Abstract
Effect of sertraline, an antidepressant, on cytosolic free Ca2+ levels ([Ca2+]i) in human cancer cells is unclear. This study examined if sertraline altered basal [Ca2+]i levels in suspended OC2 human oral cancer by using fura-2 as a Ca2+-sensitive fluorescent probe. At concentrations of 10−100 μM, sertraline induced a [Ca2+]i rise in a concentration-dependent fashion. The Ca2+ signal was reduced partly by removing extracellular Ca2+ indicating that Ca2+ entry and release both contributed to the [Ca2+]i rise. Sertraline induced Mn2+ influx, leading to quench of fura-2 fluorescence suggesting Ca2+ influx. This Ca2+ influx was inhibited by suppression of phospholipase A2, inhibition of store-operated Ca2+ channels or by modulation of protein kinase C activity. In Ca2+-free medium, pretreatment with the endoplasmic reticulum Ca2+ pump inhibitor thapsigargin or 2,5-di-(t-butyl)-1,4-hydroquinone (BHQ) nearly abolished sertraline-induced Ca2+ release. Conversely, pretreatment with sertraline greatly reduced the inhibitor-induced [Ca2+]i rise, suggesting that sertraline released Ca2+ from the endoplasmic reticulum. Inhibition of phospholipase C did not change sertraline-induced [Ca2+]i rise. Together, in human oral cancer cells, sertraline induced [Ca2+]i rises by causing phospholipase C-independent Ca2+ release from the endoplasmic reticulum and Ca2+ influx via store-operated Ca2+ channels.
Keywords
Introduction
The antidepressant sertraline has been widely prescribed as a serotonin reuptake inhibitor.1,2 Evidence is accumulating that sertraline has many in vitro effects that are unrelated to its effect on serotonin reuptake. In melanoma cells, sertraline was shown to inhibit protein expression and cause cytotoxicity. 3 Gil-Ad et al. 4 suggest that sertraline induce toxicity to human colon cancer cells and in colorectal cancer-xenografted mice. Sertraline was shown to block Na+ currents in GH3 cells, 5 to inhibit astroglial Kir4.1 channels in HEK293T cells 6 and to activate rat CYP3A level. 7 It is suggested that sertraline inhibits P-glycoprotein. 8 Regarding Ca2+, Becker et al. 9 suggest that sertraline inhibits Ca2+ flux into smooth muscle cells. Lastly, sertraline suppressed the contractile responses to noradrenaline, KCl and electrical field stimulation of rat and human isolated vas deferens10,11 and human isolated mesenteric arteries 12 ; and blocked L-type Ca2+ and transient outward K+ currents in rat ventricular myocytes. 13
A regulated rise in cytosolic free Ca2+ level ([Ca2+]i) is a crucial message for many pathophysiological responses in nearly all cell types. 14 In some cases, an abnormal [Ca2+]i rise can cause apoptosis, dysfunction of proteins, proliferation, interference of ion flux, etc. 15 Sertraline has been shown to inhibit Ca2+ entry in various studies as mentioned above. However, in human platelets it was shown that sertraline caused an increase in [Ca2+]i by activating phospholipase C. 16 In contrast, in a renal cell line (MDCK), sertraline was found to increase [Ca2+]i by causing Ca2+ entry and Ca2+ release in a phospholipase C-independent manner. 17 In oral cells, sertraline is found to cause ulceration of the oral mucosa, 18 and to kill tumor cells and decrease the levels of translationally controlled tumor protein. 19 Additionally, the serotonin precursor tryptophan and its metabolites are shown to significantly increase in patients with oral squamous cell carcinoma. 20 Thus, the present study was aimed to explore the effect of sertraline on [Ca2+]i in human oral cancer cells.
The OC2 human oral cancer cell line is a useful model for oral cancer research. It has been shown that in this cell type, several ligands can cause [Ca2+]i rise. They are thimerosal, 21 fendiline, 22 tamoxifen 23 and safrole. 24
Using fura-2 as a fluorescent Ca2+-sensitive dye, here we show that sertraline induced concentration-dependent [Ca2+]i rises both in the presence and absence of extracellular Ca2+ in OC2 cells. The [Ca2+]i rises were characterized, the concentration-response relationships in the presence and absence of extracellular Ca2+ were established, and the pathways underlying sertraline-induced Ca2+ influx and Ca2+ release were evaluated.
