Abstract
Traumatic brain injury (TBI) consists of a primary and a secondary insult characterized by a biochemical cascade that plays a crucial role in cell death in the brain. Despite the major improvements in the acute care of head injury victims, no effective strategies exist for preventing the secondary injury cascade. This lack of success might be due to that most treatments are aimed at targeting neuronal population, even if studies show that astrocytes play a key role after a brain damage. In this work, we propose a new model of in vitro traumatic brain-like injury and use paracrine factors released by human mesenchymal stem cells (hMSCs) as a neuroprotective strategy. Our results demonstrate that hMSC-conditioned medium increased wound closure and proliferation at 12 h and reduced superoxide production to control conditions. This was accompanied by changes in cell morphology and polarity index, as both parameters reflect the ability of cells to migrate toward the wound. These findings indicate that hMSC is an important regulator of oxidative stress production, enhances cells migration, and shall be considered as a useful neuroprotective approach for brain recovery following injury.
Introduction
Traumatic brain injury (TBI) is a major public health problem with limited treatment resources, 1 especially among male adolescents and elderly people. 2,3 TBI consists of a primary insult resulting from a mechanical force, and this event is amplified by a secondary insult that plays a crucial role in mediating cell damage and death following injury. This secondary insult increases reactive oxygen species (ROS) production, excitatory amino acid release, energy consumption, and perturbates calcium homeostasis. 4 Moreover, increase of energy consumption for tissue regeneration leads to decreased perilesional glucose and mitochondrial dysfunction, 5,6 and this metabolic impairment by low glucose levels may cause cell death following a TBI episode. 7 Therefore, regulation of these events is crucial for cell survival after a primary insult, as this hypermetabolic state might accelerate a secondary insult. 8,9
Despite the major improvements in the acute care of head injury victims, no effective strategies exist for preventing the secondary injury cascade and metabolic dysfunctions that occur afterward. 1,10,11 This lack of success might be due to the fact that most treatments are aimed at targeting neuronal population. 12 Although neuronal death is the main outcome that we wish to ameliorate after brain damage, several studies show that astrocytes play a key role in both normal and pathological central nervous system (CNS) functioning, 13,14 making them viable targets for manipulation after brain injury. 15 Astrocytes are the most numerous cell types in the brain and perform many functions that are essential for the normal activity of neurons in CNS. 16 There is strong experimental evidence that astrocytes protect neurons against TBI 15 providing antioxidant protection, substrates for neuronal metabolism, and glutamate clearance. 17 Therefore, protecting astrocytes and enhancing their supportive function is a promising neuroprotective strategy in TBI.
The use of bone marrow-derived human mesenchymal stem cells (hMSCs) in cerebral ischemia and other pathologies related to traumatic brain-like injuries show promising results as neuroprotective strategies. 18,19 Although hMSCs from human adipose tissue origin have a protective effect by secretion of paracrine factors, 20,21 their neuroprotective mechanisms and downstream signaling are not well understood. In the present study, we assessed the role of hMSC-conditioned medium (hMSC-CM) in scratch-injured cells, associated with metabolic impairment induced by glucose starvation.
Materials and methods
Cell culture
T98G cell line (human glioblastoma) was used as cell model system, where the stock was maintained under exponential growth in Dulbecco’s modified Eagle’s medium (DMEM) (Lonza, Basel, Switzerland), supplemented with 10% fetal bovine serum (FBS) and antibiotics (penicillin/streptomycin and amphotericin; Lonza) at 37°C and in a humidified atmosphere containing 5% carbon dioxide. hMSC was isolated from human adipose tissue and expanded in monolayer culture through passage III. hMSC markers are CD34(−), CD73(+), CD90(+), and CD105(+). hMSC-CM was generated from hMSC cultures between passage III and IV, when cultures were approximately 80% confluent in DMEM supplemented with 10% FBS. hMSC-CM was removed after 48 h and stored at −22°C. Cells were serum deprived (FBS-free medium) before experiments were carried out.
