Abstract
Arsenic trioxide (As2O3) is a known environmental toxicant and potent chemotherapeutic agent. Significant correlation has been reported between arsenic exposure (including consumption of arsenic-contaminated water and clinical use of As2O3) and dysfunction in the nervous system. In this study, we aimed to elucidate the effect of resveratrol with neuroprotective activities on As2O3-induced oxidative damage and cerebral cortex injury. Twenty-four healthy Chinese Dragon Li cats of either sex were randomly divided into four groups: control (1 ml/kg physiological saline), As2O3 (1 mg/kg), resveratrol (3 mg/kg) and As2O3 (1 mg/kg) + resveratrol (3 mg/kg). As2O3+resveratrol-treated group were given resveratrol (3 mg/kg) 1 h before As2O3 (1 mg/kg) administration. Pretreatment with resveratrol upregulated the activities of antioxidant enzymes and attenuated As2O3-induced increases in reactive oxygen species and malondialdehyde production. In addition, resveratrol attenuated the As2O3-induced reduction in the level of reduced glutathione and the ratio of reduced glutathione to oxidised glutathione, and accumulation of arsenic in the cerebral cortex. These findings support neuroprotective effect of resveratrol on As2O3 toxicity in feline brain and provide a better understanding of the mechanism that resveratrol modulates As2O3-induced oxidative damage and a stronger rational for clinical use of resveratrol to protect brain against the toxicity of arsenic.
Introduction
A summary of documented incidents throughout the world involving contamination of groundwater by arsenic has been reported by Chakraborti et al. 1 Arsenic is a potent toxic metalloid and has been shown to cause severe cytotoxicity in various types of cells. 2,3 Some clinical case reports have indicated that patients exposed to arsenicals displayed severe dysfunction in the nervous system. 4,5 In experimental animals, it has been found that arsenicals induced brain injuries that caused behavioural alterations as well as changes in the morphology and apoptosis of brain cells. 6,7 Arsenic easily crosses the blood–brain barrier 8 and accumulates in the brain leading to neurobehavioral abnormalities. 9 On the other hand, arsenic trioxide (As2O3) has been widely used and shown substantial efficacy in treating patients with relapsed or refractory acute promyelocytic leukaemia (APL). 10,11 Unfortunately, for some patients the clinical usefulness of As2O3 has been limited by its toxicity. 12
Due to increasing exposure to environmental neurotoxicants, there is an increasing interest to investigate the prophylactic and protective efficacy of natural products found in plant extracts. Usually, plant extract (green tea extract, curcumin, garlic extract, etc.) 13,14 has a benefit effects on the oxidant/antioxidant status that was observed in the previous studies. Resveratrol (trans-3, 4′, 5-trihydroxystilbene) is a natural polyphenolic compound mainly found in the skin of grapes. It is well known for its phytoestrogenic and antioxidant properties. 15,16 It has been shown to have a neuroprotective role in various models in vitro and in vivo. 17,18 Resveratrol possesses diverse biochemical and physiological properties, including estrogenic, antiplatelet, and anti-inflammatory properties. Preclinical and clinical studies have demonstrated that resveratrol has the ability to prevent a wide range of disorders, including cardiovascular diseases, cancers, diabetes, arthritis, aging-related disorders, 19,20 cerebral ischemic injury, DNA damage, apoptosis and low-density lipoprotein (LDL) oxidation. 21 Duraj et al. 22 reported that resveratrol had potential ability to interfere with the cell cycle and exert various activities in cooperated treatments with selected anticancer drugs resulting in some cases in the cell death induction.
Although liver and kidney have been suggested to be target of arsenic, 23 the metalloid is distributed into the brain regions after crossing the blood–brain barrier. In view of all above facts, the present study was undertaken to evaluate whether or not resveratrol can ameliorate the toxic effects of As2O3 and provide a better understanding of the mechanisms of the action of resveratrol in modulating As2O3-induced oxidative damage as well as a rationale for further clinical study of resveratrol used with As2O3 during chemotherapy or as a adjunctive therapy agent after As2O3 exposure.
