Abstract
Objective:
To investigate the effects of overexpression of nuclear factor E2-related factor-2 (NRF2) on lung injury in rats exposed to paraquat (PQ) poisoning.
Methods:
A mifepristone (RU486)-inducible recombinant adenoviral vector carrying the human NRF2 gene (Ad-RUNRF2) was constructed and transfected via airway into the rats 7 days before the administration of RU486. Rats were orally challenged with PQ at 20 mg/kg 24 h after the injection of RU486. On days 0.5, 3 and 21 after PQ poisoning, the expressions of NRF2 and cytokines related to inflammation and oxidation in lung tissue were examined.
Results:
RU486 remarkably enhanced NRF2 mRNA and NRF2 protein levels in Ad-RUNRF2-transfected rats in a dose-dependent manner (p < 0.01). PQ stimulated compensatory overexpression of NRF2, heme oxygenase 1 (HO-1) and NAD(P)H quinone oxidoreductase 1 (NQO-1) in lungs on days 0.5 and 3 after exposure (p < 0.05), but depleted the expression of catalase (CAT), glutathione peroxidase (GSH-Px) and glutathione (GSH), with an increased malondialdehyde (MDA) (p < 0.05). However, pretreatment with Ad-RUNRF2 and RU486 strongly enhanced the expression levels of NRF2, HO-1, NQO-1, CAT and GSH-Px in the lungs of PQ intoxicated rats, with increased GSH and decreased MDA (p < 0.05). Pretreatment with Ad-RUNRF2 and RU486 also strongly suppressed the PQ-induced activation of nuclear factor κB (NF-κB) and decreased the levels of tumour necrosis factor-α (TNF-α), interleukin-1β (IL-1β) and interleukin-6 (IL-6). In addition, Ad-RUNRF2 and RU486 induction significantly reduced PQ-induced pathological changes in lungs and attenuated lung oedema and protein leakage caused by PQ (p < 0.05).
Conclusion:
RU486-induced overexpression of NRF2 in lungs transfected with Ad-RUNRF2 can ameliorate PQ-induced lung injury by the activation of the NRF2-antioxidant response element (ARE) pathway.
Introduction
Paraquat (PQ) is one of the most widely used herbicides in the world. However, it is also toxic to humans and animals. Although several other organs including the liver, kidney, stomach, heart and thymus may also be affected, 1 –3 lungs are the main target for PQ toxicity. 4 –6 Lung injury by PQ poisoning is initially presented as acute lung injury or acute respiratory distress syndrome, which develops into extensive fibrosis. The molecular mechanisms underlying PQ toxicity have not yet been determined; however, it is generally understood that generation of superoxide radicals, activation of inflammatory cells, cell apoptosis and other processes are involved. Recognizing that PQ induces toxic effects mainly via oxidative stress-induced mechanisms, researchers and clinicians have placed great emphasis on the use of antioxidants as a treatment modality for PQ toxicity in animals or humans. However, so far none of these antioxidants have clearly shown effectiveness. 7,8 It has been suggested that the ineffectiveness might be related to properties of exogenous antioxidants including rapid hydrolysis in the circulation and the inability to cross cell membranes. 7 Recently, results from several studies have demonstrated the therapeutic potential of liposomal antioxidants or low-molecular-weight synthetic superoxide dismutase (SOD) mimetics against oxidative damage by facilitating intracellular delivery. 9,10 Therefore, improvement in the endogenous antioxidative capacity might be a promising way to antagonise the toxicity of PQ.
Nuclear factor E2-related factor-2 (NRF2) is a key transcription factor that plays a central role in the protection of cells against oxidative and xenobiotic damage. 11,12 Under quiescent conditions, NRF2 is constantly tethered in the cytoplasm and degraded via the ubiquitin–proteasome pathway in a kelch-like-ECH-associated protein 1 (Keap1)-dependent manner. Upon exposure to stresses, Keap1 is inactivated by direct modification of the cysteine thiol residues and NRF2 is subsequently stabilised. NRF2 translocates into the nucleus and binds to the antioxidant response element (ARE) in the promoter region of a number of phase 2 enzymes including NAD(P)H quinone oxidoreductase 1 (NQO-1), heme oxygenase 1 (HO-1), catalase (CAT), glutathione peroxidase (GSH-Px), etc. Through initiating the transcription of these proteins, NRF2 can enhance the levels of endogenous antioxidants such as glutathione (GSH) to withstand environmental insults. In addition, recent research has also indicated that NRF2 has anti-inflammatory and apoptosis effects. 13,14 Thus, we hypothesise that activation of the NRF2-ARE pathway is a novel protective approach that confers resistance to PQ-induced pulmonary damage.
