Abstract
Nonsteroidal anti-inflammatory drugs that are cyclooxygenase (COX) enzyme inhibitors have generally been used in short-term pain management and also to treat inflammation chronically. It is known that COX enzyme and prostaglandins play important roles in the regulation of reproductive functions in females. However, there are relatively few studies for the male reproductive system, and the results of these studies are contradictory. In this study, sperm count and motility, COX-1, COX-2, prostaglandin E1 (PGE1), prostaglandin E2 (PGE2), and prostaglandin F2α (PGF2α) levels in testis tissue, plasma follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone levels, and histopathological examination of testis tissue were evaluated after naproxen sodium and meloxicam administration in male rats. Also, testis superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx), and glutathione (GSH) levels were measured to investigate the oxidation status. According to our results, sperm count and motility were significantly decreased in treatment groups. Plasma hormone levels did not show any statistical differences between the groups. COX-1, PGE2, and PGF2α levels were significantly decreased, while the decreases in COX-2 and PGE1 levels did not show any significance statistically. Testis SOD, catalase, GPx, and GSH levels were decreased significantly. According to the results of histopathological examination, damage in seminiferous tubules, where spermatogenesis developed, was observed. In conclusion, naproxen sodium and meloxicam decreased the sperm count and motility and also induced the damage of seminiferous tubules as a direct effect without affecting plasma hormone levels in our study. The mechanism of the reproductive toxicity induced by these agents may be based on the inhibition of prostaglandin synthesis and the induction of oxidative stress can be emphasized as a secondary factor.
Introduction
Cyclooxygenase (COX) is the enzyme that mediates the formation of prostaglandins from arachidonic acid. The enzyme has two main isoforms, COX-1 and COX-2. Although COX-1 is constitutively found in most tissues, COX-2 level is known to increase in the inflammatory processes in the tissues. 1 COX-1 enzyme is involved in thromboxane A2 (TXA2) production as well as prostaglandin E2 (PGE2) production by the kidney and prostacyclin (PGI2) production that has a protective effect on gastric mucosal cells. 2 On the other hand, COX-2 enzyme is synthesized in response to pro-inflammatory and mitogenic factors including cytokines, bacterial endotoxins, interleukins, and tumor necrosis factor and is involved in the synthesis of prostaglandins which play a role in inflammatory processes. 3,4
Previous studies have demonstrated that prostaglandins and COX enzymes play an important role in human reproduction. Although prostaglandins and COX enzymes are known to have many key roles in the female reproductive system including ovulation, fertilization, and labor, their role in the male reproductive system is contradictory and remains to be elucidated. 5 –7 Female mice lacking COX-1 and prostaglandin F2α (PGF2α) have been reported to be fertile but have difficulty in labor, whereas those lacking COX-2 have been observed to be infertile. Thus, COX-2 enzyme has been suggested to play a major role in female ovulation. 7,8 On the other hand, fertility has not been affected in male mice with COX-1 or COX-2 enzyme mutations, indicating that prostaglandins may have no crucial role in the regulation of normal testicular functions in male mice. 8,9 Accordingly, COX-2 enzyme has been demonstrated to be expressed in testes with impaired spermatogenesis and morphological changes but not in healthy testes. 4,7,10 Also, Hase et al. reported that COX-2 as well as COX-1 are synthesized in human testicular cancer cells. Unlike these studies, there are several adult rat testis studies demonstrating both COX-1 and COX-2 messenger RNA (mRNA) in several testicular cell types. 6,11 However, testicular COX-2 expression has been shown to be constitutive and higher than COX-1 expression. 1,6 At this point, it has been suggested that COX-1 enzyme mediates the regulation of tissue homeostasis, and COX-2 enzyme mediates the steroid hormone biosynthesis and regulation of spermatogenesis. 1 Moreover, prostaglandin content is high in the semen of human and other species, suggesting that these endogen peptides may have an important role in fertility. 12 –15 Furthermore, adult rat testes have high PGE2 and PGF2α levels but low prostaglandin D2 (PGD2), prostaglandin E1 (PGE1), and prostaglandin F1 (PGF1) levels. 6,11 Seminal prostaglandins have been shown to affect the sperm count, 12,13 motility, 16 capacitation, 16 and the acrosome reaction 12,15 –17 in the male reproductive system. In addition, systemic administration of prostaglandins and prostaglandin synthesis inhibitors has been reported to alter the prolactin, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) levels in the circulation. 18 –21 Although the potential complexity of these molecules has been stressed in the regulation of testicular functions, it has been proposed that prostaglandins have both stimulator and inhibitor effects throughout the process of spermatogenesis. 6,13
Because of these reasons, the present study aimed to investigate the toxic effects of two commonly used agents in the routine treatment of inflammatory disease in repeated doses, the nonselective COX inhibitor naproxen sodium and selective COX-2 inhibitor meloxicam, on the male reproductive system as well as to evaluate the possible effect of selective/nonselective COX inhibition on toxicity. For this purpose, male rats were treated by naproxen sodium and meloxicam, followed by evaluation of sperm parameters including sperm count and sperm motility; COX-1 and COX-2 enzyme activities, and PGE1, PGE2, and PGF2α levels in testicular tissue; and blood FSH, LH, and testosterone levels as well as the histopathological examination. In addition, superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx), and glutathione (GSH) levels in the testicular tissue were determined in order to evaluate the oxidative status secondary to the inhibition of prostaglandin synthesis by these agents.
