Abstract
Objective:
The purpose of this study was to evaluate the patients with acute amitriptyline poisoning and investigate predictive factors for the development of life-threatening complications.
Methods:
Demographics, clinical, laboratory, and electrocardiographic (ECG) findings of 250 patients were evaluated retrospectively. Predictive parameters for the development of serious complications were studied.
Results:
Median age of patients was 14.6 years, of which, 70% of patients were female and 66% were in pediatric age group. The most common pathological clinical finding and laboratory abnormality were alteration of consciousness and hyponatremia. The rate of convulsive seizure, arrhythmia, and respiratory depression were 17 (6.8%), 16 (6.4%), and 11 (4.4%), respectively. These complications were more seen in pediatric patients than adults (15.8% and 1.2%). The incidence of hyponatremia was more in pediatric patients and severe poisoning groups (38.8 and 53.4%, respectively). The levels of amitriptyline and nortriptyline were significantly higher in the group with complications than the group without complications (p < 0.05). All adult patients were discharged with good prognosis. In pediatric age group, one patient was discharged with severe neurological sequelae and one patient died. QRS duration >100 ms, long corrected QT duration interval, and low Glasgow Coma Score (GCS) at admission were identified as independent risk factors for the development of life-threatening complications (odds ratio: 69.4, 1.9, and 1383, respectively; p < 0.05).
Conclusion:
Amitriptyline poisoning may be associated with life-threatening complications, especially in pediatric age group and in patients with hyponatremia. Low GCS, presence of hyponatremia, high serum drug levels, and pathological ECG findings on admission may be helpful in predicting the development of complications and poor prognosis.
Introduction
Because of cardiovascular toxic effects, tricyclic antidepressants (TCA) are still one of the common causes of fatal drug poisoning and manifest itself especially with electrocardiographic (ECG) abnormalities, arrhythmias, and hypotension. 1 Amitriptyline is a major TCA drug that is used widely. 2,3 Many of TCA overdose cases presented with comatose state will recover smoothly with supportive care and close monitoring. 4 However, a small group of patients develop life-threatening complications. 5 An antidepressant overdose risk assessment criteria was defined to determine the severity of poisoning with antidepressant drugs in adults. 6 However, prognostic factors for serious complications and death, especially in children, is still not clear. 2,7 Previous studies have shown that severe neurological and cardiovascular side effects arise from the blockage of voltage-dependent sodium channels providing a fast flow of sodium (Na) into the cell. 8 However, the impact level of serum Na on the weight of toxicity and prognosis is unclear. In this study, including the most extensive series of amitriptyline intoxication in pediatric patients, the clinical, laboratory, and ECG findings of patients poisoned with amitriptyline alone were evaluated. In addition, predictive factors, including the level of serum Na, were investigated to foresee the life-threatening complications.
Methods
The study was performed in the 900-bed university hospital. Medical records of patients hospitalized for amitriptyline intoxication between January 2008 and March 2013 were analyzed retrospectively. Newborns and patients poisoned with more than one drug or toxic substance were excluded. Amitriptyline intoxication diagnosis has been established with verbal expression of the patient or patient's relative who witnessed the event or by the presence of high levels of amitriptyline or its metabolite, nortriptyline, in serum samples from patients. Patients’ demographic characteristics, cause of drug use (accidental or suicide), duration between ingestion and first medical intervention, estimated dose of medication, clinical findings, ECG findings, routine laboratory results, and drug concentrations were recorded. In addition, decontamination measures, complications (if any), specific treatments, and results were recorded. Patients with a history of chronic illness or drug use, patients using antiepileptic drugs or antidepressants and patients leaving the hospital before completing the proposed follow-up period were excluded from the final evaluation. Patients were divided into three groups according to the ages and the degree of poisoning. Age groups included <8 years, 8–18 years (pediatric group), and >18 years (adults group). Patients with Glasgow Coma Score (GCS) ≤ 8 or with any serious side effects such as cardiac or respiratory failure, arrhythmias, and seizures accepted as severe, patients without serious side effect and with a GCS between 9 and 11 were defined as moderate, and patients with a GCS between 12 and 14 were accepted as mild poisoning. In addition, patients were divided into two groups according to the presence of life-threatening complications such as seizures, arrhythmias, and respiratory depression. Patients having any of this complication were defined in the group with complication. Clinical and laboratory data of all patients were obtained from hospital computer database system and medical cards. Evaluated ECG parameters consisted of heart rate, PR interval, QRS duration, corrected QT duration (QTc), and R wave amplitude in aVR or R/S ratio. Patients were divided into two groups according to QRS duration (longer than 100 ms and less than 100 ms) and into four groups according to QTc duration (<0.44, 0.44–0.46, 0.46–0.48 and >0.48 s). 9 In addition, based on R wave amplitude or R/S ratio, patients were divided into two groups: patients with and without R wave amplitude ≥4 mm or R/S ratio >0.5 in aVR. The age-matched arterial blood pressure <5 percentile was defined as hypotension and >95 percentile was defined as hypertension. Serum Na level <135 meq L−1 was defined as hyponatremia, serum glucose level of <70 mg dL−1 was defined as hypoglycemia, and serum glucose level >110 mg dL−1 was defined as hyperglycemia.
Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS 21.0, SPSS Inc., Chicago, Illinois, USA) statistical analysis program for Windows. Fisher’s exact test was used to compare the difference between the frequency ratios of the categorical data. Numerical data meeting the normal distribution was given as mean ± standard deviation; numerical data not meeting the normal distribution was given as median (minimum–maximum). The independent sample t test was used to compare the difference between the averages of data matching the normal distribution. The difference between the averages of data not meeting the normal distribution was compared using the Mann–Whitney U test. Correlations between quantitative data were analyzed by Spearman's correlation test. Logistic regression models were used to predict the risk factors for life-threatening events such as seizure, ventricular or supraventricular arrhythmias, or respiratory depression. Age, gender, cause of ingestion (accidental or suicide attempt), GCS, QRS time >100 ms, QTc interval, and presence of hyponatremia were taken as the independent risk factors. Logistic regression models were used to construct receiver-operating characteristic (ROC) curves for amitriptyline and nortriptyline. The level of statistical significance was set at p < 0.05.
Results
In the present study, 250 patients with acute amitriptyline poisoning were evaluated. During the study period, 165 pediatric and 85 adult patients were admitted to the emergency department due to amitriptyline poisoning. Of these 250 patients, 175 (70%) patients were female and their median age was 14.6 years (range, 1–87). Fifty-nine percentage of poisoning was secondary to suicide attempt. Female patients were more than male patients in all age groups. This