Materials and methods
Materials
As2O3 parenteral solution was obtained from Harbin Yida Pharmaceutical Limited Company (Harbin, China). Resveratrol (Sigma Chemical Company, St. Louis, MO) was dissolved with a mixture of dehydrated alcohol injection (5 ml) and physiological (0.9%) saline solution (5 ml), the final concentration was 10 mg/ml. Kits used for the detection of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), oxidised glutathione (GSSG), reduced glutathione (GSH) and malondialdehyde (MDA) were purchased from Jian-cheng Bio-engineering Institute (Nanjing, China). Other reagents were purchased from Sigma-Aldrich.
Animals
Since the pharmacokinetic profiles of drugs used for cats more closely correspond with that used for humans, 18,24 we selected cats for the present study. The study was performed on 24 healthy Chinese Dragon Li cats of either sex (12 males, 12 females) maintained in a air-conditioned rooms at 21°C ± 4°C and kept in individual stainless steel cages (during experimental phases). Weights and ages ranged from 2.8 to 3.5 kg and 1.5 to 2 years, respectively. The animal care and conduct of the study were performed in accordance with the Ethics Committee for Animal Experiments (Northeast Agricultural University, Harbin, China).
Experimental design
All animals were randomly divided into four groups: the control, As2O3-treated, As2O3 + resveratrol-treated and resveratrol-treated groups. All treatments were given via the foreleg vein on alternate days for 3 days (i.e., days 1, 3 and 5) with measurements made on the 6th day. Resveratrol (Sigma-Aldrich; 100 mg) was dissolved with a mixture of dehydrated alcohol injection (5 ml) and physiological (0.9%) saline solution (5 ml), and the final concentration was 10 mg/ml. The single doses of resveratrol and arsenic we selected were based on previously published dosage regimen that was shown to be biologically and therapeutically active in experimental systems. 16,25 In the control group, cats were injected with physiological (0.9%) saline (1 ml/kg); in the As2O3-treated group, cats were treated with As2O3 (1 mg/kg); and in the As2O3 + resveratrol-treated group, cats were given resveratrol (3 mg/kg) 1 h before As2O3 (1 mg/kg) administration. The resveratrol-treated group received three doses of resveratrol alone (3 mg/kg). Twenty-four hours after the last injection, all cats were killed by an overdose of pentobarbital sodium (100 mg/kg). 26 Cerebral cortex sections were collected and used for further experimentation.
Biochemical determination
The cerebral cortex were rapidly excised and homogenised in phosphate-buffered saline (PBS, pH 7.4) with an Ultra-thurax T25 homogenizer. Supernatant homogenate was collected after centrifugation at 10,000g for 10 min at 4°C. SOD, CAT and GPX activities and MDA level were measured using assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), according to the manufacturers’ instructions respectively. 27
Analysis of GSH level and GSH-GSSG ratio
The levels of GSH and GSSG content in cerebral cortex were measured by colorimetric method as described in the manufacturer’s instructions, and the ratio was calculated as follows: ratio = (GSHtotal − 2GSSG)/GSSG.
Measurement of reactive oxygen species level in the cerebral cortex
The amount of reactive oxygen species (ROS) in the cerebral cortex was measured using 2′,7′-dichlorofluorescin diacetate, which is converted into highly fluorescent dichlorofluorescin by cellular peroxides (including hydrogen peroxide). The assay was performed as described in the literature. 28 Fluorescence was determined at excitation and emission wavelengths of 488 nm and 525 nm, respectively, using a fluorescence plate reader (LS-55; PerkinElmer, Buckinghamshire, UK).
Morphological examination
The cerebral cortex sections from cats were fixed in 10% formaldehyde. Fixed tissues were trimmed, embedded in paraffin, sectioned into 2-µm sections and stained with hematoxylin and eosin (H&E) performed as described by Huang et al. 26 Morphological examination was conducted under a light microscope (BX-FM; Olympus Corp, Tokyo, Japan).
