Abstract
Hyperthermia enhanced the clastogenicity of alkylating agents. We investigated whether quercetin (QU; 3,3′,4′,5,7-pentahydroxy flavone) or naringenin (NAR) can sensitize Ehrlich ascites tumour (EAT) to cisplatin (CP) hyperthermal intraperitoneal chemotherapy treatment and whether these flavonoids in combination with CP can ameliorate CP-induced micronuclei (MNs) in peripheral blood reticulocytes of mice.
Introduction
Exposure to genotoxins causes carcinogenesis and teratogenicity. 1 Damage to the structure of the affected DNA is unambiguously the main cause of mutagenesis. 2 In addition to their generic growth properties, the majority of anti-neoplastic drugs have genotoxic effects that contribute to growth inhibition. These genotoxic effects may lead to the initiation of unrelated tumours years after the termination of chemotherapy. 3
Cisplatin (CP) or cis-diamminedichloroplatinum (II), a platinum-based anti-neoplastic agent, is one of the prominent members of the most effective broad spectrum anti-cancer drugs used against cervical, head and neck, prostate, breast, lung, testicular and ovarian cancers. 4,5 The anti-cancer activity of this drug is attributed to its capability to form covalent bonds at N-7 position of purine residues of DNA leading to the formation of 1, 2 or 1, 3-intrastrand s-links and a lesser extent of the inter-strand cross-links. These adducts of CP-DNA derail the cellular replication and transcription machinery if these lesions anyhow evade DNA repair system in the effected cells. 6–9 It is understood that non-repair or inhibition of the DNA double-strand break repair contributes to chromosomal aberrations. 8,9 This chromosomal damage could be analysed by doing the quantitative analysis of micronuclei (MNs) and now considered as a very useful parameter for the prediction and prognosis of the tumour treatment response. 3,10 Many labs including ours have demonstrated that CP generates free radicals leading to oxidative and nitrosative stress, which results into such deleterious effects in vivo. 5,11,12 Thus, full therapeutic efficacy of this drug is limited due to the development of acquired drug resistance by the cancer cells and various side effects in the host, including nephrotoxicity, damage the bone marrow cells and cumulative myelosuppression, the latter frequently presenting as severe anaemia. 5,7,9,11
The clastogenic potential of CP has become of great interest because of its serious effects on the chromosomes of non-tumour cells. Its anti-tumour activity is significantly enhanced in the presence of heat. 13,14 The mechanisms responsible for these temperature effects on cell killing by anti-cancer drugs are not entirely understood. It is not known, for instance, whether the net increase in DNA damage, which is thought to underlie the interaction between hyperthermia (HT) and many anti-cancer drugs, is due to an increase in drug uptake, 14 an alteration in intracellular distribution of drug, an alteration in drug metabolism, an increase in drug reaction rates with DNA or heat-induced inhibition of DNA repair. 15,16 Thus, hyperthermal intraperitoneal chemotherapy (HIPEC) has the advantage of a high local concentration of the cytostatic drugs with less systemic exposure compared to conventional intravenous drug administration and limited systemic side effects.
Because HT alone has produced only limited results, much attention has been focused on combining it with chemotherapy and on searching for substances able to sensitize tumour cells to HT-induced damage. 14,17–19 It has been reported that quercetin (QU; 3,3′,4′,5,7-pentahydroxy flavone), a flavonoid with a wide variety of biological activities, is a hyperthermic sensitizer in HeLa cells and synergizes the growth inhibitory effect of HT on solid tumour and leukemic progenitors. 17,18 In addition, QU sensitizes colon carcinoma to thermotherapy and thermochemotherapy in vitro and in mice models and authors suggest that QU liposome might be potentially applied for clinical cancer therapy. 20 Naringenin (NAR), the flavonoid-aglycone, in the human body acts as an antioxidant, immunomodulator and has anti-inflammatory and anti-cancer effects. 4,21–26 NAR reduces the amount of free radicals such as reactive oxygen species (ROS), thus helping to prevent the development of many chronic diseases such as cardiovascular disease and cancer. Moreover, NAR can enhance the anti-tumour effect of doxorubicin to A549 and MCF-7 cancer cells and irinotecan to VK-2 and/or Hep2 cells by selectively inhibiting the activity of multidrug resistance-associated protein but not p-glycoprotein. 4,9,11,21,27
Dietary supplements are capable of detoxifying free radicals or inactivating radicals’ intermediates generated by anti-neoplastic drugs. Moreover, experimental observations have shown that some antioxidant agents, as for example certain flavonoids, are able to enhance the cytotoxic action of the chemotherapeutic drugs without damaging normal cells. 4,12,24,26–30
Considering the widespread use of CP in clinical oncology and the ability of flavonoids to enhance the sensitivity of cancer cells to CP in normothermic or hyperthermic condition, we investigated whether non-toxic doses of QU or NAR has influence on the bone marrow toxicity induced by CP in mice. The bone marrow clastogenesis was assessed by the number of MNs in peripheral blood reticulocytes using MN assay.
