Abstract
One of the most important natural extracellular constituents is hyaluronic acid (HA) with the potential to develop a highly organized microenvironment. In the present study, we enriched HA hydrogel with tenascin-C (TN-C) and examined the viability and survival of mouse neural stem cells (NSCs) using different biological assays. Following NSCs isolation and expansion, their phenotype was identified using flow cytometry analysis. Cell survival was measured using MTT assay and DAPI staining after exposure to various concentrations of 50, 100, 200, and 400 nM TN-C. Using acridine orange/ethidium bromide staining, we measured the number of live and necrotic cells after exposure to the combination of HA and TN-C. MTT assay revealed the highest NSCs viability rate in the group exposed to 100 nM TN-C compared to other groups, and a combination of 1% HA + 100 nM TN-C increased the viability of NSCs compared to the HA group after 24 hours. Electron scanning microscopy revealed the higher attachment of these cells to the HA + 100 nM TN-C substrate relative to the HA substrate. Epifluorescence imaging and DAPI staining of loaded cells on HA + 100 nM TN-C substrate significantly increased the number of NSCs per field over 72 hours compared to the HA group (P < 0.05). Live and dead assay revealed that the number of live NSCs significantly increased in the HA + 100 TN-C group compared to HA and control groups. The enrichment of HA substrate with TN-C promoted viability and survival of NSCs and could be applied in neural tissue engineering approaches and regenerative medicine.
Introduction
A plethora of experiments have shown that neural stem cells (NSCs) have high potential for self-renewal and differentiation into multiple lineages, such as neurons, astrocytes, and oligodendrocytes. 1,2 It is believed that NSCs are one of the most important candidates for the treatment of neurodegenerative diseases, traumatic brain injury, spinal cord injury, and ischemic stroke. 3,4 These cells migrate toward damaged areas and actively respond to the clues from the surrounding microenvironment. 5,6 Along with differentiation capacity, NSCs possess the ability to secrete neuroprotective compounds in a paracrine manner. 7 -9 Despite these advantages, a main limitation and pitfall are that most NSCs undergo cell death after injection into the lesion sites. 10 According to previous experiments, most of the NSCs die soon after transplantation into the damaged sites. 11,12 The biological integration of NSCs is enhanced via the application of different types of biomaterials and substrates. 13 In this regard, recent experiments confirmed the efficiency of biomaterials in the regulation of cell behavior, viability, and differentiation capacity. 14 Hyaluronic acid (HA) is one of the natural biomaterials that has been extensively investigated in the field of stem cell biology and regenerative medicine. HA is a natural polysaccharide commonly seen in the extracellular matrix (ECM) and participates in brain development. HA maintains NSCs in the quiescence state in the neurogenic regions by the regulation of proliferation and maturation. 15 The amount of HA is high near the lateral ventricles where the NSCs reside. 16 It has been shown that substrates enriched with HA provide a platform with the ability to promote attachment of NSCs. 17,18 Also, ECM proteins are stabilized into scaffolds in the presence of HA via the promotion of covalent links between peptide motifs and HA. The application of HA has also been shown to decrease proinflammatory response by reducing reactive astrocytes around the lesions and stimulation of new ECM microfilaments. 14,19
Based on available data, the combination of HA with other molecules could increase regenerative potential. For instance, tenascin-C (TN-C), a hexametric glycoprotein, is secreted by astrocytes, neurons, and endothelial cells during embryogenesis. 20 -22 TN-C is commonly present in the ECM and participates in cell migration and angiogenesis. In some references, TN-C is termed as a potent cell survival factor. 23 Each TN-C subunit contains an N-terminal tenascin assembly domain, 14.5 epidermal growth factor (EGF)-like repeats, a variable number of fibronectin type III-like repeats (FN III), and a C-terminal fibrinogen-like domain that enables TN-C to easily attach to various cell receptors and ECM components. 24 To the best of our knowledge, there are few studies that correlated with the simultaneous application of TN-C and HA in the dynamic of NSCs. 25 Therefore, the current experiment purposed to investigate the impact of substrate consisted of 1% HA and 100 nM TN-C on NSCs viability and growth rate over 72 hours.