Materials and methods
Materials
The chemicals used were obtained from the following sources: ketamine (Ketalar®; Pfizer, Turkey); meloxicam (DEVA Holding, Turkey); naproxen sodium (DEVA Holding, Turkey); and xylazine (Bayer, Turkey). For the measurements of testosterone (Cusabio Biotech Co. Ltd., People’s Republic of China), LH (Cusabio Biotech Co. Ltd), and FSH (Cusabio Biotech Co. Ltd) levels, enzyme-linked immunosorbent assay (ELISA) kits were used according to the manufacturer’s instructions in plasma samples. For the measurements of PGE1, PGF2α, COX-1, and COX-2 levels ELISA kits from Cusabio Biotech Co. Ltd were used. PGE2 level was evaluated using ELISA kit from Enzo Life Sciences (Farmingdale, New York, USA). Testis SOD, catalase, GPx, and GSH levels were determined by ELISA kits from Cayman Chemical Company (Ann arbor, Michigan, USA).
Methods
Animals
Male Wistar rats weighing 250–300 g were obtained from our own animal facility. Rats were housed under controlled temperature (22°C) and lighting (12-h light/12-h dark cycle) with free access to food and water. The experimental protocol was approved by the Local Ethical Committee on Animal Experimentation of Anadolu University, Eskisehir, Turkey.
The experimental groups of animals were as follows: group 1, control animals treated orally with distilled water with a volume of 1.5 ml/100 g for 35 days (n = 10; C); group 2, animals treated orally with 10 mg/kg naproxen sodium with a volume of 1.5 ml/100 g for 35 days (n = 10; N); and group 3, animals treated orally with 1 mg/kg meloxicam with a volume of 1.5 ml/100 g for 35 days (n = 10; M). Control group received orally the same volume of distilled water for 35 days. The doses of meloxicam 22 and naproxen sodium 23 were determined according to the previous studies.
After 35 days of treatment, the following experimental procedure lasting for a period of 6–18 h was used.
Experimental procedure
Following 35 days of treatment, the animals were anesthetized by intraperitoneal injection of 60 mg/kg ketamine and 5 mg/kg xylazine. Under general anesthesia, the blood samples for hormonal analysis (FSH, LH, and testosterone) were collected from the right ventricle of the animals via a syringe. The blood samples were collected into the gel tubes, and plasma samples were obtained by centrifuging the tube at 2–8°C and at 1000g for 15 min. The hormonal analyses were performed by using the commercially available kits and according to the instructions of the manufacturer. The testes of the animals were removed and cleared of blood and other contaminants in the phosphate-buffered solution (PBS) (composition: sodium chloride: 8 g/l, potassium chloride: 0.2 g/l, monopotassium phosphate: 0.2 g/l, and disodium phosphate: 1.14 g/l at pH 7.4). Then, the testes were divided into equal parts and stored at −20°C after freezing in liquid nitrogen. The COX-1, COX-2, PGE1 and PGE2, PGF2
α, catalase, SOD, GPx, and GSH levels in the testicular tissue were determined by using commercially available kits and according to the instructions of the manufacturer. For histopathological analysis, the testis samples were fixed in 10% formalin for 48 h. The entire left epididymis was used for sperm count and the cauda of the right epididymis was used in order to evaluate the sperm motility.
Sperm count
Epididymis samples were chopped in 5 ml of PBS at room temperature by using anatomical scissors. This fluid-containing epididymal tissue pieces was vortexed for 10 min and allowed to stand at room temperature for 2 min. Sperm count was determined using a Makler counting chamber (Sefi Medical Instruments Ltd., Haifa, Israel) by using 50 µL of the vortexed fluid.
Sperm motility
All motility assessments were performed at 37°C. The cauda was dissected in the PBS containing 1% bovine serum albumin in order to allow the sperm to get out of the epididymis. The total and mobile sperm counts were determined using a hemocytometer by using 10 µl of this fluid. The %motility values were calculated.
COX-1 assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:10 (w:v) with cold PBS and were homogenized. The homogenates were centrifuged at 5000g for 5 min at +4°C. The supernatants were removed and were then used for COX-1 assay according to the manufacturer’s instructions. Limit of detection (LOD) of the assay was 7.8–500 pg/ml.
COX-2 assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:10 (w:v) with cold PBS and were homogenized. The homogenates were centrifuged at 5000g for 5 min at +4°C. The supernatants were removed and were used for COX-2 assay according to the manufacturer’s instructions. LOD of the assay was 1.56–100 ng/ml.
PGE1 assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:10 (w:v) with cold PBS and were homogenized. The homogenates were centrifuged at 5000g for 5 min at +4°C. The supernatants were removed and were used for PGE1 assay according to the manufacturer’s instructions. LOD of the assay was 125–2000 pg/ml.