Scratch assay
Cultured cells were trypsinized and plated into 96-well tissue culture plates at a concentration of 10,000 cells per well. After 4–5 days later, when T98G cells reach confluency, a denuded area was produced by scratching the inside diameter of the well with a 10-μl pipette tip. 22 After rinsing twice with phosphate-buffered saline (PBS) 1× buffer to remove debris, cells were treated with different concentrations of hMSC-CM, medium supplemented with 5.5 mM of glucose (balanced salt solution (BSS 5 mM)) and free glucose media (BSS 0 mM), or controls (DMEM + 10% FBS and serum-free DMEM). Since cells were serum deprived and subjected to mechanical injury, we aimed to determine whether FBS was exerting any effect on all analyzed parameters. In this case, we used DMEM + FBS as control. On the other hand, besides this mechanical stimulation, cells were also glucose deprived, and the control condition in this experiment was BSS 5.5.
Microscopy and image analysis
Wound closure was assessed by images taken at various time intervals following scratch until complete closure. The percentage of wound closure was obtained by subtracting the denuded area devoid of cells at any given time point from the original denuded area measured at 0 h in the same well. Digitized 10× magnified black and white images were captured by a digital camera (Optikam PRO 3 digital camera, Optika Digital microscopes, Italy) attached to an inverted microscope (Eclipse TS100, Nikon, Tokyo, Japan). Images were then analyzed using NIH ImageJ software 1.46r (National Institute of Health; download from http://rsbweb.nih.gov/ij/) to measure the area of scratch.
Cell proliferation and viability
The influence of hMSC-CM on T98G cell proliferation or viability was tested using MTT ((3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay (Sigma, St Louis, Missouri, USA). Cells were seeded into 96-well plates in the DMEM culture medium containing 10% bovine fetal serum at a seeding density of 10,000 cells per well, incubated for 4–5 days until they reach confluency, then were subjected to scratch injury, and finally treated according to different experimental paradigms. Viability and/or proliferation were assessed at 8, 12, 24, 48, and 72 h following injury by adding MTT solution for 3 h, and the absorbance was read at 490 nm. Each assay was performed with a minimum of six replicate wells for each condition.
In vitro model of TBI
Scratch injury and glucose deprivation was assessed as an in vitro model of TBI. Scratch assay was performed as described above. Then, to remove glucose from cell medium, wells were rinsed three times with PBS 1× buffer and placed with a glucose-free basic saline solution (BSS 0) containing sodium chloride: 116 mM; calcium chloride: 1.8 mM; magnesium sulfate: 0.8 mM; potassium chloride: 5.4 mM; monosodium phosphate: 1 mM, and 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid: 10 mM at pH 7.4 23,24 or with 5.5 mM glucose as control.
Morphometric analysis
Black and white images were analyzed to assess cell area and polarity. Cells were selected randomly using a scale generated automatically (available at http://www.random.org/integers), and images were processed using ImageJ software. The number of cells was estimated using the fractionator/dissector method by manually counting the number of cells in an area of 0.03 mm2 (300 × 100 µm2). Cell area and polarity were calculated by randomly selecting cells from the images. The polarity index was calculated as the length of the major migration axis (parallel to the direction of movement) divided by the length of the perpendicular axis that intersects the center of the cell. 25 The number of cells, cell area, and polarity were calculated in cells bordering the wound (0–100 µm) and at a dstance of 100–200 µm to measure the effect of the scratch, glucose deprivation, and hMSC-CM in cells morphology. Data were acquired from at least 6 independent cultures with a minimum of 25 cells for each condition.
Flow cytometry measurement of O2− production
To measure the effect of hMSC-CM on superoxide (O2−) production, flow cytometry analysis was assessed using dihydroethidium (DHE; Sigma) in a Guava® EasyCyte™ (Millipore, Billerica, Massachusetts, USA) cytometer. Cells were seeded into 12-well plates in the DMEM culture medium containing 10% FBS at a seeding density of 75,000 cells per well, incubated for 3 days until they reach confluency and then were subjected to scratch assay. Afterward, treatments were applied, and after 12 h, cells were incubated in 10 µm DHE in dark at 37°C for 30 min before flow cytometry analysis. Each assay was performed with a minimum of six replicate for each condition.
