Abstract
The present study was undertaken to investigate the effects of maternal lead (Pb) exposure on the expression of interleukin (IL) 1β, IL 6, and glial fibrillary acidic protein (GFAP) in hippocampus of mice offspring. Pb exposure initiated from the beginning of gestation to weaning. Lead acetate (PbAc) administered in drinking solutions was dissolved in distilled deionized water at the concentrations of 0.1, 0.5 and 1% groups ,respectively. On the postnatal day 21, the Pb levels in their blood and hippocampus were determined by graphite furnace atomic absorption spectrometry. The expression of IL 1β, IL 6, and GFAP in hippocampus was measured by immunohistochemistry and Western blotting. The Pb levels in blood and hippocampus of all Pb-exposed groups were significantly higher than that of the control group (p < 0.05). The expression of IL-1β, IL-6, and GFAP was increased in Pb-exposed groups in comparison with the control group (p < 0.05). The high expression of IL-1β, IL-6, and GFAP in the hippocampus of pups may contribute to the neurotoxicity associated with maternal Pb exposure.
Introduction
Lead (Pb) can enter the blood through skin, respiratory, and digestive tracts and cause poisoning by circulating in the blood. Its main toxic effect appears in the central nervous system (CNS). Research on Pb neurotoxicity has been of interest for many years, 1 –5 but its pro-inflammatory effects on the brain is rarely reported. This kind of effect has been observed in several other tissues and organs. The proliferation of T lymphocytes under Pb action has been observed in vitro. 6 It was reported that the young rats treated with Pb could cause the overexpression of interleukin (IL) 10 and IL-12 and increased numbers of monocytes and T lymphocytes in spleen and thymus. 5 This raises the possibility that Pb may affect immune processes in brain. Glial cells, the predominant CNS antigen presenting cells, are involved in local inflammatory processes by responding to, as well as producing, cytokines such as IL-1β, IL-6, and tumor necrosis factor α (TNF-α); both IL-1β and TNF-α are potent inducers of proteinases in the CNS. 5 Considering the critical importance of astrocytes to many developmental events, 7 a toxic insult to astrocytes in development could impair function of the CNS. Glial fibrillary acidic protein (GFAP) is the major protein of glial intermediate filaments in differentiated astrocytes. 8 One of the most commonly recognized features of astrocytes in the mature brain is the reaction to CNS damage with reactive gliosis. The latter is characterized by the presence of large numbers of reactive astrocytes, distinguished from normal astrocytes by their greater size, longer and thicker processes, and increased amount of GFAP. 9 Increased expression of GFAP is generally accepted to represent a highly sensitive marker of neurotoxin-induced CNS injury, even in the absence of an apparent pathological correlate. 10 This has been demonstrated by administration of low-level neurotoxins postnatally, 11 and our previous results also demonstrated that maternal Pb exposure could lead to behavioral deficits of offspring. 12 However, it is still not clear whether GFAP reactivity can be reliably used as a marker for prenatal neurotoxic insults. 13
The developing brain is experiencing intensive cellular proliferation, differentiation, and synaptogenesis. It is very sensitive to environmental hazard at this stage. It is known that early life Pb exposure could cause severe consequences for brain development, and hippocampus is a key point in nervous system. However, the underlying influence mechanism of Pb exposure on hippocampus is still unclear. In order to discover the potential mechanism, this study tested the expression of IL-1β, IL-6, and GFAP in hippocampus of Pb-exposed mouse pups to know whether these proteins are involved in the process of hippocampus damage.
Methods
Animals and treatment
All procedures involving animals were carried out in strict accordance with the international standards of animal care guidelines and were approved by the local Care of Experimental Animals Committee. For the purpose of this study, 40 pregnant Kunming female mice were ordered from Henan laboratory animal center. They were randomly divided into 4 groups with 10 mice in each group and caged individually. Lead acetate (PbAc; Pb(CH3OO)2·3H2O, Luoyang Chemical Reagent Factory, Luoyang, Henan, China) was dissolved into distilled water at three different concentration levels, 0.1, 0.5, and 1% for low, moderate, and high concentration levels, respectively. 14,15 Pb exposure started from the beginning of gestation and lasted until weaning (i.e. postnatal day 21 (PND 21)) by drinking Pb-containing water. The animals were maintained on a 12-h light/12-h dark cycle with food and water available ad libitum. All experiments were carried out in accordance with the regulations of the Zhengzhou University Committee on Ethics in the Care and Use of Laboratory Animals. At birth, all litters were culled to eight pups. In the process of experiment, littermates were not used within a group, but they were used across groups.