Material and Methods
Isolation, Cultivation, and Expansion of NSCs
Neural stem cells were obtained from micro-dissection of the subventricular zone (SVZ). For this purpose, male newborn mice (n = 3) were anesthetized with the mixture of 100 mg/kg/body weight ketamine and 10 mg/kg/body weight xylazine (Sigma-Aldrich) via intraperitoneal injection followed by cervical dislocation. After disinfecting heads in 70% EtOH solution (Merck), the brains were dissected and placed into cold phosphate-buffered saline (PBS) containing 1% Pen-Strep solution (Gibco). A small triangular piece of the lateral wall of the lateral ventricles was dissected and removed under a dissecting microscope. The tissue was chopped into small pieces and enzymatically digested using 0.05% Trypsin–EDTA solution (Gibco) at 37 °C for 7 minutes. The enzymatic reaction was inhibited using fetal bovine serum. Then, the cell pellet was collected by centrifugation of tubes at 4°C for 5 minutes at 1,500 rpm. The supernatant was discarded, and the cell pellet was washed twice with PBS. The cells were resuspended in a complete NSC culture medium (Gibco) supplemented 20 mM
Flow Cytometry Technique
Cells at passage 3 were subjected to the flow cytometry analysis for identification of cultured NSCs. For this purpose, 106 cells were permeabilized in PBS containing 0.01% Triton (X-100; Sigma-Aldrich) and blocked with 5% goat serum for 1 hour. Thereafter, the cells were resuspended in 100 µL PBS containing PE-conjugated antimouse nestin antibody (eBioscience) overnight at 4 °C. After washing with PBS for 3 times, the cells were analyzed using a BD FACSCalibur system and FlowJo software. 28
Preparation of HA Hydrogel
Sodium hyaluronan gels (Sigma-Aldrich) were prepared by dissolving HA powder in an NSC culture medium at a final concentration of 1% (w/v). The solution was stirred vigorously for 20 minutes. Thereafter, the mixture was transferred into sterile 10-mL syringes and stored at 4 °C overnight. The next day, sodium hyaluronan gels were poured into each well of 96-well plates (SPL Life Sciences). 29,30 The HA hydrogel was maintained on ice to prevent gelation before cell seeding and exposed to UV irradiation for sterilization.
MTT Assay
In the first series of experiments, the effect of the TN-C on the NSCs was evaluated, and in the next step, the effect of combined HA and TN-C was conducted to 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) assay. MTT assay is a colorimetric assay for assessing cell metabolic activity such as cytotoxicity (loss of viable cells) or cytostatic activity (shift from proliferation to quiescence). In this regard, various concentrations of TN-C including 50,100, 200, and 400 nM were mixed with 200 µL culture