Abstract
Background:
Polybrominated diphenyl ether (PBDE) levels in children and teenagers were higher than those of the adults and the highest levels were found in infants and toddlers. 2,2′,3,3′,4,4′,5,5′,6,6′-Decabrominated diphenyl ether (BDE-209) readily crosses the placental barrier and produces toxicity in the developing fetus, particularly to the developing brain.
Objectives:
This present study aims to investigate the potential effects of prenatal BDE-209 exposure on regulation of neurogenesis and learning function in an experimental rat model.
Methods:
Pregnant rats received BDE-209 (10, 30, or 50 mg kg−1 day−1) or vehicle (arachis oil) through gastric gavage from gestation day 1 to 14 (n = 10 per group). The embryonic hippocampal neural stem cells (NSCs) from five pregnant rats in each group were collected on day 14 and cultured in vitro to determine the cell viability, apoptosis, and differentiation of NSCs using cell counting kit 8 assay, flow cytometry, and immunofluorescence staining, respectively. In total, 20 male offspring on postnatal day 25 from each group were chosen to evaluate learning ability using a Morris water navigation task assay.
Results:
The data showed that prenatal exposure to BDE-209 decreased cell viability and differentiation of NSCs but promoted apoptosis in a dose-dependent manner. Prenatal BDE-209 exposure also impaired rat-learning acquisition in a dose-dependent manner.
Conclusions:
Prenatal BDE-209 exposure impairs rat-learning acquisition, possibly by affecting neurogenesis in the hippocampus during embryonic development.
Keywords
Introduction
Polybrominated diphenyl ethers (PBDEs) are a group of aromatic compounds that are widely used as flame retardants. 1–4 PBDEs are highly lipophilic and have long half-lives that could lead to accumulation in human tissues, increasing the risk of chronic exposure, potentially leading to toxicity. 5–7 Among these PBDEs, 2,2′,3,3′,4,4′,5,5′,6,6′-decabrominated diphenyl ether (BDE-209) is the most common and is widely used in many countries, particularly in China. 3,4,8 The annual demand worldwide was estimated as 56,100 tons in 2001. BDE-209 is not only detected in the environment and certain foods but also in human body. Recent studies showed that PBDE levels in children and teenagers were higher than that of the adults and the highest levels were found in infants and toddlers. 9 These data raised a concern about potential adverse health effects caused by exposure to BDE-209. Several studies have shown that in addition to its carcinogenic effect, 10 BDE-209 is able to damage the central nervous system (CNS), immune system, and endocrine systems in humans. 11–13 BDE-209 readily crosses the placental barrier and has been shown to produce toxicity in the developing fetus, particularly to the developing brain. 14 Previous studies have demonstrated that BDE-209 could lead to neurobehavioral derangements in adult mice during a defined period of neonatal brain development. 15,16 Another study in mice suggested that BDE-209 functions as a developmental neurotoxin, producing long-term behavioral changes following a discrete period of neonatal exposure. 17 Our own data showed that BDE-209 could induce the rate of hippocampal neurons apoptosis by increasing reactive oxygen species (ROS) and global DNA methylation. 18 We also found that BDE-209 could inhibit outgrowth and differentiation of neural stem cells (NSCs) into neurons and glial cells in a dose-dependent manner. 19 In the current study, we hypothesized that prenatal exposure to BDE-209 could impair development of cognitive function by affecting NSC growth in the brain and that BDE-209 could also continue to be effective in the developing central nervous system. We performed an animal experiment to assess the effects of BDE-209 on alteration of learning ability after the rats were prenatal exposed to BDE-209 and explored the underlying molecular mechanisms responsible for the toxic effects.
Materials and methods
Animal experiments
Pregnant Sprague-Dawley rats (gestation day 1) were obtained from the Experimental Animal Center of Southern Medical University (Guangzhou, China) and maintained in a specific pathogen-free facility with 12-h/12-h light/dark cycle. The rats received BDE-209 (Beijing J&K Chemicals, Beijing, China; 10, 30, and 50 mg kg−1 day−1) or vehicle (arachis oil) of equal volume from gestational days 1 to 14 via gastric gavage. 20 These rats were divided into the following 5 groups (with 10 pregnant rats in each group), that is, group A: blank control (only administered with animal feed); group B: vehicle group (arachis oil and animal feed); group C: 10 mg kg−1 day−1 BDE-209 (arachis oil + animal feed + 10 mg kg−1 day−1 BDE-209); group D: 30 mg kg−1 day−1 BDE-209 (arachis oil + animal feed + 30 mg kg−1 day−1 BDE-209); and group E: 50 mg kg−1 day−1 BDE-209 group (arachis oil + animal feed + 50 mg kg−1 day−1 BDE-209). 15,21 On gestational day 14 (the 14th day after pregnancy), 5 rats of every group were killed with an overdose of chloral hydrate (0.35 g kg−1 intraperitoneally). Fetal brain was dissected on ice to obtain the hippocampus for NSC culture (N = 5 rats in each group). The remaining five pregnant rats for every group were maintained until parturition, the offspring were used at postnatal day 25 (the 25th day after birth) for Morris water maze studies. The study protocol was approved by The Animal Ethics Committee of Southern Medical University, Guangzhou, China.
