Abstract
To authenticate the colon cancer preventive potential of silibinin, the efficacy of silibinin needs to be tested by evaluating an organ-specific biomarker. The aim of this study was to evaluate the impact of silibinin on the colonic expression of the caudal-type homeobox transcription factor (CDX2) an intestine specific tumor suppressor gene and its downstream targets in the colon of rats challenged with 1,2 dimethyl hydrazine (DMH). Rats of groups 1 and 2 were treated as control and silibinin control. Rats under groups 3 and 4 were given DMH (20 mg/kg body weight (b.w.) subcutaneously) once a week for 15 consecutive weeks from the 4th week of the experimental period. In addition, group 4 rats alone were treated with silibinin (50 mg/kg b.w. per os) everyday throughout the study period of 32 weeks. Histological investigation and messenger RNA and protein expression studies were performed in the colonic tissues of experimental rats. Findings of the study revealed that DMH administration significantly decreased the expression of CDX2 and Guanylyl cyclase C (GCC) in the colon of experimental rats. Further the decreased levels of CDX2 protein, colonic mucin content, and increased number of mast cells in the colon of DMH alone-administered rats reflects the onset of carcinogenesis. The pathological changes caused due to CDX2 suppression were attenuated by silibinin supplementation.
Introduction
Human colorectal cancer (CRC) is a heterogeneous disease in which genetic mutations and environmental factors contribute to tumor initiation, promotion, growth, and malignant transformation. 1,2 These genetic mutations corrupt normal homeostatic circuits, including cell cycle and proliferative regulation, DNA damage sensing and repair, and metabolic programming which, in the absence of appropriate apoptotic responses, recursively amplify the accumulation of genetic mutations. 3,4 CRCs are associated with the stepwise accumulation of genetic alterations involving many tumor suppressor genes and proto-oncogenes such as adenomatous polyposis coli (APC), Kirsten-ras, tumor protein p53, and B-cell lymphoma 2. 5 These genes are also relevant for cancers outside the gut and no intestine-specific gene has been implicated in CRC. 6 Caudal-type homeobox transcription factor 2 (CDX2), a member of the caudal-related homeobox gene family, plays a key role in early mammalian intestinal development and the maintenance of intestinal epithelia via regulation of intestine-specific gene transcription. 7,8 CDX2 may stimulate intestinal proliferation and differentiation, by transcriptional activation of intestine-specific proteins (MUC2, sucrase-isomaltase and carbonic anhydrase I). 9,10 Also, CDX2 regulates the expression of ubiquitous genes important in controlling cell dynamics, including adhesion, proliferation, cell cycle regulation, and apoptosis. 11 –13 Evidence is accumulating to suggest that the CDX2 is implicated in the pathogenesis of CRC. 14,15 Studies indicate that CDX2 performs a tumor suppressor role in human colorectal carcinogenesis. 16,17
Cancer chemoprevention aims to reverse, suppress, or prevent the development of cancer using natural, synthetic, or biologic substances. Silibinin is a naturally occurring polyphenolic antioxidant that constitutes the major portion of the dietary supplement milk thistle (Silybum marianum L.) extract. Silibinin has exhibited a significant role in cancer prevention and control, in several different rodent epithelial cancer models. 18,19 The objectives of the study were to examine whether silibinin had any effect in the gene and protein expression of CDX2 and to compare any such effect on mucin content, mast cell recruitment, and guanylyl cyclase C (GCC) expression in the colon tissue of rats challenged with the procarcinogen 1,2 di-methyl hydrazine (DMH).
Methods
Chemicals
1,2 Dimethyl hydrazine (DMH) and silibinin were purchased from Sigma Chemical Company (St Louis, Missouri, USA). All other chemicals and solvents used were of analytical grade and obtained from Hi-Media Laboratories (Mumbai, Maharashtra, India).
