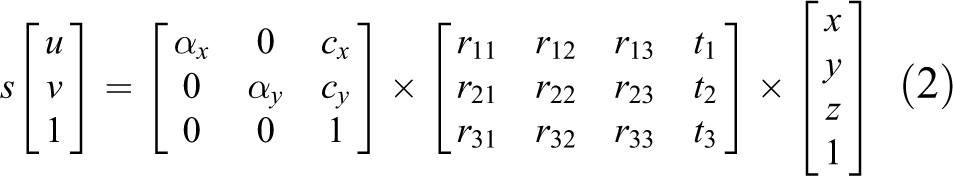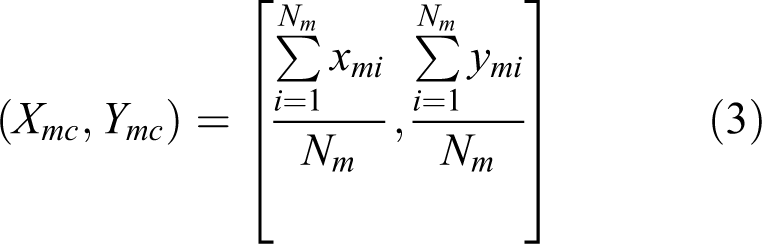Abstract
When lidocaine is locally delivered into the inner ear, it rapidly paralyzes the peripheral vestibular afferent neurons and induces unilateral vestibular loss. The goals of this study were to explore the possibility of developing intratympanic injection (IT) of lidocaine as a modality for treating acute vertigo. To evaluate the minimum concentration required, latent time, action duration, and possibility of lidocaine IT readministration to the vestibular system, we compared the development of horizontal nystagmus after IT of 2, 4, 6, 8, and 10% lidocaine solutions in rats. To identify the induction of vestibular compensation, c-Fos-like protein expression was observed in the vestibular nucleus. Results of our investigation showed that lidocaine IT concentrations greater than 4% induced vestibular hyporeflexia in the injected ear. In order to induce hyporeflexia 4 and 6% lidocaine solutions could also be repeatedly injected. Regardless of concentration, effects of the lidocaine IT dissipated gradually over time. Our findings could be used to develop novel methods for symptom control in vestibular disorder patients.
Introduction
Medications for treating acute vertiginous attacks are classified as either vestibular suppressants or antiemetics. Although their exact mechanisms of action are not known, vestibular suppressants change the distribution of neurotransmitters from vestibular hair cells to the oculomotor nucleus. 1 An ideal medication for vertigo control should correct the vestibular function without causing side effects, be persistently effective and quantitatively regulated, and not interfere with neurological examinations or vestibular compensation. Benzodiazepines are the representative vestibular suppressants that rapidly alleviate vertigo by accelerating γ-aminobutyric acid functioning in the vestibular nucleus and cerebellum. 2 However, these compounds are not recommended for long term treatments because they cause dependency and inhibit vestibular compensation. 1,3 Furthermore, benzodiazepines should not be taken by patients with glaucoma, myasthenia gravis, respiratory disease, or kidney and liver malfunction, or by females who are pregnant or breast-feeding. 4 Other medications currently used to control vertigo include anticholinergic agents, antihistamines, and calcium (Ca2+) channel inhibitors. Anticholinergic agents increase motion tolerance by affecting muscarinic receptors and are used as antinauseants or antidinics rather than treatments for acute vertigo. 5 Antihistamines, which block Ca2+ channels and have anticholinergic effects, are used to treat acute vertigo. 5 The finding that histamine H3 receptors are widely distributed from the afferent vestibular system to the central nervous system led to the use of drugs like betahistine for vertigo treatment. 6,7 Dimenhydrinate, which is frequently used in clinics, is effective within 15–30 min with oral ingestion and the drug gets metabolized afterwards or otherwise the effects last between 3 and 6 h. However, this reagent frequently causes drowsiness and can interfere with the neurologic examinations and the vestibular compensation. It should, therefore, not be used to treat acute cases of vertigo. Overall, there is currently no ideal modality for treating acute vertiginous attacks. 1
Lidocaine, a medication frequently used for inducing local anesthesia, prevents the migration of action potentials from presynaptic neurons to postsynaptic neurons by rapidly inactivating voltage-gated sodium (Na+) channels. 5 Most sensory afferent nerves respond to lidocaine selectively and sensitively. 8,9 Lidocaine may therefore temporarily relieve vestibular symptoms when injected contralaterally into the site of vestibular loss as seen in certain disorders like vestibular neuritis, or when injected ipsilaterally at the site of vestibular hyperexcitation in patients with conditions such as intractable BPPV or Meniere’s disease. 10–13
Intratympnaic injection (IT) is currently used as a common drug delivery method for patients with inner ear diseases. Although the underlying mechanism of drug absorption is unclear and a technique for regulating absorption has not been established, the round window membrane or oval window could serve as a communication channel between the middle and inner ear. 14–16 IT would have the specific advantage of repeatability because the technique is so simple and can be performed repeatedly without adverse effects or decreases in drug efficacy. In order to use lidocaine delivered via IT for the early control of vertigo, the effective lidocaine concentration and safety of repeated IT procedures must first be determined. 17 This study was therefore performed to determine the minimum requirement of lidocaine to make nystamus in rats and observe eye movement in the same animal models following a repeated delivery of lidocaine via IT at those concentrations to evaluate the possibility of developing lidocaine as a treatment for vertigo.
