Abstract
Resveratrol is a natural compound that affects cellular calcium (Ca2+) homeostasis and viability in different cells. This study examined the effect of resveratrol on cytosolic free Ca2+ concentrations ([Ca2+]i) and viability in OC2 human oral cancer cells. The Ca2+-sensitive fluorescent dye fura-2 was used to measure [Ca2+]i, and water-soluble tetrazolium-1 was used to measure viability. Resveratrol evoked concentration-dependent increase in [Ca2+]i. The response was reduced by removing extracellular Ca2+. Resveratrol also caused manganese-induced fura-2 fluorescence quench. Resveratrol-evoked Ca2+ entry was inhibited by nifedipine and the protein kinase C (PKC) inhibitor GF109203X but was not altered by econazole, SKF96365, and the PKC activator phorbol 12-myristate 13 acetate. In Ca2+-free medium, treatment with the endoplasmic reticulum Ca2+ pump inhibitor 2,5-di-tert-butylhydroquinone (BHQ) abolished resveratrol-evoked [Ca2+]i rise. Conversely, treatment with resveratrol inhibited BHQ-evoked [Ca2+]i rise. Inhibition of phospholipase C (PLC) with U73122 abolished resveratrol-evoked [Ca2+]i rise. At 20–100 μM, resveratrol decreased cell viability, which was not affected by chelating cytosolic Ca2+with 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid-acetoxymethyl ester. Annexin V-fluorescein isothiocyanate staining data suggest that resveratrol at 20–40 μM induced apoptosis in a concentration-dependent manner. Collectively, in OC2 cells, resveratrol induced [Ca2+]i rise by evoking PLC-dependent Ca2+ release from the endoplasmic reticulum and by causing Ca2+ entry via nifedipine-sensitive, PKC-regulated mechanisms. Resveratrol also caused Ca2+-independent apoptosis.
Introduction
Resveratrol is a natural dietary polyphenolic compound that was discovered in the 1940s and has cellular physiological effects on different cells. 1 Although initially used for killing cancer cells in vitro, it has shown beneficial effects against most cardiovascular and cerebrovascular diseases. 2 Through its antioxidant, anticarcinogenic, and anti-inflammatory properties, resveratrol has become a candidate for drug development in aging studies. 3 It can also improve glucose and lipid metabolism, alter cardiovascular parameters, and modify carcinogenesis. 4
Resveratrol induces death in different cancer cells. Kumazaki et al. 5 showed that resveratrol killed human colon cancer cells through modulation of signal transduction and micro RNA expression. Yang et al. 6 suggested that resveratrol induced apoptosis in hepatoma cells. In K562 tumor cells, resveratrol was shown to diminish platelet aggregation and increase susceptibility to natural killer cells. 7 Furthermore, resveratrol was shown to induce death in cervical cancer cells through apoptosis and autophagy. 8 Evidence shows that resveratrol may inhibit oral cancer. Zlotogorski et al. 9 showed that resveratrol may be applied clinically to control oral cancer. Elattar and Virji 10 showed that resveratrol might inhibit growth and proliferation of human oral squamous carcinoma cells. However, whether resveratrol alters calcium (Ca2+) ion movement and viability in cultured OC2 human oral cancer cells is unclear.
Ca2+ ions play a pivotal role in various cellular responses. A rise in cytosolic free Ca2+ concentrations ([Ca2+]i) can trigger many pathophysiological cellular processes. 11 However, an uncontrolled [Ca2+]i rise may be harmful to the cell. 12 The effect of resveratrol on Ca2+ signal has been explored in other cell types. Chang et al. 13 showed that resveratrol induced [Ca2+]i rise and apoptosis in PC3 human prostate cancer cells. Garcia-Zepeda et al. 14 suggested that resveratrol mobilized Ca2+ from intracellular stores and induced c-Jun N-terminal kinase activation in tumoral AR42J cells. Qian et al. 15 showed that resveratrol attenuated the sodium (Na+)-dependent intracellular Ca2+ overload by inhibiting hydrogen peroxide-induced increase in late Na+ current in ventricular myocytes. Hsia et al. 16 explored the effect of resveratrol on uterine contraction and Ca2+ mobilization in rats in vivo and in vitro and found that resveratrol consistently suppressed the increases in [Ca2+]i induced by PGF(2α) and high potassium (K+) concentrations. Thus, it appears that resveratrol may increase or inhibit Ca2+ movement in different cells depending on the stimuli and experimental conditions.