Experimental model
Adult male albino Wistar rats aged 5 weeks and weighing 130–150 g were obtained from the Central Animal House, Annamalai University, Annamalainagar, Chidambaram, Tamil Nadu, India. All the rats were housed in plastic cages with paddy husk for bedding in a room at 27 ± 2°C and a relative humidity of 50 ± 10% with 12-h light/12-h dark cycle. Rats were fed with standard pellet diet (M/S Sai Enterprises, Chennai, Tamil Nadu, India) and water ad libitum. This study has been approved by the Institutional Animal Ethical Committee of Annamalai University (160/1999/CPCSEA/437).
Experimental design
After allowing the rats to acclimatize for 1 week, all the animals were randomly divided into four groups with six rats in each group, and all the rats were fed with the standard pellet diet. Rats in group 1 received 0.5% carboxymethyl cellulose and served as control, and rats in group 2 received silibinin at the dose of 50 mg/kg body weight (b.w.) by intragastric intubation every day for a period of 32 weeks starting from day 1 of the experiment. Rats in groups 3 and 4 received DMH at a dose of 20 mg/kg b.w. subcutaneously once a week for a period of 15 weeks starting from week 4 of the experimental period. In addition, rats in group 4 received silibinin as in group 2 every day for a period of 32 weeks. The total experimental duration was 32 weeks.
Histopathological observations
For histopathological studies, the colon segments were dissected, fixed immediately in 10% formalin, and embedded with paraffin. Then, 4 μm sections were cut in a rotary microtome, stained with hematoxylin–eosin, and viewed under a light microscope.
Alcian blue–periodic acid–Schiff staining for mucin
Paraffin-embedded 4 μm thick colon tissue sections were incubated at 60°C for 60 min, deparaffinized in two changes of xylene, and rehydrated through graded alcohols at room temperature and stained with Alcian blue for 30 min. The sections were washed with running tap water and rinsed in distilled water followed by periodic acid treatment for 30 min. The sections were again rinsed in distilled water and incubated in Schiffs reagent for 60 min, washed in running tap water, and counterstained with hematoxylin. Presence of mucin was evidenced by the presence of Alcian blue-positive cells. The images were viewed by light microscopy and photographed.
Toludine blue staining for mast cells
After dewaxing and rehydration, the tissue sections were treated with 0.5% solution of toluidine blue dissolved in 0.5 M of hydrochloric acid (HCl; pH 0.5) for 5–7 days, filtering the solution on alternate days. Then, the slides containing the tissue sections were rinsed in distilled water and masked with 5% ammonium molybdate for 5 min, washed in running tap water for 10 min, and counterstained by 1% eosin. The sections were again washed in running tap water, then placed in n-butanol for 5 min, cleared in xylene, air-dried, and mounted using dibutyl phthalate xylene (DPX). The mucosal mast cell granules, which stained dark blue against a clean background, were photographed.
Immunolocalization of CDX2 protein
Paraffin-embedded 4 μm thick colonic tissue sections were dewaxed and rehydrated in graded alcohol. Heat-induced antigen retrieval was performed using 10 mM sodium citrate buffer (pH 6.0) in a pressure cooker.Tris–HCl buffer (0.05 M; pH 7.6) was used to prepare solutions and for washes between various steps. Sections were treated with 2% bovine serum albumin for 40 min at room temperature and incubated overnight with the appropriate primary antibody, namely, anti CDX2 (BioGenex Laboratories Inc. San Raman, California, USA). To achieve color development, samples were incubated with diaminobenzidine (DAB) using the labeled streptavidin–biotin kit (BioGenex Laboratories) and counterstained with hematoxylin.
