Abstract
Objective:
Oxidative stress plays an important role in doxorubicin (DOX)-induced toxicity. Carnosine (CAR) is a dipeptide with antioxidant properties. The aim of this study was to evaluate the decreasing or preventive effect of CAR alone or combination with vitamin E (CAR + Vit E) on DOX-induced toxicity in heart, liver, and brain of rats.
Methods:
Rats were treated with CAR (250 mg kg−1 day−1; intraperitoneally (i.p.)) or CAR + Vit E (equals 200 mg kg−1 α-tocopherol; once every 3 days; intramuscularly) for 12 consecutive days. On the 8th day of treatment, rats were injected with a single dose of DOX (30 mg kg−1, i.p.). Serum cardiac troponin I (cTnI), urea, and creatinine levels; alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities; and oxidative stress parameters in tissues were measured. We also determined thiobarbituric acid reactive substances, diene conjugate, protein carbonyl (PC), and glutathione levels and antioxidant enzyme activities.
Results:
DOX resulted in increased serum cTnI, ALT, AST, urea, and creatinine levels and increased lipid peroxide and PC levels in tissues. CAR or CAR + Vit E treatments led to decreases in serum cTnI levels and ALT and AST activities. These treatments reduced prooxidant status and ameloriated histopathologic findings in the examined tissues.
Conclusion:
Our results may indicate that CAR alone, especially in combination with Vit E, protect against DOX-induced toxicity in heart, liver, and kidney tissues of rats. This was evidenced by improved cardiac, hepatic, and renal markers and restoration of the prooxidant state and amelioration of histopathologic changes.
Introduction
Doxorubicin (DOX) is a very effective anticancer antibiotic used in the treatment of several human neoplasms; however, its use in clinical chemotherapy is limited due to its toxic effects on several organs including the heart, liver, and kidney and its hematologic and testicular toxicity. Cardiotoxicity is a major limiting factor for the clinical use of DOX and can vary from transient electrocardiographic abnormalities to cardiomyopathy and heart failure in a dose-dependent manner. 1,2 It has been reported that DOX also causes hepatocyte injury 3,4 and increased glomerular permeability and glomerular atrophy. 4,5
The mechanisms responsible for DOX-induced toxicity are not clearly known. Several explanations have been proposed to account for DOX toxicity such as free radical formation, calcium overloading, and mitochondrial dysfunction. 1 –6 However, oxidative stress is believed to be a major factor in DOX toxicity. 1 –6 Two different ways have been described for DOX-induced free radical formation. The first implicates the formation of a semiquinone free radical by the action of nicotinamide adenine dinucleotide phosphate (NADPH)-dependent reductases. In the presence of oxygen, redox cycling of DOX-derived quinine–semiguinone yields superoxide radicals. In the second, the iron-DOX complex reduces oxygen to hydrogen peroxide and other reactive oxygen species (ROS). 3,5,6 ROS can damage lipids and proteins, thereby eventually progressing to cell death and organ injury. Several investigators have reported increased production of ROS and decreased antioxidant parameters in cardiac tissue of DOX-treated animals. 2,6 –11 Significant increases in oxidative stress parameters together with tissue damage were also observed in the liver 3,4,6 and kidney 4,5,8,12 tissues of DOX-treated animals. The role of ROS in DOX-induced toxicity is also supported by the findings that several antioxidants protect organs against the toxicity of DOX. Therefore, the protective potential of several antioxidant compounds have been investigated in DOX-induced organ toxicity. 2 –12
Carnosine (CAR; β-alanyl-l-histidine) is an endogenous dipeptide that possesses various functions such as membrane stabilizing, pH buffering, metal ion chelating, antioxidant, hydroxyl radical and carbonyl scavenging, and antiglycating. These functions enable CAR to be used as an efficient antioxidant and cell protector. 13 –16 CAR exerts a protective potential against tissue damage in oxidative stress-induced pathologies and is accepted as a cardioprotective agent. 14 –17 It protected cardiomyocytes from damage and improved contractility of heart in cardiac ischemia 18,19 and DOX-induced cardiotoxicity. 20,21 We recently reported that CAR treatment diminished isoproterenol-induced myocardial damage by reducing oxidative stress in rats. 22 CAR also decreased hepatic steatosis 23,24 and necrosis. 25,26 Some investigators have suggested that CAR has renoprotective effects in ischemia/reperfusion, 27 and nondiabetic and diabetic nephropathies. 28,29 On the other hand, when CAR is used together with lipophilic agents such as vitamin E (Vit E), 30,31 melatonin, 32 or α-lipoic acid, 33 its antioxidant actions are found to increase.