In order to explore the effect of resveratrol on [Ca2+]i in human oral cancer cells, the OC2 human oral cancer cell was used because it produced measurable [Ca2+]i rises upon pharmacological stimulation. The OC2 cell is a well-established model for oral cell studies. In this cell, [Ca2+]i rises can occur under the stimulation of different compounds such as δ-methrin, 17 bisphenol A dimethacylate, 18 and carvacrol. 19
This study was aimed to examine the effect of resveratrol on [Ca2+]i and viability in OC2 cells. The Ca2+-sensitive fluorescent dye fura-2 was used to measure [Ca2+]i. The [Ca2+]i rises were characterized, the concentration–response relationship was plotted, and the pathways of resveratrol-induced Ca2+ entry and Ca2+ release was explored. The action of resveratrol on viability was assessed.
Materials and methods
Chemicals
The reagents for cell culture were purchased from Gibco® (Gaithersburg, Maryland, USA). The other reagents were purchased from Sigma-Aldrich (St Louis, Missouri, USA) unless otherwise indicated.
Cell culture
OC2 human oral cancer cells obtained from Bioresource Collection and Research Center (Taiwan) were cultured in Roswell Park Memorial Institute 1640 medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U/ml penicillin, and 100 μg/ml streptomycin.
Solutions used in [Ca2+]i measurements
Ca2+-containing medium (pH 7.4) had 140 mM sodium chloride (NaCl), 5 mM potassium chloride (KCl), 1 mM magnesium chloride (MgCl2), 2 mM calcium chloride (CaCl2), 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), and 5 mM glucose. Ca2+-free medium (pH 7.4) had 140 mM NaCl, 5 mM KCl, 3 mM MgCl2, 0.3 mM ethylene glycol tetraacetic acid (EGTA), 10 mM HEPES, and 5 mM glucose. Resveratrol was dissolved in dimethyl sulfoxide as a 1 M stock solution. The other agents were dissolved in water, ethanol, or dimethyl sulfoxide. The concentration of organic solvents in the solution used in experiments did not exceed 0.1% and did not alter basal [Ca2+]i, viability, and apoptosis measurements.
[Ca2+]i measurements
[Ca2+]i was measured as previously described by Liang et al.. 19 Confluent cells grown on 6 cm dishes were trypsinized and made into a suspension in culture medium at a density of 106/ml. Cell viability was determined by trypan blue exclusion (adding 0.2% trypan blue to 0.1 ml cell suspension). The viability was greater than 95% after the treatment. Cells were subsequently loaded with 2 μM fura-2/AM for 30 min at 25°C in the same medium. After loading, cells were washed with Ca2+-containing medium twice and was made into a suspension in Ca2+-containing medium at a density of 107/ml. Fura-2 fluorescence measurements were performed in a water-jacketed cuvette (25°C) with continuous stirring; the cuvette contained 1 ml of medium and 0.5 million cells. Fluorescence was monitored with a Shimadzu RF-5301PC spectrofluorophotometer (Shimadzu, Kyoto, Japan) immediately after 0.1 ml cell suspension was added to 0.9 ml Ca2+-containing or Ca2+-free medium, by recording excitation signals at 340 nm and 380 nm and emission signal at 510 nm at 1 s intervals. During the recording, reagents were added to the cuvette by pausing the recording for 2 s to open and close the cuvette-containing chamber. For calibration of [Ca2+]i, after completion of the experiments, the detergent Triton X-100 (0.1%) and CaCl2 (5 mM) were added to the cuvette to obtain the maximal fura-2 fluorescence. Then, the Ca2+ chelator EGTA (10 mM) was added to chelate Ca2+ in the cuvette to obtain the minimal fura-2 fluorescence. Control experiments showed that cells bathed in the cuvette had a viability of 95% after 20 min of fluorescence measurements. [Ca2+]i was calculated as previously described. 20 Manganese (Mn2+) quenching of fura-2 fluorescence was performed in Ca2+-containing medium containing 50 μM MnCl2. MnCl2 was added to cell suspension in the cuvette 30 s before the fluorescence recoding was started. Data were recorded at excitation signal at 360 nm (Ca2+ insensitive) and emission signal at 510 nm at 1 s intervals as described previously. 21
Cell viability analyses
The measurement of viability was based on the ability of cells to cleave tetrazolium salts by dehydrogenases. Increase in the amount of developed color correlated proportionally with the number of live cells. Assays were performed according to manufacturer’s instructions (Roche Molecular Biochemical, Indianapolis, Indiana, USA). Cells were seeded in 96-well plates at a density of 10,000 cells/well in culture medium for 24 h in the presence of resveratrol. The cell viability-detecting reagent, (4-[3-[4-Iodophenyl]-2-4(4-nitrophenyl)-2H-5-tetrazolio-1,3-benzene disulfonate]; 10 μl pure solution), was added to samples after resveratrol treatment, and cells were incubated for 30 min in a humidified atmosphere. The cells were incubated with/without resveratrol for 24 h. The absorbance of samples (A 450) was determined using an enzyme-linked immunosorbent assay (ELISA) reader. In experiments using BAPTA/AM to chelate cytosolic Ca2+, cells were treated with 5 μM BAPTA/AM for 1 h prior to incubation with resveratrol. The cells were washed once with Ca2+-containing medium and incubated with/without resveratrol for 24 h. The absorbance of samples (A 450) was determined using an ELISA reader. Absolute optical density was normalized to the absorbance of unstimulated cells in each plate and expressed as a percentage of the control value.