Methods
Animals
For this study, we used 12- to 16-week-old female rats (SD Rats, Koatec, Seoul, Korea) weighing 250–350 g. All experimental animals showed normal Preyer’s reflex, auditory brainstem response (ABR; Tucker-Davis Technologies, Alachua, Florida, USA), and balance function during the tail hanging test. We treated the animals in accordance with the animal use guidelines of the Gachon University Institutional Animal Care and Use Committee (GIACUCR-001), South Korea. The animals were housed with a 12-h light/dark cycle in a 100-lux and dark room kept at 23°C with 40% humidity and a noise level <50 dB.
We divided the 26 rats into 3 groups: administration (n = 18), operation (n = 3), and sham groups (n = 5). The rats in the administration group each received an IT of lidocaine (L7757-25G, Sigma Aldrich, St Louis, Missouri, USA): 4 rats received 2% lidocaine, 4 rats received 4% lidocaine, 3 rats received 6% lidocaine, 3 rats received 8% lidocaine, and 4 rats received 10% lidocaine. The sham group received an IT of saline, and the operation group underwent right side unilateral labyrinthectomy.
Anesthesia
All operations were carried out under anesthesia induced with isoflurane (Aerane, Ilsung Pharm. Co., Seoul, South Korea). Electrocardiography (THM 100-377, Indus Instrument, Houston, Texus, USA) was performed and the pulse, temperature, and oxygen saturation of the anesthetized animals were monitored to maintain a consistent physiological status.
Screw holder for head fixation
After the anesthetized animals were set in a prone position on a stereotaxic frame (Model 900, David Kope instruments, Tujunga, California, USA), a 2-cm vertical incision was made between the eyes. The muscle, connective tissue, and periosteum on the dorsal side of both frontal bones and the middle of the parietal bone were removed by electrocautery (022-100-18, Tontarra, Wurmingen, Germany). We then screwed in 6 flat-headed tap screws (5/32′′ long, #1106, 16-AT-004 flat-headed tap screws; SM Medical, Seoul, South Korea) that were used to hold additional flat-headed tap screws (30 mm tab screw, Junghwa Metal Co., Hanam, Korea) by the head. Finally, we applied instant glue (Loctite 401 20 g, Henkel, Dublin, Ireland) and dental cement (vertex self-curing; vertex-dental B.V., Zeist, Netherlands) over the screws for permanent fixation. After the procedure, the animals were allowed to recover in their normal environment for 1 week to stabilize the operation site (Figure 1(a)). Animal care after all surgical intervention was carried out by enrofloaxcin (Baytril®, 5 mg kg−1, intramuscular (IM), daily, Bayer Pharma AG, Wuppertal, Germany) with ketoprofen (2.5 mg kg−1 IM, daily, Guju pharm. Co., Seoul, Korea). All experimental animals recovered after anesthesia and the related surgical intervention without any loss.

(a) Screw implantation and animal setting for eye movement tracking. The flat-headed tap screw was fixed on the head by instant glue and dental cement over the screws for permanent fixation; (b) a bright 200-μm diameter dot was used as a marker for tracking of eye movement. It was attached at right pupil of eyeball by glue, and then upper and lower eyelids were stitched to prevent blinking. The body of rat was put into the restrainer and the previously implanted screw on the head was inserted into the holder of the sinusoidal animal rotator.
Marker production and application
A bright, 200 μm diameter dot created with Adobe Illustrator CS4, v14.0.0 (Adobe Systems Inc., Los Angeles, California, USA) was used as a marker for tracking the eye movement. After inducing topical anesthesia in the eyes of the rats with proparacaine hydrochloride (alkaine 0.5% eye drops, S.A. Alcon-Couvreur N.V., Puurs, Belgium), we applied glue (Gel-10, Daejin Chemical Co., Siheung-si, South Korea) and positioned the marker on the center of pupil under a microscope (GL-99B-V7, Daemyung Optical, Seoul, Korea). We then stitched the upper and lower eyelids open with everting sutures (D-7308M2, Deknatel, Athlone, Westmeath, Ireland) to prevent blinking. Finally, we applied greasy ointment (terramycin ophthalmic oinment, Pfizer Korea, Seoul, Korea) on the other eyeball to block visual fixation (Figure 1(b)).