mRNA expression of GCC and CDX2
Total RNA was extracted from the tissue by total RNA kit (Qiagen, Germany). The residual genomic DNA was removed by incubating the RNA with DNase (Qiagen, Valencia, California, USA). Secondary structures of RNA were denatured by incubating the RNA reaction mixture comprising of random hexamer, oligo dT primer, deoxynucleotide triphosphate (dNTP) mix, and RNA made up to 20 μL with nuclease-free water at 65°C for 5 min. Single-stranded complementary DNA (cDNA) was synthesized in a thermocycler for 1 h at 50°C from a 20-μL reaction mix comprising 5× cDNA synthesis buffer, 0.1 M dithiothreitol, RNase, nuclease-free water, reverse transcriptase, and denatured RNA. The reverse transcription polymerase chain reaction (RT–PCR) for GCC was performed with 30 cycles in a 50-μL reaction consisting of 10× PCR buffer, 50 mM magnesium chloride, 10 mM dNTP, forward primer (CCACGTGCGGTCTAAGAACT), reverse primer (GACTTGGGGGCTGGTTCTAC), Taq DNA polymerase, cDNA, and nuclease-free water. For this semiquantitative RT-PCR assay, the PCR cycle consisted of 94°C for 30 s, 55°C for 30 s, and 72°C for 45 s, with final extension at 72°C for 5 min. The PCR products (5 µL) were subjected to electrophoresis in 2% agarose gels containing 0.5 µg/mL ethidium bromide. The gels were illuminated with ultraviolet light and photographed.
For quantitative RT-PCR assay, the reaction was performed with a 20-μL mixture consisting of RNA, forward primer (CAGGAGGAAAGCTGAGTTGG), reverse primer (GCAACTTCTTTTTGTTGATTTTCC), reverse transcriptase, and SYBR green master mix made up to 20 μL with nuclease-free water. The PCR-programmed conditions to study the messenger RNA (mRNA) expression of CDX2 were reverse transcription at 55°C (10 min), initiation at 95°C (2 min), two-step cycling at 60°C (50 s), annealing at 60°C (50 s), and final extension at 70°C (20 s). The denaturation and annealing steps were carried out for 40 cycles to determine the C t values of CDX2. The relative gene expression quantification method was used to calculate the fold change of mRNA expression according to the comparative C t method using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as endogenous control.
Statistical analysis
Data are expressed as the means ± SD. Significant difference between the means of the six groups was statistically analyzed by one-way analysis of variance and Duncan’s multiple range test. The significance level was set at p < 0.05 for all the tests. Statistical analysis was performed using Statistical Package for Social Sciences Version 11.0 software package (SPSS, Tokyo, Japan).
Results
Figure 1 shows the hematoxylin and eosin-stained colonic tissue sections representing histological changes, if any. Colon of control rats showed well-defined mucosa and submucosa and epithelial cells in association with goblet cells arranged in the crypts, endocrine cells, and Paneth cells polarized at the base of the crypts. Colon section of silibinin alone-treated rats showed normal crypts comparable with that of control rats with prominent goblet cells that are characteristics of the colon. DMH alone-administered rats showed colonic adenocarcinoma. Supplementation with silibinin to DMH-exposed rats showed normal colonic crypts surrounded by lymphoid follicles, thus revealing the prevention of pathological changes induced by DMH.

Hematoxylin and eosin staining of colonic tissue of experimental rats. Colon tissue section of control rats showed normal crypt architecture and underlying muscularis mucosa, and the goblet cells are mostly adhered to epithelial cells. Colon tissue section of silibinin-treated rats showed normal crypts comparable with that of control rats with abundance of prominent goblet cells (arrows), a characteristic of the colon. There was no significant difference between control rats and control rats supplemented with silibinin. Colon tissue of DMH alone-treated rats showed infiltration of neoplastic cells through the muscularis mucosa and the submucosa indicating the genesis of malignant adenoma. Polystratification of cells and moderate dysplasia were also observed in the nearby tissues of DMH alone-administered rats. Silibinin-supplemented DMH-administered rats showed normal colonic crypt villus axis lined by epithelial cells. DMH: 1,2 dimethyl hydrazine.
Topographical representation of the mucin content in the colon of experimental rats as determined by the Alcian blue–PAS staining is shown in Figure 2. Colon section of control rats and silibinin alone-supplemented rats showed rich mucin content as evident by the presence of abundant goblet cells. Mucin-depleted foci represented as crypts containing goblet cells devoid of mucin were found in the adenoma of DMH-administered rats. Colonic sections of silibinin-supplemented DMH-administered rats showed mucin content comparable with that of control rats.
