Abstract
Nifurtimox (Nfx) and benznidazole (Bz) have serious toxic side effects. Manufacturers warn about significant adverse effects when simultaneous alcohol consumption is being made, but its mechanism is not known. The levels and toxicity of these drugs are linked to their liver microsomal nitroreduction to reactive metabolites. In this study, we analyzed whether alcohol drinking enhanced those nitroreductive processes. Male and female Sprague-Dawley rats, 5–6 weeks old (125–150 g body weight) were used. They were fed ad libitum for 28 days with Lieber and De Carli control or alcohol regular liquid diets. The rats were separated into two dietary groups: ethanol and control group. Both were pair fed with the respective diet. Their liver microsomes were isolated and the nicotinamide adenine dinucleotide phosphate-dependent nitroreduction of Nfx and Bz were determined. Alcohol drinking significantly induced microsomal nitroreduction of these drugs in male rats (11% for Nfx and 41% for Bz) but not in females. The activity observed in the alcohol-induced male rats was 100% inhibited by diphenyleneiodonium and attributable to P450 reductase. Inductive effects of alcohol drinking on nitroreductive activation of both drugs might be only partially involved in the harmful interactions described.
Introduction
A large number of medicines may lead to harmful interactions when mixed with alcohol drinking. The National Institute in Alcohol Abuse and Alcoholism from the United States recently provided a large list of commonly used medicines that clearly illustrate this problem. The significant adverse effects, including fatal consequences, resulting from those interactions were also reviewed in literature. 1 –3
Our laboratory was interested in learning about the mechanism of the clinically reported adverse effects of alcohol drinking on the trypanosomicide drugs nifurtimox (Nfx) and benznidazole (Bz). Despite the harmful effects are mentioned in the instructions given by the manufacturers of both the products, their characteristics are not available in the known medical literature and their possible mechanisms were not established. In the earlier studies by Masana and Rubio, 4 the authors reported that Bz did not change alcohol levels in blood during acute alcohol drinking experiments. No other work is available in literature specifically on this subject, despite the intensive use of those two drugs to treat Chagas’ disease. This endemic disease still affects approximately 16–18 million people in Latin America, even though the relevant advances made to achieve its eradication. 5 –7
In this article, we describe initial efforts to contribute the understanding of the nature and the mechanisms of these harmful interactions. The here described work was directed to learn about the possibility that chronic alcohol drinking induced the liver microsomal nitroreductive metabolism of both the drugs and critically involved in modulating their blood levels and their toxicity. 5
Methods
Chemicals
Nfx (3-methyl-N-[(5-nitro-2-furanyl)-methylene]-4-thiomorpholinamine-1,1-dioxide) was a gift from Bayer Laboratories (Buenos Aires, Argentina), Bz (N-benzyl-2-nitroimidazole-1-acetamide) was obtained from Roche (Buenos Aires, Argentina). Liquid diet ingredients were Lieber-De Carli Regular, #710260, for rodents ethanol group and Lieber-De Carli Regular, #710027, for rodents control group; both were purchased from Dyets, Inc. (Bethlehem, PA, USA). Nitrogen (ultrahigh purity) was obtained from AGA (San Martín, Argentina) and was further deoxygenated by bubbling it through a solution containing 0.05% 2-anthraquinone sulfonic acid sodium salt and 0.5% sodium hydrosulfite in 0.1 N NaOH. All other chemicals employed were of analytical grade.
Animals and treatment
Non-inbred Sprague-Dawley rats, 5–6 weeks old (125–150 g body weight) were used. They were kept under controlled conditions on a 12-h light:12-h dark cycle (light phase from 6 to 18 h). The room temperature was maintained at 23 ± 2°C and relative humidity was maintained at 45–65%. The procedures used for breeding, housing, and handling animals were in accordance with the guidelines set by Food, Drug and Medical Technology National Administration (Buenos Aires, Argentina). Rats were fed ad libitum with a nutritionally adequate liquid diet (Lieber and De Carli Regular rodent diet) for 28 days. 8,9 The liquid diet used provided 1 kcal/mL, where 35% calories derived from fat, 47% from carbohydrate (maltose-dextrin), and 18% from protein. The rats were housed in individual cages and separated into two dietary groups: ethanol group (alcoholic group) and control group (control). Both groups were pair fed with the same diet except that in the alcoholic group, ethanol provided 36% of the calories, replacing isocaloric amount of carbohydrate. These diets assured continued growth in all animals and normal liver in the control, whereas in the rats fed with alcohol, fatty liver developed. A record of daily liquid diet consumption, using a graduated feeding tube (Dyets, Inc. Catalog #900006), was made and their body weight changes were registered. It was started with 30 g/L ethanol of the liquid diet for 2 days, 40 g/L for the subsequent 2 days followed by the final formula containing 50 g/L during 24 additional days. The amount consumption was 13–15 g ethanol/kg/day. The animals were killed by decapitation with a Harvard guillotine and bled to minimize the potential interference of hemoglobin. Liver tissue was rapidly excised and processed.