Materials and methods
Cell culture
OC2 human oral cancer cells obtained from American Type Culture Collection were cultured in Dulbecco’s modified Eagle medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U/mL penicillin and 100 μg/mL streptomycin.
Solutions used in [Ca2+]i measurements
Ca2+-containing medium (pH 7.4) contained 140 mM NaCl, 5 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 10 mM Hepes and 5 mM glucose. Sertraline was dissolved in dimethyl sulfoxide as a 1 M stock solution. The other agents were dissolved in water, ethanol or dimethyl sulfoxide. The concentration of organic solvents in the solution used in experiments did not exceed 0.1%, and did not alter viability or basal [Ca2+]i.
[Ca2+]i measurements
Confluent cells grown on 6 cm dishes were trypsinized and made into a suspension in culture medium at a density of 106/mL. Cells were subsequently loaded with 2 μM fura-2/AM for 30 min at 25°C in the same medium. After loading, cells were washed with Ca2+-containing medium twice and was made into a suspension in Ca2+-containing medium at a density of 107/mL. Fura-2 fluorescence measurements were performed in a water-jacketed cuvette (25°C) with continuous stirring; the cuvette contained 1 mL of medium and 0.5 million cells. Fluorescence was monitored with a Shimadzu RF-5301PC spectrofluorophotometer immediately after 0.1 mL cell suspension was added to 0.9 mL Ca2+-containing or Ca2+-free medium, by recording excitation signals at 340 nm and 380 nm and emission signal at 510 nm at 1-sec intervals. Sertraline was added to cells for time periods indicated in the figures. During the recording, reagents were added to the cuvette by pausing the recording for 2 sec to open and close the cuvette-containing chamber. In Figure 1, the concentration range of sertraline-induced response was determined by testing the minimal concentration that could induce a [Ca2+]i rise, and then adding up the concentration to complete the concentration-response plot, which was 10−100 μM. In the rest of the experiments, 50 or 100 μM sertraline was used because at these concentrations sertraline induced significant [Ca2+]i rise. Cell viability was determined before and after the experiments, which was always greater than 95%. The viability was assessed by trypan blue exclusion (adding 0.2% trypan blue to 0.1 mL cell suspension. Dead cells would stain blue.). For calibration of [Ca2+]i, after completion of the experiments, the detergent Triton X-100 and 5 mM CaCl2 were added to the cuvette to obtain the maximal fura-2 fluorescence. Then the Ca2+ chelator EGTA (10 mM) was subsequently added to chelate Ca2+ in the cuvette to obtain the minimal fura-2 fluorescence. [Ca2+]i was calculated as previously described. 25 Mn2+ quench of fura-2 fluorescence was performed in Ca2+-containing medium containing 50 μM MnCl2. MnCl2 was added to cell suspension in the cuvette 1 min before starting the fluorescence recoding. Data were recorded at excitation signal at 360 nm (Ca2+-insensitive) and emission signal at 510 nm at 1-sec intervals as described previously. 26

A, Effect of sertraline on [Ca2+]i in fura-2-loaded OC2 cells. Sertraline was added at 30 sec. The concentration of sertraline was indicated. The experiments were performed in Ca2+-containing medium. Data are typical of three experiments. B, Effect of removal of Ca2+ on sertraline-induced [Ca2+]i signal. Experiments were performed in Ca2+-free medium (Ca2+ was replaced with 0.3 mM EGTA). Data are typical of three experiments. C, Concentration-response plots of sertraline-induced Ca2+ signals in the presence (filled circles) or absence (open circles) of extracellular Ca2+. Y axis is the percentage of control which is the net (baseline subtracted) area under the curve (30-250 sec) of the [Ca2+]i rise induced by 100 μM sertraline. Data are typical of three experiments. Data are mean ± SEM of three experiments. *p < 0.05 compared with open circles.
Chemicals
The reagents for cell culture were from Gibco (Gaithersburg, MD, USA). Fura-2/AM was from Molecular Probes (Eugene, OR, USA). Sertraline was a gift from Pfizer. The other reagents were from Sigma-Aldrich (St. Louis, MO, USA).