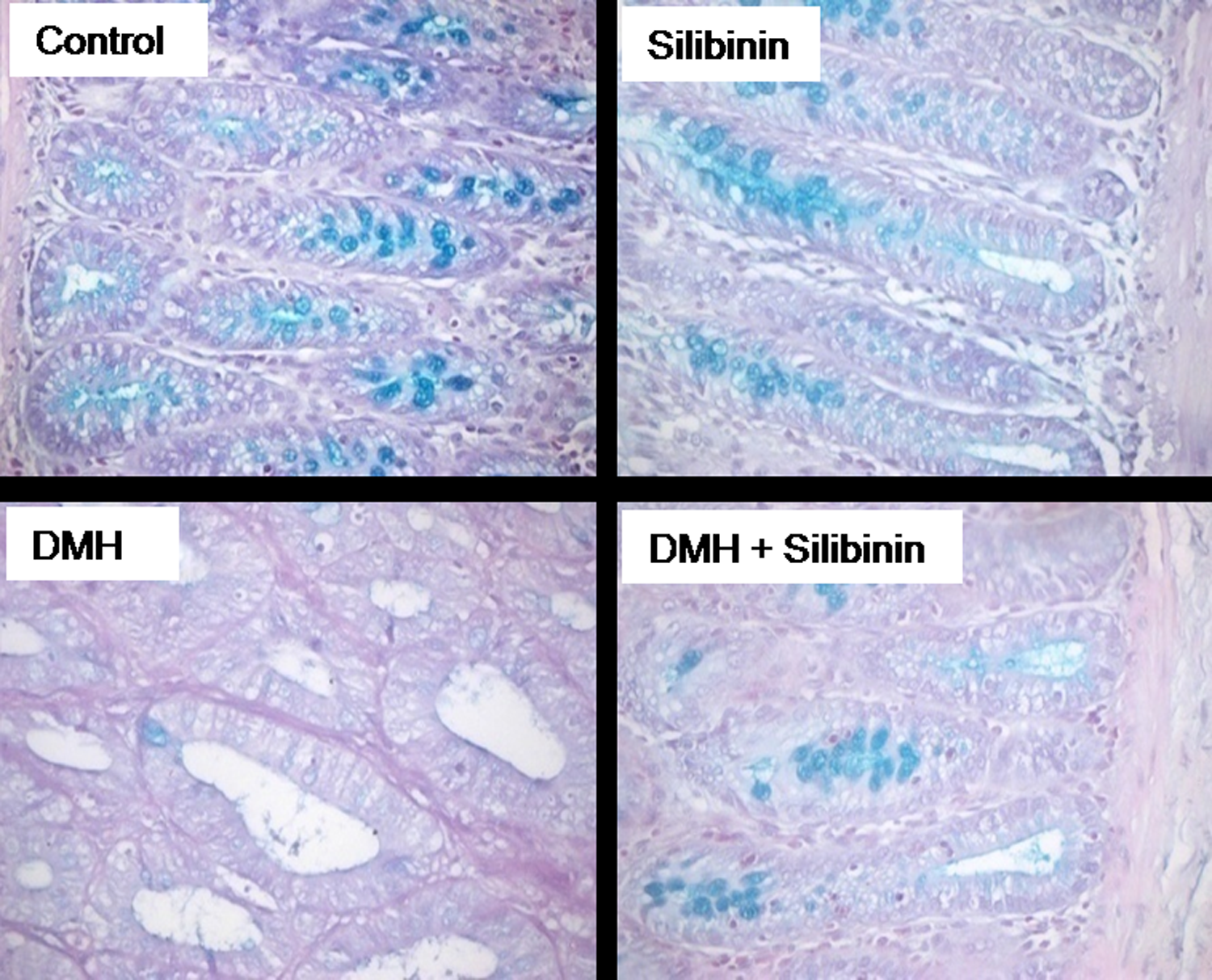
Alcian blue staining for mucin. Intense blue color seen in the colon of control rats and silibinin alone-treated rats revealed abundant mucin content. Colon tissue of DMH alone-treated rats showed mucin-depleted adenocarcinoma as evidenced by the lack of blue color. Appearance of blue coloration in the colonic tissue of silibinin-supplemented DMH-administered rats reveals reduced depletion of mucin. DMH: 1,2 dimethyl hydrazine.