Alexa®Fluor 488 annexin V/PI staining for apoptosis
Annexin V/propidium iodide (PI) staining assay was employed to further detect cells in early apoptotic and late apoptotic/necrotic stages. Cells were exposed to resveratrol at concentrations of 20 and 40 μM for 24 h. Cells were harvested after incubation and washed in cold phosphate-buffered saline (PBS). Cells were resuspended in 400 μl reaction solution with 10 mM of HEPES, 140 mM of NaC1, and 2.5 mM of CaC12 (pH 7.4). Alexa Fluor 488 annexin V/PI staining solution (Probes Invitrogen, Eugene, Oregon, USA) was added in the dark. After incubation for 15 min, the cells were collected and analyzed in a FACScan flow cytometry analyzer (BD FACSCalibur™ Catalog Number, 343524). Excitation wavelength was at 488 nm and the emitted green fluorescence of annexin V (FL1) and red fluorescence of PI (FL2) were collected using 530 nm and 575 nm band pass filters, respectively. A total of 20,000 cells were analyzed per sample. Light scatter was measured on a linear scale of 1024 channels and fluorescence intensity was on a logarithmic scale. The amounts of early apoptosis and late apoptosis/necrosis were determined, respectively, as the percentage of annexin V+/PI− or annexin V+/PI+ cells. Data were later analyzed using the flow cytometry analysis software WinMDI 2.8 (by Joe Trotter, freely distributed software). X and Y coordinates refer to the intensity of fluorescence of annexin and PI, respectively.
Statistics
Data are reported as mean ± SEM of three separate experiments. Data were analyzed by one-way analysis of variances using the Statistical Analysis System (SAS®, SAS Institute Inc., Cary, North Carolina, USA). Multiple comparisons between group means were performed by post hoc analysis using the Tukey’s honestly significantly difference (HSD) procedure. The value of p < 0.05 was considered significant.
Results
Effect of resveratrol on [Ca2+]i
Figure 1(a) shows that the basal [Ca2+]i level was 51 ± 1 nM. Resveratrol induced a [Ca2+]i rise in a concentration-dependent manner at concentrations between 5 and 20 μM in Ca2+-containing medium. At a concentration of 20 μM, resveratrol evoked a [Ca2+]i rise that attained to a net increase of 350 ± 2 nM followed by a decline to a sustained phase of 130 ± 2 nM. The Ca2+ response saturated at 20 μM resveratrol because 25 μM resveratrol did not evoke a greater response. Figure 1(b) shows that in the absence of extracellular Ca2+, 5–20 μM resveratrol induced concentration-dependent [Ca2+]i rises. Figure 1(c) shows the concentration–response relationship of resveratrol-induced [Ca2+]i rises. Removal of extracellular Ca2+ reduced the Ca2+ response by approximately 40%. The half-maximal effective concentration value was 5.2 ± 1 μM or 10 ± 2 μM in Ca2+-containing medium or Ca2+-free medium, respectively, by fitting to a Hill equation.

Effect of resveratrol on [Ca2+]i in fura-2-loaded OC2 cells. (a) Resveratrol was added at 25 s. The concentration of resveratrol was indicated. The experiments were performed in Ca2+-containing medium. (b) Effect of resveratrol on [Ca2+]i in the absence of extracellular Ca2+. Resveratrol was added at 25 s in Ca2+-free medium. (c) Concentration–response plots of resveratrol-induced [Ca2+]i rises in the presence or absence of extracellular Ca2+. Y axis is the percentage of the net (baseline subtracted) area under the curve (25–220 s) of the [Ca2+]i rise induced by 20 μM resveratrol in Ca2+-containing medium. Data are mean ± SEM of three separate experiments. *p < 0.05 compared with open circles. Ca2+: calcium; [Ca2+]i: cytosolic free Ca2+ concentrations.
