Abstract
This study aimed at determining the relationship between environmental exposure to lead (Pb) and cadmium (Cd) and blood selenium (Se) concentration in randomly selected population of children inhabiting the industrial regions of Silesian Voivodship, Poland. The study was conducted on a group of consecutive randomly selected 349 children aged below 15 years and inhabiting the industrial regions in Upper Silesia. The examined variables included whole blood Cd concentration (Cd-B), whole blood Pb concentration (Pb-B) and whole blood Se concentration (Se-B). The concentration of Cd-B, Pb-B and Se-B in the studied group of children amounted to 0.26 ± 0.14, 37.62 ± 25.30 and 78.31 ± 12.82 μg/L, respectively. In the entire examined group a statistically significant negative linear relationship was noted between Pb-B and Se-B (r = −0.12, p < 0.05). Also, a statistically insignificant negative correlation was detected between Cd-B and Se-B (r = −0.02, p > 0.05) and a statistically insignificant positive correlation between Pb-B and Cd-B (r = 0.08, p > 0.05). A multivariate backward stepwise regression analysis demonstrated that in the studied group of children higher Pb-B and a more advanced age-represented independent risk factors for a decreased Se-B. Environmental exposure to Pb may represent an independent risk factor for Se deficit in blood of the studied population of children. In children, the lowered Se-B may create one of the mechanisms in which Pb unfavourably affects human body.
Introduction
Heavy metals represent recognised toxic elements and they significantly disturb normal functioning of human body. 1 Among heavy metals to which inhabitants of Polish industrial regions are exposed, the most harmful ones include lead (Pb) and cadmium (Cd). 2 It is essential to search for the mechanisms in which heavy metals exert an unfavourable effect on the body and it is particularly important to identify such mechanisms in children.
To our knowledge, till date, the published references that analysed persons exposed to heavy metals, including children, documented a high level of oxidative stress. 3,4 In populations exposed to the metals, numerous investigations detected an increased production of reactive oxygen species (ROS) and disturbed functioning of mechanisms in which free radicals are eliminated, that is, the so-called antioxidants. 5 In other investigations, the disturbed oxidoreductive equilibrium in the body was linked to deficiency of selenium (Se). 6
Se, the element belonging to the group of trace elements, plays a significant role in numerous metabolic processes taking place in the body. 7 Studies conducted in the recent years have demonstrated a relationship between the deficiency of Se in the body and an impoverished antiviral resistance, processes of carcinogenesis, abnormal generative function and pathology in the circulatory system. 8 –12
In view of the above, it seems that lowered blood Se concentrations in children might involve one of the mechanisms in which heavy metals unfavourably affect human body. The investigations performed till now failed to explain the problem in an unequivocal manner.
This study aimed at determining the relationship between environmental exposure to Pb and Cd and blood Se concentration in randomly selected population of children inhabiting the industrial regions of Silesian Voivodship (Poland).
Material and methods
The study was conducted on a group of consecutive randomly selected 349 children aged below 15 years and inhabiting the industrial regions in Upper Silesia. The studies were conducted from 2007 to 2010. In the examined group, boys accounted for 57.31% subjects (i.e. 200 individuals) and girls accounted for 42.69% of the examined individuals (i.e. 149 individuals). The mean age group was 8.56 years, mean stature was 1.42 m, mean body weight amounted to 38.43 kg and mean body mass index (BMI) was 18.24 kg/m2. General characteristics of the studied group is presented in Table 1.
Clinical characteristics and Cd-B, Pb-B and Se-B in the whole study group (n = 349).

BMI: body mass index; Cd-B: whole blood cadmium concentration; Max: maximal value; Min: minimal value; n: number; Pb-B: whole blood lead concentration; SD: standard deviation; Se-B: whole blood selenium concentration; X: arithmetic mean; X G: geometric mean.
The examined variables included whole blood Cd concentration (Cd-B), whole blood Pb concentration (Pb-B) and whole blood Se concentration (Se-B).
Pb-B and Cd-B were determined using graphite furnace atomic absorption spectrometry with L’vov platform. The methods were based on Stoeppler et al. 13 and Stoeppler and Brandt 14 methods for determining Pb and Cd in deproteinised blood samples by electrothermal atomisation technique. After deproteinisation of blood in 5% nitric acid, the samples were analysed using an atomic absorption spectrometer (Perkin-Elmer 4100ZL instrument, Bodenseewerk Perkin-Elmer, Ueberlingen, Germany) equipped with Zeeman background correction system and an autosampler. The calibration was performed using matrix-matched calibration standards.