Determination of total arsenic in the cerebral cortex
Arsenic contents in the cerebral cortex were analysed following the method of Cui et al. 29 Briefly, approximately 0.5 g tissue sample was digested with a mixture of HNO3-HCLO4 solution (ratio 1:3 v/v) for 2 days at 130°C. After HNO3 was removed by evaporation, digested samples were diluted with deionised water. Arsenic concentrations were measured by atomic fluorescence spectrometry (AFS) (Beijing Jitian Instrument Co., Ltd., Beijing, China).
Statistical analyses
Data are the mean ± standard error. Statistical analyses were undertaken by one-way ANOVA and the Student’s t-test. A two-tailed p < 0.05 was considered significant.
Results
General health condition
Cats from As2O3-treated group showed symptoms of severe diarrhea, drowsiness, vomiting, skin pruritus and hyperpigmentation compared with the control group. The cats treated with resveratrol along with As2O3 only showed slight diarrhea and vomiting compared with the group exposed to As2O3 alone.
Effect of resveratrol on ROS production
The brain ROS level increased significantly (p < 0.05) after arsenic exposure (Figure 1). Administration of resveratrol along with As2O3 antagonised the toxic effects of As2O3 and significantly (p < 0.05) decreased the ROS levels compared with the group exposed to As2O3 alone.

Arsenic-induced change in ROS in the cerebral cortex and their response to pretreatment with resveratrol (Rev). Values are mean ± S.E.; n = 6; *p < 0.05 vs control group, # p < 0.05 vs As2O3-treated group. ROS: reactive oxygen species; As2O3: arsenic trioxide.
Effect of resveratrol on activities of antioxidant enzymes
As shown in Figure 2, the activities of GPX (Figure 2(a)), CAT (Figure 2(b)) and SOD (Figure 2(c)) in As2O3-treated group were reduced compared with the control group. However, pretreatment with resveratrol caused a significant increase (p < 0.05) in the levels of SOD and CAT compared with those seen in the As2O3-treated group.

Activities of GPX (a), CAT (b) and SOD (c) in cerebral cortex were measured. Data are the mean ± S.E.; n = 6; *p < 0.05 vs control group, # p < 0.05 vs As2O3-treated group. CAT: catalase; GPX: glutathione peroxidase; SOD: superoxide dismutase; As2O3: arsenic trioxide.
Effect of resveratrol on MDA levels
The production of MDA in As2O3-treated group was significantly higher than that in the control group (p < 0.05; Figure 3). However, pretreatment with resveratrol caused a significant decrease (p < 0.05) in the levels of MDA compared with those seen in the As2O3-treated group.

MDA levels in cerebral cortex were measured. Data are the mean ± S.E.; n = 6; *p < 0.05 vs control group, # p < 0.05 vs As2O3-treated group. MDA: malondialdehyde; As2O3: arsenic trioxide.
Effect of resveratrol on GSH level in the cerebral cortex
The contents of GSH in the control and experimental groups are shown in Figure 4(a). Significant (p < 0.05) arsenic-induced depletion of GSH was observed in the As2O3-treated group compared with the control. Simultaneous supplementation of resveratrol along with arsenic in the resveratrol + As2O3-treated group significantly (p < 0.05) elevated the levels of GSH compared with the As2O3-treated group.