Statistics
Data are reported as typical or means ± SEM of three experiments. Data were analyzed by two-way analysis of variances (ANOVA) using the Statistical Analysis System (SAS®, SAS Institute Inc., Cary, NC, USA). Multiple comparisons between group means were performed by post hoc analysis using the Tukey’s honestly significant difference (HSD) procedure. A p value less than 0.05 was considered significant.
Results
Effect of sertraline on [Ca2+]i
Figure 1A shows that before addition of sertraline, the basal [Ca2+]i level was 51 ± 3 μM (n = 3). At concentrations between 10 and 100 μM, sertraline induced [Ca2+]i rises in a concentration-dependent manner in Ca2+-containing medium. The [Ca2+]i rise induced by 100 μM sertraline attained to 90 ± 3 nM (n = 3) followed by a lasting phase within 250 sec. The Ca2+ response saturated at 100 μM sertraline because at a concentration of 200 μM, sertraline induced a similar response as that induced by 100 μM. Figure 1C (filled circles) shows the concentration-response plot of sertraline-induced [Ca2+]i response.
Two possible sources of a Ca2+ signal are extracellular medium and intracellular Ca2+ stores. Further experiments were performed to determine the relative contribution of extracellular Ca2+ influx and intracellular Ca2+ release in sertraline-induced [Ca2+]i rises. The [Ca2+]i rises induced by 10−100 μM sertraline in Ca2+-free medium are shown in Figure 1B. Removal of extracellular Ca2+ did not alter the baseline, suggesting that the amount of leaked fura-2 from the cells was minor. At a concentration of 100 μM, sertraline induced a [Ca2+]i rise by 95 ± 2 nM (n = 3) above baseline followed by a gradual phase. The concentration-response plot of sertraline-induced [Ca2+]i rises in Ca2+-free medium is shown in Figure 1C (open circles). The EC50 value is approximately 20 μM.
Sertraline-induced Mn2+ influx
Experiments were performed to confirm sertraline-induced [Ca2+]i rise involved Ca2+ influx. Mn2+ enters cells through similar pathways as Ca2+ but quenches fura-2 fluorescence at all excitation wavelengths. 26 Thus, quench of fura-2 fluorescence excited at the Ca2+-insensitive excitation wavelength of 360 nm by Mn2+ implies Ca2+ influx. Figure 2 shows that 100 μM sertraline induced an immediate decrease in the 360 nm excitation signal by 10 ± 2 (n = 3) arbitrary units. This suggests that sertraline-induced [Ca2+]i rise involved Ca2+ influx.

Effect of sertraline on Ca2+ influx by measuring Mn2+ quench of fura-2 fluorescence. Experiments were performed in Ca2+-containing medium. MnCl2 (50 μM) was added to cells 1 min before fluorescence measurements. The y axis is fluorescence intensity (in arbitrary units) measured at the Ca2+-insensitive excitation wavelength of 360 nm and the emission wavelength of 510 nm. Sertraline (50 μM) was added as indicated. Data are typical of three experiments.
Sertraline-activated Ca2+ entry pathways
Experiments were performed to explore the Ca2+ entry pathway of the sertraline-induced response. Three store-operated Ca2+ influx inhibitors: nifedipine, econczole, and SK&F96365,27–29 the protein kinase C activator phorbol 12-myristate 13-acetate (PMA), the protein kinase C inhibitor (GF109203X), and aristolochic acid (20 μM; a phospholipase A2 inhibitor) all inhibited 50 μM sertraline-induced [Ca2+]i rise in Ca2+-containing medium (Figure 3 ).

Effect of channel blockers and enzyme inhibitors on sertraline-induced [Ca2+]i rise. The [Ca2+]i rise induced by 50 μM sertraline was taken as control. In blocker- or inhibitor-treated groups, the reagent was added 1 min before sertraline. The concentration was 1 μM for nifedipine, 0.5 μM for econazole, 1 μM for SK&F96365; 20 μM for aristolochic acid, 10 nM for phorbol 12-myristate 13-acetate (PMA), and 2 μM for GF109203X. Data are expressed as the percentage of control (1st column from the left) that is the area under the curve of an interval of 100 sec of 50 μM sertraline-induced [Ca2+]i rise and are mean ± SEM of three experiments. *p < 0.05 compared to control.