Asanami and Shimono 31 reported that HT (body temperature of 39.5°C or higher) in the mouse induces MNs. HT can induce aneuploidy, microtubule depolymerisation, disturbance of the mitotic apparatus and inhibition of DNA repair; all these changes can lead to the formation of MNs. In addition, HT enhanced the clastogenicity of alkylating agents. 31
There are no examples whether flavonoids reduce the induction of MNs in CP HIPEC treatment. In this study, we investigated the effect of HIPEC treatment in mice on micronucleus (MN) induction and the ability of QU or NAR to enhance the sensitivity of cancer cells to CP as well as their effects to ameliorate CP-induced MNs in peripheral blood reticulocytes in a mouse bearing Ehrlich ascites tumour (EAT).
Materials and methods
Animals
Present study was approved by the ethical committee (Faculty of Science, University of Zagreb, Croatia). Animal studies were carried out according to the guidelines force in Republic of Croatia (Law on the Welfare of Animals, N.N. #19, 1999) and in compliance to the Guide for the Care and Use of Laboratory Animals, DHHS Publ. (NIH) # 86-23 (1986). Male albino mice of the Swiss strains, aged 3 months, weighing from 20 to 25 g from our conventional mouse colony were used. The animals were maintained on a pellet diet and water ad libitum. Experimental groups were composed of 13–15 mice each.
Tumour cells
EAT is transplantable, poorly differentiated and fast growing malignant tumour, which appeared originally as a spontaneous breast carcinoma in a mouse. EAT cells were used and maintained in male Swiss albino mice by weekly intraperitoneally (i.p.) inoculation in an ascitis form. After harvesting and preparation of cells, their total number and viability were determined by counting in Bürker-Türk chamber using trypan blue dye. Viability was always found to be at least 90%. Mice were inoculated i.p. on day 0 with 2 × 106 viable tumour cells per mouse in a volume of 0.5 mL 0.9% sodium chloride solution.
Cisplatin
The anti-cancer drug CP [cis-diamindikloroplatinum (II)] (Platimid) was purchased from Pliva (Zagreb, Croatia) and was injected into mice at doses of 5 (CP5) or 10 (CP10) mg kg− 1 (i.p.). The purity (%) of the CP used was ≥98%.
Flavonoids
QU (Quercetin dihydrate 98%, Aldrich Ch. Co. Inc. Milwauke, Wisconsin, USA) and NAR (Sigma-Aldrich Chemie GmbH, Steinheim, Germany) were used in the study as flavonoids. Before use, all test components were dissolved in ethanol and further dilutions were made in water. The final concentration of ethanol was less than or equal to 0.5% (v/v). Ethanol (0.5%) was used in the control group. Test components were given to mice i.p. at a dose of 50 mg kg− 1 body weight (b.w.).
Animal treatments and experimental design
Mice were divided into 18 experimental groups (Table 1). Tumour was generated by intraperitoneal (i.p.) injection of 2 × 106 viable tumour cells. At days 7 and 3 before the implantation of EAT cells (2 × 106), mice were injected i.p. with 50 mg kg− 1 of QU or NAR, while CP (5 or 10 mg kg− 1 i.p.) was applied 3 days after inoculation of EAT cells at normal temperature (37°C) and under hyperthermic (43°C) conditions. Intraperitoneal HT was induced by i.p. injection of 2 mL of saline (0.9% sodium chloride solution) heated at either 37°C or 43°C (hyperthermal treatment) twice, 5 min apart, followed by cytostatic CP 5 or 10 mg kg− 1 in 0.5 mL after the second hyperthermal treatment. The control animals were untreated tumour bearing mice.
Experimental design.