medium containing 1 × 104 NSCs and transferred into each well of 96-well plates and incubated over 72 hours. After completion of incubation time, the supernatant was discarded and replaced with 2 mg/mL MTT stock. The plates were kept at 37 °C for 4 hours. Dimethyl sulfoxide was then added to dissolve formazan crystals. Optical density was read at 450 nm using a Microplate reader (BioTek) and expressed as percentage of control cells. This assay was performed in triplicate. 31 For evaluation of the combinatorial effect of HA and TN-C on the NSCs survival, a 1% HA solution was placed in each well of 96-well plates and exposed to UV irradiation. Thereafter, a solution containing 100 nM TN-C was overlaid on the HA substrate followed by seeding 1 104 NSCs culture into each well. After 24 hours, the viability rate of NSCs was determined using MTT assay as mentioned earlier. 32
Scanning Electron Microscopy Imaging
After 72-hour incubation time, NSCs loaded on HA and HA + TNC surfaces were fixed with 2.5% glutaraldehyde for 2 hours. Then, the samples were immersed in 1% buffered osmic acid for 2 hours and dehydrated in 4 ascending concentrations of ethanol. The samples were freeze-dried and coated with gold-palladium using an Auto Fine Coater. 31
Investigation of the NSCs Homing and Density Using DAPI Staining
For this purpose, 1 × 105 NSCs were plated in each well of 24-well plates coated with HA and HA-modified TN-C surfaces and kept over 72 hours. DAPI (1 µg/mL for 5 minutes) was added to determine nucleus density per centimeter of the hydrogel. At respective time points, 24, 48, and 72 hours, the culture medium was discarded, and cells at the bottom of the wells were fixed with 4% paraformaldehyde solution. The cells were washed with PBS 3 times (each in 5 minutes). The images were taken by fluorescence microscope. 33
Acridine Orange/ Ethidium Bromide Assay
This assay was used to examine the live and dead NSCs under influence of HA and HA + TN-C at 24, 48, and 72 hours following cell seeding. The assay employs acridine orange (AO), which diffuses into all cells, and ethidium bromide (EB), which is unable to diffuse across a cell membrane unless it is compromised, providing an assessment of overall cell viability. After completion of NSCs culture on control (Plastic surface), HA, and HA + TN-C substrate, 100 µL of a solution containing 0.5 mg/mL AO and 100 µg/mL EB (Sigma-Aldrich) was added to each well and incubated at room temperature for 10 minutes. The cells were then subjected to light/fluorescence microscopy analyses using Olympus IX81 inverted fluorescence microscope equipped with an Olympus DP70 camera (Olympus Corp). All experiments were performed with cells harvested from passage 3 and performed in triplicate. 34