Labyrinthectomy
While the rat was in a prone position, a 2-cm vertical incision along the posterior border of the auricle was made. When the tympanic membrane was fully exposed, we removed the tympanic membrane and ossicle. After identifying the stapes and pterygopalatine artery in the middle ear, we made a small hole superior to the facial canal on the vestibular lateral wall with a 23-G needle (23G syringe needle, Shingchang Medical Co., Gumi, Korea). We expanded the perforation to completely open the lateral vestibular wall and sucked it out until the perilymph stopped leaking. To maintain the exposure of the vestibular wall, we filled the inner cavity of the vestibular system with collagen (Helitene; Intergra Life Sciences Co., Plainsboro, New Jersey, USA) and sutured the muscle and skin (Deklene® II, Deknatel). Unilateral labyrinthectomy completeness was verified by a tail-hanging test and the appearances of spontaneous nystagmus, skew, and head tilting. Furthermore, ABR were double-checked for the constant unilateral labyrinthectomy by the ABR workstation objectively.
IT technique
Initially, a 3-mm vertical incision on the inferior border of the right auricle was made under a surgical microscope (GL-99B-V7, Daemyung Optical) to expose the whole tympanic membrane. Next, 0.1 ml of lidocaine (L7757; Sigma-Aldrich) or temperature-controlled saline to eliminate the caloric effect was injected in an anterosuperior direction using a 31-G needle (31G syringe needle, Shingchang Medical Co.) and a 0.5-ml syringe (sterile insulin syringe 0.5 ml, Artsana SPA, Grandate, Italy; Figure 2).
Schematic view of intratympanic injection at right side ear. After tympanic membrane exposure completely under surgical microscope, 0.1 ml of lidocaine or temperature-controlled saline was injected in an anterosuperior direction using a 31-G needle and a 0.5 ml syringe. Black arrow: oval window; open arrow: round window; arrowhead: intratympanic injection site.
Head fixation
Each awakened rat was fixed to the Broome style restrainer (S44-RR, Plas Labs Inc., Lensing, Missouri, USA) in a prone position and the screw on the top of the skull was inserted into the holder of the sinusoidal animal rotator (M-YS4080FN001, NSK, Ukiha-shi, Fukuoka, Japan) for a complete head fixation immediately after lidocaine IT. We then fixed the restrainer to the rotator stage. We used a microcontrol XYZ linear translation stage installed on the head holder device and restrainer to move the rat vertically and laterally and bring the camera (GRAS-03K2M-C, Point Grey Research Inc., Richmond, Canada) into focus.
Measuring eye movement
We repeatedly measured the frequency and amplitude of nystagmus for 20 s at 1-min intervals after the first episode of nystagmus. When no nystagmus was detected, we observed the animal for up to 45 min to confirm that there was no further response.
All incidences of nystagmus were recorded at 120 fps (GRAS-03K2M-C, Point Grey Research Inc.) in a dark room under 10 lux using a telecentric lens (TCL1.0x-40-ST, SPO Inc., Daejeon, South Korea) with a Y2 filter and 6 extension tubes (ML-EXR5; Point Grey Research Inc.). The video was saved as 640 × 480 audio video interleave files using a 1394b cable (HAR-1394-1721, Point Grey Research Inc.) and an image-capturing board (HP NK653AA; Hewett-Packard Development Company, Palo Alto, California, USA) with Flycapture 2.0 for 64-bit (Point Grey Research, Inc.) on an IBM-compatible computer (I7, Intel, Santa Clara, California, USA). We attached a 385-nm light bulb (VAOL-3EUV8Y4; VCC Optoelectronics, San Marcos, California, USA) to the front line of the camera lens to create a fluorescent reflection from the marker attached to the eye. We used a 3.3 V battery (3.3v LiFePO4 Rechargeble Battry, Shenzhen Grenergy Tech, Guandong, China) to maintain a consistent light intensity. The calibration of the camera was carried out with 6.3 mm diameter beads (Gold tiger eye, beadbiz, Ulsan, Korea) placed in the middle of the microrotation stage and moving them vertically and laterally from 5 to 30°. We acquired calibration values of the pixel ratio to assign to the analysis program. We considered each of the theoretical spots (X, Y, and Z) to represent the actual spatial coordinates. We used matrix A, the camera matrix, to calibrate information related to the camera alone. We calibrated the focal distance using matrix A. We then calibrated the rotations and the movements using [R|t], which accounted for the effects of factors not associated with the camera itself. 18 Thus we minimized the distortion caused by projecting three-dimensional (3D) information into two-dimensions by obtaining the matrix information through calibration prior to the data analysis (equations (1) and (2)).
In this case, s is an arbitrary scale factor, m′ is a two-dimentional point projected on the camera’s image plane (u, v), A is the intrinsic parameter matrix for the camera, R is the rotation matrix applied to coincide the principal axis with Z-axis, t is the camera translation to origin of the XYZ-coordinate, and M is a 3D point in the real world (X, Y, Z).
After the calibration, we extracted information about the location of the eye from the images (equation (3)). The fluorescent reflection made it easy to accurately locate the marker. After that, we divided the image of the marker into four sections. Since the point at the center of the four sections could be considered the center of the marker, we estimated the location of center of the marker by calculating the mean of the x- and y-coordinates in terms of pixels.