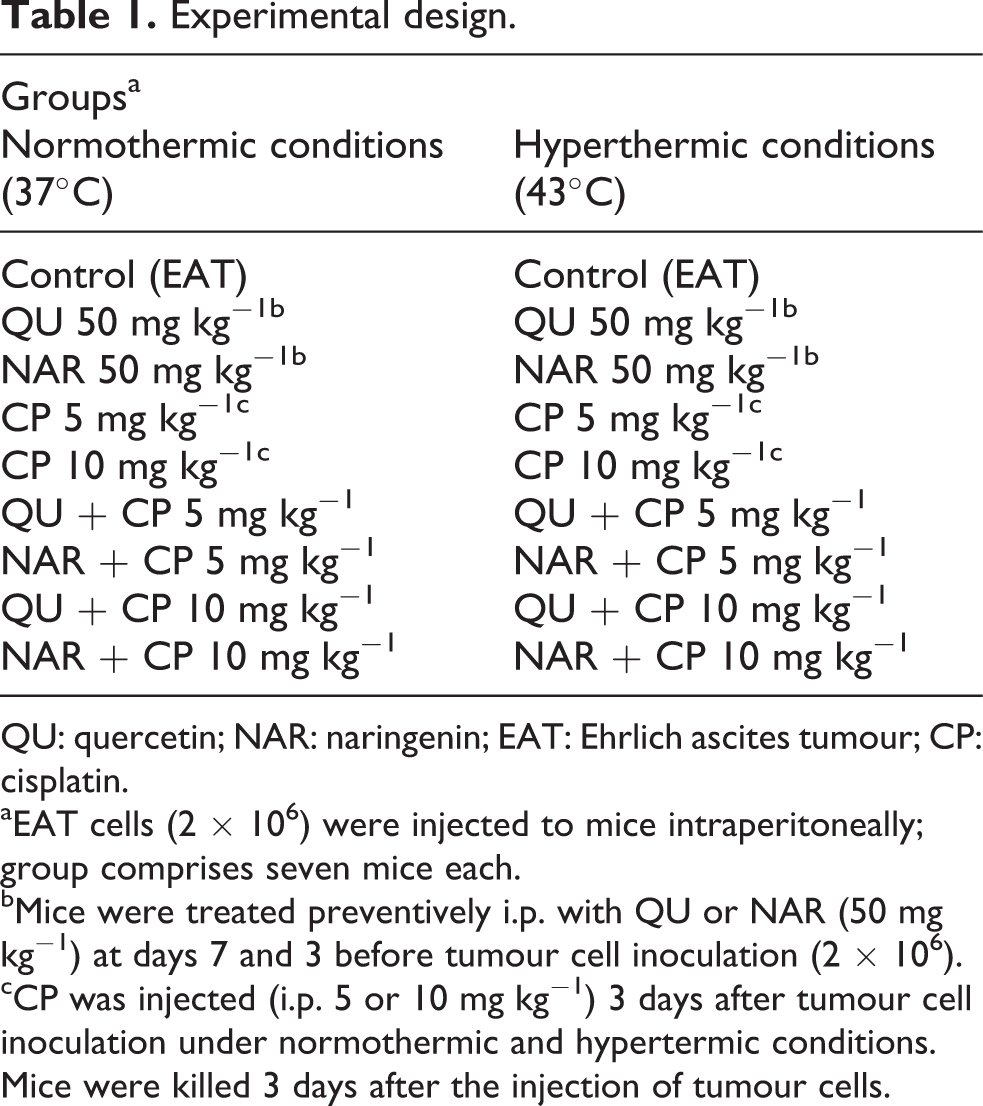
QU: quercetin; NAR: naringenin; EAT: Ehrlich ascites tumour; CP: cisplatin.
aEAT cells (2 × 106) were injected to mice intraperitoneally; group comprises seven mice each.
bMice were treated preventively i.p. with QU or NAR (50 mg kg−1) at days 7 and 3 before tumour cell inoculation (2 × 106).
cCP was injected (i.p. 5 or 10 mg kg−1) 3 days after tumour cell inoculation under normothermic and hypertermic conditions. Mice were killed 3 days after the injection of tumour cells.