Figure 3 represents the toluidine blue staining for mast cell incidence in the colon of experimental rats. The number of mast cells found in the colonic section of unsupplemented DMH-administered rats was significantly high. Moreover, the mast cells in the colonic sections of DMH-administered rats stained with toluidine blue were flat and oval. Colonic tissue sections of silibinin-supplemented DMH-administered rats and that of control rats did not show any occurrence of mast cells.
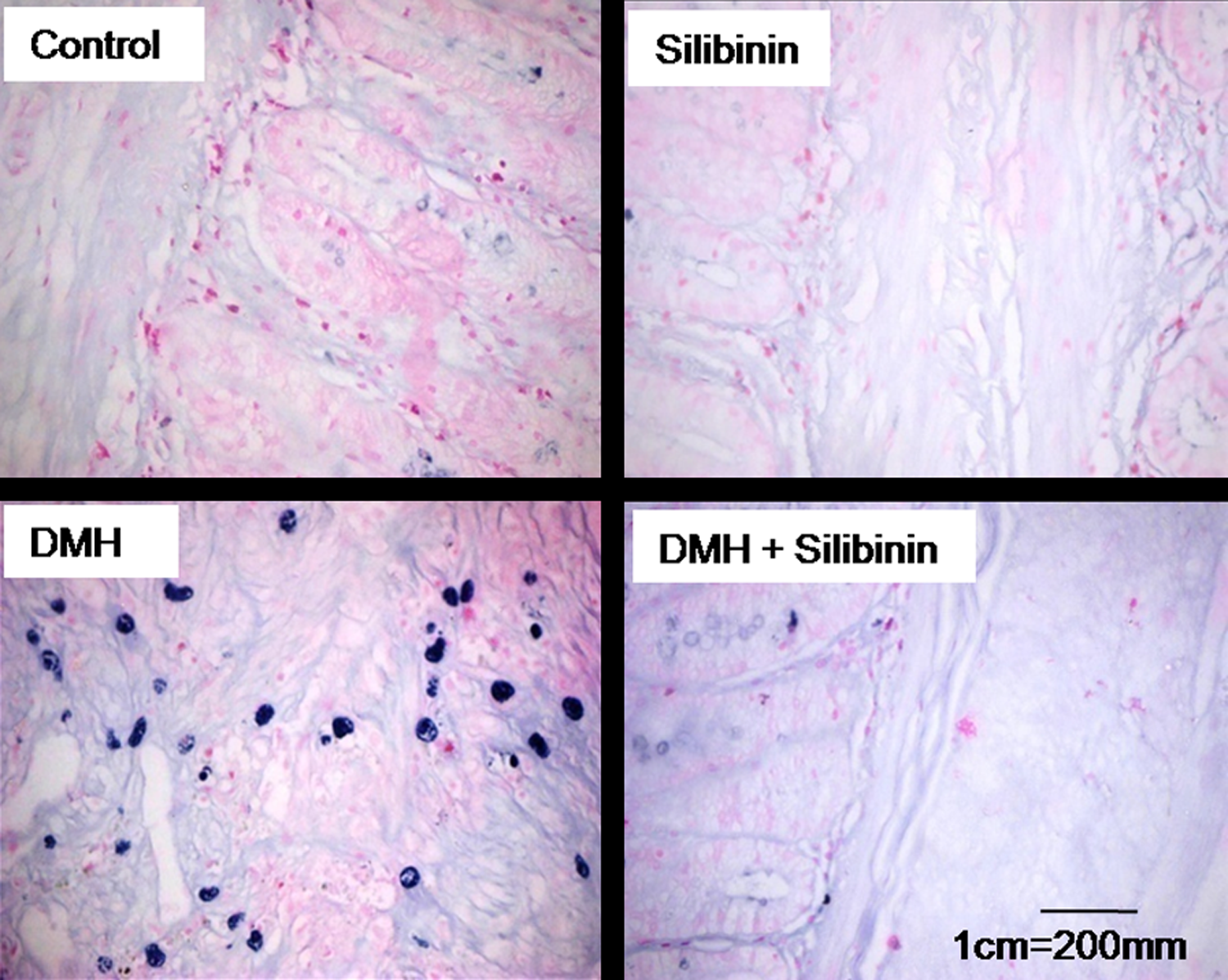
Toluidine blue staining for mucosal mast cells. Colonic tissue of control and silibinin alone-treated rats showed no evidence of the presence of mast cells. DMH alone-administered rats showed accumulation of mast cells (blue granule-like structures) in the colonic mucosa indicating the onset of pathological changes. Mast cell accumulation was markedly reduced in the colon of DMH-administered rats supplemented with silibinin. DMH: 1,2 dimethyl hydrazine.