We used LabVIEW (National Instruments, Austin, Texas, USA) to simultaneously monitor horizontal, vertical, and torsional nystagmus. 19 However, we only analyzed the horizontal nystagmus in our experiment because this condition was the most pronounced and we used animals with lateral eyes for the study. The x-axis represented the time, and the y-axis represented the distance (°) that the eyeball moved. The average slow-phase velocity (SPV, ° s−1), and the frequency of nystagmus (beats min−1) were used as the parameters for the analysis (Figure 3).

Nystagmus after 4% lidocaine intratympanic injection. The frequency and amplitude of nystagmus were repeatedly measured for 20 s at 1 min intervals after the 1st episode of nystagmus. The amplitude of nystagmus (° s−1) was plotted in open diamonds and the frequency (beats min−1) was plotted in black dots. The amplitude and frequency of nystagmus was decreased over time. ASPV: average slow-phase velocity.
Immunohistochemical staining
To investigate the effect of vestibular compensation after the lidocaine-induced temporal vestibulopathy, we stained for c-Fos-like protein in the medial vestibular nuclueus using coronal sections. 20 We killed the experimental animals with an overdose of urethane (Sigma-Aldrich) after lidocaine IT 2, 6, and 12 h and perfused transcardially with 250 ml of 0.1 M phosphate buffer (PB) after lidocaine IT. The rats were then perfused with 500 ml 4% paraformaldehyde dissolved in 0.1 M PB (pH 7.4). We removed the brains from the skulls and postfixed them with the same solution used for initial fixation for 4 h at room temperature. We immersed the fixed brains in a 30% sucrose/phosphate-buffered solution (PBS) for 2 days at 4°C for cryoprotection. We then cut sections of 35 μm thick with a freezing microtome (Leica Microsystems, Wetzlar, Germany), incubated them for 20 min at room temperature (RT) with 3% hydrogen peroxide (H2O2), rinsed them 3 times for 5 min with 10 mM PBS, incubated them for 20 min with 0.5% Triton X-100 dissolved in 10 mM PBS and incubated them for 1 h at RT with 5% skim milk. We then washed the sections in 10 mM PBS plus 0.05% Triton X-100 (PBST) and incubated them overnight at 4°C with rabbit polyclonal anti-cFos antibody (1:3,000; Oncogene Research Products, San Diego, California, USA) in an antibody diluent solution (Invitrogen, Carlsbad, California, USA). On the following day, we rinsed the tissue sections with PBST, incubated them with a biotinylated goat anti-rabbit secondary antibody (1:1,000; DAKO, Glostrup, Denmark) for 1 h at RT and incubated them for 1 h at RT with an avidin–biotin complex (Vector Laboratories, Burlingame, California, USA). We visualized the neurons that were positive for c-Fos-like protein by incubating the sections with 0.05% diaminobenzidine (DAB; Invitrogen) plus 0.003% H2O2. After the DAB reaction was completed, we rinsed the sections with 0.1 M PB, and mounted them on gelatin-coated slides. The slides were then air-dried, dehydrated, xylene cleared, and mounted on coverslips with permount (Fisher Chemical, Fairlawn, New Jersey, USA). We then examined the brain tissue sections using bright field microscopy (Olympus BX50; Olympus, Tokyo, Japan) to identify c-Fos-like protein positive neurons. We captured and digitalized images of the dark brown, c-Fos-like protein positive neurons using a microscope-mounted digital camera (Olympus DP70, Olympus) and image-capture software (Image-Pro Plus, MediaCybernetics, Inc., Rockville, Maryland, USA).
Data analysis
For statistical analysis, we created a box-and-whisker plot to visualize the data and performed Levene’s test for the equality of variances between each comparing group. We performed a two-sample t-test at the 95% confidence level to evaluate differences in mean values between the groups, and we analyzed the changes in nystagmus over time by calculating Pearson’s correlation coefficients using Statistical Package for the Social Sciences (SPSS) v17.0 (SPSS Inc., Armonk, New York, USA).
Results
Identification of the minimum lidocaine concentration that causes nystagmus
We did not detect nystagmus in the sham group or in the group treated with 2% lidocaine. The operation group showed a head tilt to the ipsilateral side, skew deviation in both eyes, and a vigorous spontaneous nystagmus toward the healthy side. The mean frequency and SPV of nystagmus in the operation group were 48.31 ± 4.13 beats min−1 and 46.28 ± 3.35 ° s−1, respectively.
Even though it did not show any abnormal head and body posture clearly, similarly to the operation group, nystagmus toward the side contralateral to the IT site was observed in all study animals that received injections of lidocaine at concentrations of 4% or more. The initial frequency and SPV of nystagmus tended to decrease over time (Figure 3, Tables 1 and 2). The directions of the induced nystagmus did not change during the experiment.
Pearson’s correlation between the frequency of nystagmus and time after the first episode of nystagmus.