Internal Ca2+ stores of sertraline-induced Ca2+ signal
Previous reports have shown that the endoplasmic reticulum is the major Ca2+ store in OC2 cells.21–24 Two inhibitors of endoplasmic reticulum Ca2+ pumps: 2,5-di-(t-butyl)-1,4-hydroquinone (BHQ) 30 and thapsigargin 31 were used as selective tools to deplete the endoplasmic reticulum Ca2+ stores to see whether sertraline utilized this store to cause intracellular Ca2+ release. Figure 4A shows that in Ca2+-free medium, addition of 1 μM thapsigargtin after 450 sec of pretreatment with sertraline failed to induce a [Ca2+]i rise. Figure 4B shows that addition of 1 μM thapsigargin evoked a [Ca2+]i rise of 10 ± 2 nM. Addition of 50 μM sertraline at 500 sec induced a tiny [Ca2+]i rise that was smaller than the control sertraline-induced response (Figure 4A) by 98 ± 2% (n = 3; p < .05; the first 250 sec interval). Figure 4C shows that addition of BHQ after 450 sec pretreatment with sertraline induced a [Ca2+]i rise of 11 ± 2 nM (n = 3). Figure 4D shows that addition of 50 μM BHQ induced a [Ca2+]i rise of 40 ± 2 nM (n = 3). Sertraline (50 μM) added at 500 sec induced only an insignificant [Ca2+]i rise.

Intracellular Ca2+ stores of sertraline-induced Ca2+ release. Experiments were performed in Ca2+-free medium. Sertraline (50 μM), thapsigargin (TG; 1 μM) and BHQ (50 μM) were added at time points indicated. Data are typical of three experiments.
Lack of a role of phospholipase C in sertraline-induced [Ca2+]i rise
Phospholipase C-dependent formation of inositol 1,4,5-trisphosphate is a key step for releasing Ca2+ from the endoplasmic reticulum.14,15 Because sertraline was able to release Ca2+ from the endoplasmic reticulum, the role of phospholipase C in this release was examined. U73122, a phospholipase C inhibitor, 32 was applied to see whether this enzyme was required for sertraline-induced Ca2+ release. Figure 5A shows that ATP (10 μM) induced a [Ca2+]i rise of 99 ± 2 nM (n = 3). ATP is a phospholipase C-dependent agonist of [Ca2+]i rise in most cell types. 33 Figure 5B shows that incubation with 2 μM U73122 did not change basal [Ca2+]i but abolished ATP-induced [Ca2+]i rises. This suggests that U73122 effectively suppressed phospholipase C activity. Figure 5B also shows that addition of 50 μM sertraline after U73122 and ATP treatments caused a [Ca2+]i rise not different from the control sertraline-induced [Ca2+]i rise.
