Abstract
The present study was designed to investigate the cardioprotective effects of betaine on acute myocardial ischemia induced experimentally in rats focusing on regulation of signal transducer and activator of transcription 3 (STAT3) and apoptotic pathways as the potential mechanism underlying the drug effect. Male Sprague Dawley rats were treated with betaine (100, 200, and 400 mg/kg) orally for 40 days. Acute myocardial ischemic injury was induced in rats by subcutaneous injection of isoproterenol (85 mg/kg), for two consecutive days. Serum cardiac marker enzyme, histopathological variables and expression of protein levels were analyzed. Oral administration of betaine (200 and 400 mg/kg) significantly reduced the level of cardiac marker enzyme in the serum and prevented left ventricular remodeling. Western blot analysis showed that isoproterenol-induced phosphorylation of STAT3 was maintained or further enhanced by betaine treatment in myocardium. Furthermore, betaine (200 and 400 mg/kg) treatment increased the ventricular expression of Bcl-2 and reduced the level of Bax, therefore causing a significant increase in the ratio of Bcl-2/Bax. The protective role of betaine on myocardial damage was further confirmed by histopathological examination. In summary, our results showed that betaine pretreatment attenuated isoproterenol-induced acute myocardial ischemia via the regulation of STAT3 and apoptotic pathways.
Introduction
Ischemic heart disease, a health problem of global concern, is characterized by a reduced blood supply (ischemia) to the heart muscle. A disparity between the oxygen requirement of the myocardium and the ability of the coronary artery to meet the oxygen need results in myocardial infarction. 1 Recent studies have demonstrated that numerous complex signaling pathways are initiated by the ischemic myocardium, and these signaling can either mediate protective responses or activate cell death events. 2
Signal transducer and activator of transcription 3 (STAT3) pathway has been shown to be an integral part of the cardioprotection. 3,4 It is one of the families of receptor-associated cytosolic tyrosine kinases that phosphorylate a tyrosine residue in cognate of STATs. 4 Activation of STAT3 in the heart has been observed in acute myocardial infarction and ischemic preconditioning, respectively, 5,6 knocking out the STAT3 gene induces myocardial ischemia in mice, 7 and compounds such as melatonin, 8 rapamycin, 4 opioids 9 and insulin 10 suppressed myocardial ischemia/reperfusion injury via increasing phospho-STAT3 expressions. In addition, apoptosis is also well known to play a key role in cell death after myocardial ischemia, and the relative ratio Bcl-2 and Bax in the cell determines to a large extent whether the cell initiates survival or apoptosis. 11 It has been reported that a decreased ratio of Bcl-2/Bax was exhibited in myocardial ischemia/reperfusion in rats. 12 Thus, activation of STAT3 pathway and modulation of Bcl-2/Bax expression may have a protective role in the treatment of myocardial ischemic injury. Betaine, a naturally occurring product with a molecular formula of C5H11NO2 (Figure 1), which derived from various varieties of plants, animals, and microorganisms, 13,14 is a methylated amino acid having a dual role as an osmolyte and as a methyl group donor in vertebrates. 15 Experimental studies have shown that long-term administration of betaine exerted a significant cardioprotective effects against isoproterenol-induced myocardial injury through suppression of oxidative stress, 16 maintenance of mitochondrial and lysosomal function, 17,18 and regulation of protein, glycoprotein, and lipid metabolism. 19,20 These results suggested that betaine might be a potential drug for antimyocardial ischemic injury. However, the signaling mechanisms in betaine-induced protection against myocardial ischemia remain poorly understood. The present study attempted to explore and evaluate the molecular mechanisms leading to the cardioprotective effects of betaine.

Chemical structure of betaine.
It is well documented that subcutaneous injection of isoproterenol, a synthetic catecholamine and β-adrenoceptor agonist, at supramaximal doses induced acute irreversible myocardial ischemia in rats, which pathophysiologically and morphologically resembles myocardial injury in human. 21 –23 Therefore, the aim of this work was to investigate the cardioprotective effects of betaine on the regulation of the STAT3 and apoptotic pathway.