Neurosphere culture and identification of hippocampal NSCs
The hippocampus of rat embryos was collected under sterile conditions and cut into 1 mm3 pieces, and a cell suspension was obtained through filtration with a 200-mesh filter. Neurospheres were cultured as follows: 22 cells were seeded in serum-free Dulbecco’s modified Eagle’s medium/nutrient mixture F-12 (DMEM/F12) medium (Gibco, Carlsbad, California, USA) containing 2% B27 (Gibco), 20 ng ml−1 basic fibroblast growth factor (bFGF), and 20 ng ml−1 epidermal growth factor (EGF) (Peprotech, Rocky Hill, New Jersey, USA) at a density of 1 × 106 ml−1 in 25 cm2 flasks at 37°C and 80% humidity with 95% air and 5% carbon dioxide. The culture medium was replenished every 3–4 days. The cells were subcultured every 7–10 days and then were fixed in 4% paraformaldehyde on polylysine-coated cover slides, which were then incubated with 0.3% TritonX-100 for 15 min at room temperature. After blocking with 10% fetal bovine serum for 30 min, the slides were incubated with a mouse anti-rat Nestin antibody (l:100; from Millipore, Billerica, Massachusetts, USA) in a humidified wet box at 4°C overnight. Then, the slides were incubated with a fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse immunoglobulin G (IgG) antibody (1:100; from Beijing Biosynthesis Biotechnology Co. Ltd, Beijing, China) at room temperature for 2 h, followed by staining with 4′,6-diamidino-2-phenylindole (DAPI; KeyGen Biotech, Nanjing, China). The slides were then observed under a fluorescent microscope (Leica, Wetzlar, Germany). For control slides, the primary antibody was replaced with 10% fetal bovine serum.
Detection of NSC purity
After 7 days of NSC culture, neurospheres were centrifuged at 1000g for 5 min and filtered through a 300-mesh filter. After that, cells were resuspended in serum-free medium at a density of 1 × 105 ml−1 prior to incubation with a mouse anti-rat Nestin antibody (1:100; Millipore) at 37°C for 2 h. The cells were then incubated with an FITC-conjugated goat anti-mouse IgG antibody (1:100; Beijing Biosynthesis Biotechnology Co. Ltd) at room temperature for 30 min, followed by flow cytometry detection of Nestin positive cells. For control slides, the primary antibody was replaced with 10% fetal bovine serum. The purity of the cultured NSCs was calculated as the percentage of positively stained cells relative to the total cell number. 23
Detection of the cultured NSC viability
The second passage neurospheres were harvested and centrifuged at 1000g for 5 min. Cells were resuspended in serum-free medium and seeded at a density of 1 × 105 ml−1 in a 96-well plate (100 μl/well) and then cultured for up to 72 h. Cells viability were counted using a cell counting kit 8 (CCK-8, Dojindo Laboratories, Kumamoto, Japan) according to the manufacturer’s instructions (n = 6). 24
Detection of the cultured NSC apoptosis rate
The second passage neurospheres were harvested and a single cell suspension was prepared for apoptosis assay using a kit from Nanjing KeyGen Biotech (Nanjing, China). 25 Briefly, cells were resuspended in 500 μl of binding buffer at a density of 1 × 105cells ml−1. To this, 5 μl of Annexin V-FITC was added to the cells, followed by 5 μl of propidium iodide. The reaction was allowed to proceed at room temperature in the dark for approximately 5–15 min, followed by flow cytometry analysis.