difference was more pronounced in suicidal poisoning. The youngest patient who attempted suicide was 8 years old. In pediatric age group, an estimated dose of drug taken by accident (13.2 mg kg−1) was significantly higher than for suicide (5.9 mg kg−1; p < 0.05). Only two patients in adult group took medication by accident; therefore, a statistical evaluation was not carried out for this group. Duration between ingestion and first medical intervention was longer in pediatric patients than adults (4.5 and 2 h; p < 0.05). Demographic and poisoning characteristics of patients are shown in Table 1.
Demographic characteristics of study patients and features of poisoning.
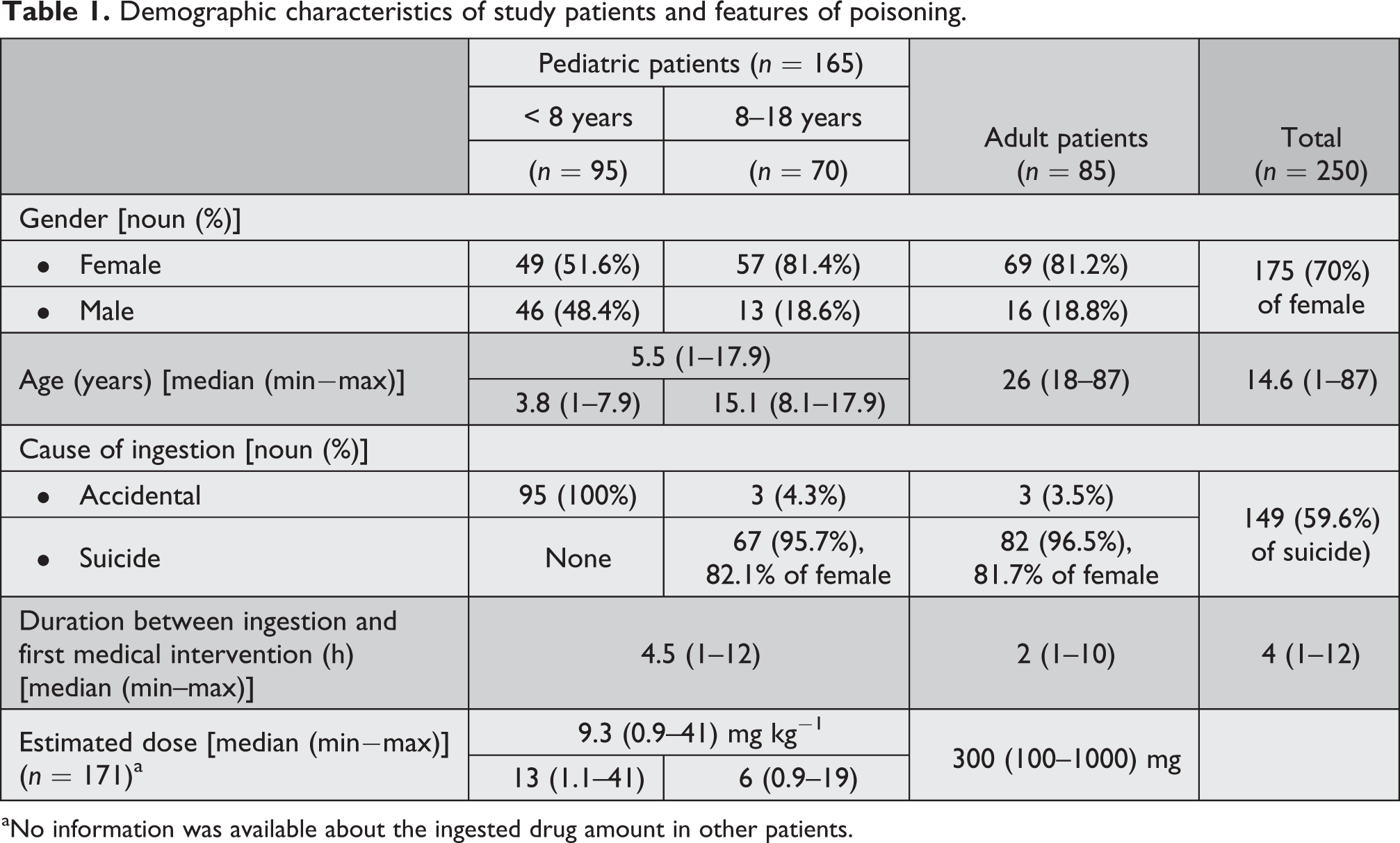
aNo information was available about the ingested drug amount in other patients.
The most common pathologic clinical findings were alteration of consciousness, sinus tachycardia, and loss of deep tendon reflexes at the time of admission. In all, 88 (53.3%) pediatric cases were admitted with coma. In adult patients, admission with coma was quite rare (3.5%). Tendency to sleep was seen in 59 (69.4%) of adult patients. In our study, the rates of serious side effects such as convulsive seizure, arrhythmia, and respiratory depression were 17 (6.8%), 16 (6.4%), and 11 (4.4%), respectively. Two adult patients had developed ventricular tachycardia. None of the other patients had developed life-threatening complications. During the time of admission, 11 patients in pediatric group and 6 patients in adult group were hypotensive. The difference was significant at p < 0.05). Clinically defined severity of poisoning was more severe in pediatric patients than adults. Clinical features of study patients are given in Table 2.
Clinical features of study patients at admission.