Materials and methods
Drugs and chemicals
Betaine (white powder, purity >99%) was purchased from Johnson Matthey Company (London, UK), lot number was C8435A. Isoproterenol hydrochloride was bought from Tokyo Chemical Industry Co. Ltd (Tokyo, Japan), lot number was MVS7F-NF. Rat cardiac troponin I (cTn I; CSB-E08594r) enzyme-linked immunosorbent assay kit was purchased from Cusabio Biotech Co. Ltd (Newark, New Jersey, USA). Total protein extraction kit and bicinchoninic acid (BCA) protein quantization kit were bought from Ken Gen Biotech. Co. Ltd (Nanjing, China). Primary antibodies to STAT3, Bcl-2, and Bax were purchased from Cell Signaling Technology (Boston, Massachusetts, USA). Antibodies to phosphorylated STAT3 (phospho-STAT3; Tyr705) were bought from Abcam (Cambridge, UK). Primary antibody to β-tubulin was purchased from Proteintech (Chicago, Illinois, USA). Primary antibodies to β-actin and secondary antibody of horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin G were bought from ZSGB-BIO (Beijing, China). Other reagents used were of commercial analytical grade.
Experimental animals
Male Sprague Dawley rats (5–6 weeks old) of 100–120 g body weight, supplied by Ningxia Laboratory Animal Centre (Yinchuan, China), were selected for the experiment. All rats were housed in independent ventilation cages (with stainless steel grill top, 48 × 35 × 20 cm3) under hygienic and standard environmental conditions .The animals were under natural light and dark cycle at 22°C and 50 ± 10% humidity for 7 days before the experiment. The rats were allowed free access to food and water. All protocols and surgical procedures were approved by the Institute of Animal Care and Use Committees of Ningxia Medical University (No. 006/2013).
Induction of myocardial injury
The myocardial injury was performed as previously described. 24 Isoproterenol hydrochloride was dissolved in normal saline and injected subcutaneously to the upper back of rats (85 mg/kg) daily for two consecutive days to induce experimental myocardial ischemia.
Experimental groups
Rats were randomized into six experimental groups as follows: Control group, rats were orally administered with 10 ml/kg physiological saline for 40 days; isoproterenol treatment group, rats were given with 10 ml/kg oral administration of physiological saline for 40 days and at the 39th day were subcutaneously injected with isoproterenol (85 mg/kg dissolved in saline, once a day at an interval of 24 h for two consecutive days); betaine pretreatment groups, rats received betaine (100, 200, and 400 mg/kg, gastric gavages, respectively) for 40 days and then at the 39th day were subcutaneously injected with isoproterenol (85 mg/kg dissolved in saline, once a day at an interval of 24 h for two consecutive days); and betaine alone, rats were given betaine (400 mg/kg) for 40 days.
Serum cTnI estimations
After 24 h from the last drug administration, rats were anesthetized with ethyl carbamate. The femoral artery blood was collected; the serum was separated and stored at −20°C freezer. According to the manufacturer’s instruction, enzyme-linked immunosorbent assay was performed for the determination of serum cTnI by Bio-Rad 680 automatic microplate reader (Berkeley, California, USA).
Tissue preparation
The heart and left ventricle were rapidly dissected and weighed after blood collection, and the heart/body weight and left ventricle/body weight, as an index of myocardial edematous were calculated. Then, the cardiac apex was excised immediately for histopathological studies. Finally, the left ventricle was frozen at −80°C for protein assays.
Histopathological studies
Hematoxylin and eosin (H&E) staining was used for morphometric measurement. The cardiac apex obtained from all experimental groups were excised and fixed in 4% buffered paraformaldehyde solution. Tissues were embedded in paraffin, sectioned at 4 µm thickness, and stained with H&E. The sections were examined under light microscope, and then photomicrographs were taken. For each section, five random noncontiguous microscopic fields were examined. As described before by Loh et al., 25 they were graded for severity of changes using scores on scale of no abnormal findings (–), mild (+), moderate (++), and severe (+++). Scoring was done on coded samples by an experienced pathologist in a blinded manner.