Statistical analysis
Data obtained from this study were tested for normal distribution by Kolmogorov–Smirnov test and homogeneity of variance by Levene’s test. Then, data were examined by analysis of variance, followed by Dunnet’s post hoc test for comparisons between controls and treatments and Tukey’s post hoc test for multiple comparisons between the means of treatments and time points. Data are presented as mean ± SEM. A statistically significant difference was defined at p < 0.05.
Results
Effect of hMSC-CM on astrocytes on wound closure and proliferation
To examine the effect of hMSC-CM paracrine factors in the wound-healing process, we cultured cells in DMEM supplemented with 10% FBS and treated with increasing hMSC-CM concentrations (0.5, 2, and 5%) in two-time scale (24 and 48 h). We observed an increase in wound closure of 14.7 (p < 0.0008), 15.1 (p < 0.0003), and 12.5% (p < 0.0045), when cells were treated with 0.5, 2, and 5% of hMSC-CM at 24 h, respectively. Furthermore, wound closure increased by 16 (p < 0.0031), 17.5 (p < 0.0007), and 13 (p < 0.0361) at 48 h, when cells were treated with 0.5, 2, and 5% of hMSC-CM, respectively (Figure 1(a)). Complete regeneration was almost reached at 48 h in cells treated with hMSC-CM (Figure 1(b)), while full regeneration in nontreated cells was only observed at 72 h following scratch (data not shown).
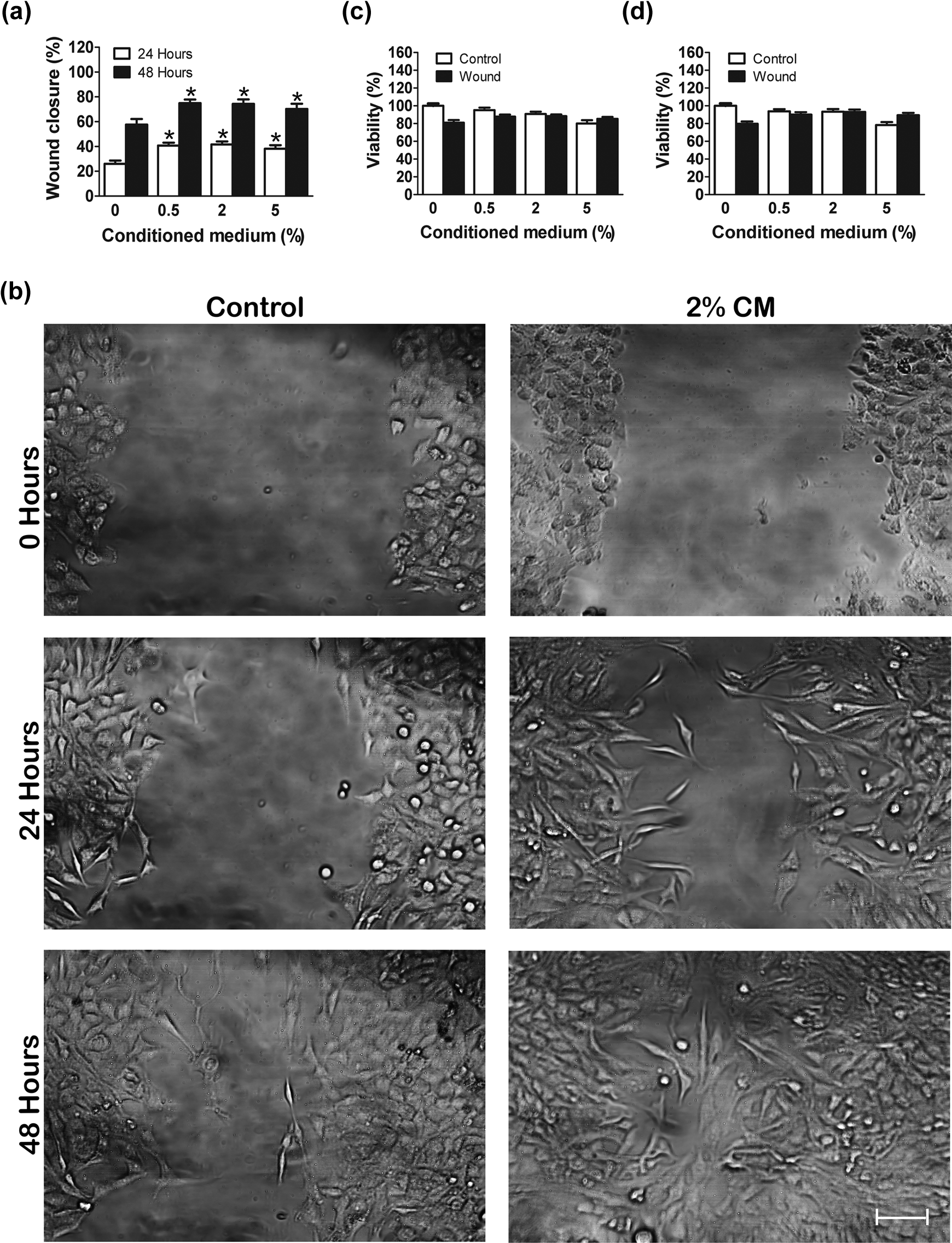
Wound closure in scratched cells supplemented with FBS and treated with different hMSC-CM concentrations at 24 and 48 h. (a) Representative images of scratch-injured T98G cells at 0, 24, and 48 h in control versus 2% hMSC-CM. (b) MTT results show no changes in cell viability with hMSC-CM at 24 (c) and 48 h (d). MTT values were normalized to control (DMEM + FBS group). Error bars indicate SEM. *p < 0.05 with respect to 0% hMSC-CM with scratch. Bar: 100 µM. FBS: fetal bovine serum; hMSC-CM: human mesenchymal stem cells-conditioned medium; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; DMEM: Dulbecco’s modified Eagle’s medium.