The adenovirus-mediated overexpression of target genes and proteins is a powerful tool for gene therapy. Although NRF2 acts as a cytoprotective factor for oxidative insults, continuous over-activation of NRF2 has been reported to be related to tumour multidrug resistance and is a possibility for tumorigenesis. 15,16 To enhance NRF2 activity in cells in a spatiotemporally controlled manner, an adenovirus containing a mifepristone (RU486)-inducible regulation system for NRF2 was constructed in our previous work. 17 We have shown that upregulation of NRF2 gene could significantly reduce PQ-induced oxidative damage, inflammatory responses and cell apoptosis in human epithelial cell line A549. 18 Here, we examined the overexpression of RU486-regulated NRF2 in lung tissue from PQ-poisoned rats and investigated the relationship between NRF2 expression and cytokines related to inflammation and oxidation. We further explored the activation of the NRF2-ARE pathway and its key effectors, HO-1 and NQO-1, and provided insights into the protective mechanism of the RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene (Ad-RUNRF2).
Materials and methods
Materials
Animals
Ethical approval for the study was obtained from the Wenzhou Medical University. One hundred and sixty-four male Sprague-Dawley (SD) rats weighing 200–240 g were provided by the animal center of Wenzhou Medical University, Wenzhou, China.
Reagents and instruments
Ad-RUNRF2 was obtained from the Laboratory of Viruses and Gene Therapy, Shanghai Eastern Hepatobiliary Surgery Hospital, Shanghai, China. PQ solution (20%) was purchased from Syngenta Nantong Crop Protection Ltd, Jiangsu, China. RU486 was obtained from Sigma (MO, USA). The NRF2 antibody was purchased from OriGene (MD, USA). The NF-κB antibody was purchased from Abcam (UK). The nuclear protein extraction kit and the BCA protein assay kit were purchased from Pierce (IL, USA). The electrophoretic mobility-shift assay (EMSA) kit was obtained from Roche (Germany). The reverse transcription kit and the polymerase chain reaction (PCR) kit were purchased from Takala Company (Japan). Tumour necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-1β (IL-1β) enzyme-linked immunosorbent assay (ELISA) kits were purchased from R&D Systems (MN, USA). Malondialdehyde (MDA), CAT, GSH-Px and GSH detection kits were obtained from Nanjing Jiancheng Bioengineering Institute (China). Ultrapure water was supplied by a Milli-Q water purification system (Millipore, MA, USA) and used for all experiments.
Primers
All primers (Table 1) were designed using Primer Premier 5.0 (PREMIER Biosoft, Palo Alto, CA, USA) software and synthesised by Sangon Biotech Co., Ltd., Shanghai, China.
Primers.

NRF2: nuclear factor E2-related factor-2; HO-1: heme oxygenase 1; NQO-1: NAD(P)H quinone oxidoreductase 1; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Construction of Ad-RUNRF2
Construction of the Ad shuttle vector pDC312-RUNRF2, identification of Ad-RUNRF2 and expression of Ad-RUNRF2 were achieved using methods described previously. 17,18
Identification of the regulatory efficiency of Ad-RUNRF2 and RU486 in vivo
A total of 44 SD rats were used to evaluate the effects of Ad-RUNRF2 and RU486 on the expression of NRF2 gene and NRF2 protein in lung tissue. Among them, 20 rats were used to assess the dose-dependent effects of Ad-RUNRF2 and RU486 on the target gene and protein expression. Rats were randomly divided into 5 groups (4 rats per group) including the normal group and RU486 0, 125, 250, 500 μg/kg groups. All rats in RU486 groups were transfected with 3 × 109 pfu Ad-RUNRF2 via the respiratory tract 7 days prior to peritoneal injection of RU486. Rats in the normal group were treated with normal saline (NS) at the corresponding time points. Three days after the administration of RU486 or NS, all rats were killed and their lung tissues were harvested. The other 24 rats were used to determine the time-dependent effects of Ad-RUNRF2 in combination with RU486 on NRF2 expression. Rats were randomly divided into 6 groups (4 rats per group) including the normal group and 0, 0.5, 3, 7 and 21 day groups. Ad-RUNRF2 and RU486 were administered as described above. The transfected rats were killed on days 0, 0.5, 3, 7 and 21, respectively, after the administration of RU486.The rats in the normal group were treated and killed as described above.