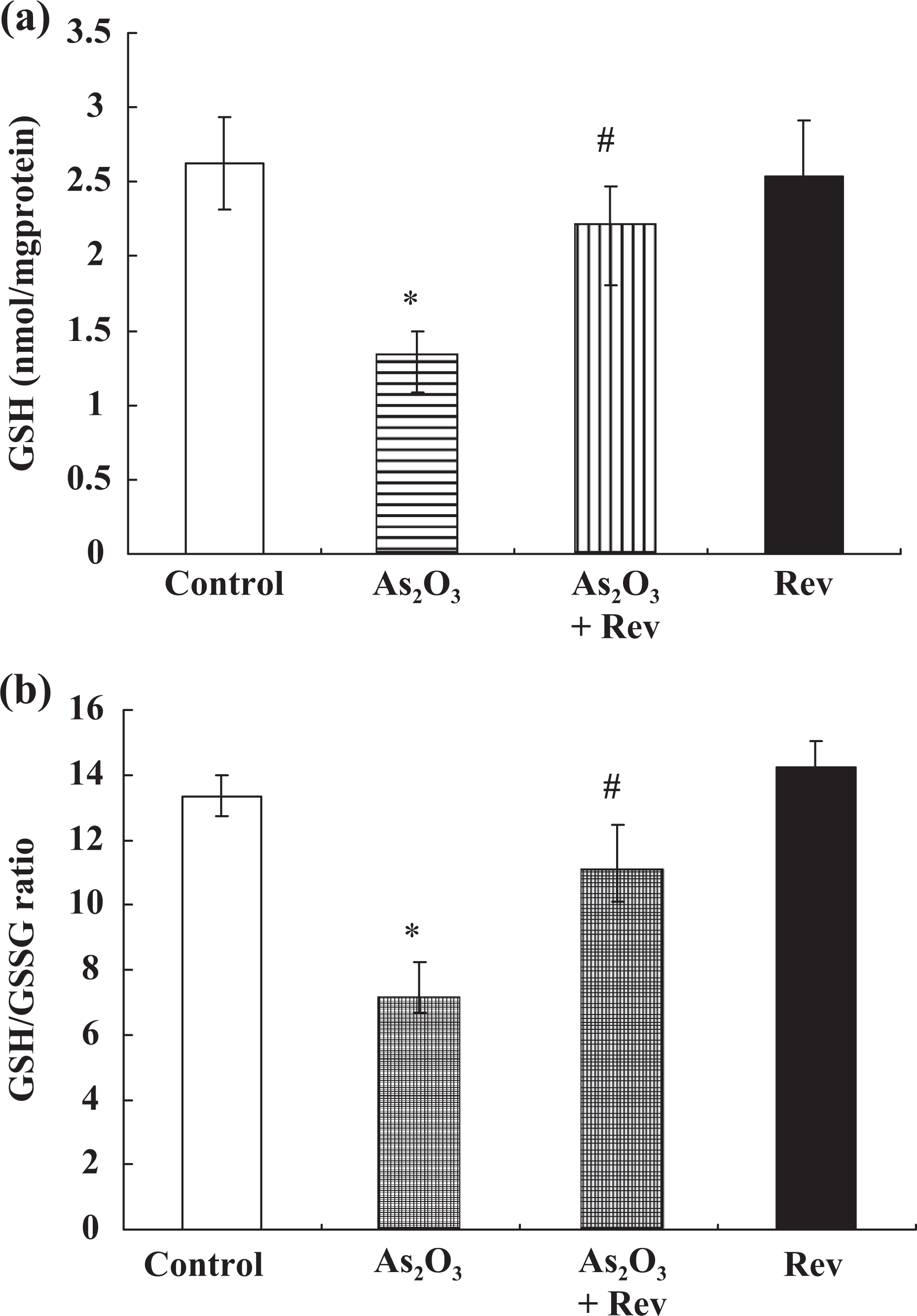
Arsenic-induced changes in the levels of GSH and GSH-GSSG ratio in cerebral cortex, and their response to pretreatment with resveratrol. Values are mean ± SEM; *p < 0.05 vs control group, # p < 0.05 vs As2O3-treated group. GSH: reduced glutathione; GSSG: oxidised glutathione; SEM: standard error of mean; As2O3: arsenic trioxide.