In the current study, we wanted to investigate whether DOX-induced oxidative stress and tissue damage in heart, liver, and kidney of rats could be prevented by treatment with CAR alone or combination with Vit E (CAR + Vit E). Therefore, we determined serum markers of cardiac, hepatic, and renal damage; histopathologic observations as well as thiobarbituric acid reactive substances (TBARS), diene conjugate (DC), protein carbonyl (PC), and glutathione (GSH) levels; and copper (Cu)–zinc superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activities in the examined organs.
Materials and methods
Chemicals
DOX (50 mg/25 mL injectable form) was obtained from Fresenius Kabi Oncology Ltd (Gurgaon, Haryana, India). Vit E was obtained from Aksu Company (Istanbul, Turkey) as Evigen ampoules containing 300 mg α-tocopherol acetate. CAR and other chemicals were supplied from Sigma-Aldrich (St Louis, Missouri, USA).
Animals and treatments
Sprague Dawley male rats that weighed 200–220 g were used in this study. They were obtained from the Institute for Experimental Medicine, Istanbul University, Turkey. The experimental procedure used in this study met the guidelines of the Animal Care and Use Committee of the University of Istanbul (no.: 173/2010). The animals were housed in a room at a mean constant temperature of 22 ± 2°C with a 12-h light/12-h dark cycle. They were allowed free access to food and water and were kept in wire-bottomed stainless steel cages.
Rats were mainly divided into four groups: (a) control group, rats were given 0.9% sodium chloride (NaCl) as vehicle; (b) DOX group, rats were injected with a single dose of DOX dissolved in normal saline (30 mg kg−1, intraperitoneally (i.p.)) and killed 4 days after the injection (this dose and examination period were in accordance with previous studies 6,8,10,34 ; (c) CAR + DOX group, rats were treated with CAR (250 mg kg−1 day−1, i.p.) for 8 consecutive days. Following a single dose of DOX on the 8th day of the pretreatment period, CAR was also administered for the next 4 days; And (d) CAR + Vit E + DOX group, rats were treated with CAR (250 mg kg−1 day−1, i.p.) plus Vit E (equals 200 mg kg−1 α-tocopherol; once in 3 days, intramuscularly) for 12 consecutive days. On the 8th day of treatment, the rats were injected with a single dose of DOX. The doses of CAR and CAR + Vit E were chosen in accordance with our previous studies. 31,35 At the end of the experimental period, the animals were fasted overnight and anesthetized with sodium thiopental (50 mg kg−1; i.p.). The rats were then killed by cardiac puncture, and blood samples were collected into dry tubes. The liver, heart, and kidney of rats were quickly excised. Both left and right ventricular walls of the heart and a longitudinal half of the liver and kidney were fixed in 10% buffered formalin for histopathologic analysis. The other parts of the organs were washed in 0.9% NaCl and kept at −70°C until analysis.
Determinations in serum
Serum cardiac troponin I (cTnI) levels were determined in an autoanalyzer (Beckman Coulter Access immunoanalyzer; Minnesota, USA). Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities and urea and creatinine levels were measured using a Cobas Integra System (Roche Diagnostics, Mannheim, Germany).