Resveratrol-induced [Ca2+]i rise involves Ca2+ influx
Experiments were performed to confirm that resveratrol-induced [Ca2+]i rise involved Ca2+ influx. Mn2+ enters cells through similar pathways as Ca2+ but quenches fura-2 fluorescence at all excitation wavelengths, quench of fura-2 fluorescence excited at the Ca2+-insensitive excitation wavelength of 360 nm by Mn2+ indicates Ca2+ influx. 21 Figure 2 shows that 20 μM resveratrol induced an immediate decrease in the 360 nm excitation signal (compared to trace a) by 82 ± 2 arbitrary units at 250 s. This suggests that resveratrol-induced [Ca2+]i rise involved Ca2+ influx from extracellular space.
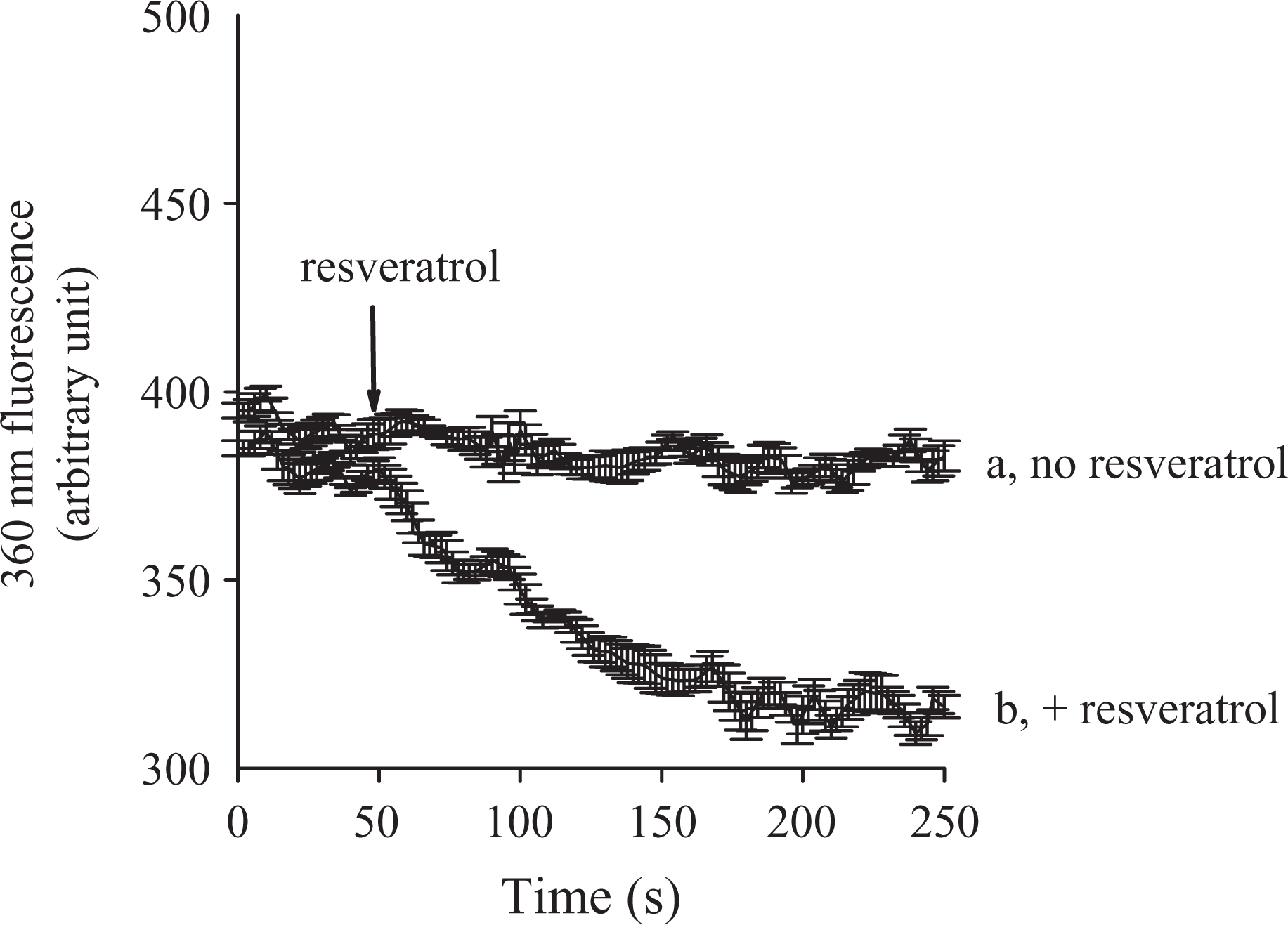
Effect of resveratrol on Ca2+ influx by measuring Mn2+ quenching of fura-2 fluorescence. Experiments were performed in Ca2+-containing medium. MnCl2 (50 μM) was added to cells 1 min before fluorescence measurements. The Y axis is fluorescence intensity (in arbitrary units) measured at the Ca2+-insensitive excitation wavelength of 360 nm and the emission wavelength of 510 nm. Trace a: control, without resveratrol. Trace b: resveratrol (20 μM) was added as indicated. Data are mean ± SEM of three separate experiments. Ca2+: calcium; Mn2+: manganese; MnCl2: manganese chloride.