SVT: supraventricular tachycardia; VT: ventricular tachycardia; DTR: deep tendon reflexes;
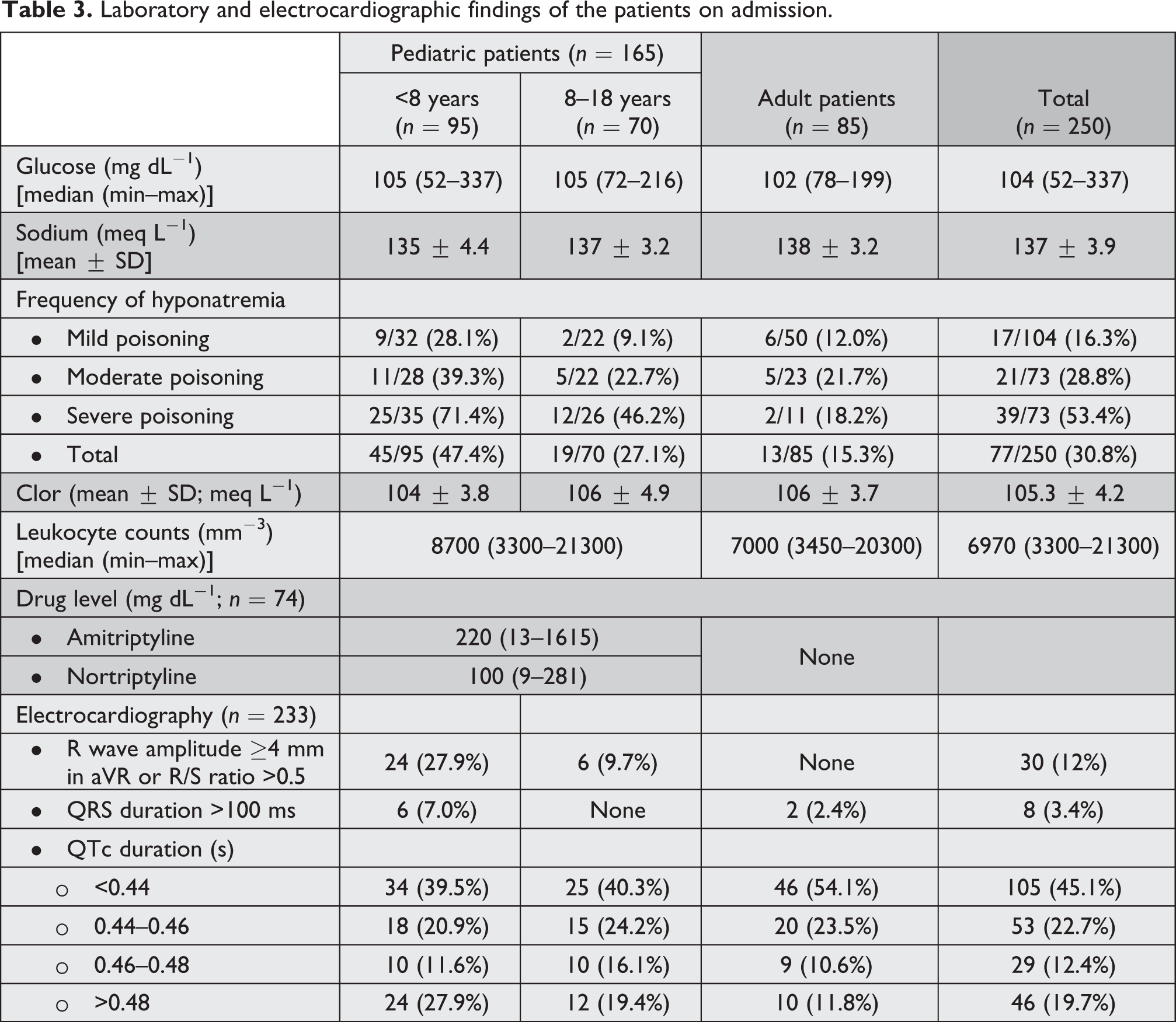
Liver and kidney functions were at normal limits in all patients. The most common laboratory abnormalities on admission to hospital were hyponatremia, hyperglycemia, and leukocytosis. Hypoglycemia was detected in three patients in pediatric age group. These three patients were under the age of three. Hypoglycemia did not persist in any of the patients. All patients were normoglycemic at follow-up. None of the patients had hypernatremia. The incidence of hyponatremia was higher in pediatric patients, in patients with complications, or in severely poisoned patients. The serum levels of amitriptyline and nortriptyline were analyzed in 74 (44.8%) pediatric patients. Drug levels were not evaluated in any of the adult patients. At the time of admission and follow-up, the levels of amitriptyline and nortriptyline were significantly higher in the group with complications than the group without complications (p < 0.05). The cut off value was 208 mg dL−1 (area under ROC curve (AUC) = 61.8%, AUC 95% confidence interval (CI) = 46.4–77.0%) for amitriptyline and 125.5 mg dL−1 for nortriptyline (AUC = 66.1%, AUC 95% CI = 51.4–80.8%; Figure 1(a) and (b)).

ROC curves for levels of (a) amitriptyline and (b) nortriptyline during the time of admission to the hospital. AUCs were 61.8%, 95% CI = 46.4–77.0% and 66.1%, 95% CI = 51.4–80.8%, respectively. ROC: receiver-operating characteristic; AUC: area under ROC curve; CI: confidence interval.