Western blot analysis
The frozen left ventricle was homogenized in cold lysis buffer using total protein extraction kit and then subjected to sonication for 5 s on ice (repeated three times), followed by centrifugation at 4°C for 5 min at 10,000g to remove cellular debris. The protein concentration was determined using a BCA protein assay reagent kit with bovine serum albumin as a standard. Equivalent amounts (50 or 60 μg) of protein samples were loaded and separated on 7.5% or 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then electrophoretically transferred to nitrocellulose membranes using an electrophoretic transfer system (Bio-Rad). Membranes were subdivided, and each protein of interest and β-actin or β-tubulin were analyzed from a single transfer. The membranes were blocked for 1 h in phosphate-buffered saline Tween 20 containing 5% nonfat milk at room temperature, then incubated with primary antibody of phospho-STAT3(Tyr705; 1:10,000 dilution), STAT3 (1:1000 dilution), Bcl-2 (1:500 dilution), Bax (1:500 dilution), β-actin (1:1000 dilution), and β-tubulin (1:2000 dilution) overnight at 4°C, followed by respective horseradish peroxidase-conjugated secondary antibodies for 2 h. Bands on blots were visualized using Super Signal West Pico Chemiluminescence kit (Thermo Scientific, Rockford, Illinois, USA, and finally were exposed to radiographic films. The films were scanned, and quantization of protein band density normalized to β-actin or β-tubulin was measured using the Quantity One software (Bio-Rad Laboratories).
Statistical analysis
The data were analyzed using the Statistical Package for Social Sciences version 11.5 software (Chicago, Illinois, USA). Statistical analyses of observed values were performed using one-way analysis of variance or nonparametric test (k-independent sample test). The value of p < 0.05 was accepted as statistically significant.
Results
Effects of betaine on serum level of cardiac injury marker enzyme
As compared to the control group, isoproterenol-treated rats showed a significant rise in serum cTnI. Betaine (200 and 400 mg/kg) lowered the level of cTnI (p < 0.05; Figure 2). There were no differences between control group and betaine (400 mg/kg) only group (p > 0.05).

Effects of betaine on serum content of cTnI from isoproterenol-induced myocardial ischemia in rats. Values are presented as mean ± SD (n = 6). # p < 0.05; ## p < 0.01: compared with control. *p < 0.05; **p < 0.01: compared with isoproterenol. cTnI: cardiac troponin I.
Effects of betaine on body and heart weight
Heart/body weight and left ventricle/body weight from isoproterenol-treated rats were significantly higher than that of the control group, whereas final body weight was markedly decreased. These parameters tended to be significantly improved by oral administration of betaine (200 and 400 mg/kg; Table 1).
Effects of betaine on body and heart weight in isoproterenol-induced myocardial ischemia in rats (mean ± SD).

a p < 0.05: compared with control.
b p < 0.01: compared with control.
c p < 0.05: compared with isoproterenol.
d p < 0.01: compared with isoproterenol.
Histopathological examination of cardiac tissues
The effects of betaine against acute myocardial injury were measured and evaluated by H&E staining (Figure 3 and Table 2). Histopathological findings of the isoproterenol-induced myocardium showed severe subendocardial necrosis, infiltration of inflammatory cells and interstitial edema as compared to the control group. Oral administration of betaine (200 and 400 mg/kg, respectively) demonstrated relative degrees of improvement in subendocardial necrosis (p < 0.01) and infiltration of inflammatory cells (p < 0.05). The section of myocardial tissues in rats that received betaine (400 mg/kg) only showed no change as compared to the control group.

Effects of betaine on isoproterenol-induced myocardial injury. Representative images of H&E (×200)-stained cardiac apexes are shown. (a) Control; (b) isoproterenol; (c) betaine (100 mg/kg) + isoproterenol; (d) betaine (200 mg/kg) + isoproterenol; (e) betaine (400 mg/kg) + isoproterenol; and (f) betaine (400 mg/kg). H&E: hematoxylin–eosin.