Determination of Pb concentrations in blood and cerebral cortex samples
Sample collection
The blood samples were collected from the tail of the pups on PND 21 after wiping the skin to remove the contaminated Pb. Then, the pups were anesthetized under ether inhalation, and the brains were dissected. The hippocampus was isolated.
Sample preparation for assay
A total of 100 μl blood sample was mixed well with 3.9 ml of 0.5 N nitric acid containing 0.01% Triton X-100 by vortexing for 10 s followed by centrifugation for 10 min at 7500 r/min at room temperature. The supernatant was collected for further analyzing the Pb content. The isolated hippocampus tissue from each sample was homogenized in a mixture of 0.5 N nitric acid, 0.5 N perchloric acid, and 0.01% Triton X-100 to make 1:10 (w/v) of diluted homogenate.
The measurement of Pb concentration in samples was performed as described. 16 –18 A 20-ml aliquot of each sample was injected into the graphite furnace atomic absorption spectrometry (Hitachi, Tokyo, Japan) by an autosampler, and 20 ml of 0.2% magnesium nitrate was added as modifier. Pb concentration was measured at 283.3 nm by a programmed heating procedure for blood and brain. Pure argon was used as the purge and protecting gas. The analysis was carried out in duplicate, and the mean peak height value was taken for calculation. For the standard curve, 1000 ppm lead chloride in 0.3 M nitric acid was diluted to 5, 10, and 20 ppm with 0.2% nitric acid containing 0.01% Triton X-100. To examine the recovery rate, standard Pb solution was added (final concentration: 10 ppm) to blood and brain samples as internal standard. The recovery rates for blood and brain homogenate were 97.5 and 98.6%, respectively.
Sample preparation and immunohistochemistry
Three pups from each group were selected for histological study. 19,20 The selected pups were anesthetized by overdose with Nembutal sodium (35 mg/kg, intraperitoneally). The whole brain was dissected. The fixed brain samples were cut at 5 μm thickness starting at 3 mm posterior to the anterior pole. The tissue slides were treated by microwave in 10 mM citrate buffer (pH = 6) for 3 min and followed by blockade of nonspecific binding by incubation in 0.1 M phosphate-buffered saline (PBS) containing 3% normal goat serum. Sections were then subsequently incubated with primary antibodies overnight at room temperature. The primary antibodies were rabbit anti-IL-1β (Santa Cruz Biotechnology, Santa Cruz, California, USA). After extensive rinsing steps in 0.1 M PBS, the sections were incubated in biotinylated goat anti-rabbit antibody (Santa Cruz Biotechnology) for 1 h at room temperature and followed by using the Vector ABC system. Subsequent incubation in diaminobenzidine was performed for visualization of the reaction product. For negative controls, the primary antibody was omitted.
Protein isolation and Western blotting
The dissected hippocampus samples were homogenized in lysis buffer containing 2% sodium dodecyl sulfate (SDS), 10% glycerol, 2% 2-mercaptoethanol, 0.002% bromphenol blue in 75 mM Tris–hydrochloric acid. 21 The samples were heated at 95°C for 10 min before separating on 10% Tris/glycine/SDS acrylamide gels. The proteins were subsequently transblotted to polyvinylidene difluoride membranes and blocked in 5% dry milk for 2 h at room temperature. The membrane was incubated with rabbit anti-IL-1β (Santa Cruz Biotechnology), anti-IL-6 (Santa Cruz Biotechnology), and anti-GFAP antibody (Santa Cruz Biotechnology) for 2 h at 37°. After three washes with Tris-buffered saline/0.05%Tween-20, the membrane was incubated with a horseradish peroxidase-conjugated goat anti-rabbit antibody (Santa Cruz Biotechnology) for 1 h at 37°C. 22 Protein signal was visualized using the SuperSignal West Pico Chemiluminescent Substrate (Pierce & Company, Stuttgart, AR) and detected with Imaging System (Syngene International Ltd., Los Altos, USA). β Actin protein was visualized and detected similar to the above-mentioned procedure.
Statistical analysis
All data were expressed as mean ± SEM. One-way analysis of variance and a post hoc Bonferroni’s test using Statistical Package for Social Sciences Version 12.0 (SPSS Inc., Chicago, Illinois, USA) software to analyze the differences in IL-1β, IL-6, and GFAP and determine the Pb content in blood and hippocampus tissue between groups. The values of p < 0.05 were considered to be significant.