Given that the scratch assay was performed in the absence of mitomycin C, we investigated whether the effect of hMSC-CM on wound regeneration was due to proliferating cells. Our results show that hMSC-CM did not have any effect on proliferation, as evidenced by viability using MTT assay when compared with control at 24 h (Figure 1(c)) and 48 h (Figure 1(d)).
Glucose deprivation affected wound closure and viability
Previous studies investigating the metabolic fate of glucose after TBI have shown that, after the initial hours following injury, interstitial glucose concentration decreases in the damaged area, 6,26 thus contributing to secondary brain damage. Therefore, impaired energy metabolism is a potential target of TBI therapy. To mimic this event, scratch-injured cells were subjected to glucose deprivation (BSS 0 mM) or cultured with 5.5 mM glucose at 12 h and 24 h. Glucose deprivation reduced wound closure by 11% (p < 0.05) at 12 h and 35% (p < 0.001) at 24 h compared with the control (Figure 2(a) and (b)). The effect of FBS on wound closure was also assessed. Interestingly, FBS facilitated wound closure by 23% (p < 0.001) at 24 h but not at 12 h, suggesting a time-dependent effect on wound regeneration. In glucose-deprived cells, viability is reduced by 50% (p < 0.001) at 12 h and 10% (p < 0.0001) at 24 h, respectively, compared with the control (Figure 2(c)). On the contrary, FBS administration did not induce differences in cell viability at 8, 12, and 24 h. Based on these observations, we assessed wound regeneration at 12 h in the next set of experiments.