ECG records of 17 patients could not be reached. Due this reason, ECG evaluation was performed in 233 patients. The most common ECG abnormality was tachycardia and prolonged QTc interval. ECG abnormalities were more frequent especially in pediatric patients, in group with severe toxicity and in patients with hyponatremia. The proportion of patients with QTc duration >0.48 s was more in the heavily intoxicated group. There was significant difference between the groups with and without complications in terms of QTc duration. Prolongation of QTc duration is found to be a risk factor for complication (odds ratio (OR) = 1.9, 95% CI = 1.089–3.196, and p < 0.05). Also, there was a significant positive correlation between QTc duration and levels of amitriptyline and nortriptyline at the time of admission (r = 0.48 and 0.52, respectively; p < 0.05). The proportion of patients both with QRS duration >100 ms and R wave amplitude ≥4 mm in aVR or R/S ratio >0.5 was more in patients with complication than patients without complication. The difference was statistically significant (p < 0.05). Laboratory and ECG findings of the patients on admission are shown in Table 3.
Laboratory and electrocardiographic findings of the patients on admission.
Of all the patients, 87 (34.8) were directly admitted to our hospital. The others were referred from an initial health care facility. A great portion of patients had undergone gastric lavage and were given activated charcoal at the first referred center (70.4 and 89.2%, respectively). The median time between admission to hospital and gastric lavage/activated charcoal application was 2 h. Among the pediatric patients, 15 (9.1%) required inotropic support and 12 (7.3%) required mechanical ventilation. None of the adult patients required inotropic support or mechanical ventilation. All adult patients were discharged with good prognosis. In pediatric age group, one patient was discharged with severe neurological sequelae and one patient has died. Treatments and prognosis of the patients are shown in Table 4.