Acute PQ poisoning
A total of 120 SD rats were used. Animals were randomised into 5 groups (Figure 1) including (1) PQ-poisoned group (PQ, n = 24): rats were orally administered with PQ at 20 mg/kg, a dose determined by our preliminary studies (data not shown). (2) Ad-RUNRF2 intervention group (Ad-RUNRF2 + RU486 + PQ, n = 24): rats were transfected with Ad-RUNRF2 at a dose of 3 × 109 pfu 7 days before the administration of RU486. Twenty-four hours after injection of RU486 at 250 μg/kg, rats were administered with PQ at 20 mg/kg. (3) Ad-RUNRF2 control group (Ad-RUNRF2 + PQ, n = 24): rats in this group underwent the same procedures as those in the Ad-RUNRF2 intervention group except that RU486 was replaced by NS. (4) RU486 control group (RU486 + PQ, n = 24): rats were treated the same as those in the Ad-RUNRF2 intervention group except that NS was used instead of Ad-RUNRF2. (5) Normal group (normal, n = 24): rats were administered with NS at the corresponding time points. On days 0.5, 3 and 21 after PQ poisoning, rats were killed and their lungs and bronchoalveolar lavage fluid (BALF) were immediately harvested.

Acute PQ poisoning and treatment. Rats were transfected with Ad-RUNRF2 at a dose of 3 × 109 pfu via the airway 7 days before the administration of RU486. RU486 was injected intraperitoneally at 250 μg/kg 1 day before the oral administration of PQ at 20 mg/kg. PQ: paraquat; RU486: mifepristone; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; a.i.: airway instillation; i.p.: intraperitoneal injection; i.g.: intragastric.
BALF collection and measurement of protein concentration
BALF was harvested from 4 rats per group. Briefly, the right main bronchus was ligated and the left main bronchus was washed 3 times with saline (2 ml/kg). BALF was collected, filtered through double gauze and centrifuged at 3000g for 15 min at 4°C. The supernatant was collected and kept at −80°C until use. The protein concentration in the BALF supernatant was determined using the BCA kit (Pierce, IL, USA) according to the manufacturer's instructions.
Detection of the lung wet to dry weight (W/D) ratio
The lung W/D ratio was determined in 4 rats per group. Rats were euthanised and the left lungs were immediately isolated. Blood and other contaminants were blotted off the harvested lungs, and the wet weight was measured. The lungs were then dried in a 70°C oven for 72 h and the dry weight was measured. The ratio of lung wet weight to lung dry weight was calculated to assess tissue oedema.
Preparation of lung tissue homogenate and determination of cytokine levels
Lung tissue was collected on 0.5, 3 and 21 days post-exposure. To obtain a 10% homogenate of lung tissue, 200 g lung tissue and 1.8 ml ice cold NS were mixed and homogenised using a high-speed homogeniser. Larger amounts of debris were cleared by centrifugation at 3000g for 15 min at 4°C. The supernatant was stored at −80°C. The concentrations of TNF-α, IL-6 and IL-1β were measured using the corresponding ELISA kits according to the manufacturer's instructions. Chemical colorimetric assays were used to detect the concentrations of CAT, GSH-Px, GSH and MDA.
Determination of the activated NRF2-ARE pathway by real-time PCR assay
Total RNA was extracted from lung tissue using TRIzol Reagent and then reverse transcribed into complementary DNA. Quantitative PCR was performed on an ABI PRISM 7300 Sequence Detector System (Perkin-Elmer Applied Biosystems, MA, USA). Quantitative values were obtained from the threshold cycle (CT) number. The expression level of target genes from each sample was normalised with respect to its glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA content.
Measurement of NRF2 and NF-κBp65 by Western blot assay
Lung homogenate was prepared using the protocol described above. Protein, 50 µg, was heated to 95°C and separated using sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE). Proteins on the gel were transferred onto a nitrocellulose membrane and incubated sequentially with milk, the primary antibody for NRF2 or NF-κBp65 and the secondary antibody. The proteins of interest were then detected by chemiluminescence assay. A molecular biology imaging system was used for quantitative analysis.
Determination of the activated NRF2 by EMSA
Nuclear proteins were extracted from lung tissue and the concentration was determined using Coomassie brilliant blue staining assay. Activated NRF2 in lung tissue was analysed using EMSA according to the manufacturer's instructions. Briefly, NRF2 was labelled with [γ- 32 P] ATP using T4 terminal ligase and purified by ethanol precipitation. Oligonucleotides of 5′-GGGACTAGTCGTCGGG-3′ sequence were used as the probes. Nuclear protein extracts and probes were mixed at room temperature, separated on a 6% polyacrylamide gel and transferred to a membrane. The membrane was cross linked, blocked, washed and then developed.
