Abstract
This in vitro study was designed to investigate the molecular mechanisms of paraquat-induced damage using cultured human fetal lung fibroblasts (MRC-5 cells), in order to promote the development of improved therapies for paraquat poisoning. Paraquat’s effects on proliferation were examined by flow cytometry, on viscoelasticity by the micropipette aspiration technique, and on connective tissue growth factor (CTGF) expression by real-time polymerase chain reaction and enzyme-linked immunosorbent assay. Paraquat was found to significantly reduce the proliferation index of MRC-5 cells in a concentration-dependent manner (p < 0.05) and to significantly impair the viscoelastic properties in a time-independent manner (p < 0.05). Exposure to paraquat led to a significant and time-dependent increase in CTGF expression (p < 0.05) and induced changes in the morphology and biomechanical characteristics of the MRC-5 cells. These findings not only provide novel insights into the mechanisms of paraquat-induced lung fibrosis but may represent useful targets of improved molecular-based therapies for paraquat poisoning.
Introduction
Paraquat (1,1-dimethyl-4-4-bipyridinium dichloride) is an effective herbicide that functions by blocking photosynthesis in plants. 1 Although paraquat is environmentally favorable, it is extremely toxic to humans and ingestion or skin absorption is associated with high rates of acute and chronic morbidity and mortality. 2 The typical symptoms of paraquat poisoning observed upon clinical presentation are skin or ocular burns, sore throat, seizures, gastrointestinal bleeding, and damage to kidney, heart, liver, and lung. 3 It is the lungs, however, that sustain the most severe injury and are associated with the remarkable physical suffering that accompanies paraquat-related death. 4
Studies of chronic exposure to paraquat have identified a primary mechanism of human toxicity involving the disruption of alveolar epithelial cells and bronchiolar Clara cells. These events are followed by hemorrhage, pneumonedema, inflammatory cell infiltration into the alveolar space, and eventually pulmonary fibrosis. Pulmonary fibrosis is a severe condition, characterized by tissue scarring inside and between the air sacs in the lung. This irreversible scarring makes the lung tissue thick and inelastic, preventing oxygen from passing through the air sac walls into the circulatory system. 1 However, in cases of paraquat ingestion by the oral route (a frequent method of suicide in economically underdeveloped countries), the major cause of death is damage to the esophagus and acute inflammation of the mediastinum. 5
Recent research efforts have focused on determining the molecular mechanisms of paraquat poisoning, with the aim of identifying useful molecular targets to develop more efficacious therapeutic strategies. Studies have revealed that in the mammalian system paraquat induces the generation of a large amount of reactive oxygen species (ROS), resulting in cell membrane lipid peroxidation and tissue injury. 6 –9 Accordingly, new clinical strategies are being developed to accelerate the elimination of circulating paraquat, since decreasing its systemic disbursement will help to minimize the amount of tissues injured. In addition, efforts to control systemic iron distribution, by manipulating the iron chelator desferrioxamine, are being developed since iron contributes to generation of ROS. Finally, therapeutic approaches using established ROS-targeting drugs or natural supplements are being considered; for example, the activities of vitamin E and N-acetylcysteine help maintain effective antioxidant levels and repair ROS-induced lesions, 10 while those of dexamethasone, methylprednisolone, and cyclophosphamide reduce inflammation. 4,5,11
Connective tissue growth factor (CTGF) is a member of the protein family comprised of the CTGF, cysteine-rich protein, and nephroblastoma-overexpressed proteins (thereby known as the CCN family). CTGF is an extracellular matrix-associated heparin-binding protein that is widely expressed in many tissues at low physiologic levels, but which has been observed as dramatically upregulated in the pathologic states of fibrosis and cancer. 12 In vitro studies have demonstrated that CTGF regulates various biological processes related to fibrogenesis, such as cell proliferation, migration, adhesion, angiogenesis, and extracellular matrix production. 13 More importantly, CTGF has been shown to exert its effects on a broad spectrum of cell types involved in the fibrogenic process, including mesenchymal stem cells, hepatic stellate cells, renal podocytes, parietal and tubular epithelial cells, pulmonary type II alveolar cells, vascular smooth muscle cells, endothelial cells, cardiomyocytes, pericytes, and fibroblasts. 13 Accumulating data have indicated that CTGF is directly related to chronic fibrotic effects mediated by transforming growth factor β (TGF-β), vascular endothelial factor, insulin-like growth factor, and angiotensin II. 14,15 For example, CTGF was shown to act through integrins and proteoglycans, as an independent proadhesive molecule and modify adhesive responses to extracellular matrix and growth factors. 16 CTGF has also been shown to function as a downstream and cooperative mediator of the TGF-β signaling pathway in many animal models of human disease, including those of diabetic nephropathies, idiopathic and nonidiopathic pulmonary fibrosis, liver fibrosis, skin fibrosis, congestive heart failure, pancreatitis, and various malignancies. 12,17,18