a r2 > 0.36 and p < 0.01
Pearson’s correlation between the slow phase velocity of nystagmus and time after the first episode of nystagmus.

a r2 > 0.36 and p < 0.01.
Characteristics of nystagmus caused by the lidocaine IT
The SPVs of nystagmus measured in the rats that received ITs of 4, 6, 8, and 10% lidocaine were 24.65 ± 9.69, 19.53 ± 10.65, 20.80 ± 8.27, and 15.41 ± 7.51 ° s−1, respectively (Figure 4). The severity of the toxic effects caused by the lidocaine was significantly concentration dependent. The SPV of nystagmus was significantly lower in the rats treated with 6,8, and 10% lidocaine compared with animals that received 4% lidocaine (p < 0.001). There was no significant difference in nystagmus SPV between the 6 and 8% lidocaine groups, but the SPV of nystagmus for the 10% lidocaine group was significantly lower than that of the 6 and 8% lidocaine groups (p < 0.0001).

The SPV of nystagmus after 4, 6, 8, and 10% lidocaine intratympanic injection. The toxic effect of lidocaine was significantly concentration-dependent. Differences in SPV between rats treated with 6% or 8% were not significant, but the SPVs of animals injected with 4 or 10% lidocaine were significantly different according to a two-sample t-test (*p < 0.001, **p < 0.0001). The nystagmus after 2% lidocaine IT was not detected. SPV: slow phase velocity; IT: intratympanic injection.
The rats that received ITs of 4, 6, 8, and 10% lidocaine showed nystagmus at frequencies of 18.32 ± 5.20, 17.65 ± 6.53, 16.80 ± 5.55, and 11.79 ± 4.70 beats min−1, respectively. There was no significant difference in the nystagmus frequency among the 4, 6, and 8% lidocaine groups (Figure 5). However, this frequency was significantly lower in the 10% lidocaine group (p < 0.0001).

The frequency of nystagmus after 4, 6, 8, and 10% lidocaine IT. The frequency of nystagmus was significantly decreased in animals treated with 10% lidocaine compared to the other experimental groups. Otherwise, the differences in beat frequency between rats treated with 4, 6, and 8% lidocaine were not significant in a two-sample t-test (*p < 0.05). The nystagmus after 2% lidocaine IT was not detected. IT: intratympanic injection.
Latency and duration of nystagmus
The latency of nystagmus was irregular and varied from 10 to 16 min (12.3 ± 2.2 min), but was not dependent on the concentration of lidocaine. The latency of nystagmus was significantly prolonged among animals in which nystagmus reappeared after repeated ITs of lidocaine at the same concentration (26.3 ± 3.4 min, p < 0.0001). For the groups that received ITs of 4, 6, 8, and 10% lidocaine, the duration of nystagmus was 25.5, 15.7, 26.3, and 23.0 min, respectively. The mean duration of nystagmus caused by the 4% and 6% lidocaine group was 22.7 min and 11.1 min respectively.
Nystagmus induction by repeated injections of lidocaine
In the 4% lidocaine group, 3 out of 4 animals developed nystagmus when lidocaine injection was repeated 4 h after the first injection. The remaining animal developed nystagmus when the injection was repeated 8 h after the first injection. In the 6% lidocaine group, 1 out of 3 animals developed nystagmus after lidocaine injection was repeated 4 h after the first injection. In the 8 and 10% lidocaine groups, nystagmus did not reappear after lidocaine injections were repeated 4 and 8 h after the first injection.
In the 4% lidocaine group, there was no difference in the frequency of nystagmus after the first injection and after the injections administered 4 and 8 h later. Mean frequencies after the first, second, and third injections were 18.19 ± 4.01, 18.19 ± 5.15, and 17.25 ± 5.28 beats min−1, respectively (Figure 6). The mean SPV of nystagmus after lidocaine injection was significantly different between the first injection and injections given 4 and 8 h later (p < 0.0001 for both comparisons). The mean nystagmus SPV after first, second, and third lidocaine injection was 15.93 ± 6.10, 29.70 ± 10.29 ° s−1, and 17.79 ± 7.80 ° s−1, respectively (Figure 7).

The frequency of nystagmus after repetitive intratympanic injections of 4% lidocaine delivered 4 and 8 h after the initial injection. There was no difference in the frequency of nystagmus after the initial injection and after the repetitive injections delivered 4 and 8 h later.