Regulation of resveratrol-induced [Ca2+]i rise
Three Ca2+ entry inhibitors nifedipine (1 μM), econazole (0.5 μM), and SKF96365 (5 μM); phorbol 12-myristate 13-acetate (PMA; 1 nM; a protein kinase C (PKC) activator); and GF109203X (2 μM; a PKC inhibitor) were applied 1 min before applying resveratrol (20 μM) in Ca2+-containing medium. Nifedipine and GF109203X inhibited resveratrol-induced [Ca2+]i rise partly (Figure 3(a)). Experiments were performed to distinguish whether GF109203X’s effect was mediated by inhibiting Ca2+ entry or Ca2+ release. Figure 3(b) shows that GF109203X also inhibited resveratrol-induced Ca2+ signal in Ca2+-free medium by about 10%.

Effect of Ca2+ channel modulators on resveratrol-induced [Ca2+]i rise. (a) In blocker- or modulator-treated groups, the reagent was added 1 min before adding resveratrol (5 μM). The concentration was 1 μM for nifedipine, 0.5 μM for econazole, 5 μM for SKF96365, 10 nM for PMA, and 2 μM for GF109203X. Data are expressed as the percentage of 20 μM resveratrol-induced response in the area under the curve (25–200 s) in Ca2+-containing medium, and are mean ± SEM of three separate experiments. *p < 0.05 compared to the 1st column. (b) In Ca2+-free medium, GF109203X was added 1 min before 20 μM resveratrol. Data are mean ± SEM of three separate experiments. Ca2+: calcium; [Ca2+]i: cytosolic free Ca2+ concentrations; PMA: phorbol 12-myristate 13-acetate.
Source of resveratrol-induced Ca2+ release
In most cell types including OC2 human oral cancer cells, the endoplasmic reticulum has been shown to be a dominant Ca2+ store. 17 –19 Thus, the role of endoplasmic reticulum in resveratrol-evoked Ca2+ release in OC2 cells was examined. To exclude the contribution of Ca2+ influx, the experiments were conducted in Ca2+-free medium. Figure 4(a) shows that addition of 50 μM BHQ, an endoplasmic reticulum Ca2+ pump inhibitor, 22 after 20 μM resveratrol-induced [Ca2+]i rise induced a small [Ca2+]i rise of 13 ± 3 nM. Figure 4(b) shows that addition of BHQ induced a [Ca2+]i rise of 110 ± 2 nM. Resveratrol added at 505 s failed to induce a [Ca2+]i rise.

Effect of BHQ on resveratrol-induced Ca2+ release. (a and b) BHQ (50 μM) and resveratrol (10 μM) were added at time points indicated. Experiments were performed in Ca2+-free medium. Data are mean ± SEM of three separate experiments. BHQ: 2,5-di-tert-butylhydroquinone; Ca2+: calcium.