Genetic approaches to knock-down CTGF in animals revealed that loss of this protein leads to dramatic suppression of pathogenic fibrotic responses, such as those in the liver and related to type 1 and type 2 diabetic nephropathies. 9 The mechanisms of these effects were found to involve CTGF-induced transcriptional activation of collagen gene type I α2 9 in the fibrosis condition and the expression of both TGF-β and its TGF-β downstream targets in the diabetic condition. 19 More recently, a study of CTGF levels in tissue, blood, and vitreal fluid identified this protein as a biomarker of fibrosis and suggested its potential for use in diagnosing or prognosing fibrotic diseases. 12
Despite the myriad evidence that has led to the characterization of CTGF as a universal mediator of fibrogenesis, the precise mechanisms that underlie its effects in this pathogenic process remain to be fully elucidated. Therefore, we designed the current in vitro study to investigate the molecular mechanisms of paraquat-induced pulmonary fibrosis, particularly by studying its effects on proliferation and viscoelasticity of lung fibroblasts. We expect that these new data will represent potential therapeutic targets by which more efficacious treatments may be developed for patients with paraquat poisoning.
Methods
Cell line and reagents
The human fetal lung fibroblast cell line (MRC-5, Cell Bank of Type Culture Collection, Chinese Academy of Sciences, China) was maintained in minimal essential media (MEM; Invitrogen, Life Technologies, Carlsbad, California, USA) supplemented with 10% fetal calf serum at 37°C under humidified atmosphere containing 5% carbon dioxide (CO2) and 95% air. For experiments, the MRC-5 cells were seeded in 10 cm dishes and grown to 50–70% confluency for 12 h of treatment with various concentrations of paraquat (0 (control), 10, 20, 50, 100, 200, or 500 mg/L in MEM). After an additional 24, 48, or 72 h incubation with fresh media (no paraquat), the cells were harvested by trypsinization and processed for cell cycle and viscoelasticity analyses (as described below), and the supernatants were collected for measuring secreted CTGF. MRC-5 cell morphology before and after treatment was evaluated under an inverted microscope (Axiovert S100; Zeiss, Jena, Germany) equipped with a charge-coupled device (CCD) camera.
Cell cycle analysis
The control- and paraquat-treated MRC-5 cells were washed and resuspended in phosphate-buffered saline (PBS) at a concentration of 1 × 106 cells/mL. For each respective group of cells, 1 mL of aliquot was mixed with cold (−20°C) absolute ethanol (3 mL), which was added dropwise during active vortexing. Each cell preparation was then fixed by incubating overnight at 4°C. After washing twice with PBS, propidium iodide (10 μL of 100 mg/L solution) and ribonuclease A (100 mg/L) were added, and the cells were incubated for 30 min at 4°C. The DNA content was then analyzed on a flow cytometer (Profile II; BD, Franklin Lakes, New Jersey, USA) at an excitation wavelength at 488 nm. The proliferation index (PI) of each sample was calculated using the following formula: (S phase + G2 /M phase)/(G0 /G1 phase + S phase + G2 phase) × 100%.
Enzyme-linked immunosorbent assay
The control and treated MRC-5 culture supernatants were analyzed for CTGF protein content using an enzyme-linked immunosorbent assay (ELISA) kit (RapidBio Lab, West Hills, California, USA) according to the manufacturer’s protocol. Briefly, the ELISA plates were precoated with a monoclonal antibody against human CTGF. The antibody-bound CTGF in the samples was then bound to the horseradish peroxidase-conjugated anti-human CTGF polyclonal antibody. After adding the substrate reagent and tetramethylbenzidine, the CTGF signals were detected using a Synergy H4 plate reader (BioTek, Winooski, Vermont, USA). Human recombinant CTGF was used as the standard. The CTGF expression for each sample was calculated based on the standard curves generated by plotting the absorbance against the known concentrations of the CTGF standard. Samples were examined in triplicate and averaged for statistical comparison.