Detection of the cultured NSC differentiation rate
The second passage neurospheres were harvested and centrifuged at 1000g for 5 min. Cells were resuspended with DMEM/F12 medium containing 2% B27, 20 ng ml−1 bFGF, 20 ng ml−1 EGF, and 10% fetal calf serum. The cell suspension was incubated in polylysine-coated six-well plates for 7 days. The differentiation of neurospheres was observed every day under an inverted microscope. Neuronal nuclear antigen (NeuN) and glial fibrillary acidic protein (GFAP) immunostaining of neurospheres were carried out on day 7, and the nuclei were stained with DAPI. 26 Six randomly selected microscopic fields were observed under a fluorescent microscope. The proportion of NeuN- or GFAP-positive cells was counted and summarized.
Morris water maze task assay
We used Morris water maze task assay to assess the learning ability in rats on postnatal day 25. 27 To avoid the influence of the sex difference to our experimental data, 20 male rats were chosen for each group. The maze (120 cm in diameter and 50 cm in height) was divided into 4 quadrants, and a platform of 9 cm in diameter and 27 cm in height was placed in the southwest quadrant. The movement of the animals in the maze was recorded using a camera connected to a computer. External cues remained constant throughout the task. White tempera paint was used to obscure the platform, and water temperature was maintained at 22 ± 1°C. The experiments consisted of four consecutive sessions daily (60 s for one session and 60 s interval between two sessions). Rats entered the maze from a different point on each daily session. If the rats failed to reach the platform within 60 s, the rats were guided to the platform by the researchers and allowed to remain on the platform for 20 s. The escape latency was defined as the time to reach the hidden platform, and the maximal escape latency was set at 60 s. The interval between two adjacent tests was no less than 60 s.
Statistical analysis
Data are summarized as means ± SD and analyzed using one-way analysis of variance of repeated measures, followed by the least significant difference test (LSD) for pairwise comparisons. All statistical analyses were performed using Statistical package for Social Sciences (SPSS) software (version 13.0, SPSS, Chicago, Illinois, USA). Statistical significance was set at p < 0.05.