Histological examination of lung tissue
Lung tissue was harvested from 2 rats per group and histologically examined. Briefly, rats were anesthetised with 10% chloral hydrate and their chests opened. Lungs were fixed by perfusion using 1% glutaraldehyde containing 4% paraformaldehyde through the main bronchus. Right lungs were then harvested and fixed with 4% paraformaldehyde. Fixed tissue was embedded in paraffin, cut into 5 μm sections and stained with hematoxylin and eosin (H&E).
Statistical analysis
All data were analysed using SPSS 16.0 (IBM). Data are presented as mean ± SD. Differences between the two groups were analysed by single-tailed Student's t-test while those between 3 or more groups were analysed by one-way analysis of variance (ANOVA). The difference of p < 0.05 or p < 0.01 was considered statistically significant.
Results
RU486 increased NRF2 mRNA and NRF2 protein expression levels by transfection in rats with Ad-RUNRF2
In order to determine whether the RU486-inducible Ad-RUNRF2 is capable of mediating the expression of NRF2 in lung tissue, real-time PCR analysis and Western blotting were performed. As shown in Figure 2, RU486 increased the target gene (Figure 2(c)) and protein (Figure 2(a)) expressions in a dose-dependent manner. Under normal conditions, the levels of both NRF2 mRNA and protein were low. After transfection with Ad-RUNRF2, the addition of RU486 significantly enhanced NRF2 mRNA and protein levels in rat lung tissue (125 μg/kg vs. normal, p < 0.01). Administration of 500 μg/kg RU486 increased the mRNA and protein levels by about 30-fold and 10-fold, respectively. However, transfection with Ad-RUNRF2 without the induction of RU486 did not affect the expression of NRF2 gene and NRF2 protein (0 μg/kg vs. normal, p > 0.05).
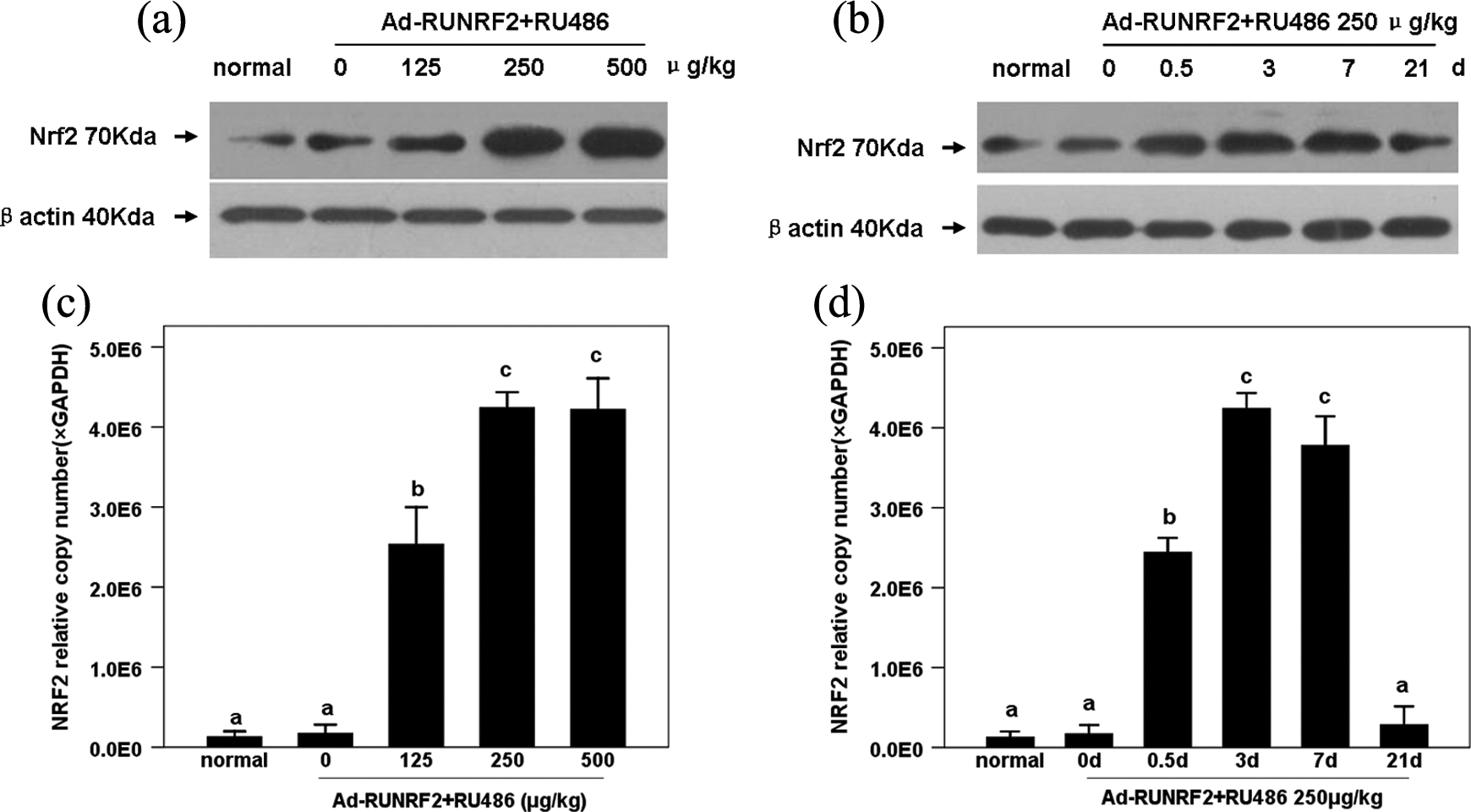
Effects of RU486 on Ad-RUNRF2-mediated NRF2 mRNA expression and NRF2 protein in normal lungs. Rats were intratracheally instilled with Ad-RUNRF2 (3 × 109 pfu) 7 days before injection of RU486 at increasing doses for 3 days in the dose-dependent effect study. Lungs were harvested for NRF2 mRNA expression by real-time PCR (C), and the NRF2 protein level by Western blot (A). And in the time-dependent effect study, rats were treated with Ad-RUNRF2 as described above and lungs were harvested at different times after injection with RU486 at a dose of 250 μg/kg. NRF2 mRNA expressions were