Determination of Se-B was performed using hydride generation atomic absorption technique. For the digestion of blood samples, the mixture of nitric acid/perchloric acid (7:3) and a standard temperature were used, and the time program was employed according to the method followed by Frank. 15 Digestion was complete within 16 h with the maximum temperature of 225°C. In order to reduce Se (VI) to Se (IV), the 2.5 M hydrochloric acid was added and the samples were warmed at 110°C for 30 min. After dilution, the samples were analysed using a continuous vapour generation system (Unicam 929, manufacturer Unicam, Cambrige, UK). The calibration was performed using calibration standards prepared in the mixture of hydrochloric acid and perchloric acid solution.
At the initial stage of the study, using cut-off points of median Cd-B and Pb-B, in the studied population of children, the subgroups were distinguished of individuals with high Cd-B and high Pb-B (group I), with high Pb-B (group II), with high Cd-B (group III), with low Cd-B and low Cd-B (group IV). Criteria for distinguishing the studied groups and numerical force of the groups are presented in Table 2. The general characteristics of the studied groups is presented in Table 3.
Inclusion criteria for the groups of children.

Cd-B: whole blood cadmium concentration; Me: median; n: number; Pb-B: whole blood lead concentration; Cd: cadmium; Pb: lead.
Clinical characteristics of the groups of children.

BMI: body mass index; Max: maximal value; Min: minimal value; n: number; SD: standard deviation; X: arithmetic mean; X G: geometric mean; n.s.: not significant.
Statistical analysis was performed using the STATISTICA 9 software (StatSoft Polska, Krakow, Ploland). For quantitative variables, geometric means (X G), arithmetic means (X), standard deviations (SD) as well as minimum (Min) and maximum (Max) values of studied variables were identified in the studied group and in distinguished subgroups. Distribution of variables was tested using Lilliefors and Shapiro–Wilk tests. Due to their abnormal distribution in further analysis, the nonparametric equivalent of analysis of variance, Kruskal–Wallis test was used. Significant differences between X were detected using the post hoc Tukey’s test. Differences for qualitative (nominal) variables were expressed in absolute values. Determination of relationships between the studied variables was defined using analysis of correlation and analysis of regression. In view of an abnormal distribution of variables, Spearman’s correlation coefficient (r) was estimated. Parameters of the model resulting from analysis of multivariable regression were estimated by the least square method. Due to the necessity of fulfilling the regression analysis criteria, in order to obtain the normal distribution of variables, the residuals analysis was performed with removal of the outliers. Results were accepted to be statistically significant at the level of p < 0.05.
The study was approved by Bioethics Committee at the Institute of Occupational Medicine and Environmental Health. Written informed consent was obtained from one parent or a caregiver and oral consent from the child.
Results
Cd-B, Pb-B and Se-B concentrations in the studied group of children amounted to 0.26 ± 0.14, 37.62 ± 25.30 and 78.31 ± 12.82 μg/L, respectively. The Min and Max values amounted to for Cd-B were 0.03 and 0.79 μg/L, respectively, for Pb-B were 9.00 and 166.00 μg/L, respectively and for Se-B were 49.00 and 137.00 μg/L, respectively (Table 1).
Comparing the mean concentrations of Se-B in the distinguished groups of children, it was found that in groups I and II, Se-B was significantly lower than in groups III and IV, (Table 4).
Se-B in the study groups of children.