Statistical Analysis
Statistical analysis was done using GraphPad Prism (ver. 8). The numerical data were expressed as means ± standard deviation. Paired t test and 1-way analysis of variance with Tukey post hoc were carried out to detect significant differences between the groups. P < 0.05 was considered statistically significant.
Results
Morphological and Flow Cytometric Analysis of NSCs
In accordance with previous experiments, we found that NSCs showed spherical floating clusters in the neural culture medium between passages 3 and 6 (Figure 1A). In the periphery of neurospheres, the outgrowth of NSCs was observed, showing the typical neurospheres morphology. The phenotypic profile of the cells was examined by flow cytometry using an anti-nestin antibody. Data showed that over 70% total population expresses nestin as an NSC marker (Figure 1B).
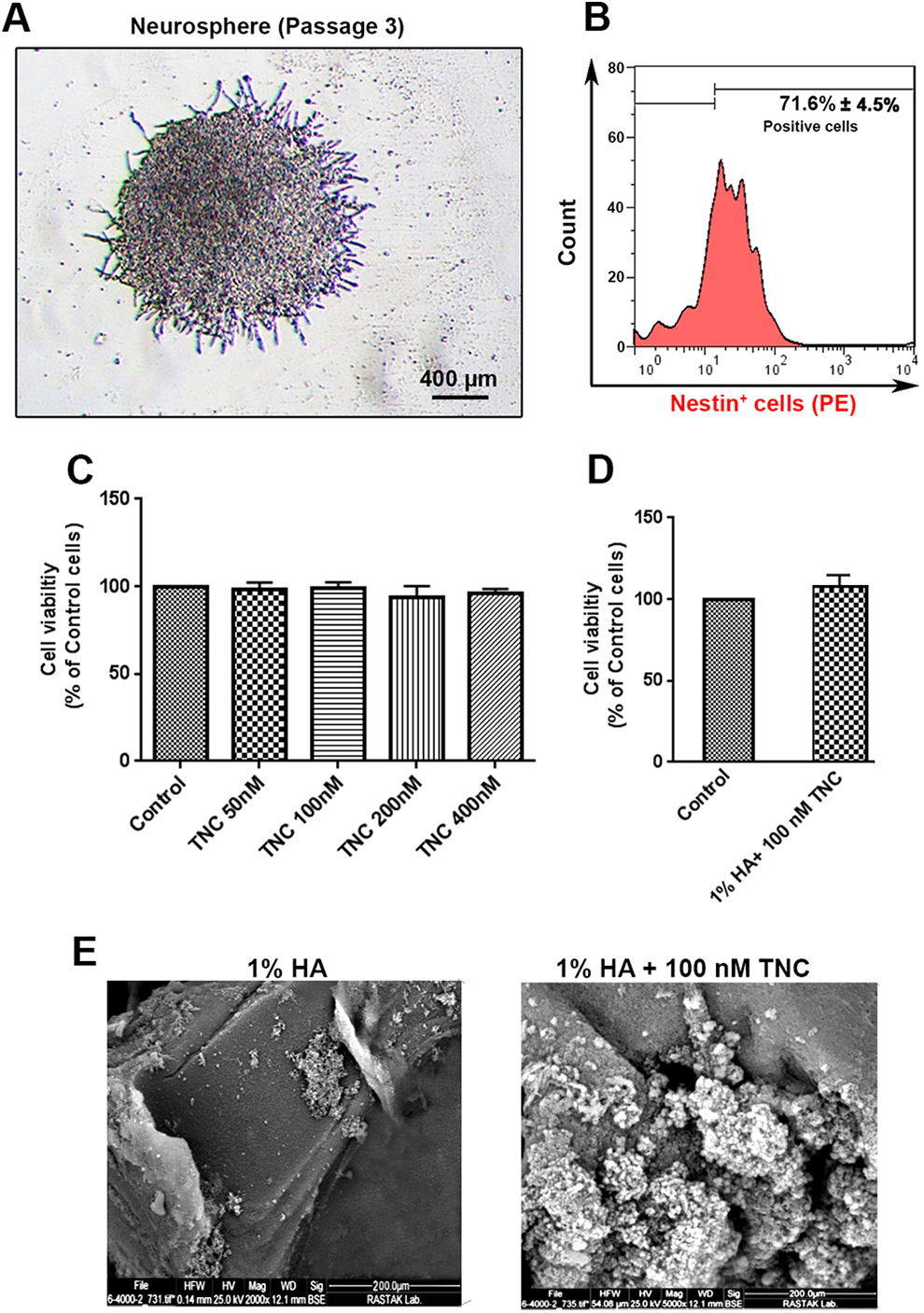
Phase-contrast images of neurospheres derived from newborn mice in passage 3 (A). Flow cytometry analysis of NSCs based on the expression of nestin (n = 3; B). Approximately 71% of NSCs were nestin positive (B). Survival rate of NSCs was evaluated using MTT in the presence of TNC (50,100, 200, and 400 nM) after 24 hours (n = 3; C). Measuring survival rate of NSCs after being plated on 1% HA and 100 nM TN-C after 24 hours (D; student t test; P > .05). The SEM images of the expanded NSCs on the 1% HA and 1% HA + 100 nM TN-C after 72 hours (E). All data are presented as mean ± standard deviation. HA indicates hyaluronic acid; TN-C, tenascin-C; NSCs, neural stem cells; SEM, scanning electron microscopy.
Effect of TN-C on Viability of NSCs
The NSCs were incubated with 4 different concentrations of TN-C including 50,100, 200, and 400 nM for 24 hours, and cell viability was evaluated using MTT assay (Figure 1C). Data showed that the increase in TN-C did not alter the NSCs’ survival rate after 24 hours in vitro. Although incubation of NSCs with 200 and 400 nM TN-C dropped the survival rate slightly, these changes did not yield statistically significant changes. According to the results, we selected 100 nM TN-C for subsequent analyses.
Effect of the Combination of HA and TN-C on Viability NSCs
To assess whether the combination of 1% HA + 100 nM TN-C could change the viability of NSCs, we analyzed the combined effect of HA and TN-C on NSC viability using MTT assay (Figure 1D). According to the data from the MTT panel, we observed NSCs plated on HA + 100 nM TN-C substrate showed a slight, but not significant, increase in survival rate compared to the HA surface (Figure 1D; P > .05). These data showed that the combination of HA and 100 nM TN-C had no cytotoxic effect on the mouse NSCs after 24 hours.