PGE2 assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:10 (w:v) with cold PBS and were homogenized. The homogenates were centrifuged at 5000g for 5 min at +4°C. The supernatants were removed and were used for PGE2 assay according to the manufacturer’s instructions. LOD of the assay was 39.1–2500 pg/ml.
PGF2α assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:10 (w:v) with cold PBS and were homogenized. The homogenates were centrifuged at 5000g for 5 min at +4°C. The supernatants were removed and were used for PGF2α assay according to the manufacturer’s instructions. LOD of the assay was 6.25–400 pg/ml.
GSH assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (50 mM 2-(N-morpholino)ethanesulfonic acid, pH 6–7, containing 1 mM ethylenediaminetetraacetic acid (EDTA)) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were deproteinated. The samples were used for total GSH assay according to the manufacturer’s instructions. The LOD of the assay was 0–16 µM.
GPx assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (50 mM Tris–hydrochloric acid, pH 7, containing 5 mM EDTA, and 1 mM dithiothreitol) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. Then, the supernatants were removed and were used for GPx assay according to the manufacturer’s instructions. The LOD of the assay was 50–344 nmol/min/ml.
CAT assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (50 mM potassium phosphate, pH 7, containing 1 mM EDTA) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were used for CAT assay according to the manufacturer’s instructions. The LOD of the assay was 2–34 nmol/min/ml.
SOD assay
The tissues were washed with PBS solution at pH 7.4. They were diluted at the ratio of 1:20 (w:v) with 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer (pH 7.2, containing 1 mM ethylene glycol tetraacetic acid (EGTA), 210 mM mannitol, and 70 mM sucrose) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. Then, the supernatants were removed and were used for SOD assay according to the manufacturer’s instructions. The LOD of the assay was 0.025–0.25 U/ml.
Light microscopic analysis
The testis samples were fixed in a 10% buffered formalin solution for 48 h and embedded in paraffin. Then, 5 μm thick slices were stained with hematoxylin and eosin and examined by light microscopy. All sections were observed under an Olympus BH-2 (Olympus Corp., Tokyo, Japan) microscope.
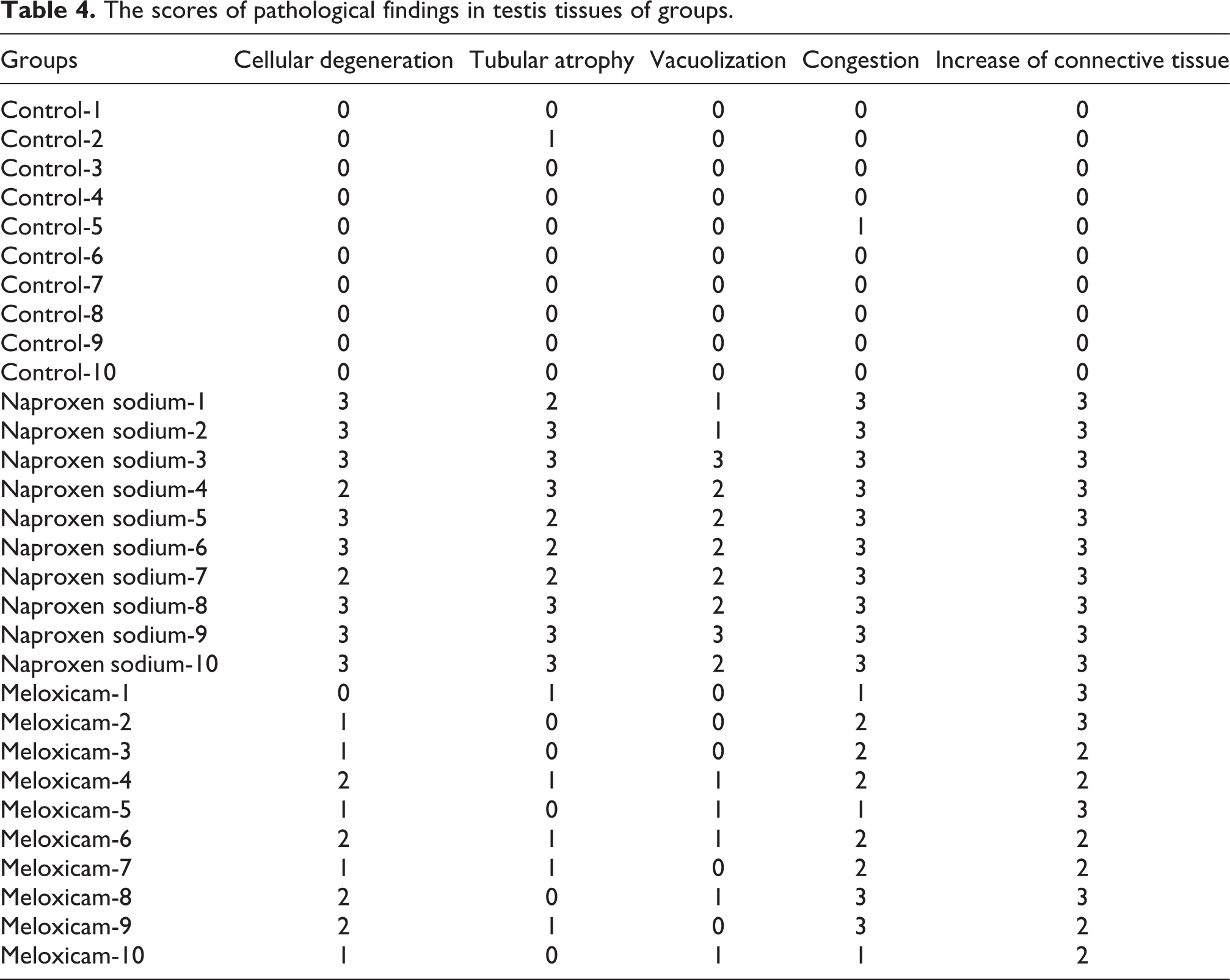
The testes were evaluated for the existence of cellular degeneration, tubular atrophy, vacuolization, congestion, and increase of connective tissue. The results were scored from 0 to 3. Scoring were performed as follows; 0: absence (no pathological finding is present in the fields); 1: slight (<25% of the fields contained evidence of any pathological finding); 2: moderate (from 25–50% of the fields contained evidence of any pathological finding); 3: severe (more than 50% of the fields contained evidence of any pathological finding; Table 4).