Systemic body temperature was determined using an electronic thermometer (BAT-10, Physitemp Instruments, Inc., Clifton, New Jersey, USA) with a rectal probe (RET-3, Physitemp Instruments) before and during HT procedure. Intra-abdominal temperature was measured by the needle probe introduced to a depth of 1 cm into the peritoneal cavity of mice treated with HT, given alone or in combination with cytostatic. Drugs were given immediately after last treatment of HT. The temperature was recorded, in peritoneal carcinomatosis-bearing mice in each group, every 2.5 min during the heating phase and every 2.5 min during the cooling phase. HT, as used in this procedure, was well tolerated by the animals. We followed the survival of animals and the side effects appearing after different treatments.
In total, 18 groups of 13–15 mice were formed for preventive i.p. treatment as given in Table 1.
Five mice of each group were killed in ether chamber on day 3 after tumour cell inoculation. After disinfection of external abdominal region, each animal was inoculated with 3 mL of saline solution and after gentle agitation of abdominal wall; the solution containing peritoneal cells was removed for cellular evaluation. The following variables were analysed: the viable and non-viable tumour cell count, mean survival time (MST), percentage increased life span (ILS%) and number of MNs in peripheral blood reticulocytes. Peripheral blood samples for MN test were collected 48 h after i.p. application of CP at 37 or 43°C.
The remaining animals, that is, seven to eight animals from each group, were used for the survival analysis.
Count of the total number of cells present in the peritoneal cavity
The total number of cells present in the peritoneal cavity was determined by counting in Bürker-Türk chamber by observing the ability of intact cells to exclude trypan blue dye and by phase contrast microscopy.
Peripheral blood MN assay
Peripheral blood smear was prepared as described by Oršolić et al. 4 Blood was collected from tail tip, and smear was prepared on pre-cleaned slides. The smears were allowed to dry at room temperature and fixed in absolute methanol for 5 min. After fixation, slides were stained with acridine orange and washed twice with phosphate buffer (pH 6.8). To determine a number of MNs, a total of 8000 reticulocytes per each experimental group (2000 reticulocytes from each sample, n = 4) were analysed using a 400 magnification fluorescence microscope (Olympus, Japan) equipped with an excitation filter of 502–525 nm.
Survival analysis
Animal life span was evaluated by surveillance of spontaneous death or by elective killing of the animal showing signs of pain or suffering according to established criteria. Long surviving mice (living more than 95 days) were euthanized on the 95th day by an overdose of anaesthetic and cervical dislocation. The results were expressed as a percent of the average life span of treated animals (T) divided by the average life span of animals in the control group (C) (T/C × 100). ILS% were calculated with the following formula: ILS% = (T − C)/C × 100. According to the criteria of the National Cancer Institute, T/C above 125% and ILS above 25% mean that the treatment had significant anti-tumour effect. 32
Statistical analysis
Results are expressed as means ± SD obtained from two experiments. Data were analysed using statistical software STATA 7.0 (Stata Press, College station, Texas, USA). Statistical significance was evaluated using ANOVA. Statistical significance was assigned at p < 0.05.
Treatment-dose specific survival curves were calculated by Kaplan-Meier method, 33 and comparison between survival curves was made by log-rank test (α = 5 %). 34
Results
QU or NAR enhance CP anti-tumour effect and life span of mice in normothermic and hyperthermic conditions
In an effort to investigate the anti-tumour activity of QU or NAR and whether the i.p. administration of these components reduces CP-induced toxicity without any loss of the anti-neoplastic capacity of CP, we initially evaluated the effects of QU or NAR (50 mg kg− 1 b.w.), CP (5 or 10 mg kg− 1 b.w.) and QU or NAR together with CP in normal (37°C) or hyperthermal (43°C) condition on tumour growth and survival time in mice model in which EAT cells were inoculated into Swiss albino mice.
Intraperitoneal treatment of animals with QU or CP (5 or 10 mg kg− 1) in the normal conditions significantly reduces the number of tumour cells in the abdominal cavity of mice-bearing EAT (QU 11.97 ± 1.31 × 106; CP5 = 7.28 ± 1.14 × 106; CP10 = 8.67 ± 1.22 × 106; vs. control = 21.10 ± 3.79 × 106; p < 0.05) and increased the life span of mice by 94.25 and 113.37% or 301.25%, respectively, as shown in Tables 2 and 3. The mice receiving QU together with CP (5 or 10 mg kg− 1) ILS of mice by 275.79% or 263.04% in normal condition while in hyperthermal condition ILS% were 346.87% in both CP concentrations.