Cell Spreading and Attachment
The attachment of NSCs on the HA and HA + 100 nm TN-C surfaces using SEM imaging (Figure 1E). SEM imaging revealed appropriate attachment and adaptation of mouse NSCs on the HA and HA + 100 nM TN-C surfaces (Figure 1E). According to our data, the density of NSCs was higher on HA + 100 nM TN-C surface compared to the HA substrate. These data showed the potency of both HA and HA + 100 nM TN-C surfaces in the attachment and flattening of NSCs in vitro.
HA+TN-C Biomaterial Increased NSCs Number Per Area of the Substrate
The NSCs were cultivated on HA hydrogel and 100 nM TN-C modified HA hydrogel over 72 hours and compared to the control group (Figure 2). Data showed that the number of NSCs was increased on the HA + 100 nM TN-C surface by time. Compared to the HA surface, we found a statistically significant difference in the number of DAPI-positive nuclei between HA and HA + 100 nM TN-C after 72 hours (P < .05; Figure 2). According to our data, we found a significant increase in the number of NSCs plated on (HA + 100 nM TN-C) surface compared to the HA group (P < .01; Figure 2).
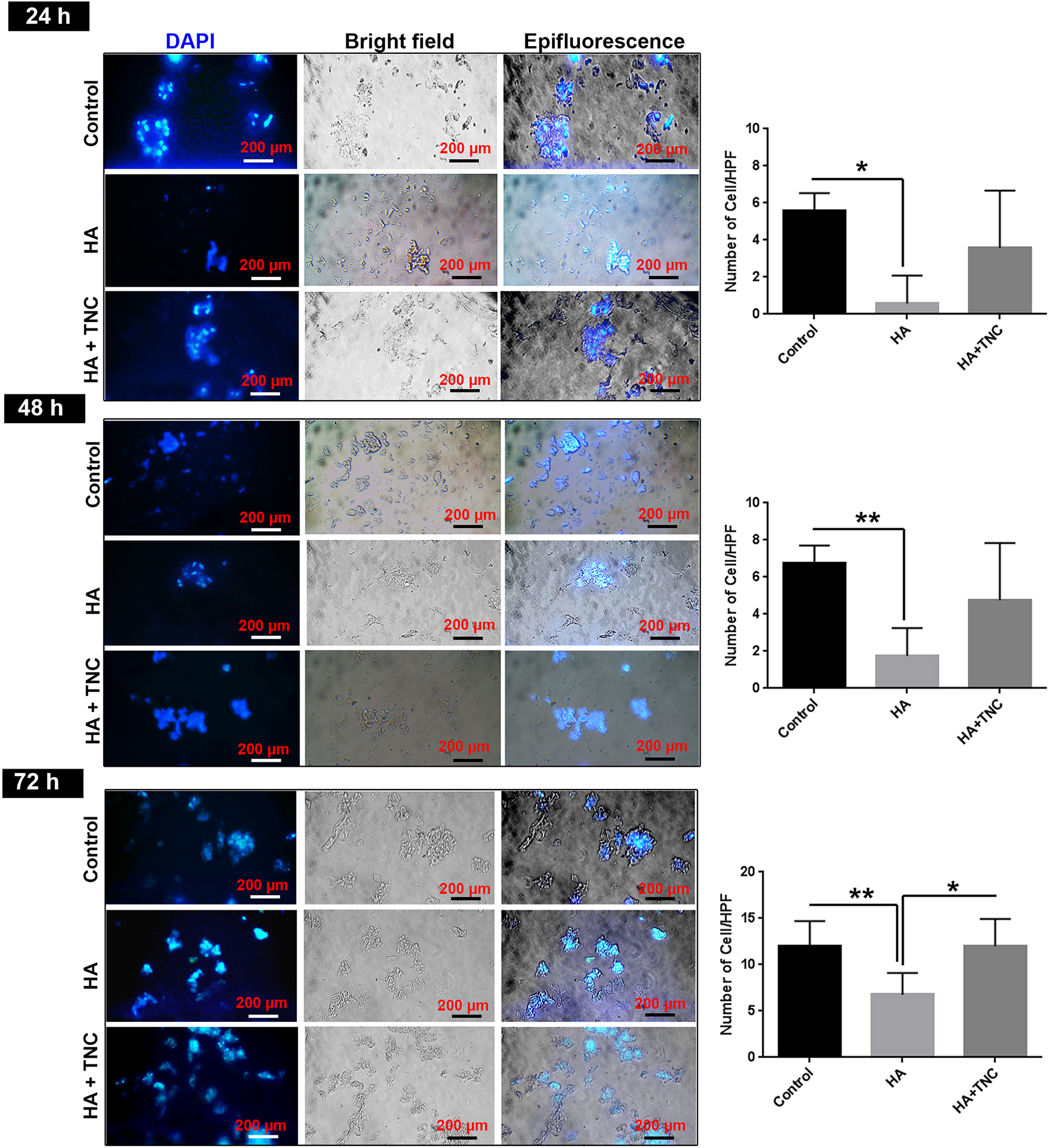