Statistical analysis
All data were expressed as mean ± standard deviation. Statistical analyses of the groups were performed on SigmaPlot v.10 package program and by using one-way analysis of variance. Tukey’s test was used as the post hoc test. The obtained scores from histological investigation were evaluated statistically via k2 tests by SPSS 19.0 program. The value of p < 0.05 was considered as statistically significant.
Results
Sperm parameters
Sperm count
Sperm count was significantly decreased in animals treated with naproxen sodium and meloxicam compared to the control animals (p < 0.001 and p < 0.001, respectively). There was no statistical difference between the treatment groups (Figure 1).

The effects of drug treatments after 35 days on sperm count in rats. C: distilled water-treated rats for 35 days, N: naproxen sodium-treated rats for 35 days, M: meloxicam-treated rats for 35 days. *p < 0.05: different from C.
Sperm motility
The percentage (%) sperm motility was found to be significantly decreased in animals treated with naproxen sodium and meloxicam compared to the control animals (p < 0.001 and p < 0.001, respectively). There was no statistical difference between the treatment groups (Figure 2).

The effects of drug treatments after 35 days on sperm motility in rats. C: distilled water-treated rats for 35 days, N: naproxen sodium-treated rats for 35 days, M: meloxicam-treated rats for 35 days. *p < 0.05: different from C.
Plasma hormon levels
Plasma FSH levels
Plasma FSH levels were not different in animals treated with naproxen sodium and meloxicam compared to the control animals. Similarly, no difference was found between the two treatment groups (Table 1).
The effects of drug treatments after 35 days on serum LH, FSH, and testosterone levels in rats.

C: control group; N: naproxen sodium group; M: meloxicam group; FSH: follicle stimulating hormone; LH: luteinizing hormone.
Plasma LH levels
Plasma LH levels were not different in the groups treated with naproxen sodium and meloxicam compared to the controls. Similarly, no difference was found between the two treatment groups (Table 1).
Plasma testosterone levels
There was no difference in plasma testosterone levels between the treatment and control groups as well as between the two treatment groups (Table 1).
COX enzyme activities in testicular tissue
COX-1 enzyme activity
COX-1 enzyme activity in the testis was significantly decreased in the animals treated with naproxen sodium and meloxicam compared to the control animals (p = 0.017 and p = 0.05, respectively). However, there was no difference between the two treatment groups (Table 2).
The effects of drug treatments after 35 days on testis COX-1, COX-2, PGE1, PGE2, and PGF2α levels in rats.

C: control group; N: naproxen sodium group; M: meloxicam group; COX-1: cyclooxygenase-1; COX-2: cyclooxygenase-2; PGE1: prostaglandin E1; PGE2: prostaglandin E2; PGF2α: prostaglandin F2α.
a p < 0.05: different from C.
b p < 0.05: different from N.
COX-2 enzyme activity
COX-2 enzyme activity in the testis was not different between the treatment and control groups as well as between the two treatment groups (Table 2).
Prostaglandin levels in the testicular tissue
PGE1 level
PGE1 level in the testes was found to be decreased with the administration of naproxen sodium and meloxicam. However, the difference was statistically significant only in the meloxicam-treated animals compared with the controls (p = 0.001). Additionally, significantly decreased PGE1 levels were found in animals treated with meloxicam compared to those treated with naproxen sodium (Table 2).
PGE2 level
PGE2 level in the testis was significantly lower in animals treated with naproxen sodium and meloxicam compared with the control animals (p = 0.041 and p < 0.001, respectively). There was no difference between the two treatment groups (Table 2).
PGF2α level
PGF2α level in the testis was also significantly lower in animals treated with naproxen sodium and meloxicam compared with the control animals (p = 0.002 and p = 0.01, respectively). There was no difference between the two treatment groups (Table 2).