Figure 4 shows the reverse transcriptase PCR analysis of GCC mRNA levels in the colon of experimental rats. Tumor-bearing rats showed downregulation of GCC mRNA level as compared to the control rats. The expression of the GCC mRNA in the colon of DMH + silibinin-administered rats showed that silibinin prevents carcinogen-induced suppression of GCC mRNA.

Expression of GCC. Agarose gel electrophoregram showing GCC messenger RNA level and the bar diagram showing the band intensities analyzed by densitometer. The data are expressed as percentage of GCC/GAPDH ratio and given as means ± SD for six experiments. Error bars sharing a common symbol do not differ significantly (p < 0.05). GCC: guanylyl cyclase C; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Figure 5 shows the immunohistochemical localization of CDX2 protein (Figure 5(a)) and real-time PCR analyses for CDX2 expression relative to GAPDH (Figure 5(b)) in the colon of experimental rats. CDX2 mRNA expression and the CDX2 protein level were decreased in the colon of tumor-bearing rats as compared to the control rats. Silibinin supplementation prevents the suppression of gene and protein expression of CDX2, a tumor suppressor, reflecting its anticarcinogenic potential.
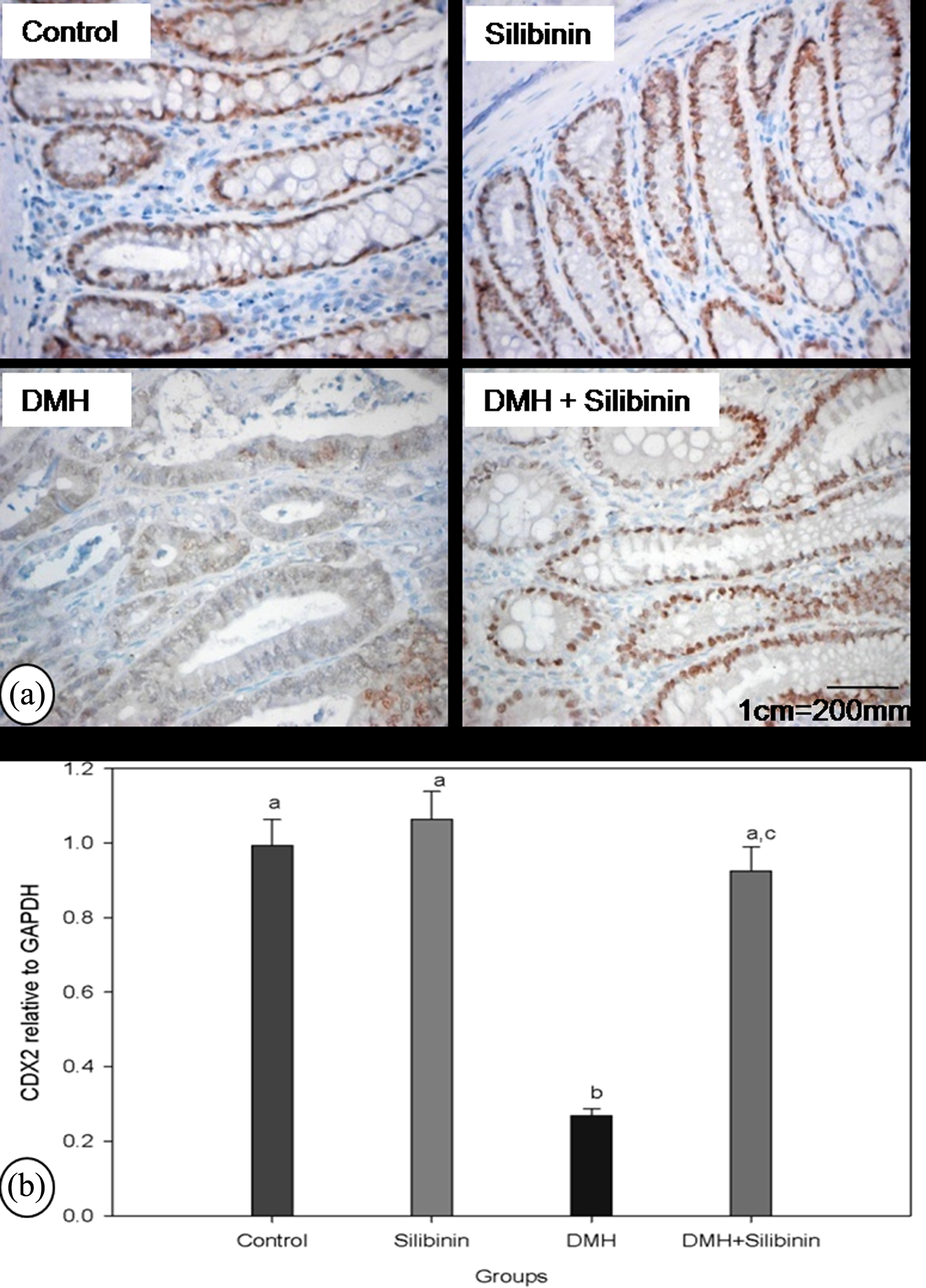
Immunolocalization of CDX2 protein and mRNA expression of CDX2. (a) Immunohistochemical localization of CDX2 protein in the colon of experimental rats. Control rats and silibinin alone-treated rats showed intense nuclear staining for CDX2 protein. Very few colonic cells of DMH alone-administered rats showed positive staining for CDX2. Silibinin-supplemented DMH-administered rats showed CDX2 protein level comparable with that of