Results
Identification of NSCs in culture
In this study, hippocampal NSCs harvested from pregnant rats exposure to BDE-209 were collected and cultured in serum-free medium for 3–5 days (Figure 1(a)). Immunofluorescence staining confirmed that the cultured cells were mostly Nestin positive (Figure 1(b)). Under DAPI staining, the nuclei were stained with blue fluorescence (Figure 1(c)). Flow cytometry revealed that 98% of the cells are NSCs (Figure 1(c)). These data indicated that this culture contained highly purified NSCs in vitro.
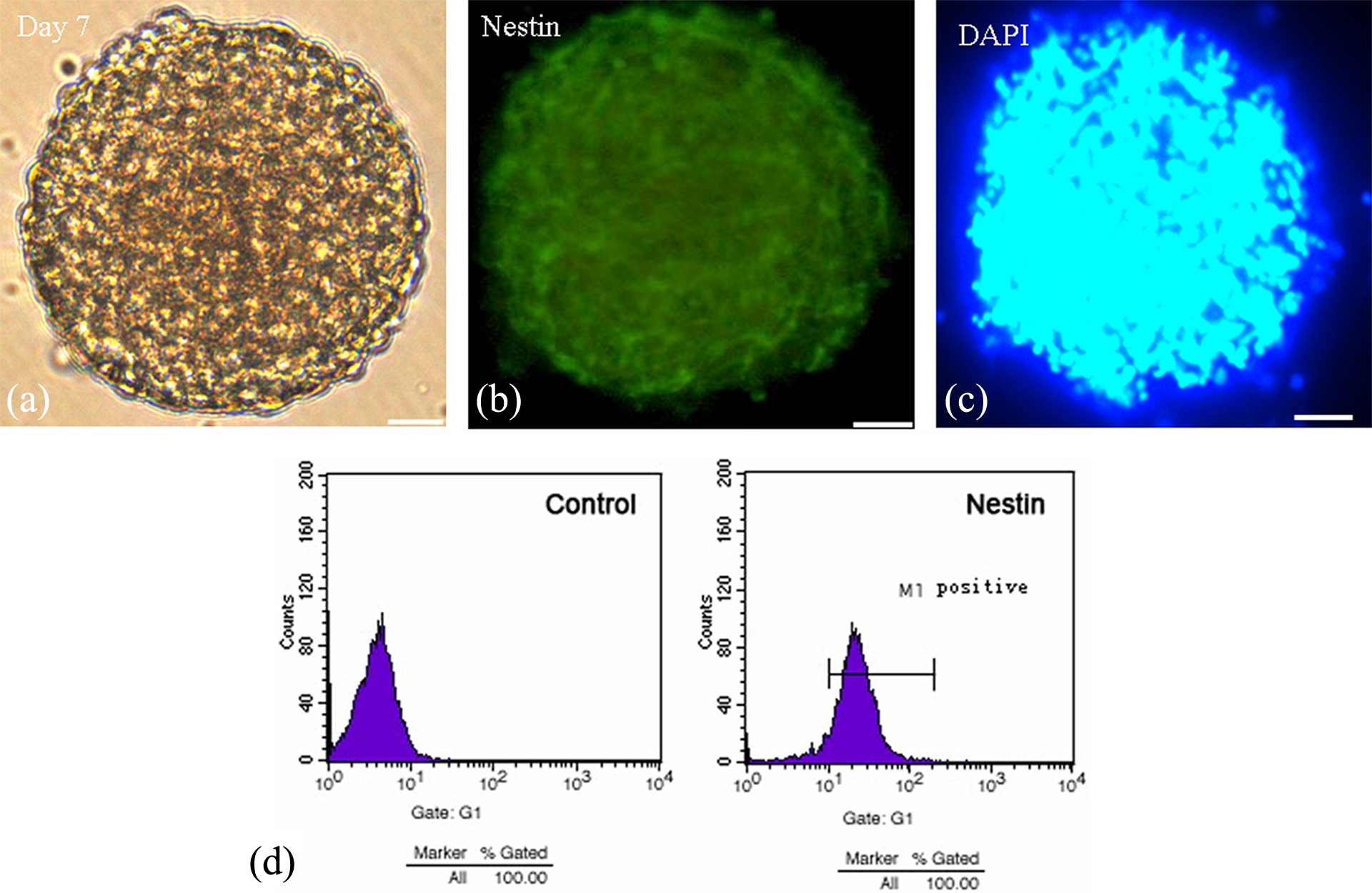
In vitro culture of hippocampal NSCs isolated from fetal rats. (a) Seven days culture in vitro observed under an inverted microscope (×200, bar = 10 μm); (b) immunofluorescence staining of Nestin protein; (c) immunofluorescence staining of DAPI; and (d) the purity of the cultured neural stem cells showed by flow cytometry analysis using an anti-Nestin antibody. DAPI: 4′,6-diamidino-2-phenylindole; NSCs: neural stem cells.
Effects of prenatal BDE-209 exposure on regulation of hippocampal NSC viability and apoptosis
A cell viability assay was performed on the NSCs, and we found that prenatal exposure to BDE-209 significantly decreased viability of these cells ( p < 0.01 vs. vehicle control for all three doses; Figure 2). In contrast, administration of vehicle alone (arachis oil) did not affect the cell viability ( p = 0.42). Moreover, the effects were dose dependent since pairwise comparison revealed significant difference between the three doses ( p = 0.006). Furthermore, prenatal BDE-209 exposure significantly increased the rate of NSCs apoptosis in a dose-dependent manner ( p = 0.034) versus the vehicle control for all three doses; Figure 3). Cells exposed to vehicle alone did not show any changes in apoptosis.
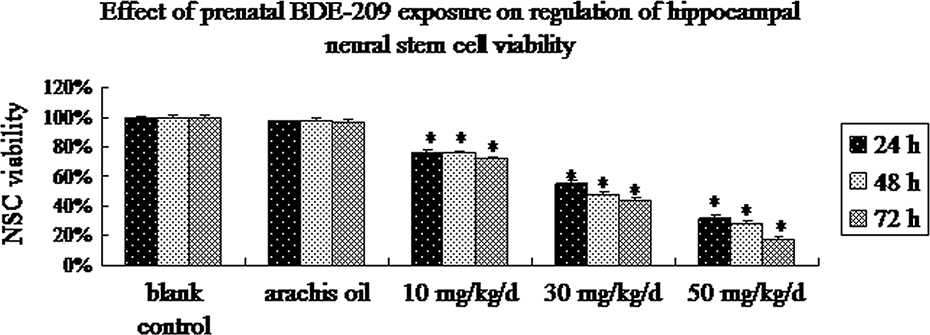
Changed cell viability of the cultured NSCs detected by CCK-8 kit. The second passage neurospheres were harvested and cultured in a 96-well plate (100 μl/well) for up to 72 h, and then cell viability was counted using a CCK-8 kit (n = 6, mean ± SD). *p < 0.01 versus the arachis oil control. CCK: cell counting kit; NSC: neural stem cell.