Antioxidant enzyme levels in the testicular tissue
GSH level
It was found that GSH level is significantly lower in the animals treated with naproxen sodium and meloxicam compared to the control animals (p < 0.001 and p < 0.001, respectively). However, no difference was found between the two treatment groups (Table 3).
The effects of drug treatments after 35 days on testis GSH, GPx, SOD, and CAT levels in rats.
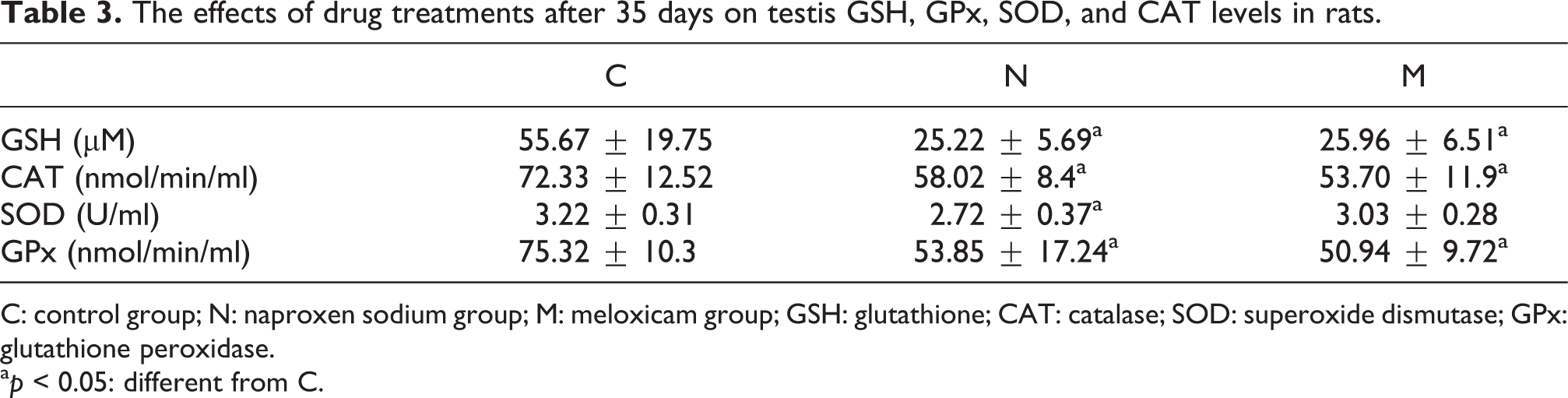
C: control group; N: naproxen sodium group; M: meloxicam group; GSH: glutathione; CAT: catalase; SOD: superoxide dismutase; GPx: glutathione peroxidase.
a p < 0.05: different from C.
Catalase level
Catalase level was found to be significantly lower in the animals treated with naproxen sodium and meloxicam compared to the control animals (p = 0.025 and p = 0.017, respectively). However, no difference was found between the two treatment groups (Table 3).
SOD level
SOD level was lower in the animals from naproxen sodium and meloxicam groups compared to the control animals. However, the difference was statistically significant only in the naproxen sodium-treated animals compared to the controls (p = 0.005). There was no difference between the two treatment groups (Table 3).
GPx level
GPx level was also significantly lower in the animals from naproxen sodium and meloxicam groups compared to the control animals (p = 0.009 and p = 0.003, respectively). There was no difference between the two treatment groups (Table 3).
Histopathological analysis of the testicular tissue
Histopathological analysis of the testicular tissue in the control group
Light microscopic examination of the testes of rats from the control group revealed normal seminiferous tubules (Figure 3).

Assessment of testis light microscopy analysis in control male rats. The testes of rats from the control group revealed normal seminiferous tubules. (a) Hematoxylin and eosin stain, scale bar 500 μm and (b) hematoxylin and eosin stain, scale bar 200 μm.
Histopathological analysis of the testicular tissue in the naproxen sodium group
In the light microscopic examination of the testes of rats treated with naproxen sodium, seminiferous tubules of the rats showed severe damage in the germinal cells in the tubule wall (*), interstitial vascular congestion (◂), and increased concentration of connective tissue (○) (Figure 4). Histological scores of cellular degeneration, tubular atrophy, vacuolization, congestion, and increase of connective tissue were significantly higher in naproxen sodium group compared to control group (Table 4).

Assessment of testis light microscopy analysis in naproxen sodium-treated male rats. The testes of rats treated with naproxen sodium, seminiferous tubules of the rats showed severe damage in the germinal cells in the tubule wall (*), interstitial vascular congestion (◂), and increased concentration of connective tissue (^). (a) Hematoxylin and eosin stain, scale bar 500 μm; (b) hematoxylin and eosin stain, scale bar 200 μm; (c) hematoxylin and eosin stain, scale bar 100 μm; and (d) hematoxylin and eosin stain, scale bar 50 μm.
The scores of pathological findings in testis tissues of groups.
Histopathological analysis of the testicular tissue in the meloxicam group
In the testes of rats treated with meloxicam, light microscopic examination revealed some seminiferous tubules containing near-normal spermatogenetic cells and ongoing spermatogenesis (*) as well as many damaged tubules (→), interstitial vascular congestion (◂), and increased concentration of connective tissue (○) (Figure 5). Histological scores of cellular degeneration, tubular atrophy, vacuolization, congestion, and increase of connective tissue were significantly higher in meloxicam group compared to control group (Table 4).