Effects of betaine on the degree of histopathological changes in cardiac apexes.a

aThe figures represent the number of rats affected. Photomicrographs were used to evaluate the damage in the cardiac apexes, and the histopathological changes were arbitrarily scored as follows: −, no abnormal findings; +, mild; ++, moderate; and +++, severe.
b p < 0.01: compared with control.
c p < 0.01: compared with isoproterenol.
d p < 0.05: compared with isoproterenol.
Effects of betaine on ventricular phospho-STAT3 and STAT3 protein expression
Compared with the control group, expression of phospho-STAT3 (Tyr705) was significantly increased, and expression of STAT3 was not significantly changed in the isoproterenol-treated rats. Betaine (400 mg/kg) increased the expression phospho-STAT3 (Tyr705; p < 0.05; Figure 4). There were no differences between control group and betaine (400 mg/kg) alone group (p > 0.05).
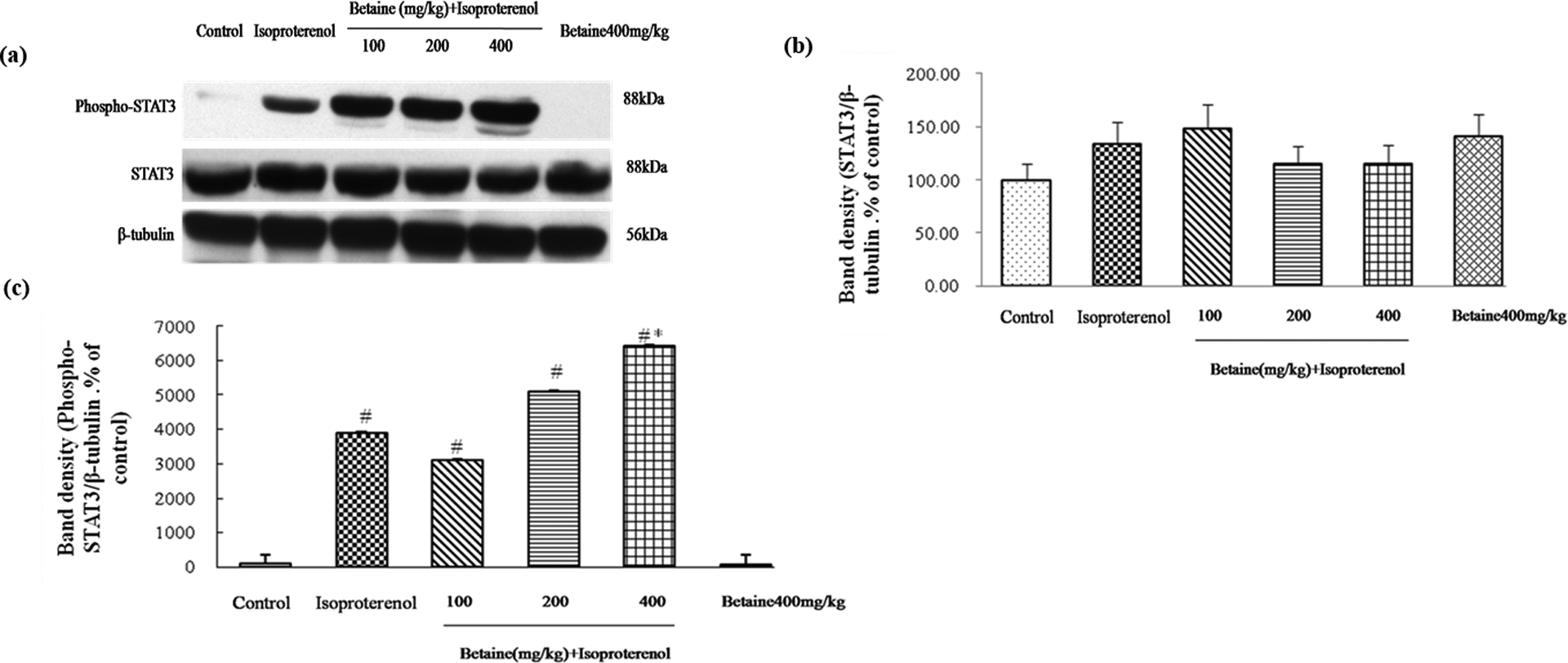
Effects of betaine on expression of STAT3 and phospho-STAT3 (Tyr705) in hearts from isoproterenol-induced myocardial ischemia in rats. (a) Represents western blot analysis of STAT3 and phospho-STAT3 (Tyr705), β-tubulin is shown as loading control. Quantitative data of STAT3 and phospho-STAT3 (Tyr705) signals were shown as percentages of the value of control rats (b and c). Values are represented as mean ± SD (n = 6). # p < 0.05: compared with control. *p < 0.05: compared with isoproterenol.