Effect of resveratrol on the ratio of GSH and GSSG in the cerebral cortex
The ratios of GSH and GSSG in cerebral cortex from different groups are shown in Figure 4(b). The glutathione redox ratio of As2O3-treated group were significantly decreased (p < 0.05) compared with the control group. However, pretreatment with resveratrol reversed the glutathione redox status of cerebral cortex tissues (p < 0.05).
Effect of resveratrol on arsenic accumulation
Arsenic accumulation in the cerebral cortex is shown in Figure 5. Although the two groups exposed to As2O3 accumulated more arsenic than those seen in the control (p < 0.01), significantly higher levels of arsenic were observed in the cerebral cortex of the As2O3-treated group than in the resveratrol + As2O3-treated group (p < 0.01).

Total arsenic accumulation in cerebral cortex was measured. Data are mean ± S.E.; n = 6; *p < 0.01 vs control group, # p < 0.05 vs As2O3-treated group. As2O3: arsenic trioxide.
Effect of resveratrol on cerebral cortex histology
Identification and alteration of histopathological changes during oxidative damage and the protective effect of resveratrol against As2O3-induced toxicity are depicted in Figure 6. Histopathological examination in the control group showed a normal structure of the cerebral cortex (Figure 6(a)). Slight neuronophagia with necrotic eosinophilic neurons was observed in the As2O3-treated group (Figure 6(b) and (c)). The cerebral cortex sections from the As2O3 + resveratrol-treated group showed good protection against the damage with almost normal neurocyte morphology (Figure 6(d)). Resveratrol-treated cats also had normal morphology.

Histopathological changes in cerebral cortex and the protection offered by resveratrol. Figure 4(a) from control group showed normal neurons with characteristic perinuclear areas (H&E, 400×). Figure 4(b) and (c) are high-power photomicrograph of the cerebral cortex from the As2O3-treated group showing slight neuronophagia and necrotic eosinophilic neurons (H&E, 400×). Figure 4(d) is high-power photomicrograph of As2O3+ resveratrol group showing almost normal neurocyte morphology (H&E, 400×).
Discussion
Brain is a soft target for arsenic toxicity as it freely crosses the blood–brain barrier. 30 Although the precise mechanism of arsenic neurotoxicity is not clearly understood, enhanced oxidative stress in the brains of rats exposed to arsenic has been suggested by several investigators. 30,31 Compared with other tissues, brain has higher probability to be challenged by ROS because it consumes more than 20% of all the oxygen utilised by other organs during mitochondrial respiration. Moreover, specific reactions, such as those catalysed by monoamine oxidases, produce H2O2, neurotransmitters, and ROS. 32
It has been shown that arsenic generates free radical species including hydroxyl radicals, superoxide anions, dimethyl arsenic peroxy radical, dimethyl arsenic radical and others and thus impairs the antioxidant system in the brain and other biological tissues. 33 Increased levels of brain ROS were reported in vitro in cultured cortical cells exposed to arsenic and gallium arsenide, an intermetallic semiconductor with extensive applications. 34 Chattopadhyay et al. 35 in an interesting study on human fetal brain explants demonstrated that arsenic exposure increased generation of ROS associated with enhanced apoptosis. Enhanced levels of ROS in rat cortex and hippocampus following treatment with arsenic and gallium arsenide in vivo were suggested to be involved in the process of neuronal dysfunction. 34 Recently, in another study by these investigators, it was observed that exposure to arsenic induced dysfunction of the enzyme activities of complexes II and IV is the main source of ROS generation in mitochondria. 36 This ROS generation results in the electrons leaked out when they were transferred from complexes I and III to complexes III and IV, respectively, increasing ROS generation. 36 Multiple pharmacological properties including antioxidant activity of resveratrol are well accepted. A number of studies suggest that resveratrol is a better scavenger of ROS due to the presence of phenolic and methylene groups on the phenyl ring and ethenyl group in its structure, responsible for its antioxidant potential. Treatment with resveratrol protected mice from Alzheimer's disease-induced oxidative stress. 37 Neuroprotective effects of resveratrol against beta-amyloid-induced neurotoxicity in rat hippocampal neurons involved with oxidative stress and protein kinase C have been reported by Han et al. 38 Moreover, resveratrol have an important role in improving the activity of mitochondrial complexes I, II, and IV, 39 which may contribute to its inhibitory effect on the overproduction of ROS. Dolinsky et al. 40 also found that resveratrol mitigates arsenic intoxication-induced oxidative damage in rats and helps to antagonise its toxicity. The neuroprotective effect of resveratrol was associated with its potential property that decrease the ROS level in the brain. Arsenic-induced decreases in brain antioxidant enzymes activity were also found to be ameliorated following resveratrol treatment in cats in the present study.