Assessment of testis light microscopy analysis in meloxicam-treated male rats. In the testis of rats treated with meloxicam, light microscopic examination revealed some seminiferous tubules containing near-normal spermatogenetic cells and ongoing spermatogenesis (*) as well as many damaged tubules (→), interstitial vascular congestion (◂), and increased concentration of connective tissue (^). (a) Hematoxylin and eosin stain, scale bar 200 μm; (b) hematoxylin and eosin stain, scale bar 200 μm; (c) hematoxylin and eosin stain, scale bar 200 μm; and (d) hematoxylin and eosin stain, scale bar 100 μm.
Discussion
Previous studies examining the effects of prostaglandins and prostaglandin synthesis inhibitors on male fertility and spermatogenesis have revealed conflicting results. Based on the results of these studies, it has been stated that the role of these agents in the regulation of testicular functions may be complex and that prostaglandins may have both stimulator and inhibitor effects on the entire process of spermatogenesis. 6 From this point of view, the present study aimed to clarify the possible effects of prostaglandins on the process of spermatogenesis. For this purpose, we evaluated the sperm count and motility as well as determined the plasma levels of the spermatogenetic hormones LH, FSH, and testosterone after administration of a nonselective COX enzyme inhibitor, naproxen sodium, and a selective COX-2 inhibitor meloxicam which are commonly used in the acute and chronic treatment of pain and inflammatory diseases. At this point, it was also aimed to clarify the contribution of selective and nonselective COX inhibition to the possible reproductive toxicity. On the other hand, the determination of COX-1, COX-2, PGE1, PGE2, and PGF2α in the testes implies that these endogen mediators are involved in the male reproductive system.
Interestingly, although nonselective COX enzyme inhibitor naproxen sodium and selective COX-2 inhibitor meloxicam led to significantly decreased COX-1 enzyme levels in the testes of the rats compared with the control animals, testicular COX-2 levels showed only slight decreases in both naproxen sodium and meloxicam groups. In the studies of Tanaka et al., 24,25 the authors have reported decreased COX-1 but increased COX-2 levels in the intestinal and gastric tissue by the administration of nonselective COX-1 inhibitor indomethacin. It has been explained by the compensatory increase of COX-2 enzyme synthesis in response to the decreased prostaglandin levels by COX-1 inhibition. In our study, we compared the groups in terms of testicular PGE1, PGE2, and PGF2α levels and found that administration of naproxen sodium and meloxicam significantly decreased the levels of prostaglandins, particularly that of PGE2 and PGF2α. It has been reposted in various tissues that naproxen leads to decreased synthesis of PGE1, 26 PGE2, 27,28 and PGF2α. 29 Similarly, meloxicam has been reported to decrease the levels of PGE1, 30 PGE2, 30 and PGF2α. 31 In our study, decreased levels of prostaglandins—the end product of COX enzyme activity—by administration of naproxen sodium and meloxicam reflects the inhibitory effects of these two agents on the enzyme.
On the other hand, we found significantly decreased sperm count and sperm motility as well as damage in seminiferous tubules in animals treated with naproxen sodium or meloxicam compared to the control animals. The negative effects of sperm parameters observed with the administration of repeated doses of naproxen sodium and meloxicam can be attributed to the decreased prostaglandin levels caused by the enzyme inhibition induced by these agents. Likewise, there are some previous studies showing that prostaglandin E and F series play a significant role in the process of spermatogenesis. 6,11 Prostaglandins have been suggested to play a role also in many processes in the male reproductive system including sperm motility and count, capacitation, and acrosome reaction. 12,14,16 In another study, decreased levels of prostaglandins have been reported in infertile men. 12 Schlegel et al. showed in their study that PGF2α has an inverse correlation with human sperm motility; however, degradation of prostaglandins also causes a significant decrease in sperm motility. Addition of PGE2 to human semen samples has resulted in increased motility and fertilization capacity in spermatozoa. 13 Kennedy et al. 16 have found that in vitro incubation of semen with indomethacin, a selective COX-1 inhibitor (resveratrol) and a selective COX-2 inhibitor (NS 398) caused a reduction in sperm motility. Patients treated with acetyl salicylic acid have been suggested to have impaired fertility due to decreased prostaglandin synthesis. The rats treated with high doses of indomethacin for 7 days have been also found to have impairment in fertility induced by decreased prostaglandin synthesis. Moreover, incubation of human and rabbit semen with the prostaglandin inhibitor 15-hydroxydehydrogenase has resulted in decreased prostaglandin synthesis and sperm motility. 13 PGE1 and PGE2 have been suggested to regulate the motility by increasing the calcium influx and the releasing the calcium from internal stores. 15,16