Survival analysis of Swiss albino mice bearing EAT after preventive treatment with QU or NAR and CP under normothermic and hypertermic conditions.
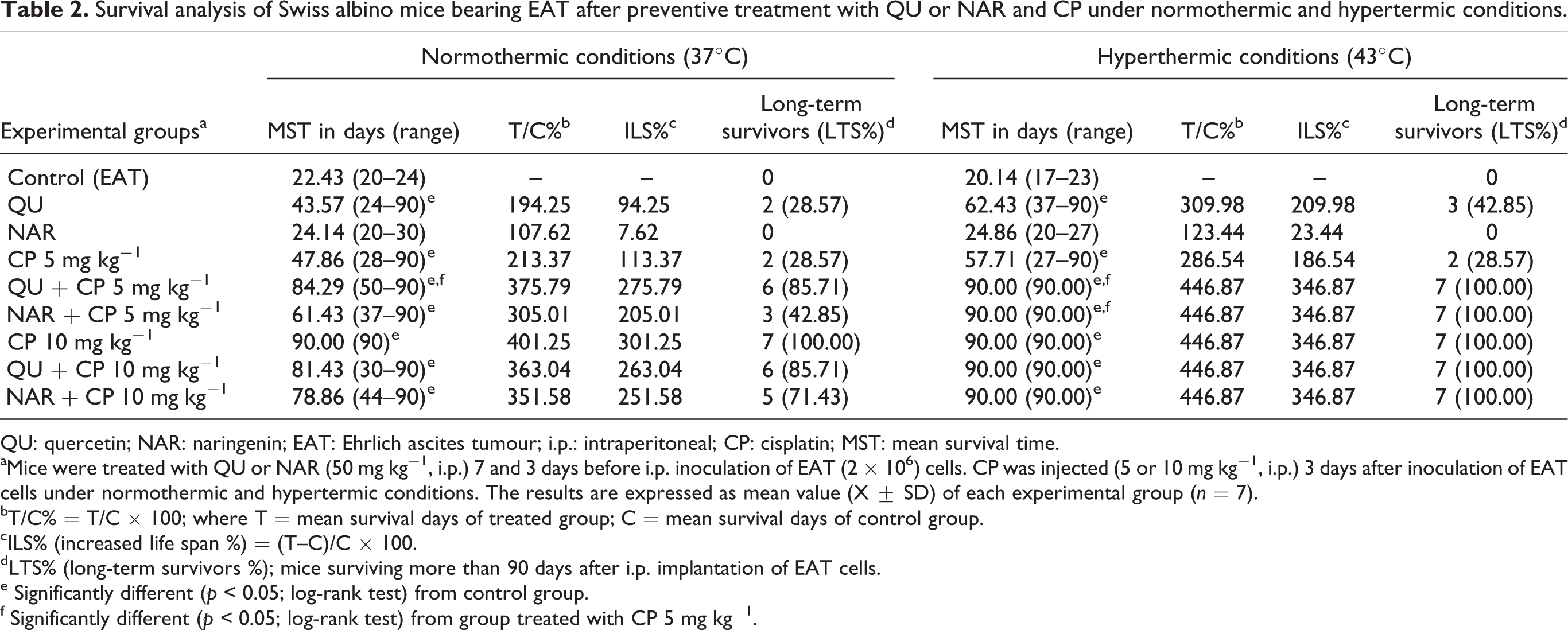
QU: quercetin; NAR: naringenin; EAT: Ehrlich ascites tumour; i.p.: intraperitoneal; CP: cisplatin; MST: mean survival time.
aMice were treated with QU or NAR (50 mg kg−1, i.p.) 7 and 3 days before i.p. inoculation of EAT (2 × 106) cells. CP was injected (5 or 10 mg kg−1, i.p.) 3 days after inoculation of EAT cells under normothermic and hypertermic conditions. The results are expressed as mean value (X ± SD) of each experimental group (n = 7).
bT/C% = T/C × 100; where T = mean survival days of treated group; C = mean survival days of control group.
cILS% (increased life span %) = (T–C)/C × 100.
dLTS% (long-term survivors %); mice surviving more than 90 days after i.p. implantation of EAT cells.
e Significantly different (p < 0.05; log-rank test) from control group.
f Significantly different (p < 0.05; log-rank test) from group treated with CP 5 mg kg−1.