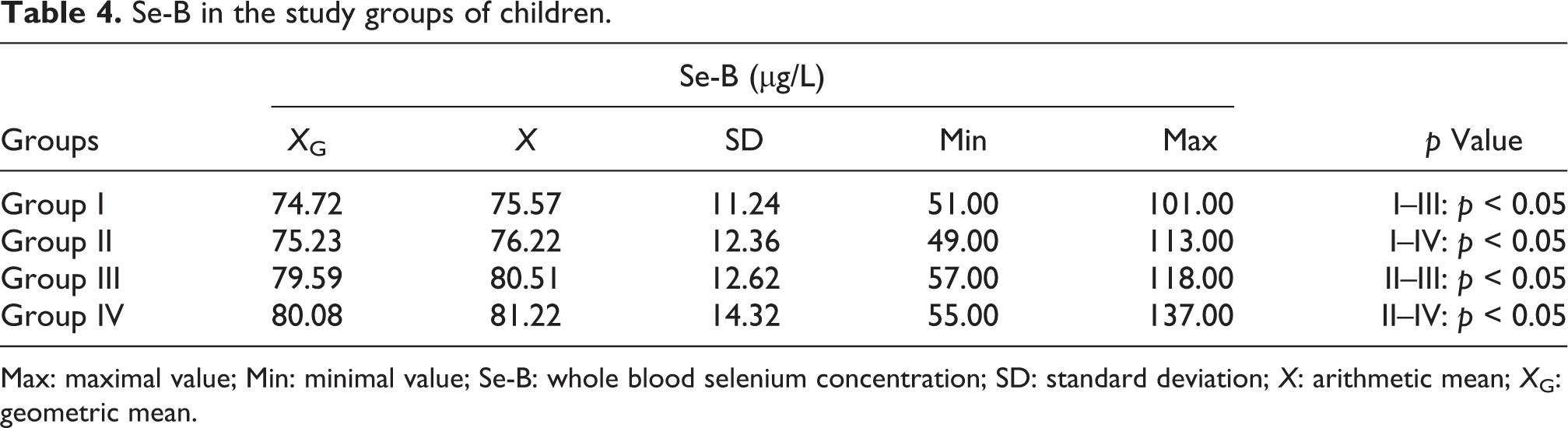
Max: maximal value; Min: minimal value; Se-B: whole blood selenium concentration; SD: standard deviation; X: arithmetic mean; X G: geometric mean.
In the studied group of children no Se deficiency (i.e. Se-B < 40 μg/L) was detected. No significant differences were demonstrated in frequency of marginally low Se concentrations (Se-B: 40–59 μg/L) between the examined groups. A suboptimal Se concentration (Se-B: 60–79 μg/L) was detected with a significantly higher frequency in group II than in groups III and IV. On the other hand, the optimum Se concentration (Se-B ≥ 80 μg/L) was noted significantly more frequently in group IV than in groups I and II and in group III as compared to groups I and II, (Table 5).
Se deficiency, marginally low Se concentration, suboptimal Se concentration and optimal Se concentration in the study groups of children.

N: number; Se-B: whole blood selenium concentration; Se: selenium; n.s.: not significant.
In the entire examined group, a statistically significant negative linear relationship was noted between Pb-B and Se-B (r = −0.12, p < 0.05). Also, a statistically insignificant negative correlation was detected between Cd-B and Se-B (r = −0.02, p > 0.05), and a statistically insignificant positive correlation between Pb-B and Cd-B (r = 0.08, p > 0.05).
A multivariate backward stepwise regression analysis yielded the following model, taking into account the principal anthropologic parameters (sex of the child, age, weight, height and BMI) and concentrations of studied heavy metals (Cd and Pb):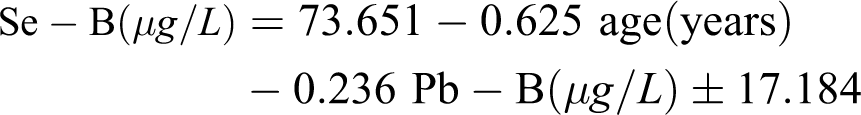
Appropriate determination of the model was confirmed by p value of the model (p < 0.05), p value for age (p < 0.05) and p value for Pb-B (p < 0.01). In the current study group of children, the obtained model demonstrated higher Pb-B and a more advanced age-represented independent risk factors for a decreased Se-B (Table 6).
Final model obtained in multivariate backward stepwise regression analysis.

Pb-B: blood lead concentration, Se-B: whole blood selenium concentration; SEM: standard error mean; Rc: regression coefficient.
Discussion
Compounds of Pb and Cd significantly disturb normal functioning of human body. Effects of acute intoxication with Pb and Cd can be noted only in exceptional situations. Clinical significance of Pb and Cd toxicity pertains first of all chronic exposures, that is, occupational exposure of individuals working in certain branches of industry and environmental exposure, in particular, children inhabiting the industrial regions.