Micropipette aspiration and viscoelasticity measurement
The viscoelastic properties of control and treated MRC-5 cells were examined using a micropipette aspiration system with refined micropipettes having a constant cell diameter to inner micropipette diameter ratio and coated with the Sigmacote siliconizing reagent (Sigma, St Louis, Missouri, USA) to prevent cell attachment. For analysis, the micropipette was filled with distilled water and placed into the micropipette holder, which was controlled by an automated micromanipulator (5173; Eppendorf AG, Hamburg, Germany). Negative pressure was applied on the surface of a MRC-5 cell, which was suspended in growth media and deposited on a glass coverslip. The suction pressure was generated and precisely controlled by the accompanying automated transjector (5246; Eppendorf AG). The initial diameter of cells and equilibrium pressure (the point at which the cells were neither aspirated nor pushed away) were obtained before starting the aspiration, in order to minimize the error. A constant step pressure (ΔP) was applied and the aspiration process was continuously recorded for 300 s using an inverted microscope (TE300; Nikon, Tokyo, Japan) equipped with a CCD camera (Zeiss). The aspirated length of the cell into the micropipette was computed using SPOT software (Diagnostic Instruments Inc., Sterling Heights, Michigan, USA) at regular intervals, with an accuracy of ±0.3 μm. The system set-up was mounted on a vibration isolation table to minimize vibration.
A linear, three-parameter viscoelastic solid model was proposed to calculate the mechanical parameters of MRC-5 cells.
20
We assumed that the pressure applied on the cell surface was uniform and axisymmetric, with a boundary condition of no axial displacement of the cell at the micropipette opening. The displacement of the cell into the micropipette can be described as a function of time (L(t)) using the following formula:
Real-time quantitative PCR
Total RNA was extracted from the control and treated MRC-5 cells using the Trizol reagent (Gibco; Life Technologies, Carlsbad, California, USA) and applied as template (1 μg) to a cDNA synthesis kit (Promega, Madison, Wisconsin, USA). The cDNA was then used to amplify CTGF and connective tissue growth factor (glyceraldehyde 3-phosphate dehydrogenase; GAPDH) (for normalization) using the following gene-specific primer sequences: CTGF: forward 5′-TTGGCAGGCTGATTTCTAGG-3′ and reverse 5′-GGTGCAAACATGTAACTTTTGG-3′; GAPDH: forward 5′-GGTGGTCTCCTCTGACTTCAACA-3′ and reverse 5′-dNTPs, 0.5 μL of TaqDNA polymerase, 0.5 μL of each primer, 5 μL of cDNA, and water (to a final volume of 25 μL). The negative control was processed without template cDNA. The polymerase chain reaction (PCR) cycling parameters were 95°C for 5 min, followed by 55 cycles of 94°C for 15 s, 63°C for 30 s, and 72°C for 45 s. The 2-ΔΔC t comparative threshold cycle method was used to calculate CTGF messenger RNA (mRNA) expression relative to GAPDH.
Statistical analysis
Statistical analyses were performed using Statistical Package for the Social Sciences (SPSS) 10.0 software (SPSS Inc., Chicago, Illinois, USA). All data are presented as mean ± SD. Student’s t tests were used to analyze the differences between two groups or two time points. The value of p < 0.05 was considered statistically significant.
Results
Paraquat significantly decreases MRC-5 cell proliferation
The effects on cell growth and morphology of MRC-5 cells in response to the various paraquat treatments used in this study were evaluated to verify the in vitro system. The 10 and 20 mg/L paraquat treatments did not cause any significant morphological changes and the number of viable MRC-5 cells was similar between these treated cells and the control cells at all posttreatment time points (data not shown). However, the 100 and 200 mg/L paraquat treatments produced significantly altered MRC-5 cell morphology and reduced cell growth at the posttreatment time point of 24 h.