Determinations in tissues
Tissues were homogenized in ice-cold 0.15 M potassium chloride (10%; w/v), and postmitochondrial fractions of tissues were obtained by centrifugation at 10,000g for 20 min. Lipid peroxidation was assessed through measurement of TBARS and DC levels in the tissue homogenates. To evaluate TBARS levels, homogenates were added to Buege-Aust reagent (15% trichloroacetic acid:0.375% thiobarbituric acid:0.25 N hydrochloric acid (HCl); 1:1:1; w/v) and heated at 100°C for 15 min, followed by centrifugation. Absorbances of supernatant were recorded at 532 nm. The breakdown product of 1,1,3,3-tetraethoxypropane was used as a standard. 36 Tissue lipids were extracted from homogenates with chloroform–methanol (2:1) mixture for the determination of DC levels. The extracted lipids were redissolved in cyclohexane, and DC amounts were calculated using a molar extinction coefficient of 2.52 × 104 M−1 cm−1 at 233 nm spectrophotometrically. 36
The oxidative protein damage was measured using spectrophotometry based on the reaction of 2,4-dinitrophenylhydrazine (DNPH) with PC groups. Organs were homogenized in 50 mM phophate buffer (pH 7.4 containing 1 mM ethylenediaminetetraacetic acid (EDTA) and a cocktail of protease inhibitors) and incubated with DNPH (10 mM in 2.5 M HCl). Then, 20% trichloroacetic acid was added, and tubes were centrifuged. The precipitates were washed in an ethanol–ethylacetate (1:1; v/v) mixture and dissolved in 6 M guanidine hydrochloride solution. The absorbance was measured at 360 nm. PC levels were determined using a molar extinction coefficient of DNPH of 22,000 M−1 cm−1. 37
GSH levels were measured with 5,5-dithiobis-(2-nitrobenzoate) (DTNB) at 412 nm in tissue homogenates. Homogenates (10%, w/v) were deproteinized with the addition of metaphosphoric acid solution (30 g NaCl, 1.67 g metaphosphoric acid, and 0.2 g EDTA in 100 mL distilled water). After centrifugation, supernatant was added to 0.3 M disodium hydrogen phosphate dihydrate and DTNB solution. Absorbances at 412 nm were measured immediately after mixing. GSH levels were calculated using extinction coefficient of 13,600 M−1 cm−1. 38 SOD and GSH-Px activities were determined in the postmitochondrial fraction of the tissues. SOD activity was assayed through its ability to increase the effect of riboflavin-sensitized photooxidation of o-dianisidine. Cuvettes containing 2.7 mL 50 mM potassium phoshate buffer (pH 7.8) with 0.1 mM EDTA, 0.1 mL of 0.39 mM riboflavin, 0.1 mL of 6 mM o-dianisidine-2HCl, and 0.05 mL postmitochondrial fraction were illuminated with 20 W fluorescence lambs at 37°C. Absorbance readings were taken before and after 8 min of illumination at 460 nm. SOD activity was calculated according to the difference in absorbances. A standard curve was prepared using bovine SOD, and the results are expressed as units per milligram protein. 39 GSH-Px activity was measured using the method of Lawrence and Burk 40 with cumene hydroperoxide as substrate. The assay mixture contained 50 mM potassium phoshate buffer (pH: 7.0), 1 mM EDTA, 1 mM sodium azide, 0.2 mM NADPH, 1 mM GSH, 0.5 IU mL−1 GSH reductase, 1.2 mM cumene hydroperoxide, and 0.1 mL diluted postmitochondrial fraction in a total volume of 1 mL. The reaction was followed spectrophotometrically (340 nm) at 37°C after the addition of cumene hydroperoxide. Results were calculated using the extinction coefficient of NADPH of 6.22 × 103 M−1 cm−1 and expressed as nanomoles per minute per milligram protein. Protein levels were determined using bicinchoninic acid. 41 In brief, 10 µL of diluted postmitochondrial fraction was added to 200 µL bicinchoninic acid that contained 0.08% copper sulfate, incubated for 30 min at 37°C, and absorbance was read at 562 nm.
Histopathologic analysis
Tissues were fixed in 10% buffered formalin, embedded in paraffin, and sectioned and stained with hematoxylin and eosin for histologic studies.
Statistical analysis
The results were expressed as mean ± SD. Experimental groups were compared using Kruskal–Wallis variance analysis test. Where significant effects were found, post hoc analysis using Mann–Whitney U test was performed. Power analysis was performed using the calculation provided in the following link: http://biostat.mc.vanderbilt.edu/twiki/bin/view/Main/PowerSampleSize.
Results
There were no significant changes in body weight and heart, liver, and kidney weights in the control and DOX-treated groups (data not shown).
Serum cTnI levels and ALT and AST activities increased in DOX-treated rats. Serum urea levels were also increased. Although there were no significant changes in creatinine levels, these values tended to increase in DOX-treated rats. CAR and CAR + Vit E treatments decreased serum cTnI levels and ALT and AST activities in DOX-treated rats. However, urea and creatinine levels also reduced, although this did not reach significance (Table 1).