Results
Pb content in blood and hippocampus of pups
Figures 1 and 2 illustrate the profile of Pb levels in blood and hippocampus of pups on PND 21 in different groups with maternal Pb exposure during pregnancy and lactation period. Only one animal per litter in each age group was used. A significant increase of Pb level in blood and hippocampus was found in Pb-exposed groups as compared to the control group (p < 0.05), and the Pb levels were positively related to the exposure dosage. The body weights of the pups were not significantly different between groups (data not shown).
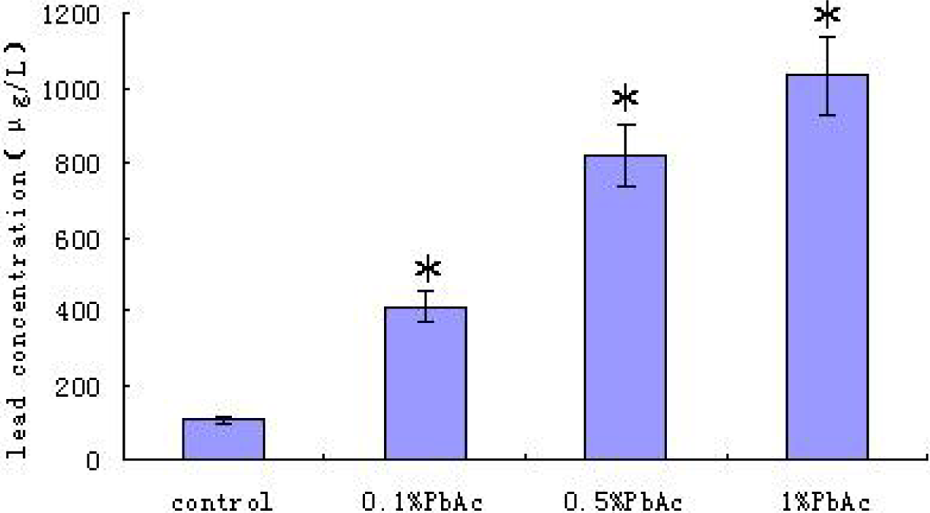
Blood Pb levels in control and Pb-exposed animals at PND 21. (n = 10 per group, *p < 0.05). Each value represents the mean ± SEM of 10 different litters in same group. Only one animal per litter in each group was used. Blood Pb levels from the control animals were significantly lower than the Pb-exposed animals. Pb: lead; PND: postnatal day.

Hippocampal Pb levels in control and Pb-exposed animals at PND 21. (n = 10 per group, *p < 0.05). Each value represents the mean ± SEM of 10 different litters in each group. Only one animal per litter in each age group was used. Hippocampal Pb level in control group was significantly lower than the Pb-exposed groups. Pb: lead; PND: postnatal day.
Effect of Pb exposure on the expression of IL-1β in hippocampus
Figure 3 illustrates the results of immunoreaction for IL-1β. Figure 3(a) shows the expression of IL-1β in hippocampus at control group. Figure 3(b) shows the influence of Pb exposure on IL-1β expression in 1% PbAc group. For an overview of the results, IL-1β was expressed in a large body of cells. Figure 4 illustrates the effects of Pb exposure on the expression of IL-1β quantitatively. Figure 4(a) is a representative result of Western blotting. Figure 4(b) shows the expression of IL-1β in hippocampus of pups in different groups. The quantitative results showed that IL-1β protein level was very low in the control; however, its expression levels were higher in 0.1, 0.5, and 1% PbAc groups in comparison with the control group, and the differences are statistically significant (p < 0.05, p < 0.01).