Nonselective COX inhibitor, indomethacin, has been shown to reduce the sperm count and motility, which were improved after discontinuation of the indomethacin therapy. Unlike other studies, this has been associated with a reduction in testosterone levels in plasma, testes, and testicular interstitial fluid. 11 Normal testicular development and spermatogenesis are under the control of testosterone, LH, and FSH during fetal and postnatal life. 32,33 According to the results of the present study, treatment with naproxen sodium and meloxicam resulted in no significant difference in plasma testosterone, LH, and FSH levels. In a study of Gates et al. examining the relationship between the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and human steroid hormone levels, drugs have been found to have no effect on the hormone levels. On the other hand, systemic administration of prostaglandins has been suggested to increase the plasma prolactin, testosterone, LH, and FSH levels. 11 In another in vitro study examining the relationship between COX-2 enzyme expression and testosterone biosynthesis in the rat Leydig cells, testosterone biosynthesis has been shown to be decreased or increased with increasing expression and inhibition of COX-2 enzyme, respectively. 21 In the in vitro study of Frungieri et al. on Leydig cells of hamsters, testosterone production has been found to decrease with the administration of PGF2α. Moreover, high doses of prostaglandins have been shown to decrease the spermatogenesis in rats and the plasma testosterone levels in mice, rats, and hamsters. 18 Similarly, administration of PGF2α has resulted in decreased plasma testosterone level in healthy male rats, which was prevented by coadministration of LH. PGE2 has led to no significant change in plasma testosterone level, however, in vivo administration of prostaglandin synthase inhibitor, indomethacin, has been found to result in increased plasma testosterone level. 19 StAR gene expression and testosterone biosynthesis are regulated by two pathways mediated by release of LH from the pituitary gland. Firstly, LH causes an increase in the release of arachidonic acid and consequently metabolites of arachidonic acid increase, which regulates StAR gene expression. Secondly, LH regulates the StAR gene expression and steroidogenesis through protein kinase A. Thus, the changes occurring in these two pathways will affect the hormone levels. Previous studies have suggested that lipooxygenase- and epoxygenase-mediated arachidonic acid metabolites increase the steroid hormone synthesis and that COX-2-mediated metabolites inhibit the StAR gene expression and steroidogenesis. 20,21 Our study shows no significant difference in LH, FSH, and testosterone level between the study groups, which contrasts with the results of previous studies reporting increased and decreased testosterone levels with COX enzyme inhibition and activation, respectively. This may be attributed to the differences in the major factors affecting toxicity such as experimental model, animal species, drug dose, and exposure time. According to the results of present study, it is not possible to associate the negative effects on sperm parameters induced by naproxen sodium and meloxicam with the levels of hormones playing a role in spermatogenesis, the testosterone, LH, and FSH.
Although it is well-known that chronic administration of high doses of NSAIDs such as paracetamol and phenacetin to the male rats lead to testicular atrophy and inhibition of spermatogenesis, some authors have suggested this to be a nonspecific part of the toxicity syndrome induced by many drugs or chemical reagents that have no effect on prostaglandin synthesis. 13 Löscher and Blazaki (1986) have showed that indomethacin, naproxen, acetylsalicylic acid, and phenylbutazone resulted in decreased PGE2 levels in rat seminal fluid, but only indomethacin decreased the fertility. The authors have attributed the decreased fertility to the secondary toxic effects of indomethacin. 13 In various toxicity models such as gastric mucosal injury, hepatic injury, and pancreatitis induced by meloxicam 34,35 or naproxen sodium, 36 –38 the toxic effects of these agents have been associated with decreased prostaglandin synthesis as well as with secondarily induced free radical formation and decreased antioxidant defense mechanisms.
Oxidative stress is resulted from the imbalance between the antioxidants and the free radicals (reactive oxygen species (ROS) and reactive nitrogen species). 39 The cells are protected from oxidative stress by enzymatic (catalase, SOD, and GPx) and nonenzymatic (GSH) complex antioxidant systems. 34,40 Increased oxidative stress in the cells, tissues, and organs are known to have the potential to cause fetal abnormalities and infertility. 39,41 –46 Oxidative stress is considered as the most important factor in the etiology of male infertility. 47 It has been shown that ROS levels are increased in 25–40% of infertile men, which have a negative effect on the morphology and motility of spermatozoa. ROS-induced toxicity in spermatozoa begins with lipid peroxidation, followed by impaired motility, DNA damage, and impaired ability to promote the normal embryologic development. 47 –51 Moreover, ROS impairs the steroidogenetic capacity of Leydig cells in testes. The presence of multiple factors that may trigger the oxidative stress renders the testicular tissue to be highly dependent to the oxygen for spermatogenesis and also to be very sensitive to the toxic effects of reactive oxygen metabolites, which is very similar to the brain in this context. 47