determined by real-time PCR (D), and NRF2 protein levels were detected by Western blot (B). Rats administered with NS and intratracheal instillation of NS acted as the control group. Data were represented as mean ± SD (n = 4). Bars with different characters are statistically different at the p < 0.01 level. RU486: mifepristone; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; NRF2: nuclear factor E2-related factor-2; NS: normal saline.
RU486 also increased NRF2 expression in Ad-RUNRF2-transfected rat lungs in a time-dependent manner (Figure 2(b) and (d)). Real-time PCR results (Figure 2(d)) demonstrated that the NRF2 mRNA level significantly increased at 12 h after the administration of RU486 (p < 0.01). The NRF2 mRNA level peaked on day 3 (about a 30-fold increase), then decreased gradually and returned to the normal level 3 weeks after RU486 induction. The expression of the NRF2 protein showed similar time-dependent changes as that of NRF2 mRNA (Figure 2(b)). These results suggest that Ad-RUNRF2 and its inductor RU486 can time-dependently upregulate NRF2 mRNA and NRF2 protein levels in vivo.
RU486 stimulated NRF2-ARE pathway activation and attenuated oxidative damage in Ad-RUNRF2-transfected lungs in rats with PQ poisoning
NRF2 binds to ARE and causes transcriptional activation of the protective genes. Activation of the NRF2-ARE pathway protects against oxidative stress-induced cell death. In order to verify the protective mechanism of RU486 against PQ-induced lung injury, the activation of the NRF2-ARE pathway and oxidative damage in lungs were explored on days 0.5, 3 and 21 in rats exposed to PQ with pretreatment of Ad-RUNRF2 and RU486 (250 μg/kg). As shown in Figure 3(d), PQ increased the NRF2 mRNA level by 5 to 6 folds in lung tissues on days 0.5 and 3 (p < 0.01). Pretreatment with Ad-RUNRF2 and RU486 further enhanced the NRF2 mRNA expression on days 0.5, 3 and 21 (p < 0.01), with the maximal NRF2 mRNA level observed on day 3. However, pretreatment with Ad-RUNRF2 or RU486 (250μg/kg) alone did not obviously affect the expression of NRF2 mRNA in rat lungs exposed to PQ. RU486 and Ad-RUNRF2 also increased the translocation of NRF2 in lungs on days 0.5, 3 and 21, respectively, as determined by the EMSA (Figure 3(a)–(c)). These results suggest that the RU486-induced overexpression of NRF2 gene can enhance the activity of NRF2 in lung tissue from PQ-intoxicated rats.