The dominant role of PLC in resveratrol-induced [Ca2+]i rise
PLC is one of the pivotal proteins that regulate the release of Ca2+ from the endoplasmic reticulum. Because resveratrol released Ca2+ from the endoplasmic reticulum, the role of PLC in this process was explored. U73122, 23 a PLC inhibitor, was applied to explore whether the activation of this enzyme was required for resveratrol-induced Ca2+ release. Figure 5(a) shows that adenosine triphosphate (ATP; 10 μM) induced a [Ca2+]i rise of 95 ± 2 nM. ATP is a PLC-dependent agonist of [Ca2+]i rise in most cell types. 24 Figure 5(b) shows that incubation with 2 μM U73122 did not change basal [Ca2+]i but abolished ATP-induced [Ca2+]i rise. This suggests that U73122 effectively suppressed PLC activity. The data also show that incubation with 2 μM U73122 also abolished 20 μM resveratrol-induced [Ca2+]i rise. U73343 (2 μM), a U73122 analogue, failed to have an inhibition (not shown).

Effect of U73122 on resveratrol-induced Ca2+ release. Experiments were performed in Ca2+-free medium. (a) ATP (10 μM) was added as indicated. (b) U73122 (2 μM), ATP, and resveratrol (20 μM) were added as indicated. Data are mean
Effect of resveratrol on viability in OC2 cells
Cells were treated with 0–100 μM resveratrol for 24 h, and the tetrazolium assay was performed. In the presence of 20–100 μM resveratrol, cell viability decreased in a concentration-dependent manner (Figure 6(a)).

Effect of resveratrol on viability of OC2 cells. (a) Cells were treated with 0–100 μM resveratrol for 24 h, and the cell viability assay was performed. Data are mean ± SEM of three separate experiments. Each treatment had six replicates (wells). Data are expressed as percentage of control that is the increase in cell numbers in resveratrol-free groups. Control had 10,254 ± 711 cells/well before experiments, and had 13,111 ± 788 cells/well after incubation for 24 h. In each group, the Ca2+ chelator BAPTA/AM (5 μM) was added to cells followed by treatment with resveratrol in Ca2+-containing medium. Cell viability assay was subsequently performed. *p < 0.05 compared to control. #p < 0.05 compared to pairing group. (b) After cells were treated with 5 μM BAPTA/AM for 24 h, cells were loaded with fura-2/AM as described in the Materials and methods. [Ca2+]i was measured. Resveratrol (20 μM) was added as shown in Ca2+-containing or Ca2+-free medium. Data are mean ± SEM of three separate experiments. (c) Cells were treated with GF109203X (2 μM) or U73122 (2 μM) for 24 h, and the cell viability assay was performed. Data are mean ± SEM of three separate experiments. Ca2+: calcium; BAPTA/AM: 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid-acetoxymethyl ester.
Lack of a role of Ca2+ in resveratrol-induced cell death
The intracellular Ca2+ chelator BAPTA/AM25 was applied to prevent a [Ca2+]i rise during resveratrol pretreatment, in order to explore the role of Ca2+ in resveratrol-induced cell death. Figure 6(a) also shows that 5 μM BAPTA/AM loading did not change the control value of cell viability. Resveratrol (20 μM) did not evoke [Ca2+]i rise in BAPTA/AM-treated cells in both Ca2+-containing and Ca2+-free solutions (Figure 6(b)). This suggests that BAPTA loading for 25 h still effectively chelated cytosolic Ca2+. Figure 6(a) shows that in the presence of 20–100 μM resveratrol, BAPTA loading did not reverse resveratrol-induced cell death.
Lack of a role of PKC or PLC inhibitor in OC2 cell viability
In Figures 3 and 5, PKC inhibitor GF109203X (2 μM) and PLC inhibitor U73122 (2 μM) were shown to inhibit resveratrol-induced [Ca2+]i rises. Therefore, the role of PKC or PLC activation in OC2 cell viability was examined. Cells were treated with GF109203X (2 μM) or PLC inhibitor U73122 (2 μM) for 24 h, and the tetrazolium assay was performed. GF109203X or U73122 loading did not induce cell death (Figure 6(c)).
Effect of resveratrol on apoptosis in OC2 cells
The cytotoxic effect was most significant between 20 μM and 40 μM resveratrol (Figure 6(a)). Therefore, concentrations of resveratrol at 20 μM and 40 μM were chosen for apoptotic experiments. Annexin V/PI staining was applied to detect apoptotic/necrotic cells after resveratrol treatment. Figures 7(a) and (b) show that treatment with 20 or 40 μM resveratrol significantly enhanced early apoptosis in a concentration-dependent manner, but did not enhance late apoptosis.