Measuring the NSCs density using DAPI staining after 24-, 48-, and 72-hour time points. Fluorescence imaging revealed the increase in NSCs per area of HA + 100 nM TN-C substrate compared to HA group. One-way ANOVA and Tukey post hoc analysis (n = 4). *P < 0.05; **P < 0.01. All data are presented as mean ± SD. HA indicates hyaluronic acid; TN-C, tenascin-C; NSCs, neural stem cells; ANOVA, analysis of variance; SD, standard deviation.
Evaluation of Live and Dead NSCs Using AO/EB Assay
Cell viability of cultured NSCs within every 3 groups (control, HA hydrogel, and TN-C modified HA hydrogel) was measured at respective time points, including 24, 48, and 72 hours (Figure 3A and B). Similar to DAPI staining, we found that the NSC density was more in control and (HA + 100 nM TN-C) groups compared to the HA substrate (P < 0.05). According to our data, we found a necrotic change in NSCs cultured on the HA surface compared to the control and (HA + 100 nM TN-C) surface. Of note, we found late apoptotic changes in NSCs culture on (HA + 100 nM TN-C) surface. Although both (HA + 100 nM TN-C) and HA substrates exhibited necrotic changes, the density of NSCs on (HA + 100 nM TN-C) surface was more compared to the HA-alone surface (Figure 3A and B). According to our data, the number of live NSCs was significantly increased in (HA + 100 nM TN-C) surface at 3 time points compared to the HA group (Figure 3B; P < 0.05). No significant difference was found in terms of live cells between the control and (HA + 100 nM TN-C) group (Figure 3B; P > 0.05).