Isolation of liver microsomal fraction
Each microsomal sample was obtained from the homogenate of the total livers from five animals per group (alcoholic group and control). The liver tissue of five rats from each group (alcoholic group and control) was homogenized (1:4 w/v) in a Teflon-Glass-Potter-Elvehjem homogenizer with 0.25 M sucrose, 5 mM EDTA, 20 mM Tris/HCl, pH 7.4. The homogenate was centrifuged 20 min at 9000g, and the supernatant was further centrifuged for 1 h at 100,000g to obtain the microsomal pellets. All procedures were performed at 0–4°C. 10
Microsomal nitroreductase activity
All incubations were run anaerobically (N2) in 20 mL septum-sealed flasks, with shaking (150 oscillations/min) in a covered Dubnoff shaker, to ensure darkness, at 37°C for 20 min. The incubation mixtures contained the microsomes suspended in 20 mM phosphate buffer, pH 7.4; 0.5 mM β-nicotinamide adenine dinucleotide phosphate (NADPH), reduced form of NADPH-generating system, and 50 µM of Nfx or Bz are dissolved in N, N-dimethylformamide (final concentration 2%) in a final volume of 2.5 mL. To test the inhibition of the microsomal activity, 10 µM diphenyleneiodonium chloride (DPI) was added to another set. After incubation, the reaction was interrupted simultaneously by placing the flasks on ice and by adding 1 mL of 15% (w/v) zinc sulfate. A fraction of each incubation mixture was poured over 1 g of NaCl and extracted with 4 mL of ethyl acetate. An aliquot of the organic layer was evaporated under N2. The residue dissolved in mobile phase was analyzed by high-performance liquid chromatography (HPLC). Nitroreductase activity was followed by substrate decrease, calculated from the difference in concentration of Nfx or Bz at the beginning and at the end of the reaction. 11,12 Recoveries of known additions of each drug to incubation mixtures were more than 95%. The data from each group (alcoholic group and control) were the mean ± SD from three separate experiments. In each one, five separate microsomal incubations and HPLC analysis were made.
HPLC assay for Nfx and Bz
Samples were analyzed at 40°C in a Hewlett Packard model 1090 series II HPLC with a HP ODS Hypersil column (200 × 2.1 mm inner diameter, 5 mm particle size) and a HP diode array detector. The mobile phase, consisting of 60% methanol–water, was delivered at a constant flow-rate of 0.2 mL/min. The column effluent was monitored at 400 nm for Nfx and at 320 nm for Bz. 11,12 Drugs were quantified by peak-area ratio with reference to standards identically treated. The concentration range of the calibration curves were 10–70 µM for both the drugs. At these selected wavelengths, the others components of the incubation mixtures did not interfere with Nfx and Bz HPLC chromatograms.
Protein concentration
Protein concentration was determined using the method of Lowry, using bovine albumin (from bovine plasma) as standard. 13
Statistical analysis
Significance differences between mean values with their respective SDs were assessed by the Students’ t test and two-sided p value. Calculations were performed using GraphPad Software. 14 For all the analysis, the criterion of significance was set at p < 0.05.