In order to protect tissues against the deleterious effects of ROS, all cells possess numerous defence mechanisms that include enzymes such as SOD, CAT, glutathione reductase and GPX. Increases in antioxidant enzymes such as CAT and SOD have been shown to suppress arsenic-induced sister chromatid exchanges in human lymphocytes. 41 Similarly, CAT-deficient Chinese hamster ovary cells were hypersensitive to arsenite insult, demonstrating arsenic-mediated ROS. 42 There was report about rats provided with As2O3 exhibited decrease in serum and tissue SOD as well as GPx and CAT activities, 16,43 which was consistent with our present results. Meanwhile, it was reported that resveratrol increased the amounts of several antioxidant enzymes, including GPX, glutathione S-transferase and glutathione reductase. 44 In another study, it was indicated that treatment with resveratrol immediately after traumatic brain injury reduces oxidative stress and lesion volume. 45 Park et al. 46 proposed that resveratrol indirectly activated sirtuin SIRT1/PGC-1α through a signalling cascade involving cyclic AMP (cAMP), exchange protein activated by cAMP 1 (Epac1) and AMP-activated protein kinase (AMPK) and increase in PPARγ Coactivator (PGC)-1α levels was reported to upregulate the expression of many ROS-detoxifying enzymes, including SOD, CAT and GPx in cultured neural cells from oxidative-stressor-mediated cell death, 47 which may contribute to the increase in the enzymes in present study. Therefore, the recovery of activity of SOD, CAT and GPX in present study could be correlated with decreased generation of ROS and increased expression of these antioxidant enzymes by resveratrol treatment.
Arsenic produces oxidative damage by disturbing the pro-oxidant–antioxidant balance, partly because it has very high affinity for sulfhydryl groups in GSH, which might have implications in the maintenance of thiol-disulphide balance. 48 Several pathways have been proposed to elucidate the depletion in arsenic-induced cellular GSH level. First, according to Radabaugh and Aposhian, 49 GSH can act as an electron donor for the conversion of arsenic (V) to arsenic (III). Second, arsenite has strong affinity towards GSH, which is necessary for arsenic methylation. Third, GSH may be oxidised due to the interaction with the free radicals induced by arsenic. During the metabolic action of GSH, its sulfhydryl group becomes oxidised resulting in the formation of corresponding disulphide compound, GSSG (oxidised form). Thus, depletion of GSH content is associated with an increase in GSSG concentration, resulting in the depletion of GSH-GSSG ratio. Previous experimental studies have demonstrated that treatment with resveratrol upregulates GSH-GSSG ratio. 50,51 In addition, resveratrol has been reported to preserve antioxidant defence by preventing depletion of total glutathione and GSH levels. 52 Because of the antioxidant properties of resveratrol against oxidative stress mentioned earlier, the GSH level and the ratio of GSH-GSSG were maintained approach to normal level.