Effects of RU486 on the NRF2 mRNA level and NRF2 translocation in Ad-RUNRF2-transfected lungs with the PQ challenge. Rats were intratracheally instilled with Ad-RUNRF2 (3 × 109 pfu) 7 days before the administration of RU486 (250μg/kg). And 24 h later, the rats were orally ingested with PQ (20 mg/kg). Lungs were harvested on days 0.5, 3 and 21 after PQ intoxication for NRF2 mRNA expression by real-time PCR (d), and NRF2 translocation assays with EMSA ((a) 0.5 days; (b) 3 days; (c) 21 days). Rats that were intratracheally instilled with Ad-RUNRF2 and induced with NS acted as the Ad-RUNRF2 control group and those intratracheally instilled with NS and induced with RU486 acted as the RU486 control group. The rats in the normal control group were treated with NS for both transfection and induction. Data were represented as mean ± SD (n = 8). Bars with different characters are statistically different at the p < 0.05 level. RU486: mifepristone; NRF2: nuclear factor E2-related factor-2; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; PQ: paraquat; PCR: polymerase chain reaction; EMSA: electrophoretic mobility-shift assay; NS: normal saline.
NRF2 downstream effectors including HO-1, NQO-1, CAT and GSH-Px were also analysed by real-time PCR analysis and chemical colorimetric assays. As shown in Figure 4, PQ stimulated compensatory overexpression of HO-1 and NQO-1 on days 0.5 and 3 (p < 0.05), with a decreasing trend (Figure 4(a) and (b)). PQ also significantly suppressed CAT and GSH-Px as shown by a decrease in GSH and an increase in MDA (Figure 4(c)–(f)). However, pretreatment with Ad-RUNRF2 in combination with RU486 strongly enhanced the expression of HO-1, NQO-1, CAT and GSH-Px, showing an increase in GSH and a decrease in MDA (p < 0.05).

Effects of RU486 on ARE-responsive phase 2 enzymes and antioxidants in Ad-RUNRF2-transfected lungs exposed to PQ. Rats were intratracheally instilled with Ad-RUNRF2 (3 × 109 pfu) 7 days before the administration of RU486 (250μg/kg). And 24 h later, the rats were orally ingested with PQ (20 mg/kg). Lungs were harvested for analysis on days 0.5, 3 and 21 after PQ intoxication. HO-1 and NQO-1 mRNA expressions were determined by real-time PCR (a and b), and the other enzymes and antioxidants, CAT (c), GSH-Px (d) and GSH (e) were measured by chemical colorimetric assays. MDA, the indicator of lipid peroxidation, was analysed by chemical colorimetric assays (f). Data were represented as mean ± SD (n = 8). Bars with different characters are statistically different at the p < 0.05 level. RU486: mifepristone; ARE: antioxidant response element; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; PQ: paraquat; HO-1: heme oxygenase 1; NQO-1: quinone oxidoreductase 1; PCR: polymerase chain reaction; CAT: catalase; GSH-Px: glutathione peroxidase; GSH: glutathione; MDA: malondialdehyde.
These results suggest that the RU486-induced overexpression of NRF2 can protect lungs from PQ-induced oxidative damage by the activation of the NRF2-ARE pathway.
RU486-induced overexpression of NRF2 suppressed NF-κB activation and attenuated inflammation in lung injury evoked by PQ
As shown in Figure 5, PQ remarkably activated the NF-κB-signalling pathway in a time-dependent manner, as shown by the increase in the expression level of NF-κBp65 on days 0.5, 3 and 21 (Figure 5(a)—(d)). NF-κB-regulated cytokines, such as TNF-α, IL-1β and IL-6, were also increased (Figure 5(e)—(g)). However, pretreating rats with Ad-RUNRF2 in combination with RU486 strongly suppressed PQ-induced activation of the NF-κB-signalling pathway, as demonstrated by the decrease in the expression levels of NF-κB and its downstream cytokines, TNF-α, IL-1β and IL-6. It is also worth mentioning that pretreatment with RU486 or Ad-RUNRF2 alone did not alter the expression levels of these proteins. These results indicate that the activation of NF-κB and its downstream cytokines play a role in PQ-induced inflammatory injury in lungs, and RU486-induced overexpression of NRF2 effectively reduces inflammation by the inhibition of the PQ-activated NF-κB-signalling pathway.

Effects of RU486 on NF-κB and its downstream proinflammatory cytokines in Ad-RUNRF2-transfected lungs exposed to PQ. Rats were intratracheally instilled with Ad-RUNRF2 (3 × 109 pfu) 7 days before the administration of RU486 (250 μg/kg). And 24 h later, the rats were orally ingested with PQ (20 mg/kg). Lungs were harvested for analysis on days 0.5, 3 and 21 after PQ intoxication. The levels of NF-κBp65 were determined by Western blot in lungs harvested on days 0.5 (a), 3 (b) and 21 (c), and the quantitative assays were analysed using a molecular biology imaging system (d). The concentrations of TNF-α, IL-1β and IL-6 were measured using ELISA kits (e, f and g). Data were represented as mean ± SD (n = 8). Bars with different characters are statistically different at the p < 0.05 level. RU486: mifepristone; NF-κB: nuclear factor κB; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; PQ: paraquat; NF-κBp65: nuclear factor κB p65; TNF-α: tumour necrosis factor-α; IL-1β: interleukin-1β; IL-6: interleukin-6; ELISA: enzyme-linked immunosorbent assay.