Effects of glucose deprivation in (a, b) wound closure and (c) cell viability at 12 and 24 h. Glucose withdrawal reduces wound closure and viable cells at these time points. MTT values were normalized to control cells (DMEM + FBS). Error bars indicate SEM. *p < 0.05 and **p < 0.01 with respect to control. Bar: 100 µm. MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; DMEM: Dulbecco’s modified Eagle’s medium; FBS: fetal bovine serum.
hMSC-CM increased viability in glucose-deprived cells at 12 h
First, we determined whether hMSC-CM protects cells from glucose deprivation. We observed that hMSC-CM did not affect viability in both controls and BSS 5.5 mM-treated cells at 12 h. However, hMSC-CM at 0.5, 2, and 5% increased viability in glucose-deprived cells by 13 (p < 0.05), 35 (p < 0.001), and 30% (p < 0.004), respectively, when compared with nontreated cells. Interestingly, we found that 2 and 5% hMSC-CM induced a greater cell viability compared to other concentrations (Figure 3(a)), though no statistical differences were seen when comparing 2% hMSC-CM group with 5% hMSC-CM group. Therefore, 2% hMSC-CM was used in next experiments.

(a) Dose–response of hMSC-CM in glucose-deprived cells at 12 h. 2% hMSC-CM facilitates wound closure and increases cell viability (b, c) almost to control conditions (d). MTT values were normalized to control cells. Error bars indicate SEM. *p < 0.05 with respect to control cells; #p < 0.05 with regard to non-hMSC-CM-treated cells. Bar: 100 µm. hMSC-CM: human mesenchymal stem cells-conditioned medium; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
2% hMSC-CM facilitated wound closure and proliferation in a TBI in vitro model at 12 h
We proposed a new model of TBI using a scratch assay combined with glucose deprivation. We found that 2% hMSC-CM increased by 12% wound closure compared with the control (p < 0.004; Figure 3(b) and (c)). To determine whether this increase in wound closure was due to either migration, proliferation, or both, we assessed the MTT viability assay. No significant differences were found in cells subjected only to scratch injury; however, 2% hMSC-CM increased viability nearer to 35% (p < 0.002) and 40% (p < 0.001) in scratched- and glucose-deprived cells or only deprived cells, respectively (Figure 3(d)). To assess whether proliferation might be implicated in wound closure, we determined the relative number of cells according to the distance from the wound. Our results showed that the relative number of cells bordering the injury was 25 ± 1.0/0.03 mm2 and 26 ± 1.8/0.03 mm2 at 100–200 µM far from the wound in TBI cells. Upon hMSC-CM treatment, no statistical differences were observed in cells bordering the lesion (24 ± 1.8/0.03 mm2) or those located at 100–200 µM (30 ± 2.1/0.03 mm2).
2% hMSC-CM induced changes in cell morphology at 12 h
To assess the effect of 2% hMSC-CM in cell morphology, serial pictures were taken using a bright-field microscope according to the distance from injury. hMSC-CM-treated-TBI cells bordering injury (0–100 µM) have augmented cell area when compared to those located at 100–200 µM from the border (p < 0.05; Figure 4(a)). The polarity index indicates a state of migrating cells, that is, when the higher this index is, the more is the disposition to migration. 25 Following injury, cells tend to migrate in order to close wound and promote regeneration. 27,28 In this context, we did not find statistical differences in cell polarity in hMSC-CM-treated cells subjected only to scratch injury; in contrast, when cells are submitted to both scratch injury and glucose deprivation, hMSC-CM treatment increased cell polarity (p < 0.001), indicating a higher capability of migration in cells toward injury. Additionally, the polarity index was higher in TBI-treated cells bordering the lesion in comparison with those located at 100–200 µM distance (p < 0.04; Figure 4(b) and (c)).