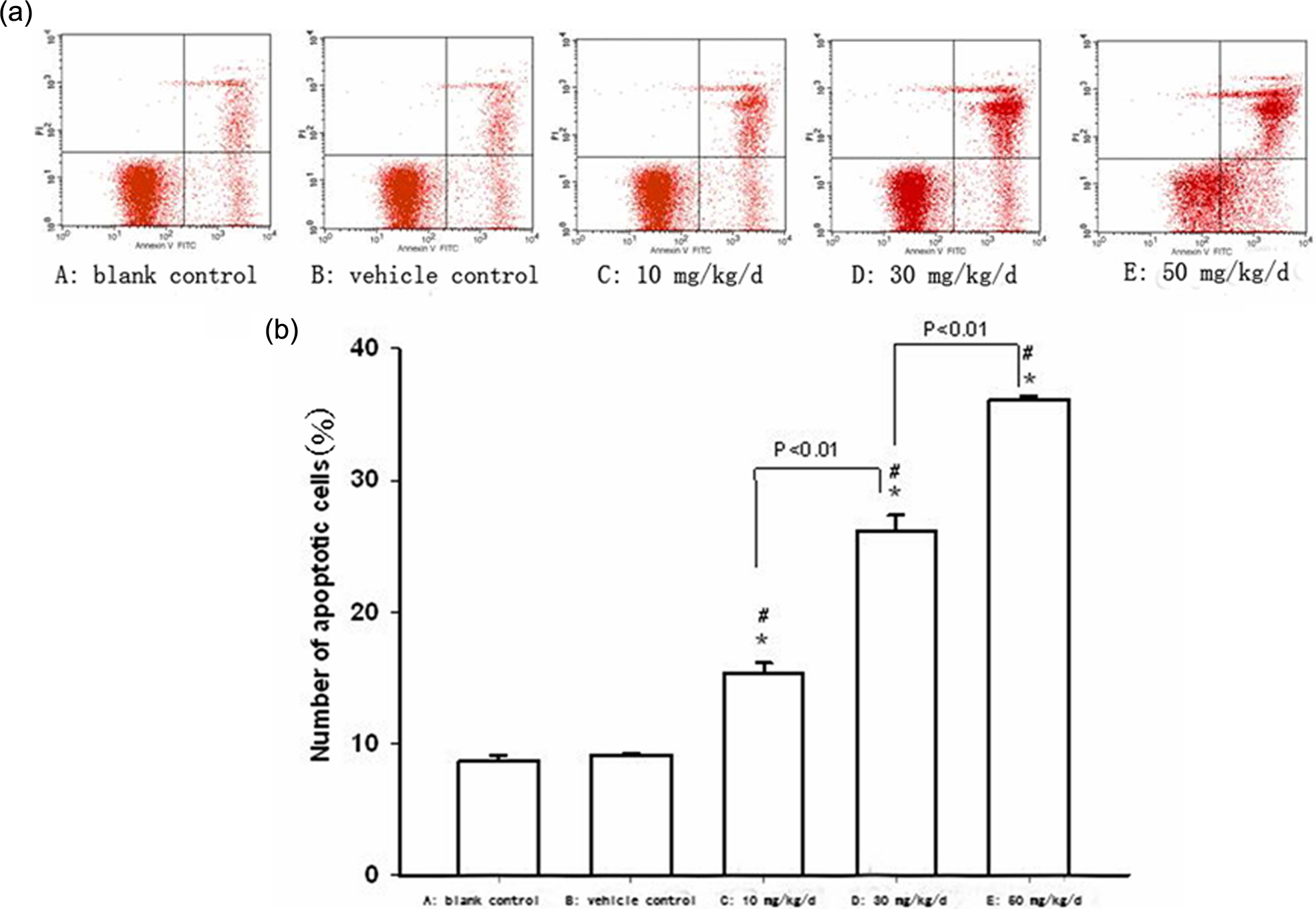
Effect of prenatal BDE-209 exposure on regulation of hippocampal NSC apoptosis measured by flow cytometry. (a) Flow cytometry profile for each group; (b) summarized data from (a) (n = 6). *p < 0.05 versus blank control; # p < 0.05 versus arachis oil control. BDE-209: 2,2′,3,3′,4,4′,5,5′,6,6′-decabrominated diphenyl ether; NSC: neural stem cell.