Leelank and Bansal 53 reported that GSH depletion decreased the GSH-GSSG ratio, which lead to the production of free radicals. ROS intermediates and other oxygen radicals are shown to be associated with cellular injury and further necrosis due to peroxidation of cellular membrane lipids, which in turn result in altered cellular function and structure. Arsenic-induced ROS directly attacks polyunsaturated fatty acids and form MDA, 4-hydroxynonenal, 2-propenal (acrolein) and isoprostanes, which can be measured in plasma and tissue as indirect indicators of oxidative stress. 54 According to Yadav et al., 55 an increase in MDA and protein carbonyl levels was observed in frontal cortex, corpus striatum and hippocampus regions of the brain in arsenic-treated rats. Similarily, increased levels of MDA in the cerebral cortex in arsenic-treated cats as observed in the present study are consistent with these reports. Reduced level of MDA for pretreatment with resveratrol in the present study could be correlated with decreased generation of ROS and increase of these antioxidant enzymes by resveratrol.
Moreover, arsenic could inhibit cell energy metabolism. Arsenate is a phosphate analog, which replaces phosphate in glycolytic and cellular respiration pathways. 56 Arsenite can react with the sulfhydryl groups of proteins and inhibit cellular glucose uptake, gluconeogenesis, fatty acid oxidation, and activity of pyruvate dehydrogenase, which results in decreased citric acid cycle activity and production of cellular ATP. 57 However, resveratrol improves mitochondrial function and increases mitochondrial biogenesis by activating SIRT1 and PGC-1α, 58 which may contribute to preventing arsenic-induced ATP depletion and supplying sufficient energy for arsenic excretion.
The result of histopathology suggested that arsenic damaged the normal architecture of the lung (Figure 6). The cerebral cortex histological and pathological alterations seem to be correlated with other biochemical results in our study. The effect might result from the ability of resveratrol’s above protective effect.
Another interesting finding in the present study is the ability of resveratrol to reduce arsenic burden in the target tissues, which may play a key role in resveratrol against As2O3-induced toxicity. It has also been found the arsenic-GSH complex transported by multidrug resistance-associated protein 2 is one of the forms of arsenic efflux from cells, and the transportation of the arsenic-GSH is an ATP-dependent process decreases arsenic accumulation in cells. 59 It has also been reported resveratrol possesses affinity-chelating properties for metals, which might cause the reduction in brain arsenic levels. Based on the above studies, resveratrol decreased arsenic concentration in the brain possibly through three pathways. First, GSH (which helps in the efflux of arsenic) could be overexpressed in brain cells after pretreatment with resveratrol. 45 Second, resveratrol has been reported to enhance the number and function of mitochondria, 58 which supply more ATP for the efflux of arsenic from the brain cells. Last, the level of arsenic might be decreased by resveratrol for its affinity-chelating properties of metals. Those studies are in accordance with the results of the present research that resveratrol can decrease the concentration of arsenic in the brain.
The limitations of the study are that different doses of resveratrol and its effect on long-term arsenic exposure and also lack the application of As2O3 combination with resveratrol on animal model of tumours are not investigated.
In conclusion, the present study supports the notion that the protective properties of resveratrol against As2O3-induced oxidative stress and toxicity were realised through decreasing the accumulation of arsenic and modulating cellular redox status in the brain. Therefore, further studies are needed to investigate the effect of different doses of resveratrol and the effect of systemic administration. On the other hand, taken together the safety profile of resveratrol in other studies, 60 we plan to try in preclinical studies to provide data on clinical treatment with resveratrol, especially when the use of resveratrol as post-remission therapy for cancer patients with As2O3 chemotherapy as well as adjunctive therapy in patients with exposure to arsenic.
Footnotes
Authors’ Note
The authors Yanyan Cheng and Jiangdong Xue and the universities made an equal contribution to this study.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by the National Science Foundation Committee of China (31101868), Heilongjiang Province Foundation for Young Scholars (QC2010057), Chinese Postdoctoral Science Foundation (20100481040), Special Foundation of China Postdoctoral Science Foundation (2012T50302), Heilongjiang Province Postdoctoral Science Foundation (LBH-Z10256), and Northeast Agricultural University Doctoral Foundation (2010RCB41).