The control MRC-5 cells showed the normal morphology of long and spindle shape with a clear edge. Treatment with 50 mg/L paraquat led to a reduced number of live cells (by 15.00 ± 2.27%) at the posttreatment 24 h time point but without producing any remarkable effects on the cell morphology. Treatment with 100 mg/L paraquat led to abnormally round-shaped cells and substantial cell death (by 48.00 ± 4.13%) at the posttreatment 24 h time point. Treatment with 200 mg/L paraquat treatment resulted in similar morphological changes but even more cell death (74.00 ± 3.84%). Not surprisingly, the 300 mg/L paraquat treatment was absolutely cytotoxic (100%).
We next questioned how paraquat affects cell proliferation by flow cytometric analysis. Increasing concentrations of paraquat were found to decrease the number of MRC-5 cells in S-phase and increase the number of cells in G 0/G 1 phase, compared with control cells. In addition, the calculated PIs indicated that, unlike the control MRC-5 cells which actively proliferated, the cells treated with paraquat had significantly reduced cell proliferation (p < 0.05; Table 1). In particular, at posttreatment 72 h, the cells treated with 50 or 100 mg/L paraquat showed significantly increased proliferation (p < 0.05) but those treated with 200 mg/L showed significantly reduced cell proliferation (p < 0.05). We also observed that the paraquat treatment-induced MRC-5 cells to undergo apoptosis in a concentration-dependent manner (data not shown).
The proliferation index of MRC-5 cells in the presence or absence of paraquat.

a p < 0.05 compared with control cells.
b p < 0.05 compared with the previous time point, respectively.
c p < 0.05 compared with the previous time point, respectively.
Paraquat significantly reduces MRC-5 cell viscoelasticity
The control MRC-5 cells showed an instantaneous viscoelastic response to the applied suction pressure, as evidenced by induced shape deformity followed by a subsequent decrease in deformation rate until eventually reaching a steady state (Figure 1). The calculated values of apparent viscosity μ, the instantaneous Young’s modulus E 0, and the equilibrium Young's modulus E ∞ combined with the response curve of treated and control MRC-5 cells are presented in Table 2. Statistical analyses revealed that paraquat treatment significantly reduced all viscoelastic parameters of MRC-5 cells (vs. control cells, p < 0.05). The concentrations of 100 or 200 mg/L paraquat, in particular, led to dramatic, but similar, decreases in MRC-5 cell viscoelasticity (vs. 50 mg/L paraquat-treated cells, p < 0.05).

Paraquat treatment reduces MRC-5 cell viscoelasticity. A representative curve of aspirated length at constant negative pressure over time (24 h) of MRC-5 cells treated with various concentrations of paraquat is shown.
Viscoelastic parameters of MRC-5 cells before and after paraquat treatment.

a p < 0.05 compared with control cells at the same time point.
b p < 0.05 compared with 50 mg/L paraquat treatment at the same time point.
Although the high levels of paraquat produced toxicity in a large percentage of the MRC-5 cells, we were still able to collect enough healthy cells from these treatment groups to evaluate their viscoelasticity. The paraquat-induced biomechanical changes in the MRC-5 cells were not significantly different among the three time points at each concentration (p > 0.05; Table 2). We concluded that the impaired biomechanical properties induced by paraquat resulted in deformed MRC-5 cells, and that paraquat significantly alters lung fibroblast viscoelasticity.
Paraquat significantly enhances CTGF expression in MRC-5 cells
The quantitative PCR experiments revealed that paraquat treatment led to significantly reduced CTGF mRNA expression at 24 h posttreatment (vs. control cells, p < 0.05; Table 3) but not at 48 h posttreatment. Intriguingly, the paraquat-treated cells showed significantly increased CTGF mRNA expression level at 72 h posttreatment (vs. control cells, p < 0.05). For the control cells, the CTGF mRNA level was significantly higher at 48 h compared with 24 h (p < 0.05), but the increasing trend did not continue through to the 72 h time point. In general, the paraquat-induced CTGF mRNA expression followed a time-dependent pattern (p < 0.05).
CTGF mRNA expression in MRC-5 cells in the presence or absence of paraquat.

CTGF: connective tissue growth factor; mRNA: messenger RNA
a p < 0.05 compared with control group.
b p < 0.05 compared with the previous time point.