Effects of prenatal BDE-209 exposure on regulation of hippocampal NSC differentiation
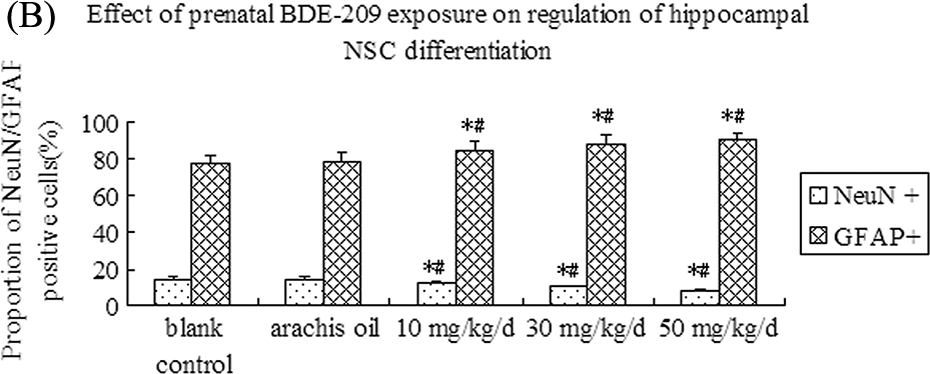
Next, we assessed the effects of prenatal BDE-209 exposure on regulation of hippocampal NSC differentiation. The control group showed that the cells after 24 h seeding were adherent to the culture flask, and these processes gradually extended outward and afterward and formed a network at day 7. NeuN-positive cells were neurons of triangle, round, or conical shape with large nuclei, and GFAP-positive cells were long spindle-shaped or polygonal with long processes and small oval-shaped nuclei. We found that prenatal BDE-209 exposure decreased proportions of NeuN-positive cells and increased GFAP-positive cells ( p = 0.024) versus vehicle control for all three doses (Figure 4). BDE-209 exposure changed the proportion of differentiated cells in a dose-dependent manner (Figure 4).
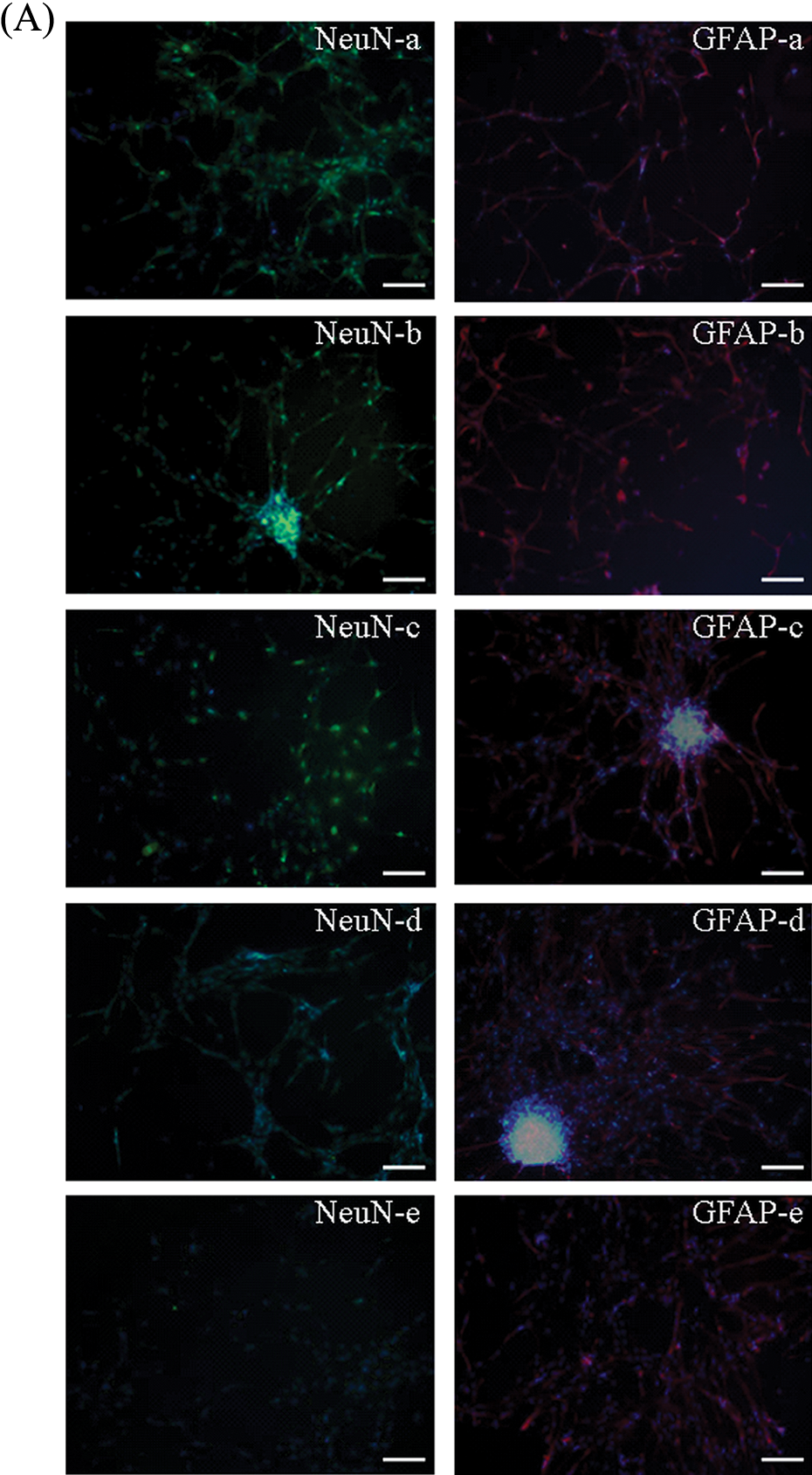
NeuN and GFAP expression measured by immunofluorescence staining in each of the different groups after prenatal BDE-209 exposure on regulation of hippocampal NSC differentiation. (A) NeuN and GFAP expression measured by immunofluorescence staining in all the groups (×200): (a) blank control; (b) vehicle group; (c) 10 mg kg−1 day−1 BDE-209 group; (d) 30 mg kg−1 day−1 BDE-209 group; and (e) 50 mg kg−1 day−1 BDE-209 group. (B) Summarized data of proportion of NeuN-positive cells and GFAP-positive cells in different