The effect of CAR + Vit E treatments on cTnI levels, ALT, and AST activities; urea; and creatinine levels in serum of rats treated with DOX.a
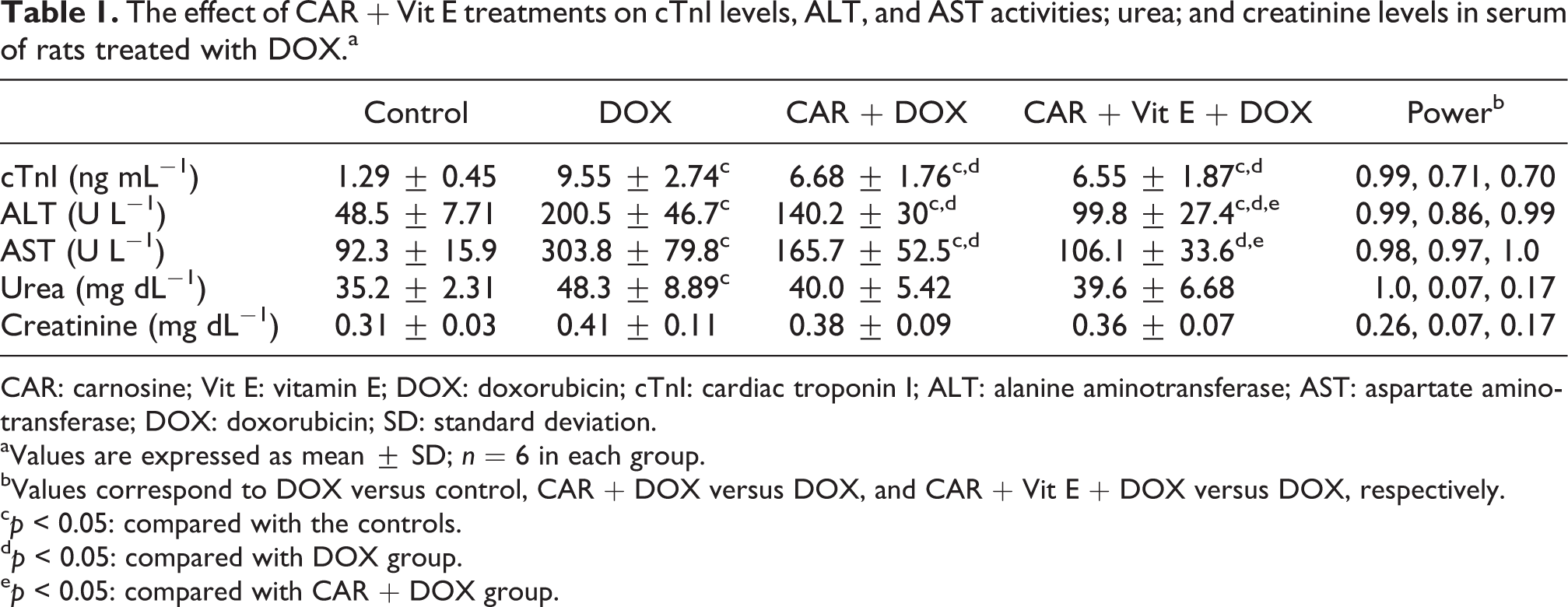
CAR: carnosine; Vit E: vitamin E; DOX: doxorubicin; cTnI: cardiac troponin I; ALT: alanine aminotransferase; AST: aspartate aminotransferase; DOX: doxorubicin; SD: standard deviation.
aValues are expressed as mean ± SD; n = 6 in each group.
bValues correspond to DOX versus control, CAR + DOX versus DOX, and CAR + Vit E + DOX versus DOX, respectively.
c p < 0.05: compared with the controls.
d p < 0.05: compared with DOX group.
e p < 0.05: compared with CAR + DOX group.
Levels of TBARS, DC, and PC were found to have increased in the heart, liver, and kidney of DOX-treated rats. CAR and CAR + Vit E treatments decreased cardiac TBARS, DC, and PC levels in DOX-treated rats. PC levels decreased in CAR + DOX and CAR + Vit E + DOX groups, but hepatic TBARS and DC levels remained unchanged. Renal TBARS, DC, and PC levels were also observed to decrease in DOX-treated rats due to CAR or CAR + Vit E treatments (Table 2).