The paraquat-induced protein expression profiles detected by ELISA followed a similar pattern. In the control cells, CTGF expression progressively increased after 48 and 72 h (p < 0.05). CTGF protein expression was significantly lower in the paraquat-treated cells at 24 and 48 h posttreatment (vs. control cells, p < 0.05; Table 4). However, at 72 h posttreatment, the CTGF protein expression was significantly higher in the paraquat-treated cells (vs. control cells, p < 0.05; Table 4). Progressive increases in CTGF expression were observed at 48 and 72 h with the 50 and 100 mg/L concentrations (p < 0.05); however, expression was only significantly increased after 72 h with the 200 mg/L treatment (p < 0.05). Altogether, these findings suggest that paraquat induces CTGF gene expression in MRC-5 cells.
CTGF protein expression in MRC-5 cells in the presence or absence of paraquat.
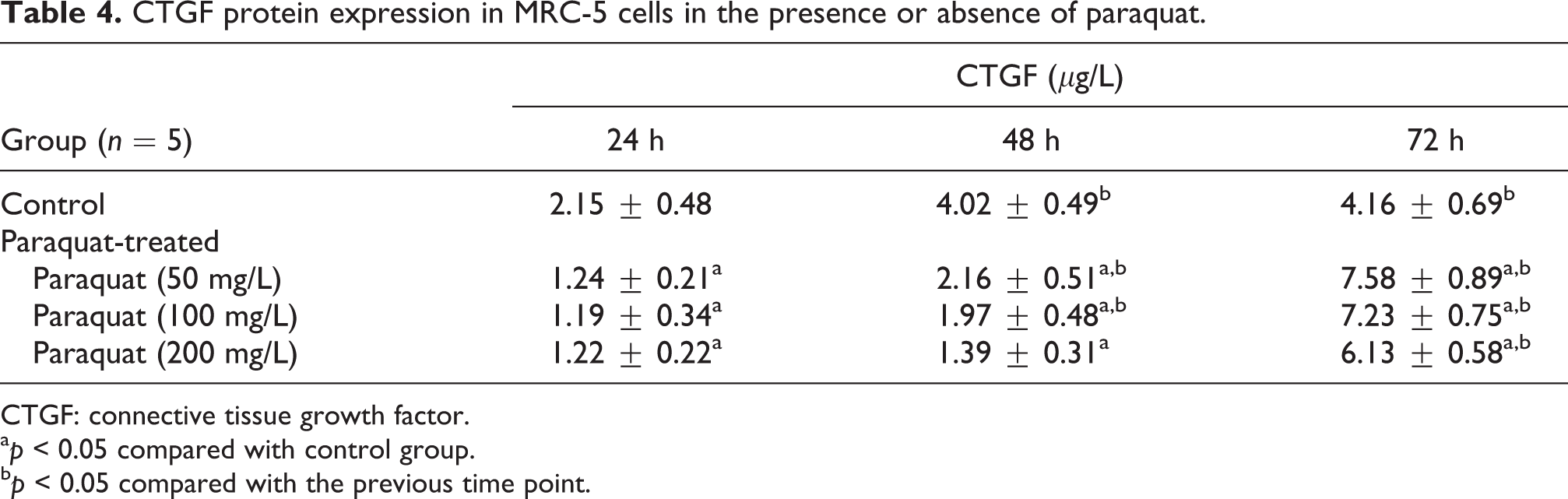
CTGF: connective tissue growth factor.
a p < 0.05 compared with control group.
b p < 0.05 compared with the previous time point.
Discussion
In the current study, we demonstrated that paraquat significantly reduces lung fibroblast proliferation and profoundly affects the cell’s viscoelastic properties, both in a concentration-dependent manner, as well as causing dramatic alteration of the cell’s morphology. We also show that paraquat profoundly affected the viscoelastic properties of MRC-5 cells in a concentration-dependent manner. More importantly, our in vitro studies show that paraquat significantly enhances CTGF transcription and translation in lung fibroblasts. Furthermore, we demonstrated a time-dependent effect, with and initial suppression of expression, followed by enhanced expression after 72 h. The examination of protein changes did not reveal a dose-dependent effect for the concentrations and time-points selected. However, as the reflection of changes in protein expression levels is delayed with respect to mRNA changes, it is possible that a later time point is needed to observe an effect. It is also possible that the lowest concentration of paraquat used in our study fell below the linear range for regulating CTGF protein expression in MRC-5 cells after 24 h. Nonetheless, this work suggests that the CTGF signaling pathway may be an important mechanism for the progression of lung fibrosis caused by paraquat poisoning.