groups. *p < 0.05 versus blank control; # p < 0.05 versus arachis oil control. NeuN: neuronal nuclear antigen; GFAP: glial fibrillary acidic protein; BDE-209: 2,2′,3,3′,4,4′,5,5′,6,6′-decabrominated diphenyl ether; NSC: neural stem cell.
Continued
Effects of prenatal BDE-209 exposure on regulation of rat learning ability
A Morris water maze task was used to determine the effects of prenatal BDE-209 exposure on regulation of rat learning ability. The data showed that the escape latency over the four sessions differed significantly among the five treatment groups (F = 71.686, p = 0.0006). The escape latency decreased over time in all five different treatments (F = 6.863, p = 0.00023). There was also a significant difference among the treatment conditions (F = 32.352, p = 0.00034). The post hoc LSD analysis indicated that prenatal BDE-209 exposure decreased the acquisition of the task at 30 and 50 mg kg−1 day−1 ( p = 0.016) versus vehicle and the blank control for both doses, but not at a lower dose of 10 mg kg−1 day−1 (Figure 5).
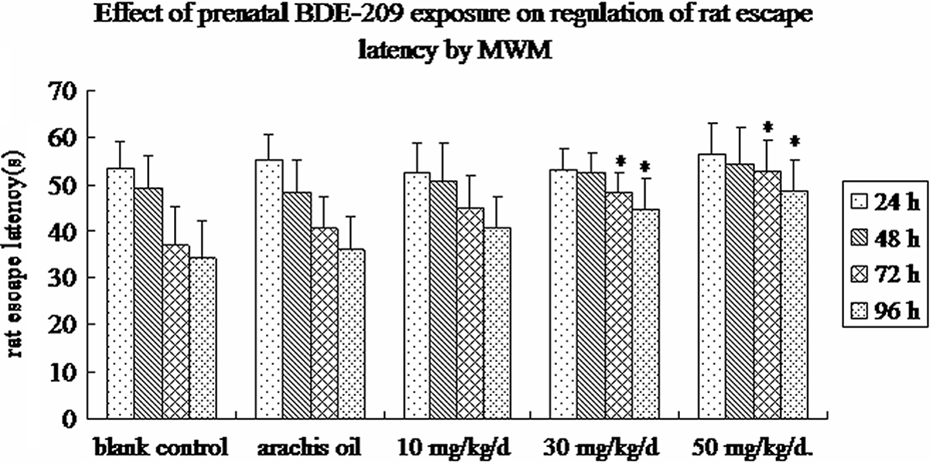
Effects of prenatal BDE-209 exposure on regulation of rat escape latency. Morris water maze task assay was used to assess the learning ability in rats on postnatal day 25 (for review see details in the “Materials and Methods” section). n = 20, data (rat escape latency) shown are mean ± SD. *p < 0.05 versus blank control. BDE-209: 2,2′,3,3′,4,4′,5,5′,6,6′-decabrominated diphenyl ether.