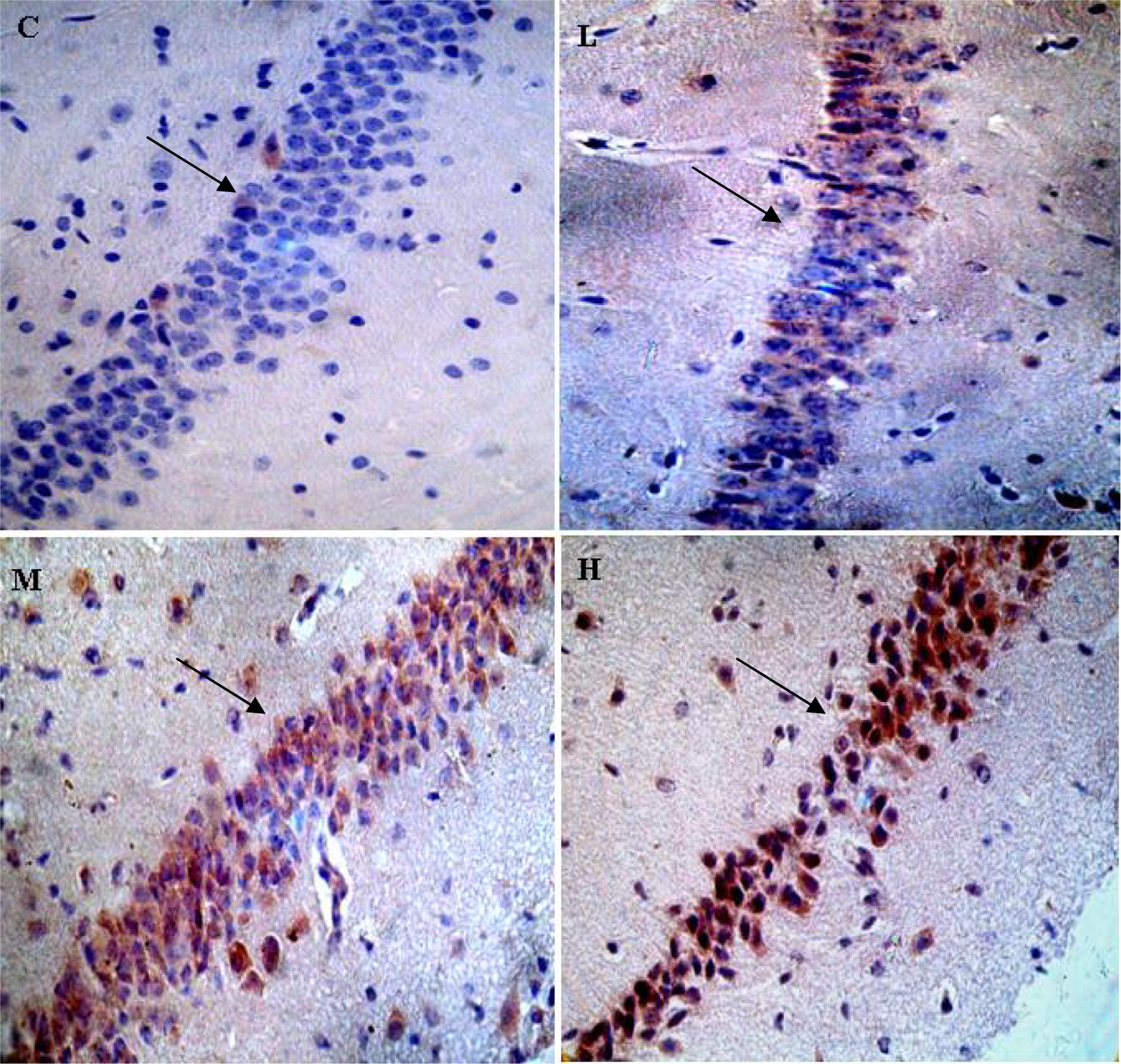
IL-1β immunoreactivity in CA1 region of hippocampus. (n = 3 per group). C: control group; L: 0.1% PbAc group; M: 0.5% PbAc group; H: 1% PbAc group (×400 magnification). IL-1β: interleukin 1β; PbAc: lead acetate.

Western blot analyses of IL-1β protein in hippocampus tissue. (a) IL-1β protein was analyzed by Western blot in hippocampus of pups in different groups. β actin is shown as a loading control. (b) IL-1β protein expression levels were analyzed using Western blot in each group. The values represent the ratio of IL-1β/β actin intensity (n = 10 per group; *p < 0.05; **p < 0.01). C: control group; L: 0.1% PbAc group; M: 0.5% PbAc group; H: 1% PbAc group; IL-1β: interleukin 1β; PbAc: lead acetate.
Effect of Pb exposure on the expression of IL-6 in the hippocampus
Figure 5 illustrates the effects of Pb exposure on the expression of IL-6 quantitatively. Figure 5(a) is a representative result of Western blotting. Figure 5(b) shows the expression of IL-6 in each group. The expression of IL-6 in the hippocampus of pups was significantly increased in 0.1, 0.5, and 1% PbAc groups when compared with the control group (p < 0.05, p < 0.01).

Western blotting analyses of IL-6 protein in hippocampus tissue. (a) IL-6 protein was analyzed using Western blotting technique in hippocampus of pups in different groups. (b) IL-6 protein expression levels were analyzed using Western blot in each group. The values represent the ratio of IL-6 /β actin intensity (n = 10; *p < 0.05; **p < 0.01). C: control group; L: 0.1% PbAc group; M: 0.5% PbAc group; H: 1% PbAc group; IL-6: interleukin 6; PbAc: lead acetate.