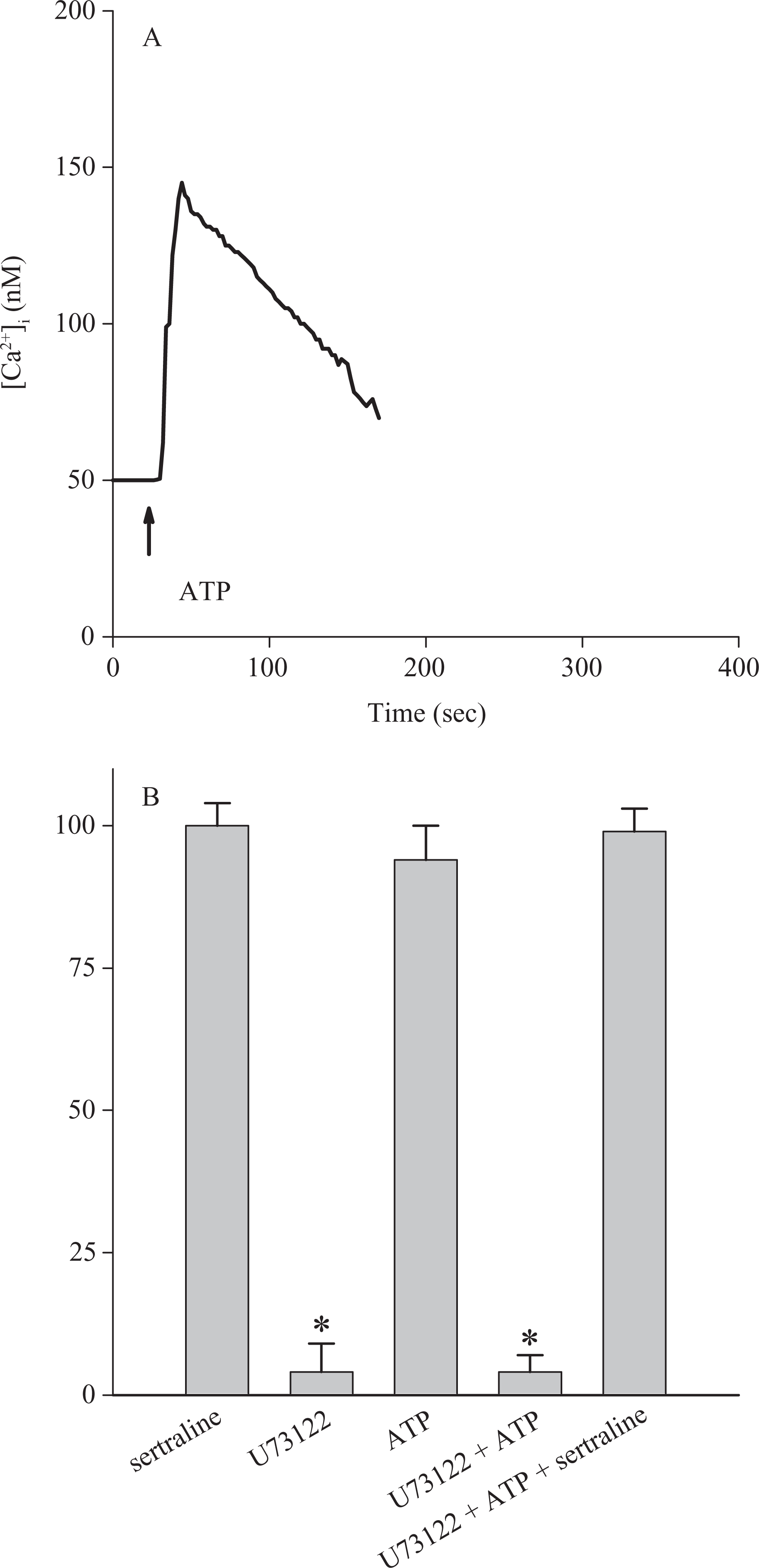
Lack of effect of U73122 on sertraline-induced Ca2+ release. Experiments were performed in Ca2+-free medium. (A) ATP (10 μM) was added as indicated. Data are typical of three experiments. (B) Experiments were performed by adding U73122 (2 μM; for 1 min), ATP (10 μM; for 100 sec), and sertraline (50 μM; for 100 sec) sequentially. U73122 did not increase [Ca2+]i while abolished ATP-induced [Ca2+]i rise. Subsequently added sertraline induced a [Ca2+]i rise similar to control response. Data are mean ± SEM of three experiments.
Discussion
Ca2+ signaling plays a crucial role in the function of almost all cell types including oral cancer cells. For instance, berberine induces apoptosis in human HSC-3 oral cancer cells via simultaneous activation of the death receptor-mediated and mitochondrial pathway that are accompanied by a rise in [Ca2+]i. 34 Thus, elucidation of the mechanisms underlying a compound-induced Ca2+ signal is important for understanding the downstream cellular responses.
Our study is the first to show that sertraline evoked [Ca2+]i rises in OC2 human oral cancer cells and explored the underlying mechanisms. The data show that sertraline induced a concentration-dependent [Ca2+]i rise in OC2 cells between 10 μM and 100 μM. The evidence suggests that sertraline increased [Ca2+]i by depleting intracellular Ca2+ stores and inducing Ca2+ entry from extracellular solution because removing extracellular Ca2+ decreased about 20% of sertraline-induced [Ca2+]i rises. Removal of extracellular Ca2+ reduced the sertraline-induced response throughout the measurement period, suggesting that Ca2+ entry occurred during the whole stimulation interval. The ability of sertraline to induce Ca2+ entry was also independently demonstrated by sertraline-induced Mn2+ quench of fura-2 fluorescence.