RU486-induced overexpression of NRF2 improved histopathological changes in lungs and reduced lung oedema and the protein level in BALF
The typical pathological features of acute lung injury include alveolitis, leukocyte infiltration, increase in alveolar wall thickness, protein leakage, and oedema. As shown in Figure 6(a), several histopathological alterations including cell structure destruction, alveolar wall thickening, lung oedema, and neutrophil infiltration were observed in the lungs after poisoning with PQ. These morphological alterations were more obvious on days 3 and 21. Pretreatment with Ad-RUNRF2 and RU486 significantly improved PQ-induced pathological signs in lungs.

Effects of RU486 on histopathological changes, protein accumulation in BALF and the wet/dry weight ratio in Ad-RUNRF2-transfected lungs with the PQ challenge. Rats were intratracheally instilled with Ad-RUNRF2 (3 × 109 pfu) 7 days before the administration of RU486 (250μg/kg). And 24 h later, the rats were orally ingested with PQ (20 mg/kg). Lungs were harvested for analysis on days 0.5, 3 and 21 after PQ intoxication. The histopathological assays (40× magnification) in lungs were performed at 0.5, 3 and 21 days after PQ exposure in the normal control group, PQ group and Ad-RUNRF2 + RU486 + PQ (a). The pulmonary oedema was determined by the wet/dry weight ratio (b) and the protein level in BALF (c) was measured at 0.5, 3 and 21 days after PQ exposure. Data were represented as mean ± SD (n = 4). Bars with different characters are statistically different at the p < 0.05 level. RU486: mifepristone; BALF: bronchoalveolar lavage fluid; Ad-RUNRF2: RU486-inducible recombinant adenoviral vector carrying the human NRF2 gene; PQ: paraquat.
In addition, the effects of NRF2 overexpression induced by RU486 on lung W/D ratio and protein level in BALF were investigated in this study. Compared with the control group, oral ingestion of PQ resulted in a significant increase in the protein level of BALF and lung oedema evidenced by the elevation in lung W/D ratio. Pretreatment with Ad-RUNRF2 and RU486 remarkably attenuated the increase in lung oedema and protein leakage caused by PQ (Figure 6(b) and (c)). These findings indicate that RU486-induced overexpression of NRF2 via Ad-RUNRF2 transfection could be a novel protective way for PQ-induced pulmonary injury.
Discussion
In this study, we demonstrate for the first time that pretreatment with a RU486-inducible recombinant adenovirus Ad-RUNRF2 significantly inhibited PQ-induced pulmonary injury in rats. Furthermore, the protection effect of Ad-RUNRF2 against PQ-induced pulmonary damage was mediated by activation of the NRF2-ARE pathway and suppression of oxidative damage and NF-κB-mediated inflammatory responses.
The RU486-inducible gene regulatory system is widely used in many animal models. 19,20 Its regulatory protein consists of the yeast galactose 4 (GAL4) DNA-binding domain, the truncated human progesterone receptor ligand-binding domain and the activation domain of the p65 subunit. 21 Initially produced as an inactive monomer, it is activated in the presence of RU486, which triggers a conformational change and causes the formation of an active homodimer. The active homodimer then binds to the GAL4 sites in the inducible promoter and stimulates the transcription of the transgene, leading to increased production of the protein of interest. This is a powerful system that enables target genes to be expressed within a certain range in vivo in proportion to the concentration of RU486 used for induction. Due to the absence of GAL4 in mammalian cells, RU486 cannot activate any endogenous gene, therefore making the system safe. Additionally, the amount of RU486 required to activate the system is very low, approximately 1000 times lower than that used for abortion (10 mg/kg). Here, we show that the expression of NRF2 in Ad-RUNRF2-transfected lungs was dependent on the dose of RU486, and both NRF2 gene and NRF2 protein were expressed at low levels in the absence of RU486. Therefore, our studies have shown that rat lung tissue was successfully transfected with Ad-RUNRF2 via airway instillation (a.i.) and expression of NRF2 was regulated by RU486.