Effects of betaine on ventricular Bcl-2 and Bax expression
Compared with the control group, the expression of Bcl-2 significantly decreased, whereas the expression of Bax markedly increased in the isoproterenol-treated rats. Betaine (400 mg/kg) markedly increased the level of Bcl-2 expression and betaine (200 and 400 mg/kg) significantly decreased Bax expression. Compared with the control group, the ratio of Bcl-2/Bax significantly decreased in the isoproterenol-treated rats (p < 0.01). Betaine (400 mg/kg) elevated the isoproterenol-induced decrease in the ratio of Bcl-2/Bax levels (p < 0.05).There were no differences between the control group and betaine (400 mg/kg) alone group (p > 0.05; Figure 5).
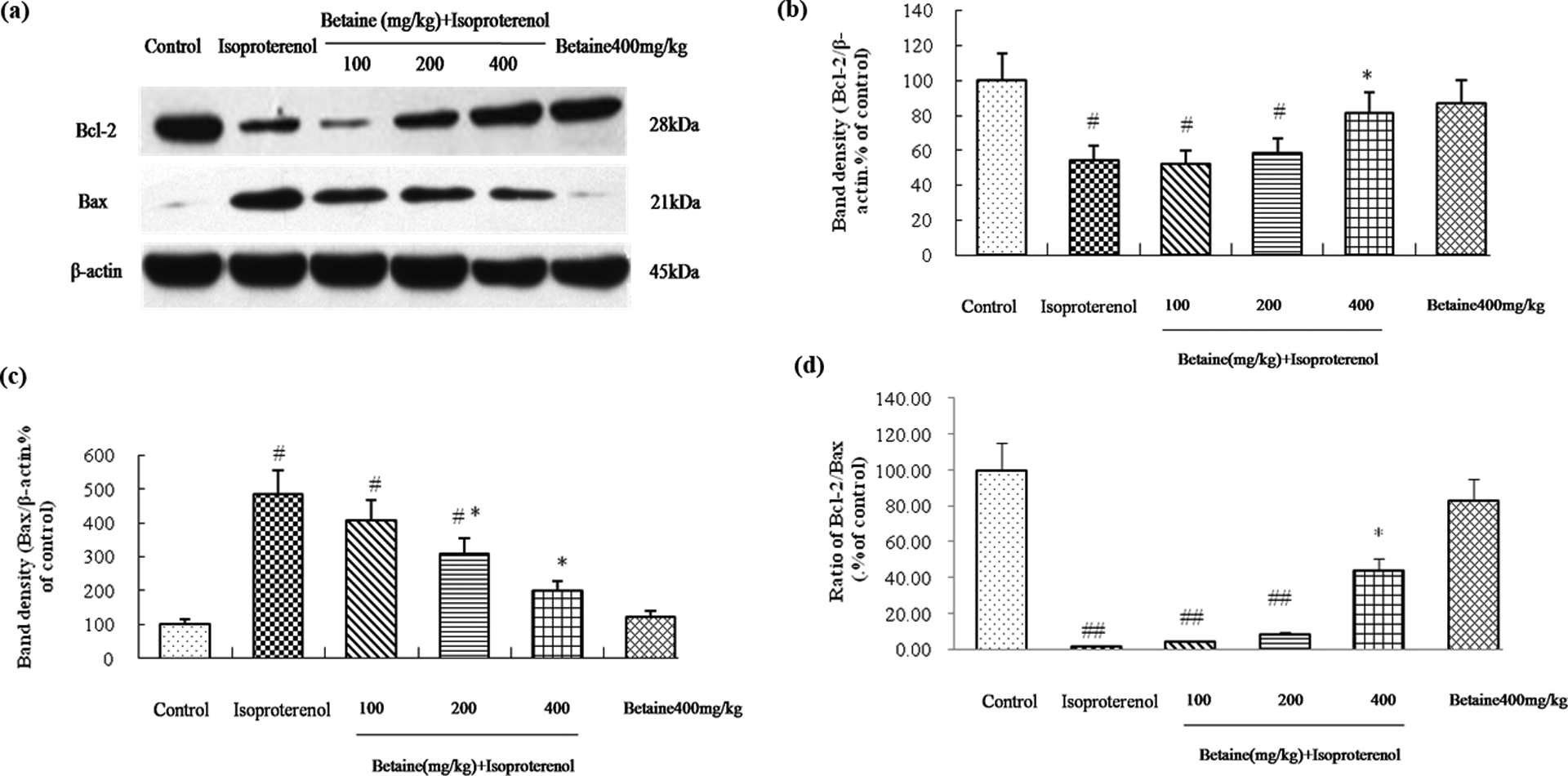
Effects of betaine on expression of Bcl-2 and Bax in hearts from isoproterenol-induced myocardial ischemia in rats. (a) Represents Western blot analysis of Bcl-2 and Bax, β-actin is shown as loading control. Quantitative data of Bcl-2, Bax, and ratio of Bcl-2/Bax signals were shown as percentages of the value of control rats (b, c, and d). Values are represented as mean ± SD (n = 8). # p < 0.05; ## p < 0.01: compared with control. *p < 0.05: compared with isoproterenol.
Discussion
Myocardial infarction is experimentally caused by occluding coronary vessels or administering chemical substances. One frequently used model consists of ligaturing the descending branch of the left coronary vessel in anesthetized animals. 26,27 However, this method of inducing myocardial ischemia is invasive and require open chest surgery and dedicated instrumentation. Alternatively, noninvasive animal models have been used to mimic the pathophysiological characteristics of myocardial injury. It is well documented that rats treated with low (0.02–3.0 mg/kg) doses of isoproterenol was a useful model to assess the mechanisms of myocardial growth and the progression of cardiac diseases, 28 whereas subcutaneous injection of very large (20–100 mg/kg) doses of isoproterenol to experiment animals were found to induce subendocardial myocardial ischemia, hypoxia, necrosis, and finally fibroblastic hyperplasia, and the pathophysiological and morphologic alterations in the heart of this noncoronary myocardial necrotic rat model were comparable with those taking place in human myocardial infarction. 