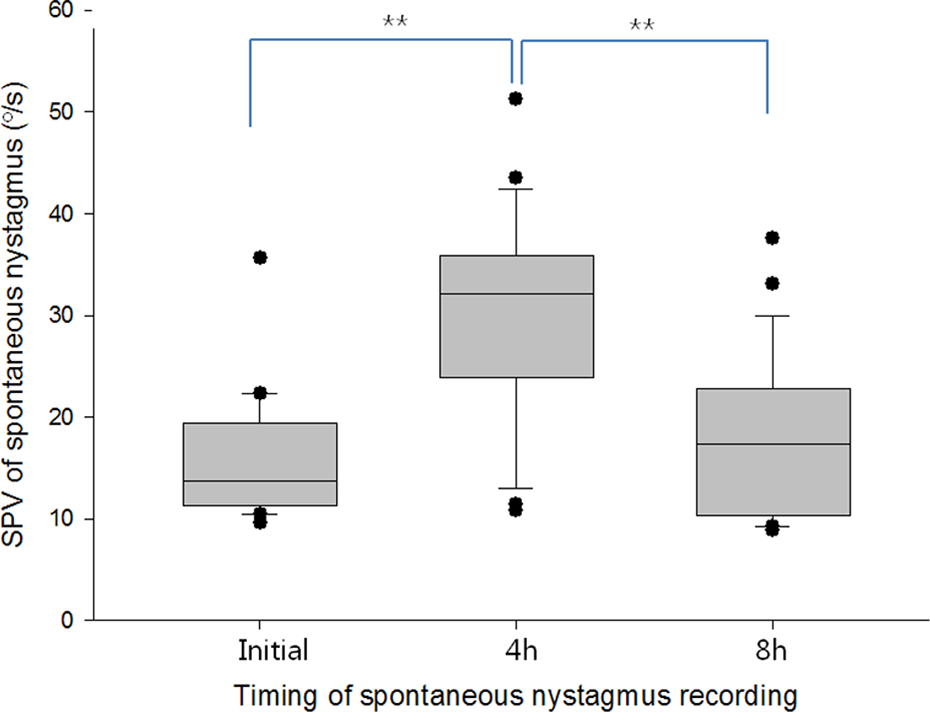
The SPV of nystagmus after the repetitive intratympanic injection of 4% lidocaine 4 and 8 h after the initial injection. The average SPV of nystagmus after lidocaine injection was significantly different between the initial injection and the repetitive injections administered 4 and 8 h later in a two-sample t-test (**p < 0.0001). SPV: slow phase velocity.
In the 6% lidocaine group (Figure 8), the mean frequency of nystagmus was significantly lower after the second injection (6.36 ± 3.33 beats min−1) than after the first injection (11.25 ± 5.48 beats min−1; p = 0.01844). Nystagmus SPV for the 6% lidocaine group decreased significantly from the 1st injection to the 2nd (p = 0.022). Mean SPVs for the 6% lidocaine group after the 1st and 2nd injections were 12.37 ± 3.82 and 6.05 ± 2.18 ° s−1, respectively (Figure 9).

The frequency of nystagmus associated with the repetitive intratympanic injection of 6% lidocaine delivered 4 h after the initial injection. The beat frequency significantly decreased with the intratympanic injection 4 h later in a two-sample t-test (*p < 0.001). The nystagmus associated with the repetitive intratympanic injection of 6% lidocaine 8 h after the initial injection was not detected.

Changes in SPV of nystagmus after repetitive ITof 6% lidocaine delivered 4 h after the initial injection. SPV was significantly decreased with repetitive IT administered 4 h after the initial injection in a two-sample t-test (*p < 0.001), but it was not significant. The nystagmus associated with repetitive IT of 6% lidocaine delivered 8 h after the initial injection was not detected. IT: intratympanic injection; SPV: slow phase velocity.
Changes in nystagmus frequency and SPV according to the duration of nystagmus
Tables 1 and 2 show the regression analyses results for the nystagmus frequency and SPV over time. No clear trend for nystagmus SPV over time was observed. Only 2 animals with r 2 values >0.36 showed the decreased SPV. The frequency of nystagmus decreased among approximately half of the animals (8 out of 14) for which the r 2 values were >0.36. Additionally, these eight animals had significant r 2 values showing a downward slope over time.
c-Fos-like protein expression induced by lidocaine ototoxicity
The IT delivery of 4% lidocaine did not induce c-Fos-like protein expression bilaterally in the medial vestibular nuclei on 2, 6, and 12 h after lidocaine injection compared to the sham group (Figure 10).

Immunohistochemical staining of FLP expression in bilateral MVN in coronal midbrain sections (a) after saline only IT, (b), (c), and (d) on 2, 6, and 12 h after right side ULX respectively, and (e), (f), and (g) after 4% lidocaine intratympanic injection delivered 2, 6, and 12 h, respectively after the initial injection. Following lesion, animals were anesthetized, perfused, and fixed with 4% paraformaldehyde. Brains were removed, immersed in 30% sucrose, sectioned with a thickness of 40 μm, and incubated with cFos antibody. After subsequent reactions, FLP expression was visualized under bright microscope. The induction of FLP expression in the bilateral MVN caused by ULX was not detected in saline IT and 4% lidocaine IT groups on 2, 6, and 12 h. IPSI: ipsilateral to ULX or IT; CONTRA: contralateral to ULX or IT. Scale bar, 400 μm. FLP: cFos-like protein; MVN: medial vestibular nuclei; ULX: unilateral labyrinthectomy; IT: intratympanic injection.