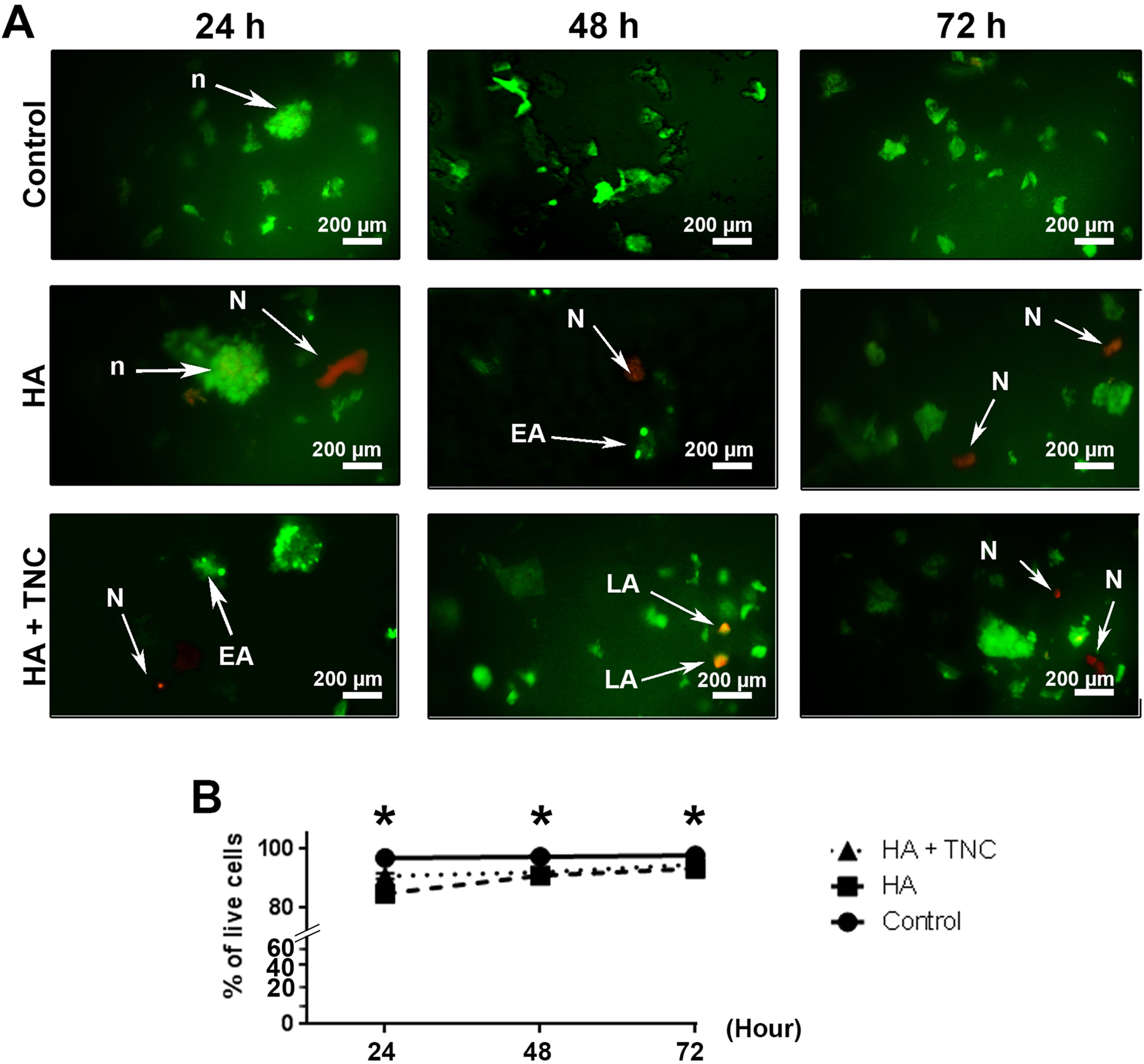
AO/EB staining of cultured NSCs on HA and HA + 100 nM TN-C surfaces after 24, 48, and 72 hours (A and B). Significant difference was observed in the level of live cells between controls, HA, and HA + 100 nM TN-C groups. AO/EB staining revealed the existence of necrotic and late apoptotic cells on the HA and HA + 100 nM TN-C groups. One-way analysis of variance (ANOVA) and Tukey post hoc analysis. *P < 0.05 (n = 5). (Time point 24 hour: Control: 96.7 ± 0.29; HA: 84.65 ± 0.98; and HA + 100 nM TNC: 90.62 ± 0.87; Time point 48 hour: Control: 97.16 ± 0.88; HA: 90.08 ± 1.3; HA + 100 nM TNC: 91.94 ± 0.8; Time point 72 hours: Control: 97.6 ± 0.61; HA: 93.17 ± 0.75; and HA + 100 nM TNC: 94.57 ± 0.61). All data are expressed as mean ± standard deviation (SD). HA indicates hyaluronic acid; TN-C, tenascin-C; NSCs, neural stem cells; AO, acridine orange; EB, ethidium bromide.