Tumor cells viability and mortality in the intraperitoneal cavity of Swiss albino mice bearing Ehrlich ascites tumor after preventive treatment with QU or NAR and cisplatin under normothermic and hypertermic conditions
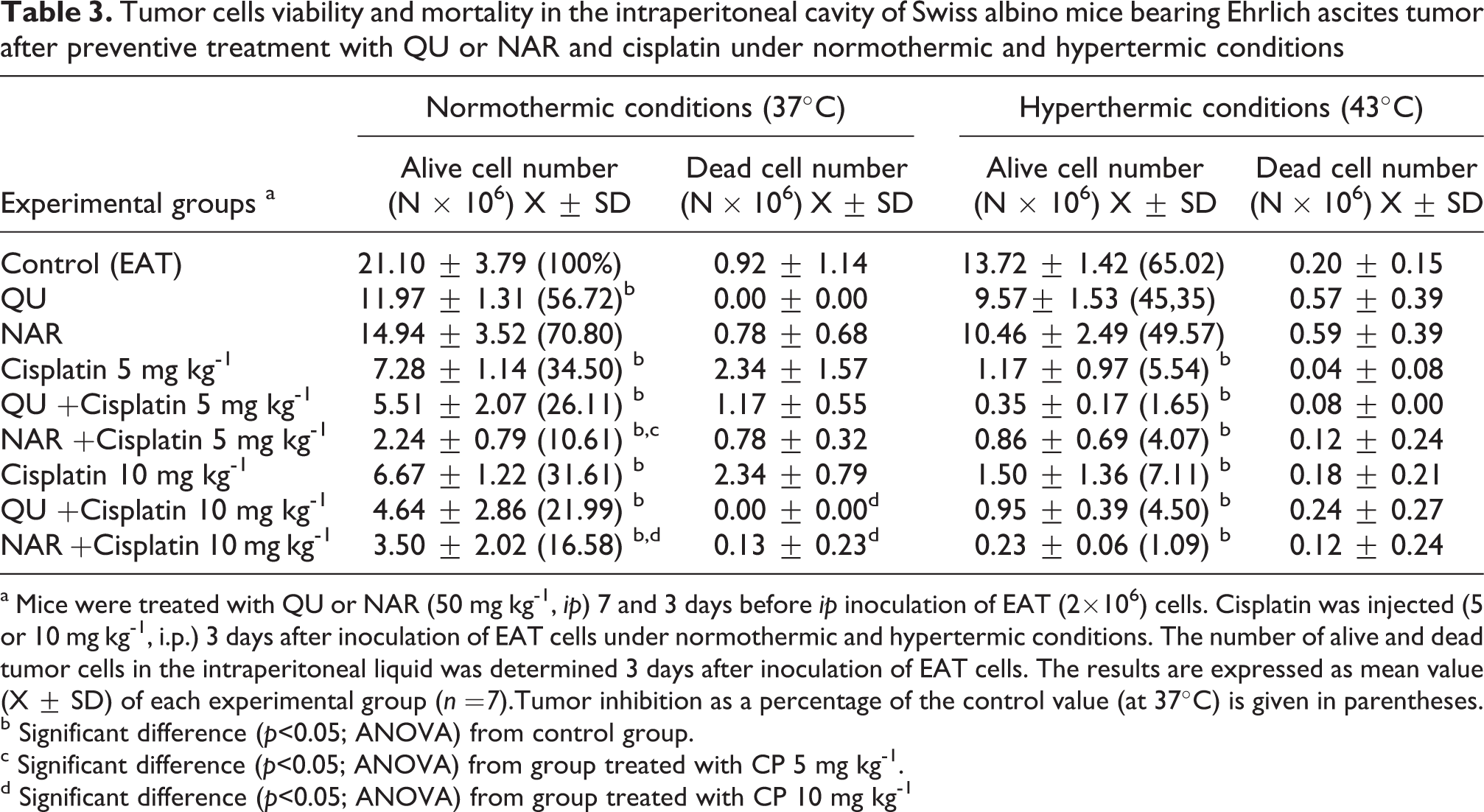
a Mice were treated with QU or NAR (50 mg kg-1, ip) 7 and 3 days before ip inoculation of EAT (2×106) cells. Cisplatin was injected (5 or 10 mg kg-1, i.p.) 3 days after inoculation of EAT cells under normothermic and hypertermic conditions. The number of alive and dead tumor cells in the intraperitoneal liquid was determined 3 days after inoculation of EAT cells. The results are expressed as mean value (X ± SD) of each experimental group (n =7).Tumor inhibition as a percentage of the control value (at 37°C) is given in parentheses.