The effect of CAR + Vit E treatments on TBARS and DC levels in heart, liver, and kidney tissues of rats treated with DOX.a

CAR: carnosine; Vit E: vitamin E; DOX: doxorubicin; DOX: doxorubicin; TBARS: thiobarbituric acid reactive substances; DC: diene conjugate; SD: standard deviation.
aValues are expressed as mean ± SD; n = 6 in each group.
b p < 0.05: compared with the controls.
c p < 0.05: compared with DOX group.
d p < 0.05: compared with CAR + DOX group.
eValues correspond to DOX versus control, CAR + DOX versus DOX, and CAR + Vit E + DOX versus DOX, respectively.
DOX treatment was observed to cause significant decreases in cardiac, hepatic, and renal GSH levels and increases in hepatic SOD activities. However, GSH-Px activity remained unchanged in examined tissues. Although CAR and CAR + Vit E treatments did not alter hepatic GSH levels, these treatments elevated cardiac and renal GSH levels and decreased hepatic SOD activities in DOX-treated rats (Table 3).
The effect of CAR + Vit E treatments on GSH levels and GSH-Px and SOD activities in heart, liver, and kidney tissues of rats treated with DOX.a

CAR: carnosine; Vit E: vitamin E; DOX: doxorubicin; GSH: glutathione; GSH-Px: glutathione peroxidase; SOD: superoxide dismutase; SD: standard deviation.
aValues are expressed as mean ± SD; n = 6 in each group.
b p < 0.05: compared with the controls.
c p < 0.05: compared with DOX group.
dValues correspond to DOX versus control, CAR + DOX versus DOX, and CAR + Vit E + DOX versus DOX, respectively.
Histologic examination of hearts in the DOX-treated rats revealed a mild degree of interstitial edema and lymphocyte infiltration, irregular clusters of myocardial fibers, and necrotic changes. In the liver, necrobiotic parenchymal cells were seen in the midzonal area of the liver