Effect of Pb exposure on the expression of GFAP in the hippocampus
Figure 6 illustrates the effects of Pb exposure on the expression of GFAP quantitatively. Figure 6(a) is a representative result of Western blotting. Figure 6(b) shows the expression of GFAP in each group. The expression of GFAP in the hippocampus of pups was significantly increased in 0.1, 0.5, and 1% PbAc groups when compared with the control group (p < 0.01).

Western blotting analyses of GFAP protein in hippocampus tissue. (a) GFAP protein was analyzed using Western blotting in hippocampus of pups in different groups. (b) GFAP protein expression levels analyzed using Western blot in each group. The values represent the ratio of GFAP/β actin intensity (n = 10; **p < 0.01). C: control group; L: 0.1% PbAc group; M: 0.5% PbAc group; H: 1% PbAc group; GFAP: glial fibrillary acidic protein; PbAc: lead acetate.
Discussion
Pb neurotoxicity has been defined as a significant pediatric health problem. The fetal stage is a very susceptible period to Pb exposure even at the level under 10 μg/dl, which is the lower bound threshold for Pb neurotoxicity in children. 23 The nervous system is the primary target for the Pb exposure and the developing brain appears to be especially vulnerable. 24 However, the mechanisms of Pb exposure on brain deficits remain unclear. 24
Neuronal damage has long been recognized to play a key role in Pb-induced impairment of brain function. 25 However, recent attention has focused on neuroglia as a target of subclinical Pb exposure. 26 Microglia represent a source of many cytokines and thus may exert a strong regulatory influence on other brain cells, including astrocytes. Glial cells play an important role in local inflammatory processes by producing cytokines such as IL-1β, IL-6, and TNF-α. IL-1β is a specific cytokine involved in intercellular communication between glial cells in injured brain and plays a key role in the regulation of inflammatory processes. Interestingly, in the present study, the immunoactivities of IL-1β and IL-6 were significantly increased in hippocampus of mouse pups with maternal Pb exposure. Pathologically, inflammatory cell infiltration at different acute experimental autoimmune encephalomyelitis (EAE) stages was observed during EAE episodes and correlated strongly with the reduction in synaptic protein immunoactivity. 27
GFAP is an intermediate glial filament which expressed late in hippocampus development during astrocytic maturation. 28 The developmental profile of GFAP expression in control animals is similar to that reported in other studies. 29 GFAP is not exclusively localized to astrocytes but has been shown to be a minor component present in Bergmann glia and oligodendrocytes; 30 thus, the changes in GFAP expression could also be a reflection of different glial population with differing temporal sensitivities to Pb exposure. Furthermore, the changes in GFAP expression may be a by-product of disturbances in oligodendroglial integrity and the process of myelination. The pathological consequences of the decreased level of GFAP gene expression are not yet clear. In the adult animal, GFAP expression is maintained at a low level; however, insults to the brain dramatically elevate its concentration. 28 While the role of GFAP during brain damage is not clear, these data indicate that Pb may impair the capacity of hippocampus in responding to injury in the mature rat brain.
The functional role of pro-inflammatory factors induced by Pb is not completely defined and studies are still being carried out. On one hand, pro-inflammatory events may have a pathological effect by interfering with the function of residual neuronal circuits, thus, preventing axonal remyelination and inhibiting axonal regeneration. On the other hand, following CNS injury, pro-inflammatory factors may also participate in wound healing. 31,32 Reactive astrocytes serve to regulate molecular and ionic contents of the extracellular space at injury site and induce secretion of neurotrophic factors.
The present study demonstrates that the immunoactivities of IL-1β, IL-6, and GFAP were significantly increased in hippocampus of Pb-exposed groups. Thus, enhanced levels of this cytokine observed after Pb treatment signal the induction of mechanisms leading to the inflammatory cascade and indicate potential proinflammatory action, and our previous results also demonstrated that maternal Pb exposure could lead to behavioral deficits of offspring. 12 As hypothesized, increase of immunoactivity for IL-1β, IL-6, and GFAP may contribute to the progression of brain damage in mouse pups with maternal Pb exposure. It is concluded that the increased IL-1β, IL-6, and GFAP expression in hippocampus with hippocampal neural damage as a consequence of Pb exposure during pregnancy and lactating period are excessively expressed in hippocampus of the offspring. The results imply a potential mechanism for early life Pb exposure on neuropathological processes under conditions of maternal Pb exposure. Future studies should place particular emphasis on the conditions that promote cytokine-mediated neuronal damage. Defining specific changes in the CNS environment responsible for enhanced detrimental effects of pro-inflammatory cytokines may also reveal new targets in the treatment of Pb neurotoxicity.
Footnotes
Funding
This research was supported by National Natural Science Foundation of China (NNSFC) by grant NNSFC-31201878.