Discussion
In this study, we exposed pregnant rats to BDE-209 and then tested the effects of BDE-209 exposure on alteration of rat learning ability. Our data showed that prenatal BDE-209 exposure decreased rat learning capacity. Meanwhile, we also cultured hippocampal NSCs in vitro to assess the effects of prenatal BDE-209 exposure on regulation of cell viability, apoptosis, and differentiation of these cells. Our data showed that prenatal BDE-209 exposure suppressed neural cell viability and differentiation and promoted apoptosis in a dose-dependent manner. From these data, we presumed that prenatal exposure to BDE-209 impairs rat learning capacity by inhibition of neurogenesis in the rat hippocampus.
Nestin is a type VI intermediate filament protein expressed in the dividing cells during the early stages of development in the CNS. However, after neural differentiation, nestin will be downregulated and replaced by tissue-specific intermediate filament proteins. For example, during neurogenesis and gliogenesis, expression of nestin is replaced by cell type-specific intermediate filaments, for example, neurofilaments NeuN and GFAP. Thus, detection of these two proteins is used to confirm neuron differentiation. So, our study utilized these three proteins as markers to identify neural cells and their differentiation.
Increased studies have shown that BDE-209, the most commonly used PBDE, causes biological toxicity in humans. For example, a study by Viberg et al. showed that BDE-209 caused long-term changes in locomotor activity, spontaneous behavior, and learning and memory alterations. 30 BDE-209 also inhibited the secretion of neurotransmitters, and exposure of newborn rats to BDE-209 resulted in behavior alterations in adulthood, indicating the neurodevelopmental toxicity of BDE-209. 11,16,29,30 A study conducted by Riva et al. revealed that BDE-209 was able to damage cell DNA. 30 Indeed, our data revealed that BDE-209 suppressed cell viability and differentiation of cultured hippocampal NSCs in vitro.
Moreover, previous studies from our laboratory revealed that BDE-209 was able to cross the placenta and accumulate in a variety of fetal tissues. 20,31–34 Thus, BDE-209 exposure could contribute to abnormal development of nervous system in rats. Our current study demonstrated that prenatal BDE-209 exposure inhibited cell viability and development of NSCs during the embryonic development. Moreover, the current data also showed that prenatal BDE-209 exposure impaired learning and memory ability and capacity.
The hippocampus is a key anatomical basis of learning and memory ability. 35–37 Capilla-Gonzalez et al. demonstrated that N-ethyl-N-nitrosourea exposure induced mouse neurogenesis impairment in the hippocampus and subventricular zone in vivo and in vitro, and the mice also demonstrated defects in memory and olfaction. 38 Thus, our study focused on the effects of prenatal BDE-209 exposure on regulation of hippocampal NSC viability, differentiation, and apoptosis. Our results showed a link between a dose-dependent decrease in hippocampal NSC viability and their differentiation into neurons by prenatal exposure to BDE-209 with reduced learning capacity of the rats. The combination of reduced cell viability/differentiation and increased apoptosis of hippocampal NSCs perhaps could result in decrease in the number of differentiated neurons and thus impair the subsequent learning and memory capacity of the rats.
The results from the current study clearly demonstrated the effects of prenatal BDE-209 exposure on hippocampal NSCs and learning capacity in rats. However, the defined mechanisms underlying these changes still need to be elucidated. To date, the precise effect of BDE-209 on humans remains to be defined. Epidemiological data has shown that populations featuring high BDE-209 levels were afflicted with high morbidity rates of lung, kidney, and liver cancers (e.g. Zhejiang, China). 39 Further studies have shown that BDE-209 blood levels are higher in the cancer patients than that of normal inhabitants. 39 Our data demonstrated that low-dose PBDE-209 (0–100 nM) induced growth of human breast, cervical, and ovarian cancer cells in a dose-dependent manner in vitro. 10 However, to date, there are no toxicokinetic data available in humans. For this reason, future studies need to clarify the effects of BDE-209 on humans, such as metabolism of BDE-209 in humans and potentially synergistic effects of BDE-209 and other environmental pollutants on neurodevelopment.
Footnotes
Authors’ Note
The authors YHC and ZHL contributed equally to this work.
Acknowledgment
We thank Medjaden Bioscience Limited (Hong Kong, China) for assisting in the preparation of this manuscript.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported in part by grants from Guangzhou Education Bureau (B94119), the Guangdong Province Nature Science Foundation (S20120010008932), the China Bureau of Education (20114423110004) to DJC, and the Guangdong Province Technology and Science Agency to YHY (2011A030400008).