21,22,28,29 In our study, acute myocardial injury was induced in rats by subcutaneous injection of 85 mg/kg of isoproterenol daily for two consecutive days, 21 –24,29 and the efficacy of betaine on protecting the heart against myocardial infarction were observed on this noninvasive myocardial necrotic rat model.
Our study demonstrated that betaine administration for 40 days was effective in myocardial injury in isoproterenol-treated rats. The major findings of this study were that betaine protected against isoproterenol-induced myocardial ischemia via regulation of STAT3 and apoptotic pathways.
cTnI and cTnT that were released from myocardium in proportion to the degree of myocardial ischemia have become well established as blood biomarkers for cardiac injury. 30 The previous studies demonstrated 31 that cTnI was a sensitive and specific biomarker of cardiac injury in laboratory animals, because the amount of cTnI released and detected with injury is substantially more than for cTnT. Thus, serum cTnI is used as the gold standard serum biomarker for cardiotoxicity in clinical examinations and in laboratory animals. 32,33 The significantly increased level of this enzyme indicated the severity of isoproterenol-induced myocardial infarction. 34,35 In line with the previous findings, our results showed significant elevation in the levels of cTnI in serum of isoproterenol-injected rats. Oral administration of betaine (200 and 400 mg/kg) for 40 days markedly reduced the elevation of cTnI induced by isoproterenol, indicating that betaine attenuated isoproterenol-induced acute myocardial injury.
In the present study, isoproterenol caused a significant increase in heart/body weight and left ventricle/body weight as reported in previous studies, 36 and these increases appeared to be a hypertrophic response. Treatment with betaine lowered the heart to body weight and left venticle to body weight ratio, and these data confirmed that betaine offered preventive effects on isoproterenol-induced myocardial ischemia featured as heart and left ventricular hypertrophy.