Effect of 2% hMSC-CM in cell area (a) and polarity index (b) in TBI cells at 12 h, according to the distance of wound. (c) 2% hMSC-CM increased cell area and polarity in cells bordering the wound. Arrows indicate non-migrating cells (left) and a migrating cell (right) with altered cellular morphology. Error bars indicate SEM. &p < 0.05 with respect to the distance from the wound; #p < 0.05 compared to non-hMSC-CM-treated cells. Bar: 200 µM indicating 0–100 and 100–200 µM distance from the wound. hMSC-CM: human mesenchymal stem cells-conditioned medium; TBI: traumatic brain injury.
hMSC-CM reduced O2− production in an in vitro model of TBI at 12 h
In TBI, energy depletion is the result of mitochondria impairment and ROS production is one of the major causes of mitochondrial damage, 4 especially the free radical, O2−. To examine a possible mechanism by which hMSC-CM is increasing wound closure, migration and proliferation, and changes in cell morphology, we assessed its effect in the intracellular production of O2− by DHE fluorescence using flow cytometry. To observe whether hMSC-CM was affecting production of O2−, we performed a dose–response curve using 0.5, 2, and 5% of hMSC-CM. We found a dose–response relation in 0.5% of hMSC-CM (a reduction by 100× O2− production; p < 0.001) and 2% hMSC-CM (a decrease by 200× O2− production; p < 0.0001). Moreover, our findings showed similar results when glucose-deprived cells were treated either with 2 or 5%, demonstrating that higher hMSC-CM concentrations did not reflect on a better reduction of O2− levels (Figure 5(a)); therefore, 2% hMSC-CM was later used in all experimental groups. hMSC-CM at different concentrations did not induce changes in O2− production in control conditions (DMEM + FBS; data not shown). We found an augmented production of 200× in O2− generation compared with the control (p < 0.0001) in TBI cells. Moreover, O2− levels are decreased by 200× when cells are treated with 2% hMSC-CM (p < 0.0001), indicating a full recovery to basal levels. On the other hand, scratch injury alone did not affect production of O2− (Figure 5(b) and (c)).
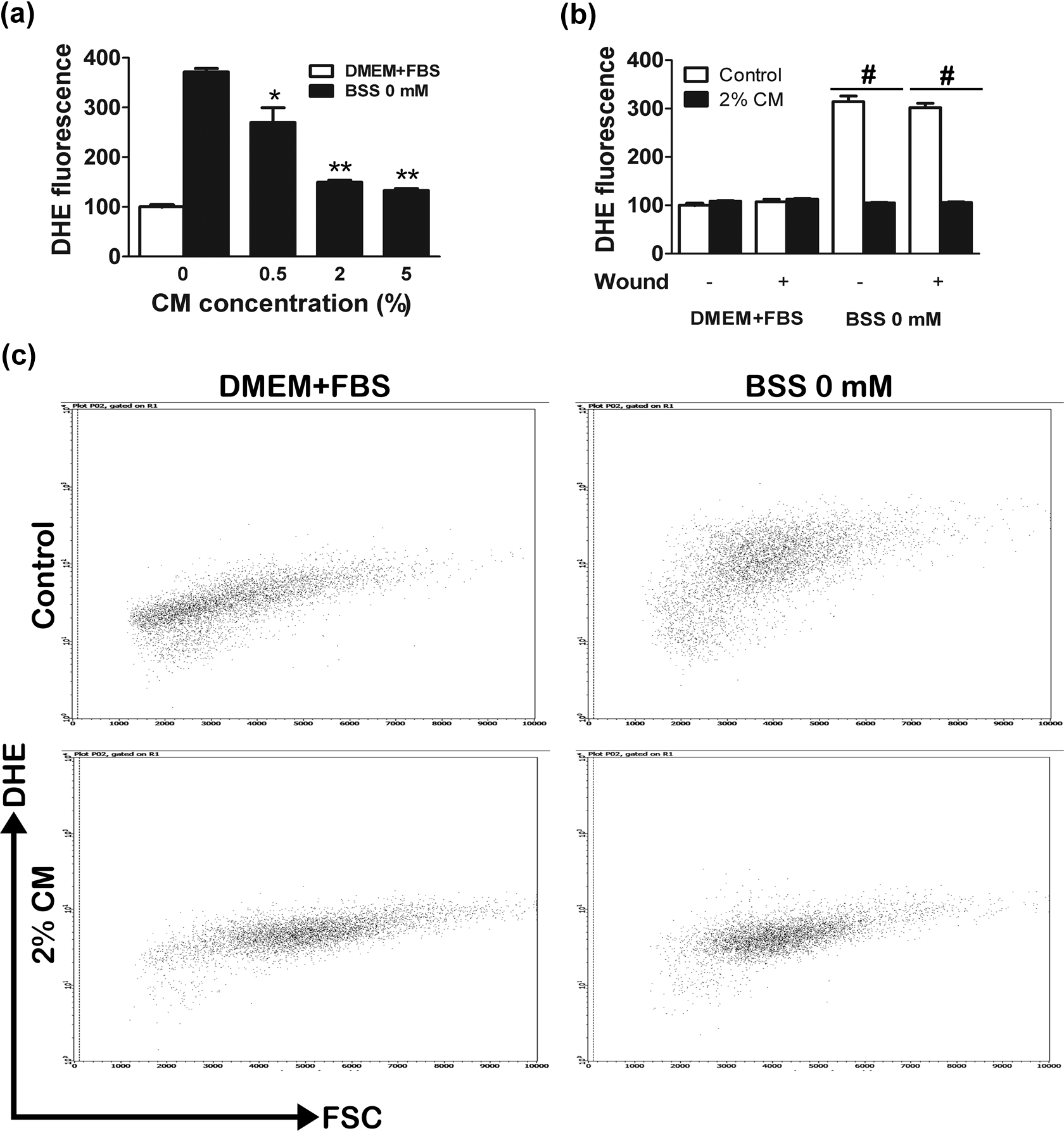
(a) Dose–response of hMSC-CM in O2− production in glucose-deprived cells. (b, c) 2% hMSC-CM reduces O2− production to control conditions in TBI cells at 12 h. DHE relative values were normalized to control cells. Error bars indicate SEM. *p < 0.05 and **p < 0.01 compared with glucose-deprived cells with 0% of hMSC-CM. #p < 0.05 compared with non-hMSC-CMtreated cells. hMSC-CM: human mesenchymal stem cells-conditioned medium; O2−: superoxide; TBI: traumatic brain injury; DHE: dihydroethidium.