parenchyma. Infiltration of lymphocyte and plasma cells was scant. In addition, swelling and hydropic degeneration in renal tubular epithelium were observed in DOX-treated rats. Congestion of medullarly areas and necrotic changes that resemble acute tubular necrosis were also detected. According to the histopathologic observations, cardiac, hepatic, and renal lesions were observed to be reduced in groups treated with CAR or CAR + Vit E compared with the DOX group. Necrobiotic changes in cardiomyocytes and liver parenchyma were not seen. In addition, there were no changes that resembled acute tubular necrosis in the CAR + DOX- or CAR + Vit E + DOX-treated rats (Figure 1).
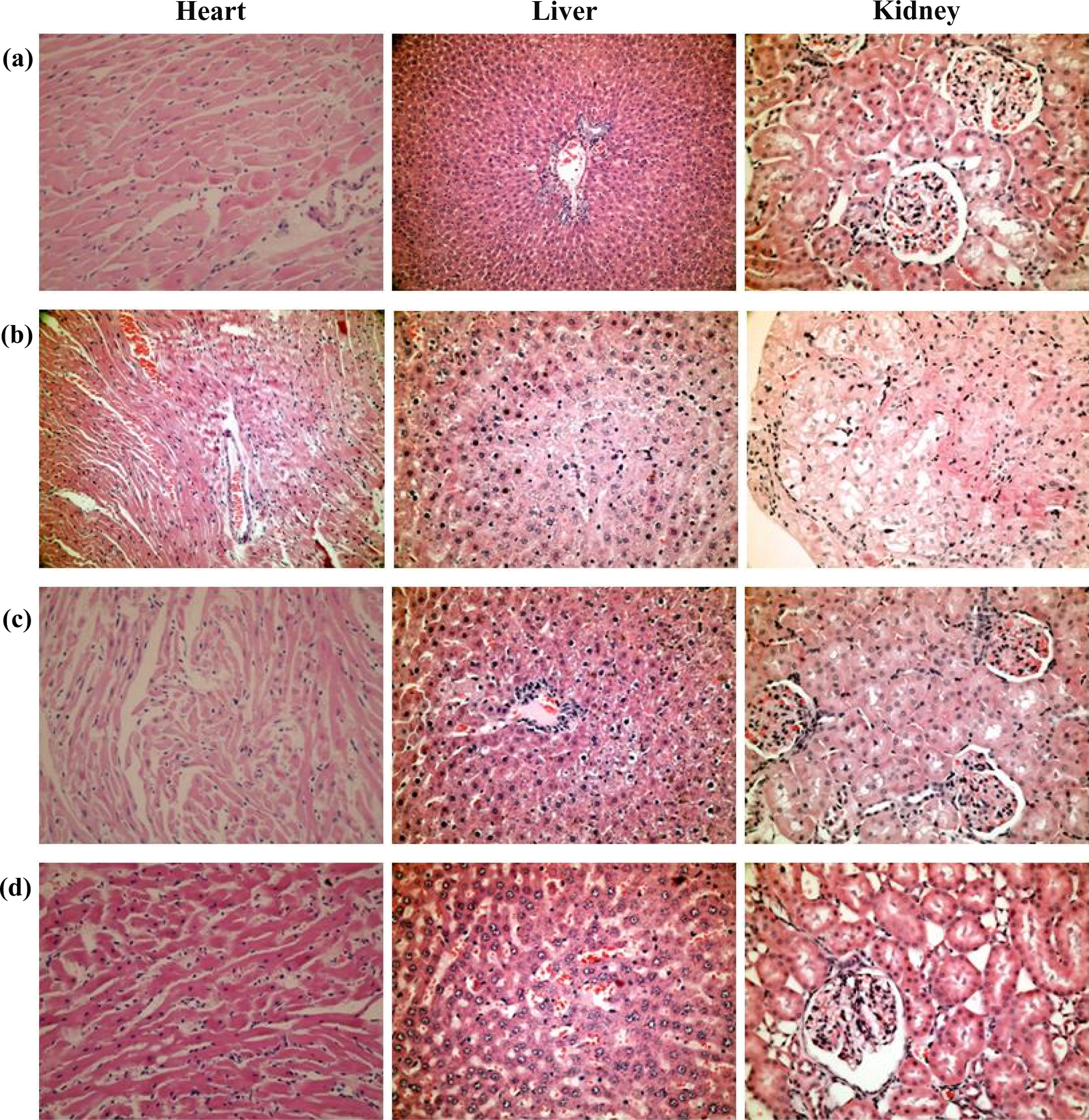
The effect of CAR + Vit E treatments on DOX-induced histopathological alterations in the heart, liver, and kidney tissues (H&E). (a) Control group: normal cardiac muscles, liver, and kidney histologies (×400). (b) DOX group: interstitial edema, lympocyte infiltration, and irregular clusters of myocardial fibers of cardiac muscles (×200); necrotic–necrobiotic changes and cellular swelling in midzonal areas of the liver (×400); and congestion and necrotic changes resembling acute tubular necrosis in kidney (×400). (c, d) CAR + DOX and CAR + Vit E + DOX groups: interstitial edema but not necrobiotic changes in cardiac muscle (×200); cellular swelling in hepatocytes, scanty lymhocytic infiltration but not necrobiotic changes in the liver (×400); and hydropic degeneration and swelling of tubular epithelium in kidney (×400). CAR: carnosine; Vit E: vitamin E; DOX: doxorubicin; H&E: hematoxylin and eosin.
On the other hand, there were no changes in examined parameters or histopathologic appearence of tissues in DOX-treated rats due to Vit E treatment alone (data not shown).