To better understand the protective effect of betaine on acute myocardial injury, cardiac histopatholgical examination was evaluated. A number of investigations have suggested that supramaximal doses of isoproterenol produce myocardial necrosis. 22 –25,29,37 In line with the previous studies, the present study showed that isoproterenol-induced myocardial ischemia in rats demonstrated subendocardial necrosis, infiltration of inflammatory cells, and interstitial edema, which indicated the presence of cardiac ischemic injury. Betaine (100, 200, and 400 mg/kg) significantly ameliorated the histological damage of the hearts as evidenced by the reduction of subendocardial necrosis and inflammatory cells in the myocardium, indicating that betaine exerted pronounced protective effects on myocardial ischemia.
STAT3 pathway is now recognized as an important membrane-to-nucleus signaling pathway for a variety of stress responses including ischemia, hypoxia, and oxidative stress. 38 In response to ischemic stress, cytosolic STAT monomers occur followed by tyrosine phosphorylation and dimerization. The STAT dimers readily translocate to the nucleus, bind to the promoter regions of the DNA, and regulate transcription of genes. 39 The previous studies showed that activation of the STAT3 pathway during ischemia/reperfusion injury protected against ischemic stress. 40,41 It is well documented that treatment with isoproterenol induce cardiac hypertrophy and subendocardial myocardial ischaemia. Increased phospho-STAT3 (Tyr705) protein levels were observed in isoproterenol-induced hypertrophic H9c2 cardiomyoblast cells and neonatal rat cardiomyocyte, and treatment of mice with isoproterenol (15 mg/kg body weight, intraperitoneal) also markedly increased phospho-STAT3 (Tyr705) protein in myocardium. 42 –44 Our present study found that treatment of rats with isoproterenol (85 mg/kg body weight, subcutaneous) for 2 days markedly increased phospho-STAT3 (Tyr705) in myocardium without altering the protein abundance of STAT3. Oral administration of betaine (100 and 200 mg/kg) maintained the high level of phospho-STAT3 (Tyr705) in isoproterenol-induced myocardium, and betaine at a dosage of 400 mg/kg further increased this protein expression. These findings suggested that betaine may protect against myocardial injury via STAT3 pathway.
It has been reported that phospho-STAT3 dimerizes and translocates from the cytoplasm into the nucleus to induce transcription of target gene Bcl-2. 45,46 Bcl-2 is required for the survival of mature cells, and the pro-survival activity of Bcl-2 has been clearly demonstrated in overexpression. 47 In addition, Bax, a soluble monomeric protein which mainly resides in the cytosol of healthy cells, also plays a key role in cardiomyocyte apoptotic processes. 48 The previous studies showed that the decreased gene or protein expression of myocardial Bcl-2 as well as the increased expression of myocardial Bax were observed in isoproterenol-induced myocardial ischemia. 35,49 In line with the previous findings, our study showed that Bcl-2 protein expression was markedly decreased, whereas Bax protein expression was increased, thus, the ratio of Bcl-2/Bax significantly decreased in the heart of isoproterenol-induced myocardial ischemia. Oral administration of betaine (200 and 400 mg/kg) attenuated the reduction of isoproterenol-induced Bcl-2 protein, the elevation of Bax protein, and increased the ratio of Bcl-2/Bax. All these results indicated that betaine inhibited isoproterenol-induced cardiomyocyte apoptosis through upregulation of Bcl-2, downregulation of Bax, and elevation of the ratio of Bcl-2/Bax.
In summary, the present study provided experimental evidence that long-term oral administration of betaine offers protection to acute myocardial ischemia. The mechanism responsible for this beneficial effect may involve regulation of STAT3 and apoptotic pathways. These findings might be rational to understand the cardioprotective effects of betaine on myocardial injury and to provide a preventive potential for future pharmacological management of myocardial ischemic diseases.
Footnotes
Authors’ note
Authors P Zheng and J liu contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of People’s Republic of China [grant no. 81160403].