Discussion
One of the most important brain ECM is HA, especially in the stem cell niche. In this regard, this substrate is commonly used to fabricate scaffolds for regenerative strategies. 35 The main purpose of CNS regenerative medicine is to arrange the transplanted cells inside structures similar to the naive tissue. 36 Accordingly, we performed an in vitro experiment to assess the combined effect of TN-C and HA on the survival rate and viability of mouse NSCs. MTT analysis and DAPI staining showed increased survival and density rates of NSCs in (HA + 100 nM TN-C) group compared to the HA-plated group.
Previous experiments showed various amounts of HA exist in different parts of the brain tissue. HA could attach to different receptors on the cell surface, including monomeric and clustered CD44, RHAMM, and TLR2/4. 37 In the current experiment, we used low-molecular-weight HA alone or in combination with the TN-C in vitro. The reduction of NSCs proliferation rate in the proximity of HA could be related to the simultaneous activation of CD44 and RHAMM on NSCs surface that could modulate proliferation rate and mitosis, leading to maintenance of stemness. 37 It is thought that HA encloses the brain NSCs and is thought to be involved in quiescence and stemness of NSCs. 38 HA with molecular weights could promote diverse responses from single cells, and even different cell types could respond differently to HA molecular weights. 39 The 3D conformational changes of HA with different molecular weight subunits and unique viscoelastic properties could also affect the biological activity of HA on target cell types. 15 The attachment and enrichment of HA with glycoproteins and linker proteins, such as TN-C, could mimic a condition that triggers proliferation and survival signals in NSCs to respond appropriately to the pathological cues. 40 In support of this claim, the addition of laminin to HA hydrogels increased migration of NSCs in response to stromal cell-derived factor 1-α (SDF1α) through the CXCR4 receptor. 41 It has been demonstrated that TN-C possesses unique EGF-like repeats with the ability to attach EGF receptors. The interaction of TN-C with the EGF receptor promotes survival signaling without receptor internalization in mesenchymal stem cells. 42 It also demonstrated that the sensitivity to some growth factors such as fibroblast growth factor-2 and bone morphogenetic protein 4 increased in the presence of 100 nM TN-C. 43 Of note, the suppression of TN-C in mice abrogated the ability of NSCs to express the EGF receptor in in vitro and in vivo conditions. These data demonstrated that TN-C could increase viability and proliferation of NSCs via the stimulation of receptors and sensitivity to the low dose of cytokines and factors. 44 The current experiment is a preliminary study to show the combined effect of 1% HA and 100 nM TN-C on survival and viability of mouse NSCs. To precisely show the dynamic activity of HA and 100 nM TN-C and distinct concentration, a plethora of experiments is highly needed.
Conclusion
The current experiment showed a positive and stimulatory effect of TN-C in combination with HA on the survival and proliferation of NSCs. The TN-C can increase the modulatory effect of HA on mouse NSCs in vitro. Taken together, it is concluded that the combination of biomaterials and NSCs could propose new and promising therapeutic strategies in neural tissue engineering and regenerative medicine.
Footnotes
Acknowledgment
The authors wish to thank the personnel of the Stem Cell Research Center.
Author Contributions
Shahi, M. contributed to acquisition and drafted manuscript; Mohammadnejad, M. contributed to acquisition and drafted manuscript; Karimipour, M. contributed to conception and design, contributed to interpretation, and critically revised manuscript; Rahbarghazi, R. contributed to conception and design, contributed to acquisition and interpretation, and critically revised manuscript; Abedelahi, A. contributed to conception and design, contributed to acquisition, and drafted manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant (No. 60862) from Tabriz University of Medical Sciences and confirmed by the local ethics committee of Medical Sciences University of Tabriz (IR.TBZMED.VCR.REC.1397.420).