The majority of paraquat poisoning mortalities are due to a lack of effective treatments. 6 Unfortunately, the currently available elimination therapies and therapies against the related inflammatory responses have displayed little efficacy, with paraquat tissue accumulation and the related pathological responses continuing in treated patients. 11 Furthermore, histopathological analyses of clinical samples have revealed a surprising incidence of critical structural changes that occur in response to the paraquat-induced severe inflammatory responses. 5 The lung has been identified as an especially vulnerable organ to paraquat poisoning, becoming heavily congested and edematous with development of large numbers of plump fibroblasts in alveolar walls and alveolar spaces. The observed changes of pronounced vascular congestion, intra-alveolar diffuse coagulation, and macrophage infiltration suggest an abnormal activation of the nuclear factor-κB signaling pathway. Indeed, gross examination of patients with severe paraquat poisoning has shown dark gray lungs with reduced size and increased stiffness, characteristic of pulmonary fibrosis. 22
The molecular mechanism of fibrosis involves accumulation of extracellular matrix due to excessive tissue repair and impaired matrix turnover. CTGF is a cysteine-rich protein, which stimulates extracellular matrix production and up-regulation of collagen and fibronectin expression. Recent studies of the profibrotic properties of CTGF have indicated that this factor promotes accumulation of several extracellular matrix proteins, such as collagen, fibronectin, and tenascin C. 23 In addition, silencing of CTGF using small interfering RNA was shown to significantly decrease the expression of collagen in skin fibroblasts, 19 while proteomic analyses of CTGF-treated normal lung fibroblasts demonstrated a dramatic increase in extracellular matrix and cytoskeletal proteins. 24
Overexpression of collagen and other extracellular matrix proteins in fibrotic lesions is considered a biomarker of fibrosis. Several groups have demonstrated that CTGF induces collagen I by stimulating collagen I transcription in various cell lines. 13,25 Lung fibrosis is also accompanied by abnormal expression of myriad cytokines and enzymes, including matrix metalloproteinases. 16 Our results are consistent with the conclusion that CTGF is a major mediator of paraquat-induced lung fibrosis. Yet further studies are necessary to elucidate precisely how paraquat promotes CTGF transcription and translation as well as which signaling pathways are responsible for CTGF upregulation, in addition to the TGF-β pathway.
High viscoelasticity is an important property of lung tissue. Viscoelastic substances do not maintain a constant stress under constant deformation. In addition, under constant stress, viscoelastic materials undergo a continuous deformation over time. 26 In fact, viscoelasticity represents an important component of respiratory mechanics for lung deformation caused by respiratory movement. Lung fibrosis is known to impair the dynamic mechanical movement necessary for proper lung function. Mechanical stimuli, in turn, can affect fibroblast proliferation and cytokine expression. 27 Therefore, mechanical activity plays a critical role in the process of pulmonary fibrosis. The relationship between CTGF expression and biomechanical properties of the lung in pulmonary fibrosis pathogenesis remains unclear. In future studies, we plan to determine the mechanisms by which CTGF and other cytokines and extracellular matrix proteins affect the lung biomechanical properties as these data will be useful in helping to resolve the life-threatening pathological processes of paraquat poisoning.
In summary, our in vitro data suggest that paraquat poisoning significantly reduces lung fibroblast proliferation and alters cellular morphology. Paraquat is critical for CTGF overexpression, which may contribute to altered viscoelasticity of lung fibroblasts. These findings represent empirical evidence to understand paraquat-induced damage in human lung fibroblasts. Inhibiting CTGF overexpression and accumulation in lung tissue could be a potential therapeutic strategy for paraquat poisoning. Certainly, identifying the signaling pathways involved in the regulation of CTGF expression and degradation would provide further insights for efficacious treatments. The future studies investigating the mechanisms by which CTGF affects lung biological function in animal models are likely to shed light on other modes of therapy for paraquat poisoning.
Footnotes
Acknowledgment
We would like to thank the staff of the Department of Emergency Medicine at the First Hospital of Jilin University for their contribution.
Authors’ Note
Authors NZ and YPX contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by a grant from the Youth Science and Technology Foundation of Jilin Province (No. 20100138).