control rats. (b) Bar diagram representing fold change of CDX2 expression in experimental rats normalized to the endogenous reference gene GAPDH and relative to the control rats was calculated by the ΔΔC t method. Thus, the ratio in control rats equals 1 by definition. For CDX2, mRNA isolated from six rats in each group were analyzed and paired with control rats. The data are expressed as means ± SD for six experiments. Error bars sharing a common alphabet do not differ significantly (p < 0.05). CDX2: caudal-type homeobox transcription factor 2; mRNA: messenger RNA; DMH: 1,2 dimethyl hydrazine; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Discussion
This study investigated the effect of silibinin on the pathogenesis of colon adenocarcinoma, with emphasis on the expression of CDX2 gene and CDX2 protein level. The main finding of the study is silibinin that prevents suppression of CDX2 expression in the presence of DMH and inhibits DMH-induced tumorigenesis in the rat colon. The mechanism by which silibinin exerts this preventive effect is obscure. But still it has been shown that CDX2 is frequently inactivated by methylation of its promoter in gastric carcinoma and CRC cells, and moreover, green tea polyphenol was reported to decrease the CDX2 methylation frequency in a dose-dependent manner. 20 In this context, Kauntz et al., 21 reported that silibinin was able to significantly inhibit DNA methyl transferase activity (DNMT) in both the primary adenocarcinoma cells SW480 and their derived metastatic cells SW620. Hence, it was hypothesized that silibinin could exert its anticarcinogenic potential by inhibiting CDX2 methylation, thereby preventing the suppression of CDX2 gene expression.