Discussion
Adipose hMSC represents an alternative source of multipotent cells. 29 These cells are easier to isolate and are relatively abundant compared with bone marrow MSCs, 30 which make them a better stem cell source for wound repair and regeneration after a TBI episode. On the other hand, T98G cells are an astrocyte-derivative cell intensively used as a model to study about astrocytes 31 –33 due to its biological resemblance with primary astrocyte. These cells express the glial fibrillary acidic protein, a specific marker of astrocyte and share other phenotypes to primary astrocyte such as CD68 and human leukocyte antigen-I. 31 Previous studies also reported the use of other tumor cell lines for migration study purposes. 34,35 In this study, we have investigated whether T98G cells responses to injury are altered by paracrine factors from hMSC, which are known for protecting neurons, fibroblasts, and keratinocytes. 36,37 We show that paracrine factors signaling from hMSC can regulate wound closure, proliferation, morphological changes, and oxidative stress in an in vitro TBI model.
The TBI in vitro model proposed in this work is not considered a novel approach to study TBI. In a previous work by Allen et al., 7 a TBI in vitro model that combines a mechanical trauma and a metabolic impairment in neurons was proposed, as both phenomena are common and associated features in traumatic-injured mammals. 38,39 Although they use a glucose deprivation to mimic metabolic impairment, the trauma per se was generated by a mechanical punch device, which is a different experimental paradigm from ours, a scratch assay approach instead. 22
Since hMSC-CM was collected in a nonfree serum medium, the effects of FBS in our experiments were assessed. Previous reports have shown that FBS may affect proliferation and cell migration in various experimental studies. 40,41 Despite these observations, we did not observe any effect of serum in all analyzed parameters, demonstrating that FBS is not playing a role in cell proliferation and migration at 12 h. hMSC-CM, as like other mediums, contains some trophic factors that might interfere with some cellular functions. 20,42 Despite this caveat, we tested low hMSC-CM concentrations (2%), which contains approximately 0.001% FBS, and observed that it did not exert any effect on cells response following injury, contrasting to a previous study reporting FBS-induced proliferation in other cell types. 40,43 Furthermore, serum at 0.5 and 10% concentrations did not exert effect on viability of deprived astrocytes for 24 h, 41 thus corroborating with our results.