Discussion
DOX resulted in increases in the production of ROS and inflammatory cytokines, inflammatory reactions, and apoptotic and necrotic changes in tissues and body fluids. 2 –12,34 In our study, serum markers that indicate cardiac, hepatic, and renal damage increased significantly 4 days after 30 mg kg−1 DOX was administered, compared with the controls. In the light microscopic examination, histopathologic changes were seen in tissues of the DOX-treated rats. In addition, TBARS, DC, and PC levels increased, GSH levels decreased, and SOD and GSH-Px activities remained unchanged in tissues due to DOX, and only increased hepatic SOD activity was detected. This increase may be related to an adaptive increase against oxidative stress in the liver. Our results reported here agree with those of previous studies, which showed that DOX produces a prooxidant status in the tissues of rats.2–12
CAR and related derivatives of histidine are found in high concentrations in cardiac muscle. CAR is considered a multifunctional molecule with antioxidant action and operates as a selective inhibitor of protein glycation and protein–protein cross-linking. CAR has a powerful antioxidant activity and can serve as an efficient electron donor, which prevents lipid peroxidation. It also quenches singlet oxygen and interacts with superoxide. CAR can also chelate Cu and iron, and this chelation prevents these ions from catalyzing Fenton chemistry, hence blocking production of hydroxyl radicals.
13
–16
Moreover, CAR-Cu complexes possess SOD activity and bind covalently to reactive degradation products of lipid peroxides, which prevent them from reacting with other cellular targets. CAR performs an effective buffering action that binds a proton reducing the pH value.
13
–16
These physiologic roles of CAR are based on its chemical structure.
16
The imidazolic ring is required for the antioxidant, metal ion chelating, and buffering activity. The β-alanine moiety regulates the metabolic fate of the
CARlikely seems to be cardioprotective. 14 –17 It has regulatory effects on myocardial calcium levels, an increase in sensitivity of calcium release channels and of the contractile apparatus, with favorable effects on cardiac contractility and function. 17 It has been reported that exogenous CAR has been shown to protect ischemia–reperfusion 18,19 and isoproterenol-induced 22 cardiac toxicities. It is also protective for DOX-induced cardiomyopathy. CAR prevented cardiac oxidative stress and inactivation of SOD and GSH-Px after a single dose of DOX. 21 CAR treatment also normalized cardiac hemodynamic parameters and histopathologic changes in congestive heart failure due to repeated DOX treatment in rabbits. 20 It has recently been reported that CAR combined with conventional therapy has beneficial effects on exercise performance and quality of life in patients with chronic heart failure. 42 In the current study, CAR caused significant decreases in high serum cTnI levels, amelioration in prooxidant–antioxidant balance, and cardiac histopathologic findings in DOX-treated rats. These results clearly indicate that CAR reduced DOX-induced oxidative stress and cardiotoxicity in rats. However, whether CAR can protect liver and kidney tissues against DOX has not yet been investigated. CAR treatment resulted in decreased serum ALT and AST activities. Urea and creatinine levels also reduced, although not significantly in DOX-treated rats. However, amelioration in prooxidant–antioxidant balance and histopathologic findings were also detected in the liver and kidney tissues. These results clearly indicate that CAR also reduced DOX-induced oxidative stress and tissue damage in the liver and kidney of rats.
On the other hand, the combination of two different antioxidants can be beneficial for limiting oxidative stress-induced pathologies. 30 –33 Lipophilic Vit E easily penetrates the cell membranes, and the combined effects of CAR and Vit E may enhance the total antioxidative potential. 30,31,35 Water-soluble CAR functions in the cytosol, but lipophilic agents are effective in all parts of the cell. This state may account for the synergistic effect of the combined use of CAT and Vit E. This combination was detected to be more effective in decreasing the lipid peroxides in liver and heart tissues of rats than dietary CAR or Vit E alone. 30,31,35 In the current study, no amelioration was observed in the disturbed serum and tissue parameters in rats in the DOX group treated with Vit E alone. However, when CAR was used together with lipophilic Vit E, this combined treatment was observed to be more effective than CAR treatment alone on DOX-induced oxidative stress and injury in the examined tissues.
In conclusion, our results may indicate that CAR alone, especially its combination with Vit E, protects against DOX-induced toxicity in heart, liver, and kidney tissues of rats as evidenced by improved cardiac, hepatic, and renal markers and restoration of the prooxidant state and amelioration of histopathologic changes. This protective potential of CAR can be attributed to prevent oxidative stress through several mechanisms such as scavenging/neutralizing free radicals, regulating enzymatic/nonenzymatic antioxidants, and metal chelating. Thus, CAR or CAR + Vit E treatment could be an effective regimen to reduce toxic adverse effects and to enhance the therapeutic efficiency of DOX in clinical chemotherapy. However, future work will be needed to confirm our results.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Fund of University of Istanbul (Project No: T-11683).