Biological effects of chronic exposure to Pb in adults involve sequels developing in cardiovascular system, immune system, central and peripheral nervous system, haemopoiesis, kidneys, alimentary system and fertility. 16 –19 In children, chronic exposure to Pb results in abnormalities manifesting both in the prenatal and postnatal periods. Already women in the 19th century with occupational exposure to Pb were found to suffer from more frequent abortions, stillbirths and deliveries of newborns with inborn errors. 20 Inborn errors linked with exposure to Pb in the prenatal period were defined using the acronym VACTREL, which denotes V: vertebral anomalies, A: anal atresia, C: cardiac abnormalities, T: tracheoesophagal fistula, E: oesophagal atresia, R: renal abnormalities or radial dysplasia and L: limb abnormalities. 19 Exposure to Pb in the prenatal period also increases the risk of obstetrical complications, that is, for precocious delivery, precocious rupture of foetal membranes, intrauterine growth retardation syndrome and a low birth weight. 21 In the postnatal period, the children suffer from an incompletely developed blood–brain barrier and the Pb compounds exert the most pronounced effects on central nervous system. Therefore, chronic environmental exposure to Pb compounds results in children first of all in a disturbed mental and psychomotor development, most frequently manifested by attacks of aggression, emotional liability, a disturbed memory, difficulties in learning to read and write and a lowered achieved intelligence quotient. 22,23
In adults, chronic exposure to Cd results in lesions located in kidneys and osseous system; also, abnormalities in the alimentary system, respiratory system and circulation may appear. 24 In 1960s, in Japan, an endemic disease of osseous system was described in individuals environmentally exposed to Cd, including osteomalacia with fractures of long bones or the so-called itai-itai disease. 25 Moreover, Cd has been recognised to exert proven carcinogenic effects. 26 In children, chronic exposure to Cd results in prenatal and postnatal abnormalities. The few data gathered till now obtained during observations of children delivered by mothers exposed to Cd compounds demonstrated that intrauterine exposure to Cd may result in an increased frequency of obstetrical complications. Experimental investigations on animal models demonstrated teratogenicity and mutagenicity of Cd compounds. 27 Exposure to Cd in the postnatal period was linked for the potential development of abnormal psychomotor development and a disturbed behaviour (an increased risk of attention-deficit hyperactivity disorder manifestation). 28 Reports are also available on a negative correlation in the children between environmental exposure to Cd and efficiency of the immune system. 29
In 1950s, for the first time, the problem of an unfavourable effect of Se deficiency in the body, including the child’s body, was brought up. On the animal models, a decrease in blood Se concentration was found to promote manifestation of muscular diseases, such as the so-called white muscle disease and paediatric age cardiomyopathy, the so-called Kashin disease, a degenerative disease of blood vessels within bones, termed the Kashin–Beck’s disease. 30,31 Studies conducted in the subsequent years demonstrated beyond doubt that Se deficiency represents a significant factor in the pathogenesis of abnormalities in antioxidative protection. Se represents a component of more than 10 enzymes, the so-called selenoproteinases, representing catalysers of oxidation/reduction reactions. Insufficient blood Se concentrations result in a decreased activity of selenoproteinases and, therefore, in an augmented production of free oxygen radicals in the body. 6,8 The available literature contains confirmation of the hypothesis on the relationship between deficiency of Se and a reduced immunity, particularly in cases of viral infections, that is, in Varicella, Molluscum contagiosum, HIV-1 infections, infection with Coxsackie type B3 virus, with hepatitis viruses B and C and with measles virus. 9,32 In adults with Se deficiency, an increased propensity for neoplastic diseases was detected (particularly for malignant tumours of prostate, breast and colon), certain thyroid diseases, neuropsychiatric disturbances and disturbance of fertility proved to be more frequent. 12,33 –36 A lowered serum Se concentration was detected in women with frequent abortions, in men with diagnosis of oligospermia. 11 In recent years, deficiency of Se was found to be significant in pathogenesis of circulatory diseases. In studies on the population of Finland inhabitants with low blood concentration of Se introduction of supplementation with selenium resulted with the decreaese in mortality rate due to circulatory diseases. 36