Over the past few decades, research on the mechanisms of PQ toxicity has identified several toxic outcomes of the redox cycling reaction, including the generation of reactive oxygen species (ROS), depletion of NADPH, oxidation of GSH and oxidative damage to lipids, proteins and DNA. 8 The importance of oxidative stress as a mechanism of PQ toxicity has been demonstrated in plants and bacteria, both in vitro and in vivo. 7,22 Thus, in theory, antioxidant therapy should be effective if appropriate measures are taken. The NRF2-ARE pathway plays a central role in the protection of cells against oxidative and xenobiotic damage. Target genes of this pathway are involved in ROS elimination, GSH synthesis and xenobiotic detoxification. 23 More recently, we and others have demonstrated that some natural extracts and compounds, such as cycloartenyl ferulate, α-lipoic acid and rosiglitazone, showed cytoprotective effects on PQ-exposed human cell lines via activating NRF2-ARE pathway, 24 –26 suggesting that NRF2 is an effective target for protection against oxidative damage induced by PQ. In this study, oral ingestion of PQ resulted in an overwhelming oxidative damage to the lung tissue in rats, as demonstrated by the significantly increased MDA, lung oedema and protein leakage in BALF and decreased GSH, CAT and GSH-Px. The Ad-RUNRF2-mediated overexpression of NRF2 gene could dramatically enhance the activities of NRF2 and its downstream cytoprotective genes and proteins including HO-1, NQO-1, CAT and GSH-Px, remarkably increase the GSH level, suppress the marker of oxidative damage and attenuate histopathological changes. These results indicate that RU486-inducible Ad-RUNRF2 protects the rats against PQ-induced pulmonary damage through suppression of oxidative stress by activation of the NRF2-ARE pathway.
Activation of the NF-κB and overexpression of its downstream cytokines plays an important role in the pathogenesis of acute lung injury induced by a variety of factors. 27,28 It has been shown that excessive activation of the NF-κB-signalling pathway resulted in overly exuberant inflammatory injury of lungs after exposure to PQ. 29,30 Similarly, we showed in this study that PQ significantly stimulated the expression of NF-κB and its downstream proinflammatory cytokines (TNF-α, IL-1β and IL-6) and caused obvious leukocyte infiltration in the lungs. These, however, were reversed by pretreating rats with Ad-RUNRF2, which regulated the target gene expression in vivo. Potential crosstalk between NRF2 and NF-κB pathways might be the mechanism for this finding. ROS have been recognised as important inducers of NF-κB. 31 Increased intracellular superoxide concentrations induced by PQ enhanced nuclear accumulation of NF-κB in neutrophils via a p38MAPK-dependent mechanism and increased the expression of NF-κB-dependent proinflammatory cytokines. 32 The activation of NRF2 and ARE responsive intracellular antioxidants can abolish the activation of NF-κB by depleting ROS. Increasing the intracellular GSH level after activating the NRF2-ARE pathway can decrease the release of cytokines and chemokines from lung cells by decreasing NF-κB activation. 33 NRF2-mediated HO-1 induction has been demonstrated to exert a protective effect against inflammation. 34 Previous studies have indicated that the anti-inflammatory activity of HO-1 is expressed by the regulation of immune-mediated inflammation and the suppression of proinflammatory cytokines, chemokines, ROS formation and iNOS through attenuation of NF-κB activation in several inflammatory diseases. 35,36 Recently, cyclo (His-Pro), an endogenous cyclic dipeptide, has also been shown to inhibit NF-κB nuclear accumulation induced by PQ in rat pheochromocytoma PC12 cells via NRF2-mediated HO-1 activation. These results again suggest the effective suppression of NF-κB activation by HO-1. 37 Thus, Ad-RUNRF2-mediated overexpression of NRF2 and the subsequent activation of NRF2-ARE pathway inhibit NF-κB inflammatory pathways by a multiway and multilevel mechanism and play a protective role in PQ-induced pulmonary inflammatory injury.
In summary, these results indicate that RU486 can successfully control the expression of NRF2 in lung tissue transfected with Ad-RUNRF2. RU486 induced overexpression of NRF2 to ameliorate PQ-induced lung injury in rats by mediating oxidation and NF-κB-dependent inflammatory responses via activating the NRF2-ARE pathway. This study therefore provides an important insight into the potential clinical treatments that may attenuate pulmonary injury caused by PQ poisoning.
Footnotes
Acknowledgements
We thank Professor Qijun Qian, the director of viral gene therapy laboratory and the staff of Shanghai Hepatobiliary Surgery Hospital for helpful technical guidance on the construction of Ad-RUNRF2.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by the Natural Science Foundation of Zhejiang Province (Y2080977), the Core Project of Medicine and Health Care Platform in Zhejiang Province (2012RCA040), the Key Discipline Construction Project of Colleges and universities in Zhejiang Province and the key construction academic subject (medical innovation) of Zhejiang Province (11-CX26).