Results and discussion
In the present study, we observed a significant inductive effect on the nitroreductive metabolic transformation of Nfx and Bz by liver microsomes from male rats drinking a Lieber-De Carli regular diet for 28 days. The inductive effect was more intense for the case of the Bz nitroreductase activity (e.g. 41%) than for the one of the Nfx metabolizing enzyme (e.g. 11%). The inductive effect of alcohol drinking was not observed in the microsomal nitroreductive metabolism of Nfx or Bz from female rat livers (Table 1). The liver microsomal nitroreductase activity from control rats of Nfx was more intense than the equivalent one for Bz (3.1 times in the case of males and 2.3 times in females). Furthermore, the enzymatic activity for each drug was higher in males than in females (1.5 times in the case of Nfx and 1.1 times for Bz). In the case of values obtained from alcohol-treated animals, the difference between the metabolic rates of Nfx against Bz was smaller than the one observed in controls (2.5 times for males and 2.1 times for females). These differences might be attributable to the inductive effects of alcohol drinking on the respective enzymatic activities (Table 1). There was a significant difference in the male liver microsomal nitroreductase absolute activities values from control and ethanol animals when experiments were performed 8 months apart each other (spring values from Table 1 and autumn results Table 2). However, the values for the inductive effect of ethanol were of an approximate similar order (19% instead 11% for Nfx and 34% instead of 41% for Bz). The reasons for that difference are not known by us at present. The animals and their strain, sex, and weight were the same in both the experiments. Furthermore, the animal food and general procedure for breeding, housing, and handling animals were exactly the same. The induced enzyme activity in male rats was 100% inhibited by 10 µM DPI (Table 2). At this concentration, DPI is a well known inhibitor of P450 reductase activity. 15
Nitroreductase activity in hepatic microsomes of male and female rats drinking an alcoholic liquid diet for 28 days.a
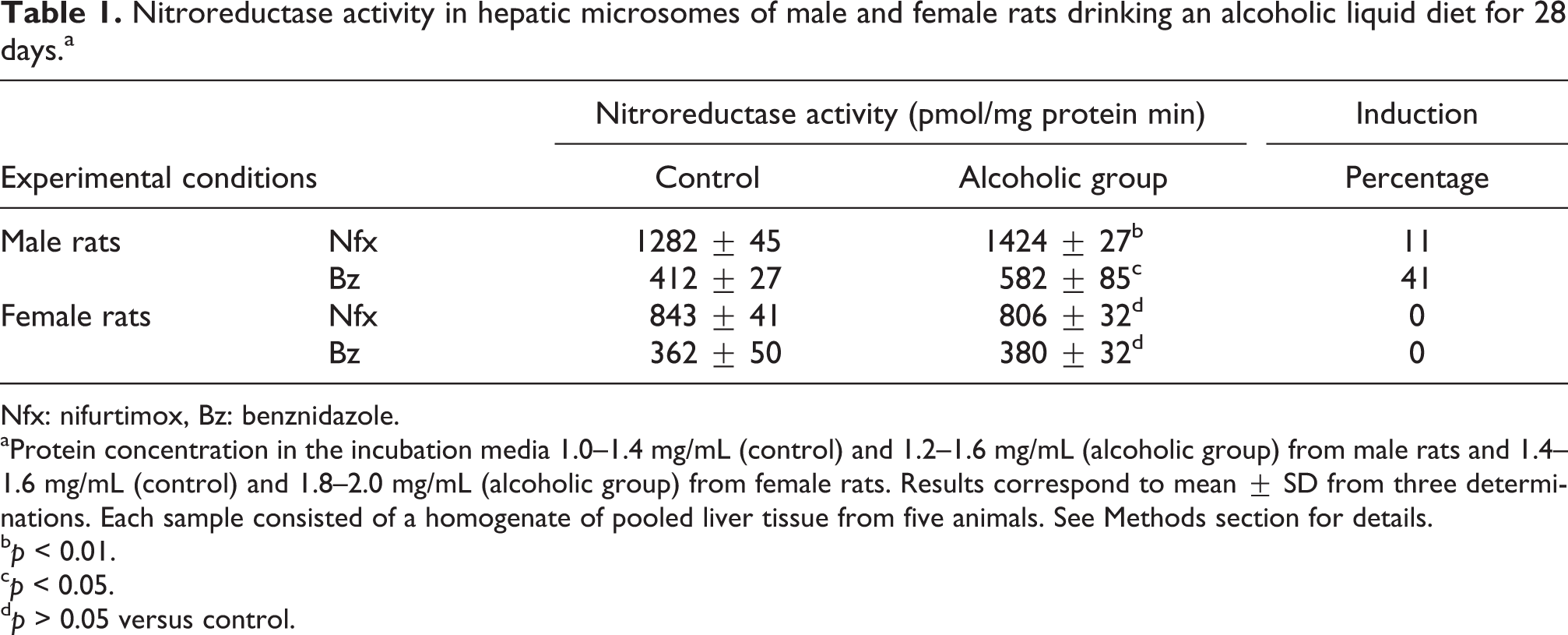
Nfx: nifurtimox, Bz: benznidazole.
aProtein concentration in the incubation media 1.0–1.4 mg/mL (control) and 1.2–1.6 mg/mL (alcoholic group) from male rats and 1.4–1.6 mg/mL (control) and 1.8–2.0 mg/mL (alcoholic group) from female rats. Results correspond to mean ± SD from three determinations. Each sample consisted of a homogenate of pooled liver tissue from five animals. See Methods section for details.
b p < 0.01.
c p < 0.05.
d p > 0.05 versus control.