Performed treatment attempts and prognosis.
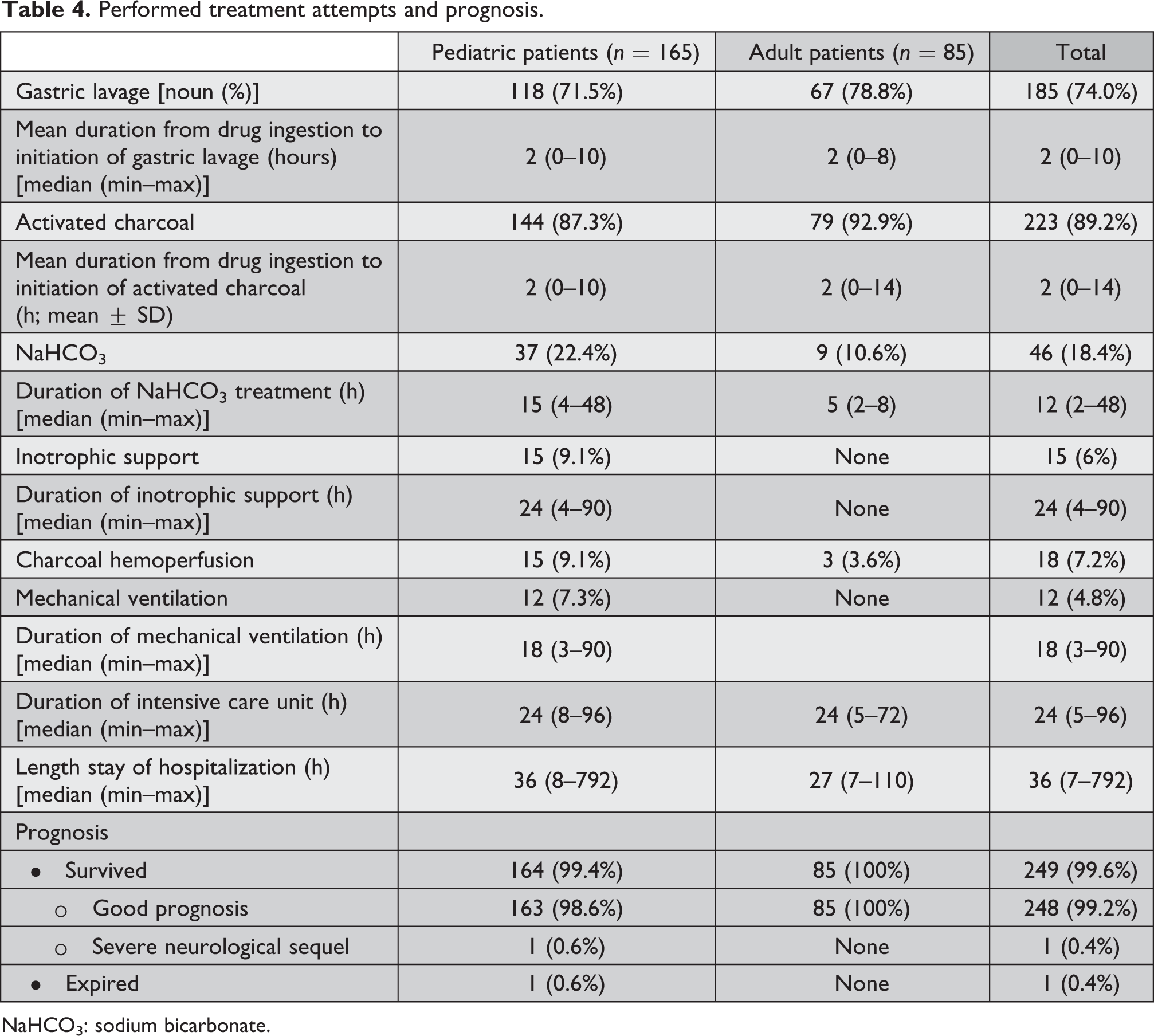
NaHCO3: sodium bicarbonate.
In all, 27 patients had developed at least one life-threatening complication such as seizure, arrhythmia, or respiratory depression. Of these patients, 25 (92.6%) were at pediatric age group. General characteristics of patients who had developed complications are shown in Table 5.
Characteristics of patients with severe complications.

RD: respiratory depression; S: seizure; A: arrhythmia; QTc: corrected QT interval; NS: neurologic sequel; Na: sodium; GCS: Glasgow Coma Score; HP: hyponatremia.
The effect of factors like age, sex, cause of ingestion (accidental, suicide), admission GCS, ECG, QRS duration >100 ms, QTc duration, and presence of hyponatremia on the development of life-threatening complications such as seizure, arrhythmia, or respiratory depression was examined by binary logistic regression analysis. QRS duration >100 ms, QTc interval, and GCS were identified as independent risk factors. The correct classification rate of test was found to be 93.1% (Table 6).
Results of logistic regression analysis to determine predictive markers for developing severe complications.

QTc: corrected QT, GCS: Glasgow Coma Score; OR: odds ratio; CI: confidence interval.
a p = 0.057.
Discussion
In this study, similar to previous studies, evaluation of symptomatic patients associated with amitriptyline overdose was found to be the most common pathological clinical signs such as tachycardia, altered consciousness, and coma, and the most common pathologic laboratory findings were leukocytosis, hyperglycemia, and hyponatremia. 2,4,10 We found that duration between ingestion and first medical intervention was significantly longer in pediatric patients. This may be because a large portion of poisonings are accidental and the recognition of such situation by families can be delayed.