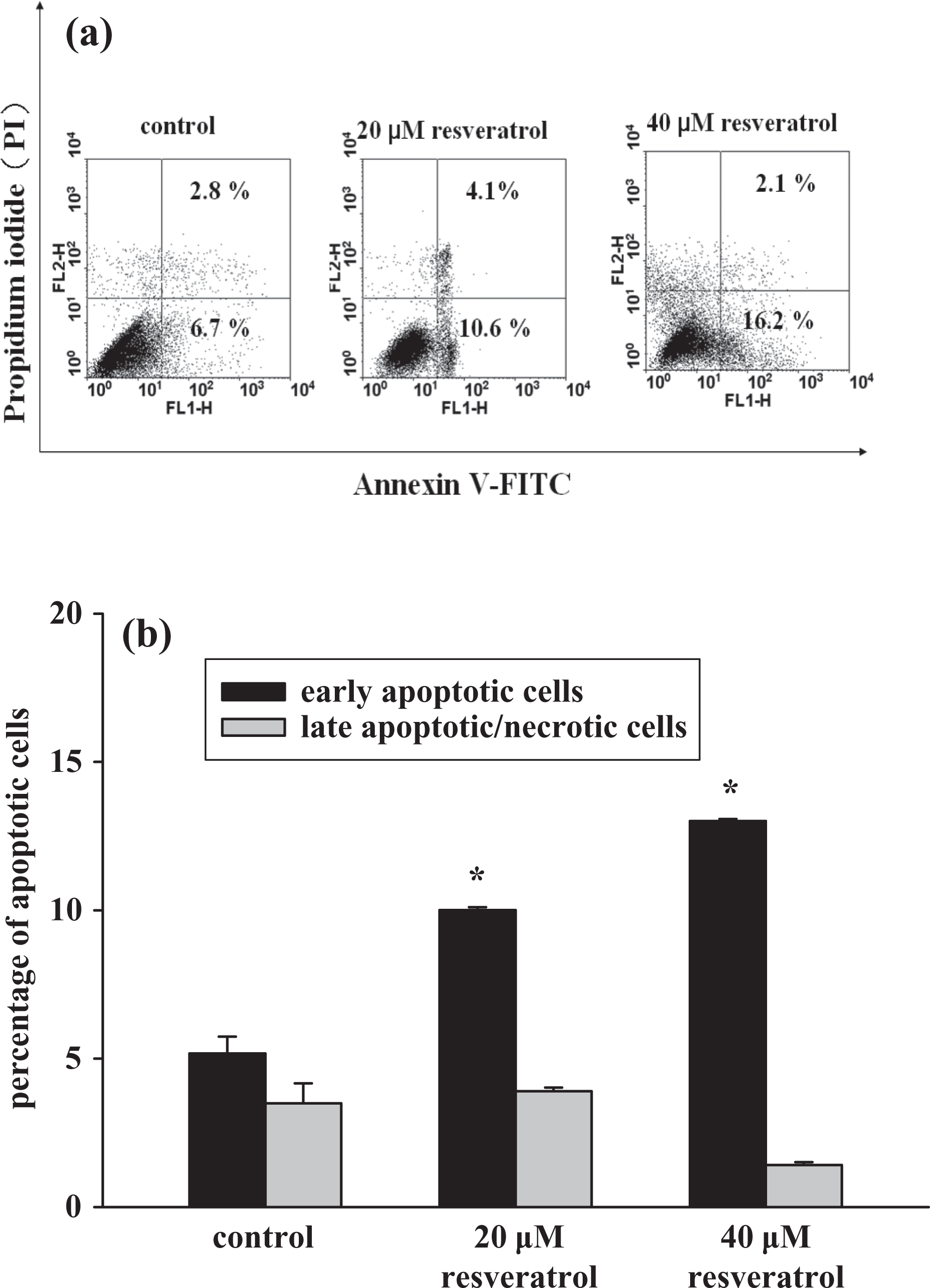
Resveratrol-induced apoptosis as measured by annexin V/PI staining. (a) Cells were treated with 0, 20, and 40 μM resveratrol for 24 h. Cells were then processed for annexin V/PI staining and analyzed by flow cytometry. (b) The percentage of early apoptotic cells and late apoptotic/necrotic cells. Data are mean ± SEM of three separate experiments. *p < 0.05 compared to control. PI: propidium iodide.
Discussion
Our study shows that resveratrol increased [Ca2+]i and caused cell death in OC2 human oral cancer cells. Garcia-Sanchez et al. 8 suggested that resveratrol at higher concentrations (0.1–1 M) mobilized Ca2+ from intracellular stores in tumoral AR42J cells. However, Padín et al. 26 showed that resveratrol at 1 μM caused Ca2+ release in chromaffin cells. Thus, it appears that resveratrol induces [Ca2+]i rise in different cell types at a large concentration range. Furthermore, resveratrol increased [Ca2+]i in OC2 cells by depleting Ca2+ stores and causing Ca2+ influx because the resveratrol-induced Ca2+ signal was partly inhibited by removal of extracellular Ca2+.