Inhibitory effects of DPI on the Nfx or Bz liver microsomal nitroreductase activities from male rats chronically drinking a Lieber-De Carli diet for 28 days.a

DPI: diphenyleneiodonium chloride; Nfx: nifurtimox, Bz: benznidazole, ND: no detectable activation.
aProtein concentration in the incubation media 0.66–0.69 mg/mL for Nfx and 1.1–1.2 mg/mL for Bz. Results correspond to mean ± SD from three determinations. Each sample consisted of a homogenate of pooled liver tissue from five animals. See Methods section for details. DPI final concentration was 10 µM.
b p < 0.05.
c p < 0.005 versus control.
The potent inhibitory effects of low concentration of DPI, on the induced and control nitroreductive metabolism of Nfx and Bz by liver microsomes from male rats, clearly show that P450 reductase enzyme is involved. This enzyme was previously reported, by our laboratory, to be of major relevance in the liver microsomal metabolism of both the nitroheterocyclic drugs. A smaller fraction of the nitroreductive process requires cytochrome P450 (CYP). 10,16 Our results, in Table 2, also show that chronic alcohol drinking was able to induce to some extent P450 reductase activity (19% when tested via Nfx metabolism and 34% when determined through Bz metabolism). This observation is in accordance with previous findings made already in 1968 by Sasame et al., 17 who reported that even after a single dose of alcohol, there was at 24 h a small but significant increase in NADPH cytochrome c reductase activity in male liver microsomes. NADPH-dependent P450 reductase is able to nitroreduce both the drugs without participation of P450 itself. The fraction of activity requiring participation of CYP, however, also involves NADPH-dependent P450 reductase, which is needed to change the CYP to its reduced form, the latter being the active species reducing Nfx or Bz. 10,16 In fact, both processes, direct and indirect, appear to play a role. Concerning the specific nature of the P450 isoenzyme involved in the smaller fraction of the metabolism of both the drugs, we are not able, at present, to evidence which one is participating in this anaerobic pathway. However, it is known that alcohol drinking is able to induce mostly CYP2E1 but also possibly less of CYP1A2 and CYP3A4. 18 Notwithstanding, their participation in the ethanol-inductive process was established via determination of the aerobic transformation of ethanol to acetaldehyde.
At present, we are not able to fully evaluate the reasons for the relative differences between the liver microsomal nitroreductive enzymes activity of both drugs and their toxicity. One reason is that their mechanism of action differs at steps after the nitroreductive stage. 5 In addition, it is not known whether other predictable reductive processes occur and about their relevance (e.g. a reduction in Nfx hydrazo group to a hydrazine) or if metabolites formed are able to feed back inhibit the nitroreductive metabolism of both drugs.
Another interesting observation is that there was a gender difference in the effect of chronic alcohol consumption on the nitroreductive metabolism of Nfx and Bz by liver microsomes. The enzymatic nitroreductase activity for both the drugs was higher in control males than in females but the difference between both is not as large as that previously reported for other substrates. 19 For chronic alcohol drinking, the difference in the inductive response of males to that observed in females might reasonably be linked to the effect of alcohol drinking on microsomal drug metabolizing enzymes. 18,20 Although the exact mechanism responsible for gender differences in the response of liver to alcoholic group injury remains unclear, the available findings strongly suggest an involvement of sex hormones in the pathophysiology of alcohol-induced liver disease. 21 In fact, it is very well known that chronic alcohol drinking provokes intense changes in the hormonal milieu of blood and the liver in both sexes. 21 Studies in men and in male rats evidenced that during chronic alcohol drinking, they cannot maintain an appropriate hormone balance, leading to low serum testosterone and increased estrogen levels. Decrease in serum testosterone occurs in the early stages of alcohol drinking and is attributed to the toxic effects in the testes. 21 –23
In women and female animals increased levels of estrogen after alcohol drinking were also reported by other workers. 21 Studies in our laboratory showed that chronic alcohol drinking produces severe ultrastructural alterations in different cell types of the rat ovarium. 24 Furthermore, in both female rats and women, progesterone, the “counter -hormone to estrogen”, is reduced with alcohol consumption. Thus, the estrogen present is less opposed by progesterone. 21
In summary: The different response to chronic alcohol drinking found in our studies in relation to the liver microsomal nitroreductive metabolism of Nfx and Bz suggests that hormonal factors might be involved. These hormonal changes might perturb enzymes involved in the liver processing of steroid hormones and also modulate in a different mode the gene expression leading to synthesis of drug metabolizing enzymes. These enzymes are critically involved in Nfx and Bz metabolic activation to reactive toxic metabolites which lead to relevant adverse effects of both drugs. 5
Footnotes
Conflicting of Interests
The authors declared no conflicts of interest.
Funding
This work was supported by a grant from FONCyT Argentina (PICT/05 38235) and by the San Martín University, Argentina (SP06/090).