The lowered blood Se concentration seems to represent one of the mechanisms in which heavy metals exert an unfavourable effect on child’s body. In order to verify such a hypothesis, concentrations of Pb, Cd and Se were estimated in blood of 349 children inhabiting the industrial regions of the Silesian Voivodship (Poland) and suffering from environmental exposure to Pb and Cd compounds. In the studied group of children, the mean Cd-B, Pb-B and Se-B concentrations amounted to0.26 ± 0.14, 37.62 ± 25.30 and 78.31 ± 12.82 μg/L, respectively. Regression analysis allowed to finding out that independent factors of risk for a decreased Se-B in the group of children environmentally exposed to Pb and Cd were a more advanced age and a higher Pb-B. An increase in Pb-B by 10 μg/L was accompanied by a decrease in Se-B by the mean of 2.36 μg/L. The observed values in regression analysis inverse the relationship between Pb-B and Se-B and are confirmed by the results obtained using correlation analysis and by comparisons between the distinguished groups. In the entire studied group of children environmentally exposed to Pb and Cd, a significant inverse linear relationship has been documented between Pb-B and Se-B (r = −0.12, p < 0.05). Moreover, in groups of individuals with high Pb-B (groups I and II), the mean Se-B has been significantly lower than in groups of individuals with low Pb-B (groups III and IV). The mean Se-B has proven to be significantly lower in the group of children with high Cd-B and Pb-B (group I) and in the group of children with high Pb-B (group II) as compared to the group of children with high Cd-B (group III) and as compared to the group of children with low Cd-B and Pb-B (group IV). Analogous results have been obtained evaluating the Se-B in the groups distinguished according to the accepted standards for blood Se concentration. In the examined group of children, no Se deficit (Se-B < 40 μg/L) has been detected and no significant differences have been detected in frequency of a marginally low Se concentration (Se-B: 40–59 μg/L). Suboptimal concentration of Se (Se-B: 60–79 μg/L) has been significantly more frequent in the group of individuals with high Pb-B (group II) than in groups with low Pb-B (groups III and IV). Optimum Se concentration (Se-B ≥ 80 μg/L) has been noted significantly more frequently in groups of individuals with low Pb-B (groups III and IV) than in groups of individuals with high Pb-B (groups I and II). Optimum Se concentration has been noted significantly more frequently in the group of children with low Cd-B and Pb-B (group IV) and in the group of children with high Cd-B than in the group of children with high Cd-B and Pb-B (group I) and in the group of children with high Pb-B (group II). As already mentioned above, the obtained results unequivocally point to an inverse linear relationship between Pb-B and Se-B in the examined group of children environmentally exposed to Pb and Cd compounds.
On the other hand, analysis of the results obtained in the examined group of children environmentally exposed to Pb and Cd compounds seems to deny existence of significant relationships between Cd-B and Se-B. The analysis of regression has excluded that Cd-B might represent an independent risk factor for changes in Se-B. Moreover, the correlation analysis has failed to document a significant linear relationship between Cd-B and Se-B. Also, the absence of the analysed relationship was confirmed by the results of comparisons between the distinguished groups. Although in comparisons of groups differing in Cd-B (the high Cd-B – groups I and III vs. low Cd-B – groups II and IV), the mean Se-B has been significantly lower in the group of individuals with high Cd-B and whole Pb-B (group I) than in the group with low Cd-B and Pb-B (group IV). However, in the absence of significant Pb-Se relationships in analyses of correlation and regression and in view of the absence of significant differences in blood Se concentration between the group of children with high Cd-B and Pb-B (group I) and the group of children with high PB-B (group II), and also, between the group of children with high Cd-B (group III) and the group of individuals with low Cd-B and Pb-B (group IV), the obtained difference in Se-B between the group of individuals with high Cd-B and Pb-B (group I) and the group of individuals with low Cd-B and Pb-B (group IV) should be interpreted only as the effect of differences in Pb-B. The results obtained during the evaluation of Se-B should be analogously interpreted in the groups distinguished taking into account the accepted standards of blood Se concentration. Also, in this analysis significant differences have been obtained between the group of individuals with high Cd-B and Pb-B (group I) and the group with low Cd-B and Pb-B (group IV), stemming exclusively from differences in whole Cd-B, because no significant differences have been detected between the group of children with high Cd-B and Pb-B (group I) and the group of children with high Pb-B (group II), as well as between the group of children with high Cd-B (group III) and the group of individuals with low Cd-B and Pb-B (group IV). It should be stressed again that the obtained results unequivocally point to an absence of a relationship between Cd-B and Se-B in the studied group of children environmentally exposed to compounds of Pb and Cd.