GCC, a downstream transcriptional target of CDX2, maintains intestinal homeostasis by restricting proliferation and promoting differentiation. 22,23 Elimination of GCC expression induces genomic instability in intestinal epithelial cells, increasing DNA double-strand breaks, loss of heterozygosity, and point mutations in genes central to tumorigenesis, including APC and β-catenin. 24 Earlier reports from our laboratory also revealed the accumulation of β-catenin in the cytoplasm of colonic tissues challenged with DMH reflecting tumorigenesis. 25 In the present study, we observed decreased GCC mRNA expression in the colon of unsupplemented rats challenged with DMH. Since the intestine-specific expression of the GCC promoter requires CDX2, 26 downregulation of CDX2 in the colonic tissue of DMH-administered rats could also contribute to the downregulation of GCC and thereby disruption of intestinal homeostasis and the promotion of carcinogenesis.
Mucins are glycoproteins made up of chains of sugars attached to the polypeptide backbone 27 and are the main structural components of the mucus layer that acts as a barrier against various toxic insults including carcinogens. 28 Mucins secreted by adenomas and normal colorectal mucosa differ in quality and quantity. 29 In the present study, we observed depletion of mucin content in the adenoma of colonic epithelial cells challenged with DMH reflecting reduction in goblet cells and mucus secretion as a consequence of adenoma carcinoma sequence. In this context, it has been reported that targeted elimination of GCC signaling in mice (Gucy2c−/−) increases crypt length reflecting hyperplasia of the proliferating compartment, with an increase in rapidly cycling progenitor cells and reciprocal reduction in the differentiated cells, including Paneth and goblet cells. 22 Earlier reports from our laboratory showed that DMH-administered rats showed increased activity of mucinase, a microfloral enzyme, which hydrolyzes the protective mucin coat in the intestinal wall and exposes the underlying mucosa to luminal carcinogens released by β-glucuronidase activity and thus increased mucinase activity could lead to mucin degradation and induction of carcinogenesis. 30 Furthermore, MUC2, a predominant mucin secreted by goblet cell, is also shown to be a target gene of CDX2. 31 Since CDX2 interacts with the MUC2 promoter and activates MUC2 transcription, it can play an important role in the differentiation of goblet cells. Hence, modulation in the expression of CDX2 and thereby GCC could contribute to the changes in the colonic mucin content.
Mast cells are innate immune cells that may modulate colonic adenoma to carcinoma development. Since mast cells promote the progression of experimental tumors by triggering inflammation, it might be a valuable therapeutic target. 32 Further a linkage between the key pathways involved in inflammation and regulators of homeostasis is often seen, 33 supporting the hypothesis that there is a connection between inflammation and CDX2 expression. In the current study, increased numbers of mast cells were found in the colonic tissue of unsupplemented DMH-administered rats, implicating the initiation of inflammatory cascade which was attenuated in the presence of silibinin.
Histological assessment carried out by hematoxylin and eosin staining revealed that supplementation with silibinin protects against DMH-induced tumorigenesis as evidenced by the normal mucosal and submucosal layers with localization of endocrine cells and Paneth cells toward the base of the crypts. Colonic tissue of unsupplemented DMH-administered rats showed adenocarcinoma. Restoration of normal colonic histomorphology and protection against mucin depletion in the colon of silibinin-supplemented DMH-administered rats reveal the protective effect of silibinin.
Findings of our study suggest that the expressions of CDX2 and GCC are remarkably decreased in the colon of DMH alone-administered rats, promoting inflammation, mucin degradation, and thereby interruption of mucosal homeostasis and promotion of the adenoma to carcinoma sequence. These pathological changes caused due to CDX2 suppression were attenuated on supplementation with silibinin. Silibinin might preserve colonic epithelial cells by modulating DNA methyl transferase (DNMT) activity that should be studied in future.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The author NS has been financially supported by the Council of Scientific and Industrial Research in the form of Research Associate [9/003(0011)2k12-EMR-I].