Discussion
The results of our investigation showed that lidocaine IT concentrations greater than 4% induced vestibular hyporeflexia in the injected ear and that 4 and 6% lidocaine solutions could also be repeatedly injected to make hyporeflexia. Regardless of concentration, the effects of the lidocaine IT dissipated gradually over time.
Since 1978, lidocaine has been evaluated as a treatment for vertigo and motion sickness. 17 Temporary hyporeflexia by lidocaine IT on vestibular afferent nerve was reported in animals 8,11,12,21 and humans. 17 Even for lidocaine delivered through intravenous (IV), the action time on vestibular system was less than 4 h and was completely recovered until 6 h. 8 Even unilateral labyrinthectomized cats were able to ambulate freely without falling within 4 h of a 4-mg kg−1 IV lidocaine injection. 9 In addition to providing inconsistent results, IV lidocaine injection also alters the cerebellar inhibitory signal by causing an abnormal regulation of Deiters’ neuron. 21–23 This would in return affect vestibular compensation. Another drawback of IV lidocaine is that it would necessitate a relatively high dosage or concentration of lidocaine to achieve the desired effects. Due to the rapid metabolism of lidocaine by hepatic blood flow rather than hepatic enzymes, the action time of lidocaine delivered via IV is less than 2 h and the plasma concentration is not stably maintained. Therefore, a high dose of the drug is invariably required. 23 High doses of IV lidocaine may lead to adverse effects such as feelings of dissociation, paresthesia, mild drowsiness, agitation, multiple twitching, convulsions, disorientation, and decreased hearing at plasma levels of 5 mg 1−1 24,25 and above or with the dose higher than 1%, 7.6 mg kg−1. 26 Another one of the most overlooked potential problems is that when lidocaine is absorbed into a fat layer, the high lipid solubility of the drug delays its release into the systemic circulation. The lipid solubility of lidocaine further increases with alkalosis because lidocaine is a weak base. Alkalosis thus increases the diffusion of lidocaine across the neural membranes. 10 This occurrence makes it difficult to maintain the appropriate concentration of lidocaine in the body.
Despite these unresolved problems, lidocaine is used in the clinics to treat tinnitus 22 and control vertigo associated with Meniere’s disease. 13,27 However, the dose or concentration of lidocaine IT is empirical. The possible side effects of lidocaine IT on hearing are unclear. However, in a previous human study, the auditory threshold decreased after lidocaine IT (40 mg in 1 ml of saline) by 2–10 dB at transient-evoked otoacoustic emission frequencies of 1–3 kHz. This reduction in hearing ability reached a maximum 30 min after IT. 17 In other words, the cochlear effect preceded the vestibular effect. To quantify the minimum concentration of lidocaine required for vestibular function modification, the mechanism of action and target of lidocaine along with the effectiveness of repeated administration and minimum concentration of lidocaine IT that does not affect to vestibular compensation must therefore be studied in advance.
It is unclear which organ in the vestibular system is targeted by lidocaine. However, the vestibular evoked potential (VsEP) study showed that lidocaine injected into the middle ear cavity would affect mechanoelectrical transduction in the vestibular end organs and afferent vestibular nerve. 28,29 In our study, the lidocaine-induced nystagmus was directed away from the side of the IT, which was also observed in the operation group, and the direction of nystagmus never changed throughout the experiment. Additionally, the transient vestibular effect of lidocaine IT at the minimum concentration was not linked to c-Fos-like protein expression until 12 h after the drug administration. Thus we assume that the target of action would be the peripheral end organ of vestibular system alone.
The precise underlying mechanism of lidocaine IT is also unclear. In a previous investigation, 30 a scanning electron microscopy revealed adhesion and fusion among the outer hairs of the organ of Corti as well as the sensory hairs of the crista ampullaris and otolith organ after transtympanic infusion of 10% lidocane. It implies an irreversible change of inner ear at higher concentration of lidocaine. However, in our experiement, the lidocaine concentrations of 10 and 4% caused significantly different frequencies and SPVs of nystagmus. Therefore, we still do not know the minimal concentration or amount of lidocaine to make hyporeflexia of vestibular system. In our study, we found that nystagmus developed with lidocaine concentrations of 4% or higher. Although the total volume of lidocaine that can be injected into the tympanic cavity is 0.1 ml, the total amount of lidocaine delivered cannot be determined because the mechanism of lidocaine delivery into the perilymphatic space is unclear and the drug solubility is affected by the pH of peri- and endolymphatic fluid. Another limiting factor is the contact time between the round window membrane and the injected lidocaine. The lidocaine injected could easily drain out through the eustachian tube. Additionally, the diameter and shape of the eustachian tube in rodents is quite different from that in humans. Thus the contact time or absorption ratio of lidocaine delivered via IT could vary greatly. Additional studies are needed to achieve the intended vestibular toxicity with the minimal amount of drug.