In this study, administration of naproxen sodium and meloxicam resulted in significantly decreased GSH, catalase, SOD, and GPx levels in the testes. Meloxicam has been previously shown to increase the myeloperoxidase and xanthine oxidase activity that catalyzes the formation of radicals and to decrease the levels of antioxidant enzymes SOD and GSH in gastric mucosa. The authors have suggested that meloxicam-induced gastric mucosal injury resulted from the inhibition of prostaglandin synthesis and formation of free radicals and that decreased GSH and SOD levels have also contributed to the pathogenetic process. 34 In a study of gastric mucosal injury induced by a nonselective COX inhibitor piroxicam and selective COX-2 inhibitor meloxicam conducted by Villegas et al., 35 increased mucosal lipid peroxidation and decreased SOD, GSH, and GSH reductase levels have been reported. Accordingly in an experimental gastric antral ulceration model induced by naproxen, the authors have reported increased lipid peroxidation and MDA levels and decreased SOD, catalase, and GPx levels in gastric mucosa. 36 Indomethacin-induced gastric mucosal injury has been also associated with increased mucosal lipid peroxidation and decreased GSH and GPx levels. 52 Increased production of free oxygen radicals and lipid peroxidation induced by naproxen and the resultant oxidative stress caused by the depletion of GSH stores have been identified as major contributors to the pathogenesis of acute pancreatitis. 38 Administration of naproxen has been shown to induce oxidative stress by increasing the lipid peroxidation and depleting the GSH stores in isolated perfused rat liver. 37
Nonselective COX inhibitors, diclofenac and naproxen, and selective COX-2 inhibitors, rofecoxib and celecoxib, induce oxidative stress by increasing the formation of free radicals in cardiac vascular structures (aorta and heart) of hypertensive rats. 53 Doxorubicin-induced cardiac toxicity exacerbates with the administration of naproxen due to increased myocardial lipid peroxidation and decreased levels of GSH, catalase, and SOD. 54 Moreover, administration of naproxen to the rats infected with Helicobacter pylori have resulted in exacerbation of gastric ulceration by increased MDA levels and decreased GSH levels in gastric tissue. 55 In contrast, meloxicam has been shown to have protective antioxidant effect by decreasing lipid peroxidation in the tissues in Freund’s adjuvant-induced rat arthritis model, 56 amyloid-β-peptide-induced mouse Alzheimer’s model 45 and scopolamine-induced mouse cognitive dysfunction model. 57 Similarly, naproxen has shown a protective effect by decreasing lipid peroxidation in the tissues in rat immobilization stress model, 58,59 ethanol-induced oxidative stress, 23 and model of colchicine-induced cognitive dysfunction. 60 The protective effects of naproxen and meloxicam against lipid peroxidation have been suggested to be associated with anti-inflammatory effects of the agents and inflammation has been shown to induce the oxidative stress. Thus, in these experimental models involving inflammatory processes, antioxidant effects of meloxicam and naproxen may be attributed to the anti-inflammatory activities of the drugs. 45,56,57,60 It is well-known that besides impairing the integrity of membrane, ROS-induced lipid peroxidation also has chemotactic effects on inflammatory cells and initiates the inflammatory reaction by attracting these cells to the tissues. 61 On the other hand, peroxidase activity of COX is also known to increase ROS production. 60 It is not surprising for our results contrasting with the protective effects observed previously with the administration of meloxicam and naproxen in oxidative stress-induced experimental models. As mentioned, anti-inflammatory agents decreased the oxidative stress in experimental models involving inflammatory processes. Another important point to note here is that recently proposed antitumoral effects of NSAIDs may be associated with induction of oxidative stress related to many signaling pathways including the apoptosis. 62 In our study examining the relative effects of naproxen sodium and meloxicam on oxidative parameters in testicular tissue, we found increased oxidant/antioxidant balance in favor of the oxidants.
It should be noted that various testicular damages were observed in our study including cellular degeneration, tubular atrophy, congestion, and increase of connective tissue. Histopathological findings have been widely used as significant biomarkers. The severe histological changes of testes could accompany with the decreases of sperm quantity and quality. 63 Previous studies showed seminiferous tubules damage and degenerative changes in the germinal cells after exposure to agents. 64 –66 It is well known that human testes and spermatozoa are extremely sensitive to oxidative stress-induced damage. 63 Prolonged oxidative stress in testes can cause significant histopathologic alterations in tissue. 66 –68 Free radicals induce lipid peroxidation of membrane-bound polyunsaturated acids of mammalian testes and other biomembranes leading to impairment of the membrane integrity and degeneration of seminiferous tubules. As a result, testicular atrophy and tissue degeneration occur. 69 Our histopathologic findings showed that altered testicular antioxidant status might be associated with the testicular damage investigated in our study.
In conclusion, prostaglandins were found to have direct effects on sperm count and motility in the process of spermatogenesis in the male reproductive system without affecting the plasma hormone levels. Inhibition of the synthesis of these endogen mediators by administration of NSAIDs including naproxen sodium and meloxicam resulted in decreased sperm count and motility and in damage to seminiferous tubules, which were independent of plasma hormone levels. On the other hand, the similarity of the inhibitor effects of a nonselective COX enzyme inhibitor naproxen sodium and a selective COX-2 inhibitor meloxicam implies that both COX-1 and COX-2-mediated prostaglandins play a structural role in the male reproductive system. Although our results indicate that decreased prostaglandin synthesis is the major pathogenetic mechanism in reproductive toxicity induced by these agents, oxidative stress may also have a key role. It is an inevitable consequence of the toxic effects of oxidative stress on spermatogenesis to affect the male fertility negatively.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