hMSC-CM, as we expected, increased wound closure in scratch–injured cells but not glucose-deprived cells in control conditions. This healing process might be attributed to migration, as CM did not induce proliferation (Figure 1(c) and (d)) at 24–48 h, as supported by other studies on fibroblasts and keratinocytes. 36,44 In TBI cells, MTT results showed increased proliferation at 12 h, despite the unchanged relative number of cells bordering the lesion and those located at 100–200 µM. Although these differences may not be attributed only to migration, and MTT assay is reporting a whole cell population assessment, cell counting is reflecting only a small percentage of these cells. In this context, this process cannot be attributed only to migration, since hMSC-CM induces proliferation in the TBI group at 12 h (Figure 3(d)). In hypoxic–ischemic conditions, bone marrow MSCs release several paracrine factors that are known to regulate cell proliferation, 45 –47 supporting both the idea of a paracrine-mediated effect on cell proliferation.
Changes in cell morphology and polarity may be required for cells to migrate. Cells may acquire a spatial asymmetry to turn intracellular forces into cell body translocation. 48 One manifestation of this asymmetry is a polarized morphology (Figure 4(c)), and an increase in cell size is associated with biosynthetic activity and might be correlated to migration and proliferation phenomena. 49 We observed that cell area and polarity are decreased in cells nearby the injury (0–200 µM distant), and we hypothesize that this might be due to glucose withdrawal, as no changes in morphology were seen in cells subjected only to scratch. Conformational modifications in cellular structures are energy demanding, 48 and glucose deprivation markedly reduces cellular adenosine triphosphate levels. 50 These reduced changes in morphology and polarity are rescued when cells are treated with CM, until it reach control values (nondeprived cells), and this effect is tightly dependent upon distance from the injury, with higher values at cells bordering the lesion. Cells surrounding lesion area present augmented cell area and polarity index, demonstrating that these cells might be directly contributing to cover the denuded area. In fact, administration of 2% CM brings polarity index values close to control levels in cells surrounding injury, suggesting that most cells located in this area might be migrating toward the lesion in order to regenerate the wounded area. Since migration and proliferation might be playing a role in wound regeneration, it is possible that both processes are acting synergically, and this effect is potentiated by CM.
It is possible that increased O2− production, a common secondary mechanism following brain injury, 51,52 could affect wound closure, viability, and induce morphological changes in T98G cells. According to this, we found an increased O2− production, and CM administration was able to reduce it to basal levels, suggesting its ability to regulate oxidative stress. Previous studies have shown that bone marrow-derived hMSC transplantation reduces ROS following myocardial infarction, 19 cerebral ischemia, 18 and diabetes, 53 possibly by paracrine and autocrine mechanisms. Furthermore, ROS production may reduce cell migration 25,54 and viability, 55 and changes in both, spatial and temporal organization of the actin filaments leading to altered cytoskeleton shape. 56 Therefore, alterations in cell cytoskeleton, that is, actin filaments, by ROS, and the fact that hMSC-CM rescued cell morphology and polarity almost to control values in our study, demonstrates that hMSC-CM may reduce ROS thus affecting cell morphology. In conclusion, hMSC-CM is a source of paracrine signal that may regulate T98G cell regeneration via proliferation and migration, effectively reduce oxidative stress, and induce important morphological changes in cells surrounding the wound. Altogether, our results give support to the idea that endogenous application of hMSC paracrine factors shall facilitate brain recovery following traumatic injuries.
Footnotes
Funding
This work was supported in part by grants PUJ IDs 4327 and 4367 to GEB.
