis on the effects of thiopurines on birth outcomes. They included both exposed male and females with IBD. They found an association between the use of thiopurines during pregnancy in IBD women with preterm birth. Despite our results, they did not report any effect on birth weight of newborns or congenital abnormalities. 51 Of course, that study’s inclusion criteria was different from ours as we included one more cohort study. 41 The other difference was that they included two studies, 27,49 while we recognized them bias and thus excluded. The latter study by Zelinkova et al. 49 compared pregnant women to women with a delivery not having received azathioprine during conception or pregnancy. In order to consider the similar disease condition in exposed women, they compared the results to an IBD pregnant group who used medications other than azathioprine. Regarding the importance of disease activity, IBD women who were not on medication for remission were excluded from the study. Francella et al. reported no increase in adverse birth outcomes following 6-mercaptopurine treatment in a retrospective cohort of 171 pregnant women with IBD. However, the study reported the birth outcomes following both maternal and paternal exposure to 6-mercaptopurine whereas it was excluded from our meta-analysis. 28
In a prospective cohort, azathioprine prescription in pregnant women led to a decrease in birth weight, gestational age, and made an increase in occurrence of prematurity but birth defects. It should be noted that 83 participants had IBD. We excluded this cohort because IBD women data were not reported separately. 52 Two other cohorts evaluated the effects of azathioprine and 6-mercaptoprine during pregnancy for treatment of different diseases including IBD. When the comparison was restricted to controls with the same underlying disease, the increase in the risk of preterm birth was significant following thiopurines exposure during pregnancy. 51 Low birth weight, congenital malformation, and preterm birth were observed in pregnant women who consumed thiopurines during pregnancy. 31,53
A long-term follow-up study has been performed in children with in utero exposure to thiopurines due to maternal IBD. Maternal exposure to thiopurines during pregnancy was not associated with long-term development or immune function of children. 54 In order to assess adverse pregnancy outcomes in IBD women, Dotan et al. concluded that besides the significant higher incidence of low body weight and congenital abnormalities in IBD women than healthy controls, IBD women receiving medication during pregnancy showed a higher risk for preterm birth and infants with lower birth weight in comparison with pregnant IBD women who was not on medication. 55
A review of the safety of anti-TNFs during pregnancy based on FDA database reported a higher number of congenital malformations such as vertebral abnormalities, anal atresia, cardiac defect, tracheoesophageal, renal, and limb abnormalities (VACTERL) in newborns exposed to infliximab and etanercept in comparison with historical control data. 56 A clinical review suggested that knowledge about the extent of placental transfer of anti-TNFs in each pregnancy stage could be helpful in moderating decision. 57 Zelinkova et al. determined the intrauterine exposure to infliximab during pregnancy in IBD patients. They reported that before gestational week 30, infliximab use led to higher fatal exposure than maternal peripheral blood. Although they did not observe any complications, they presented the concern about its high concentration in long term on the development of immune system in infants. 49 However, current opinion regarding the increased risk of active UC and CD during pregnancy is to control the relapse rate by use of drug therapy such as anti-TNFs. 1,58
Evidence supports an increased risk of adverse pregnancy outcomes in women with IBD. It has been demonstrated that disease activity may increase the risk of low birth weight and prematurity in comparison with healthy women. 1,46,59 Several studies have reported the harmful effect of disease relapse during pregnancy on the infant in an extent more than continuing maintenance therapy. Therefore, an effective medical treatment is of great importance and necessary during pregnancy. However, there is a great concern about safe choices during pregnancy in IBD patients. 1,27,59 –61
Few cohorts and small sample sizes limit us to make an absolute conclusion about safety of these drugs during pregnancy. In the present study, we have reported OR to warn practitioners or researchers about chance of an incidence but the risk relevancy. Absolutely, more reliable information can be presented if more clinical and epidemiological studies would be available.
Noteworthy in comparing data, not all studies have included disease activity. In fact, grading disease activity in involved patients may be impossible for researchers due to lack of information in retrospective cohorts and use of different proxies for diseases activity such as hospitalization, surgery, and the medications like immunomodulators and thiopurines. In addition, most of the involved study populations suffered from CD who have different response to biological therapies comparing UC 62 and thus might influence the results according to disease activity. Cleary et al. excluded IBD women who did not report use of IBD medications in early pregnancy considering this group as patients with inactive disease. 63 Nørgård et al. have also stated that their reference group who received no IBD medicine was likely to present no disease activity. They considered disease activity through definition of drug subcohorts and adjusting to number and length of admissions for CD during pregnancy. However, they have mentioned that there is residual confounding by disease activity that may have been left in their analyses. 43
However, some studies included both healthy and same underlying disease groups as control groups. In addition, the distinct dose of drugs used for each participant was not clear, and the outcomes were not reported separately according to the type of IBD (CD or UC).
Since all IBD drugs are not analyzed for their safety during pregnancy, further cohort studies on safety of more several medicines during pregnancy would be valuable in addition to follow-up visits of newborns to evaluate the risk of adverse pregnancy outcomes such as prematurity and low birth weight for consequent complications in infants and its correlation to the cord blood concentration of drugs. Moreover, to execute valuable and helpful studies, specifying IBD subtypes (UC and CD) with their associated drugs would be of value. In addition, as mentioned earlier, the disease activity in included participations should be considered to decrease confounders.
Taking collectively, a full safe drug therapy cannot be introduced for IBD patients during pregnancy and thus the benefit–risk ratio should be always considered by physicians.
Footnotes
Acknowledgment
This paper is the outcome of an in-house financially non-supported study. The authors thank the National Elite Foundation for supporting the first author's postdoc program.
Authors’ contribution
SM drafted the manuscript, collected the data, and prepared the tables. AHA helped in bibliography and drafting the manuscript. SN did the meta-analysis, wrote, and reviewed the article. MA gave the idea, supervised the whole study, edited, and reviewed the article. All authors have read and approved the final version of the article.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