The obtained results may prove useful in the currently ongoing discussion on the relationship between exposure to Pb and Se-B. The studies conducted in the past do not clarify the problem in an unequivocal way. A relationship between Pb-B and blood Se concentration has been documented, inter alia, in reports of Gustafson et al. 37 , Osman et al. 38 , Kapka et al. 39 and those of Bárány et al. 40 In the studies conducted by Gustafson et al., persons occupationally exposed to Pb were found to be characterised by significantly lower plasma Se concentrations than persons not exposed to Pb. 37 Moreover, a statistically significant negative correlation has been detected between Pb-B and plasma Se concentration (r = −0.33). Osman et al., Kapka et al. and Bárányet al. demonstrated the relationship in studies performed on children. 38 –40 Osman et al. documented significant inverse linear relationships between Pb-B and Se-B (r = −0.38) and between Pb-B and serum Se concentration (r = −0.56). 38 Studies of Kapka et al. and those of Bárány et al. confirmed the existence of a negative correlation between Pb-B and serum Se concentration (r = −0.23 and −0.33, respectively). 39,40 The studies are also available, however, which provide grounds for negating such relationships. According to Ellis et al., Pb-B of randomly selected adults failed to correlate with either Se-B or serum Se concentration. 41 The study by Telisman et al. also confirm the absence of linear relationships between Pb-B and serum Se concentration in randomly chosen adults. 42 An absence of relationships between Pb-B and plasma Se concentration in persons occupationally exposed to Pb was indicated by studies published by Kasperczyk et al. 43 The results obtained in this study, similarly to results published by Osman et al., Kapka et al. and by Bárány et al. point to existence of an inverse direct relationship between Pb-B and Se-B in children environmentally exposed to Cd and Pb.
The results of this study may prove useful in the ongoing discussion on the relationship between exposure to Cd and blood Se concentration. The relationship has not been till now unequivocally clarified in the available literature. A significant negative linear relationship between Cd-B and serum Se concentration was demonstrated in the studies conducted on children by Osman et al. 38 and in adults by Telisman et al. 42 (r = −0.30 and −0.19, respectively). An analogous correlation between Cd-B and Se-B was demonstrated in children by Osman et al. 38 and in adults by Ellingsen et al. 44 (r = −0.18 and −0.80, respectively). Moreover, the study using regression analysis published by Ellingsen et al. documented that Cd may constitute an independent risk factor for a lowered Se-B. 44 Reports are also available in which no relationship could be detected between Cd-B and blood Se concentration. Such a relationship could not be detected in randomly selected adults either for the blood Se concentration or for serum Se concentration i.a. in the studies of Ellis et al. 41 In the study conducted on children by Bárány et al., the relationship could not be detected. 40 Results obtained in this study seem to fit those of Bárány et al. and they indicate absence in children population of a relationship between Cd-B and Se-B.
Summing up the above analysis, the current study in the children population has unequivocally proven existence of an inverse linear relationship between Pb-B and Se-B. In view of the obtained results, the environmental exposure to Pb should be considered to represent an independent risk factor for Se deficiency in the body. Therefore, it seems probable that in children the lowered Se-B may create one of the mechanisms in which Pb unfavourably affects human body. Our inability to demonstrate a relationship between Cd-B and Se-B with high probability precludes in children the chance that a lowered blood Se concentration represents a significant mechanism in which Cd unfavourably affects human body. In the case of Cd, other mechanisms of its toxicity should be looked for.
Conclusions
In the studied population of children an inverse linear relationship was documented between Pb-B and Se-B. Environmental exposure to Pb may represent an independent risk factor for Se deficiency in blood of the studied population of children.
Footnotes
Acknowledgements
The authors are indebted to Adam Prokopowicz, PhD, for estimating the whole blood cadmium concentrations, whole blood lead concentrations and whole blood selenium concentrations.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the European Union through its Sixth Framework Programme for Research and Technological Development (contract no FOOD-CT-2006-016253). It reflects only the authors’ views. The Community is not liable for any use that may be made of the information contained therein.