Cases with TCA drugs (including amitriptyline) overdose usually refer with comatose state. The majority of these patients recover well with supportive therapy. Potentially life-threatening complications occur in a small number of cases. Absence of early clinical and laboratory findings in identifying the patients at risk of life-threatening complications such as arrhythmia, seizure, and respiratory depression during the amitriptyline poisoning is a serious problem. Amitriptyline toxicity usually occurs at doses exceeding 10 mg kg−1. 2,11 However, serious signs of toxicity can occur even in low doses. 12 –14 For this reason, estimated amount of drug taken based on anamnesis alone may be misleading. For amitriptyline, serum levels of 50–200 ng mL−1 is considered to be therapeutic, but >1000 ng mL−1 is a toxic concentration. 8,15,16 However, correlation between serum level of drugs and severity of clinical findings is weak and the predictive value of single drug level is low. 16 –18 For this reason, studies have focused especially on ECG findings. 2,7,18 –20 Serum drug levels were analyzed in about half of the pediatric cases. Our results suggest that the serum drug levels may be useful in demonstrating the presence and predicting the severity of poisoning especially in pediatric age group. TCA drugs often change cardiac conduction, contractility, and speed. 8 Cardiotoxicity caused by the blockage of Na flow into cell through the voltage-dependent Na channels is the most important factor associated with mortality. 3,8,14,21 Inhibition of fast Na channels in His-Purkinje cells results in delay of depolarization and conduction abnormalities. 8 The most common ECG finding is sinus tachycardia. 6,10,22,23 Other frequent findings are prolongation of PR, QRS, and QTc intervals, R wave amplitude in aVR ≥ 4 mm or R/S ratio >0.5, and right axis deviation of the terminal 40 ms. 2,4,8,24,25 Local changes in conduction system of the heart may predispose to ventricular dysrhythmias by forming reentrant cycles. 8 This effect is more pronounced in the case of rapid heart rhythm, hyponatremia, and acidosis. 8 A fast flow of Na into the cell is required for the release of intracellular calcium and eventually for the cardiac contractility. 8 A decrease in Na entry into cells decreases the myocardial contractility and leads to hypotension. 8
Many previous studies have investigated the relationship between ECG findings and development of serious complications in TCA overdose. 2,7,18,24 However, the results are conflicting. Some researchers reported that the QRS duration >100 ms is predictive for seizures, ventricular arrhythmias, 15,18 and coma. 2 Foulke and Albertson 26 have reported that any clinical finding or length of QRS is not predictive for complication development and these are not sufficient for risk grouping. Liebelt et al. 20 have found the R wave amplitude ≥4 mm in aVR to be the only predictive ECG finding for the development of seizures and arrhythmias. Similarly, Olgun et al. 2 have reported that none of the patients with R wave amplitude ≥4 mm in aVR has developed seizures (negative predictive value 100%). Despite that, this parameter was not predictive for serious adverse events in other studies. 7,27 QTc duration is usually prolonged in TCA poisoning. 7,28 However, this prolongation is usually associated with enlargement of QRS and it alone is not a predictor. 7 The each of three ECG parameters in our study was found to be associated with the development of life-threatening serious complications and with the severity of poisoning. Compared with other studies, we observed that the QTc duration was correlated with the serum levels of amitriptyline and nortriptyline on admission.
Possible mechanisms for TCA-induced neurotoxicity include γ-aminobutyric acid receptor antagonism, neuronal Na channel blockage, central anticholinergic activity, and the effects of biogenic amines. 8 Almost all patients had various degrees of altered consciousness. However, patients who had not suffered with cardiopulmonary arrest and cerebral hypoxia (including patients who developed status epilepticus) had no permanent neurological impairment. As we did not know the previous serum Na levels of the patients, we could not evaluate whether the changes in serum Na levels or hyponatremia plays a role in the ethiopathogenesis of the seizures. Hyponatremia is the most common electrolyte disorder in hospitalized critically ill patients. 29 –31 This condition, arose from Na deficit or free water excess, is a cause of significant morbidity and mortality. 29,31 Impaired fluid excretion, inappropriate antidiuretic hormone secretion, the use of hypotonic fluids, medications and primary diseases can be the cause of hospital-acquired hyponatremia. 29 In addition, vomiting during acute intoxication or gastric lavage with hypotonic fluids may increase the risk of hyponatremia. One of the interesting results of this study was the close relationship among the severity of poisoning, ECG findings, and presence of hyponatremia. The emergence of the side effects of drug, mainly via the blockage of Na channels, makes this result more meaningful. However, it is not clear whether hyponatremia is a result or reason. To our knowledge, we did not find any study evaluating the relationship between the hyponatremia and the severity and incidence of intoxication in the literature.
Strengths of this study include being one of the largest series related to amitriptyline intoxication in the literature, an exclusion of patients with secondary drug or substance use that can affect prognosis and an evaluation of only symptomatic patients. Also, we think that this study may shed light on the differences between pediatric and adult patients in amitriptyline poisoning. Being a retrospective study and absence of evaluation of relationship between the changes in serum Na and clinical and ECG changes during the follow-up period are major limitations.
In conclusion, although life-threatening complications such as seizures, arrhythmias, and respiratory depression are relatively frequent in pediatric patients than adults with amitriptyline intoxication, the prognosis is generally good in both groups. We found correlation between the serum drug level and complication rate which is contrary to some previous studies. The results of this study suggest that there may be a relationship between the serum Na levels or presence of hyponatremia and the severity of poisoning, ECG findings, and development of complications. Prospective-controlled studies are needed to further investigate this topic.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