Transcriptional factors such c-Fos, FosB, Jun, and Krox are expressed in the central nervous system from several minutes to hours after excitatory or inhibitory vestibular stimulator. 31,32 Eventually c-Fos like protein as a kind of immediate early gene protein is commonly used as a marker of compensation including cellular metabolism in the vestibular nucleus. 33 In this present study, the decreased frequency and SPV of nystamgus observed with higher lidocaine concentrations were not considered to be a vestibular compensation effect because c-Fos-like protein was not expressed 2, 6, and 12 h after IT of the minimal concentration of lidocaine. The reason for such decreases is not clear but we assumed that the nonlinear internal property of the lidocaine concentration is a factor.
Past studies concerning with lidocaine IT did not use an accurate measurement method to estimate the vestibular function: they only counted the frequency of nystagmus. 11,12 However, we measured both frequency and SPV of nystagmus to obtain the best possible objective measurements of vestibular function. Although we tried to exclude the influence of anesthesia while evaluating nystagmus, there was still a possibility of measurement error. We minimized the error of the overall estimate associated with errors for the SPV measurements because the frequency of fine nystagmus is relatively easy to determine. Moreover, in a clinical setting, the frequency and intensity of nystagmus are the indicators of the degree of dizziness. On the other hand, the SPV of nystagmus reflects the pure tonic imbalance of vestibular nucleus, which is vulnerable to the anesthesia or awakeness. Consequently, expressing nystagmus frequency and SPV as separate parameters might be complementary relation. Eventually, the frequency of nystagmus depends on the SPV of nystagmus, recoiling power-related muscular fatigability, and corrective saccadic activity. 34 Attention level and degree of cerebellum inhibition may also play important roles in determining nystagmus frequency. 35,36 Our results also showed some differences between the frequency and the SPV of nystagmus in comparison of concentration and repeatitive IT results. The authors suspected it to be caused by the fine amplitude of nystamgmus and remaining anesthetic effect on vestibular nucleus. Therefore, the authors inferred the results that it had an unique consistency between the frequency and the SPV of nystagmus. For example, meaningful nystagmus was promoted by lidocaine ITs at concentrations of 4% and higher in both the frequency and SPV analyses (p < 0.0001) and the frequency and SPV of nystagmus was significantly lower after the 2nd injection than after the first injection in the 6% lidocaine group (p < 0.05).
The development of nystagmus after lidocaine IT was latent. It was irregular and varied from 10 to 16 min (12.3 ± 2.2 min), but was not dependent on the concentration of lidocaine. Nystagmus latency was significantly prolonged; however, only among the animals in which nystagmus reappeared after repeated ITs of lidocaine at the same concentration (26.3 ± 3.4 min, p < 0.0001). This could be due to the ability of lidocaine to block Na+ channels that leads to the inhibition of peripheral nerve conduction.
The effect of repetitive drug administration is very important to develop a modality for treating vertigo because the symptoms can last over 24 h or easily reappear as seen in the cases of benign paroxysmal positional vertigo. In previous rat studies, 8,12 the nystagmus frequency saturated on a significantly lower level than during the first blockade when the transient vestibular blockade was repeated by a second infusion during the following day. Also, serial single infusions, with recovery between each functional vestibular loss, gave rise to a less severe nystagmus. These results were completely same to our results of single repeated injection. Although we could not understand this phenomenon sufficiently, the authors speculated that the rapid decline (6% lidocaine) or inconsistent (4% lidocaine) resulting in the frequency and SPV of reappearanced nystagmus may reflect the degree of end organ damage caused by the initial injection. 12 The increased intensity of nystagmus in this study of the pure vestibuar tonic imbalance can be explained by increased neural damage caused by the accumulation of low lidocaine concentrations. Therefore, the repeated injections of lidocaine at low concentrations over a short period of time must be reconsidered. Furthermore, the frequency response to repeated injections might be affected to a great extent by environmental factors such as ocular–muscular fatigue or central modulation.
In the groups that received ITs of 4, 6, 8, and 10% lidocaine, the duration of nystagmus was 25.5, 15.7, 26.3, and 23.0 min respectively. The mean durations of the nystagmus caused by the 4% and 6% lidocaine group were 22.7 min and 11.1 min, respectively. The frequency and the SPV of nystagmus significantly decreased over time if the p-value was less than 0.01 and r 2 was over 0.36. Overall, the effects of lidocaine IT were temporary regardless of concentration and disappeared within 30 min at the experimental concentration.
Conclusion
In the current study, lidocaine IT at concentrations greater than 4% caused nystagmus. Repetitive injection was feasible at 4 and 6% concentrations of lidocaine IT. The effects of lidocaine IT were temporary regardless of drug concentration and gradually disappeared over time at the experimental concentrations. Our findings could help to develop a method of symptom control for patients with vestibular disorders.
Footnotes
Authors’ Note
The authors HHL and MJK contributed equally to this work.
Conflict of interest
The authors declared no competing financial interests. The authors alone are responsible for the content and writing of the paper.
Funding
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2010-0011858).