Our data show that resveratrol-evoked [Ca2+]i rise was sensitive to nifedipine but insensitive to econazole and SKF96365. These three compounds have been used to inhibit store-operated Ca2+ entry. 27 –29 Store-operated Ca2+ channels have been shown to play a role in [Ca2+]i rise stimulated with several compounds in OC2 cells. 17 –19 Transient receptor potential channels are a possible pathway for Ca2+ entry in nonexcitable cells, 30 but no evidence suggests that OC2 cells possess this channel. In OC2 cells, δ-methrin-induced [Ca2+]i rise was also not inhibited by econazole and SKF96365 but was inhibited by nifedipine. 17 However, in the same cells, bisphenol A dimethacylate-evoked Ca2+ entry was suppressed by nifedipine, econazole, and SKF96365, 18 but carvacrol-induced Ca2+ entry was not altered by these compounds. 19 Thus, OC2 cells appear to have at least three kinds of Ca2+ entry pathways: nifedipine-sensitive Ca2+ channels, store-operated Ca2+ channels, and channels insensitive to these three compounds.
Ca2+ signaling is tightly regulated by PKC activity. 31 Our data show that resveratrol-evoked [Ca2+]i rise was decreased when PKC activity was inhibited by GF109203X (but not activated by PMA). Note that GF109203X inhibited resveratrol-induced [Ca2+]i rise to a similar level in Ca2+-containing medium and Ca2+-free medium, suggesting that GF109203X inhibited resveratrol-induced Ca2+signal via inhibiting Ca2+ release, but not Ca2+ entry. The data that PMA did not increase resveratrol-induced [Ca2+]i rise might be because 20 μM resveratrol had induced the maximum Ca2+ response. However, in PC3 cells, resveratrol-induced [Ca2+]i rise was abolished by PMA but was not altered by other compounds. 13
Regarding the Ca2+ stores involved in resveratrol-evoked Ca2+ release, the BHQ-sensitive endoplasmic reticulum stores seemed to be the dominant one. The data further show that the Ca2+ release was via a PLC-dependent mechanism, given the release was blocked when PLC activity was inhibited. Thus, our data show that resveratrol induced Ca2+ release in a PKC- and PLC-dependent manner. Conversely, in PC3 cells, resveratrol-induced Ca2+ release was PLC independent. 13
Cell viability could be altered in a Ca2+-dependent or Ca2+-independent fashion. 32,33 Our findings showed that resveratrol between 20–100 μM induced cell death in a concentration-dependent manner. BAPTA/AM loading for 25 h did not reverse resveratrol-induced cell death. Therefore, resveratrol-induced cell death did not appear to be caused by a preceding [Ca2+]i rise. Furthermore, 20–60 μM resveratrol-induced cell death was decreased when cytosolic Ca2+ was chelated by BAPTA/AM. This implies that in this case, Ca2+ may play a protective role in resveratrol-induced cell death. Additionally, since BAPTA/AM treatment failed to reverse resveratrol-induced cell death, there was no reason to explore the effect of BAPTA/AM treatment alone on apoptosis. Annexin/PI staining data suggest that apoptosis was involved in resveratrol (20–40 μM)-induced cell death. Based on these results, it is concluded that resveratrol induced Ca2+-independent cell death via apoptosis. Evidence showed that resveratrol induced cell death in other human cancer cells, such as prostate cancer cells, 13 breast cancer cells, 34 and bladder cancer cells. 35 Conversely, resveratrol was shown to cause cell proliferation in PC3 human prostate cancer cells. 13
Clinical evidence shows a single 5 g dose of resveratrol resulted in a maximum concentration (C
max) of 5 μM after 24 h.
36
Consistently, our data show that resveratrol at concentrations between
Conclusion
The findings suggest that via a PKC- and PLC-sensitive pathway, resveratrol evoked Ca2+ release from the endoplasmic reticulum and also caused Ca2+ influx via a nifedipine-sensitive pathway in OC2 human oral cancer cells. Resveratrol also induced Ca2+-independent cell death via apoptosis.
Footnotes
Funding
This work was supported by Kaohsiung Veterans General Hospital (VGHKS100-081, VGHKS101-082, and VGHKS102-094) to HJC.