The pathway of sertraline-evoked Ca2+ entry was explored. The data indicate that sertraline might induce Ca2+ entry via stimulating store-operated Ca2+ entry because nifedipine, econazole, and SKF96365 all inhibited the [Ca2+]i rise. Nifedipine was originally thought to be a selective blocker of L-type voltage-gated Ca2+ channels. Recent evidence suggests that nifedipine also blocks store-operated Ca2+ channels.27–29 Econazole has been shown to inhibit store-operated Ca2+ channels in different models. SKF96365 is also applied as a store-operated Ca2+ entry blocker.35–37
Our data show that aristolochic acid, a phospholipase A2 inhibitor, significantly inhibited sertraline-induced [Ca2+]i rise. Evidence shows that phospholipase A2 activity is coupled to Ca2+ fluxes. Tedesco et al. 38 suggest that snake PLA2 neurotoxins evoked Ca2+ overload in nerve terminals of cultured neurons. Lupescu et al. 39 thought that human parvovirus B19 capsid protein VP1-induced Ca2+ entry was inhibited when phospholipase A2 activity was suppressed. Thus, these findings are consistent with our data that phospholipase A2 activity might be required for sertraline-induced Ca2+ signal in OC2 cells.
Sertraline has been shown to increase protein kinase C activity in rat cortical synaptoneurosomes 40 ; however, another study showed that sertraline did not alter protein kinase C activity in the soluble and particulate fractions in the frontal cortex. 41 Thus, the effect of modulation of protein kinase C activity on sertraline-induced [Ca2+]i rise was examined in OC2 cells. Both activation and inhibition of protein kinase C decreased sertraline-induced [Ca2+]i rise. The effect of activation is greater. These results are in contrast to that found in MDCK renal cells in which case sertraline-induced [Ca2+]i rise was not affected by store-operated Ca2+ channel blockers or protein kinase C modulators. Thus, the mechanisms underlying sertraline-induced Ca2+ signal in these two cells are apparently different. One of the possible mechanisms that might contribute to sertraline-induced [Ca2+]i rise is that sertraline inhibited plasma membrane Ca2+ ATP pump such that Ca2+ could not leave the cells and [Ca2+]i would rise via Ca2+ leaks across the plasma membrane.
The thapsigargin-sensitive endoplasmic reticulum stores might be the dominant stores for sertraline-induced Ca2+ release because thapsigargin/BHQ pretreatment nearly abolished sertraline-induced [Ca2+]i rise; and conversely, pretreatment with sertraline inhibited thapsigargin-induced [Ca2+]i rise. Furthermore, it seems that phospholipase C-dependent pathways did not have a role in sertraline-induced Ca2+ release, since the response was not much changed when phospholipase C activity was inhibited by U73122. This is in contrast to the data reported in human platelets 16 that sertraline activated PLC. This might be because that the effect of sertraline varies with cell types. In MDCK cells, sertraline also induced Ca2+ release in a phospholipase C-independent manner. 17 How sertraline released Ca2+ from thapsigargin-sensitive store was unclear. Because two kinds of endoplasmic reticulum Ca2+ pump inhibitors both inhibited sertraline-induced [Ca2+]i rise, one possibility was that sertraline also induces Ca2+ release by inhibiting endoplasmic reticulum Ca2+ pumps.
It is interesting that in contrast to the data from renal tubular cells (MDCK) and platelets, both non-excitable cells, several previous studies9–13 performed in muscle cells and myocytes showed that sertraline blocked Ca2+ entry via inhibiting L-type Ca2+ and transient outward K+ currents. Thus, it appears that sertraline affects Ca2+ homeostasis differently in cells derived from tissues of different function.
In a study in which 21 adults were treated with 25−150 mg of sertraline once a day for a month, the plasma level of sertraline was found to be 8 nM−0.32 μM. 42 Our data show that sertraline was able to increase [Ca2+]i at 10 μM. Thus, in situations where sertraline is administered, the interference of sertraline on Ca2+ handling in oral cancer cells might be ignored.
Together, the data show that sertraline evoked Ca2+ release from endoplasmic reticulum and also caused Ca2+ influx in a PLC-independent, phospholipase A2-dependent manner. Since a rise in [Ca2+]i can alter many cellular responses, the effect of sertraline on physiology of different cells needs to be further examined.
Footnotes
Acknowledgement
We thank Pfizer for supplying sertraline to us.
This work was supported by grants from Kaohsiung Veterans General Hospital (VGHKS98-100) to CR Jan and PS98004 to JM Chien.