b Significant difference (p<0.05; ANOVA) from control group.
c Significant difference (p<0.05; ANOVA) from group treated with CP 5 mg kg-1.
d Significant difference (p<0.05; ANOVA) from group treated with CP 10 mg kg-1
Treatment with NAR at the doses of 50 mg kg− 1 reduced the viable tumour cell count as compared to that of the EAT control group in normothermic or hyperthermic condition (at 37°C, 14.94 ± 3.52 vs. 21.10 ± 3.79; at 43°C, 10.46 ± 2.49 vs. 13.72 ± 1.42) but without statistical significance.
Treatment with NAR + CP at the doses of 5 or 10 mg kg− 1 significantly (p ≤ 0.05) reduced the viable tumour cell count as compared to that of the EAT control group in normothermic or hyperthermic condition (at 37°C NAR + CP5 = 2.24 ± 0.79 or NAR + CP10 = 3.50 ± 2.02 vs. 21.10 ± 3.79; at 43°C, NAR + CP5 = 0.86 ± 0.69 or NAR + CP10 = 0.23 ± 0.06 vs. 13.72 ± 1.42).
In the EAT control group, the MST was 22.43 (at 37°C) or 20.14 (at 43 °C) days, while it increased to 24.14 or 24.86 days, respectively, in the NAR-treated groups, whereas the combined treatment group had a MST of 61.43 days (at 37°C NAR + CP5) or 78.86 days (NAR + CP10). In hyperthermal treatment groups MST were 90 days in both CP doses; ILS% were 346.87% in both doses of CP and all 7 mice were long time survivors (p = 0.0010, Kaplan-Meier analyse) while in normothermic groups ILS% were 205.01% (NAR + CP5) or 251.58 (NAR + CP10); 3 or 5 mice from total 7 were long time survivors.
Among test components, QU + CP (5 mg kg− 1) was the most potent in inhibiting proliferation of EAT cell under normothermic and hyperthermic conditions; the percent of EAT cells compared to control was less for 95.9% and/or 97.17%; 6 or 7 mice were long time survivors (p = 0.0010, Kaplan-Meier analyse).
Moreover, in tumour-bearing mice treated with QU and in combination of QU with chemotherapeutic drug CP, a delay in tumour growth and significant increase in survival without mortality or body weight loss was noted during experimental period.
It is important to mention that during the heating phase, the intra-abdominal temperature was stable, ranging from 42.5-43°C, while rectal temperature was not significantly affected. During the cooling phase the temperature in the peritoneal cavity of treated mice decreased to normal values approximately 15 minutes after applying the hyperthermal procedure as in our previous experiments.14
Flavonoids attenuate the number of MNs in the peripheral blood reticulocytes induced by CP in normothermic condition but enhance in hyperthermic condition
As shown in Table 4, pre-treatment of mice with EAT tumour with QU or NAR decreased the frequencies of MN in blood reticulocytes induced by CP; QU or NAR protect reticulocytes from toxicity by CP in normothermic condition (QU + CP5 = 7.0 ± 1.4 or NAR + CP5 = 3.5 ± 0.7 vs. CP5 = 11.0 ± 1.4 and QU + CP10 = 4.5 ± 0.7or NAR + CP10 = 3.5 ± 0.7 vs. CP10 = 15.0 ± 2.8. Interestingly, in hyperthermal condition, QU or NAR in combination with CP significantly increased number of micronucleated cells indicating prooxidative effect of QU; number of micronucleated reticulocytes was higher in combined treated groups than in group treated with CP alone (QU + CP5 = 103.5 ± 24.3 or NAR + CP5 = 104.0 ± 32.6 vs. CP5 = 59.3 ± 5.6 and QU + CP10 = 154.3 ± 35.6 or NAR + CP10 = 97.5 ± 39.0 vs. CP10 = 65.5 ± 12.8) (Table 4).
Micronucleus assay on peripheral blood reticulocytes of Swiss albino mice bearing Ehrlich ascites tumor after preventive treatment with QU or NAR and cisplatin under normothermic and hypertermic conditions.
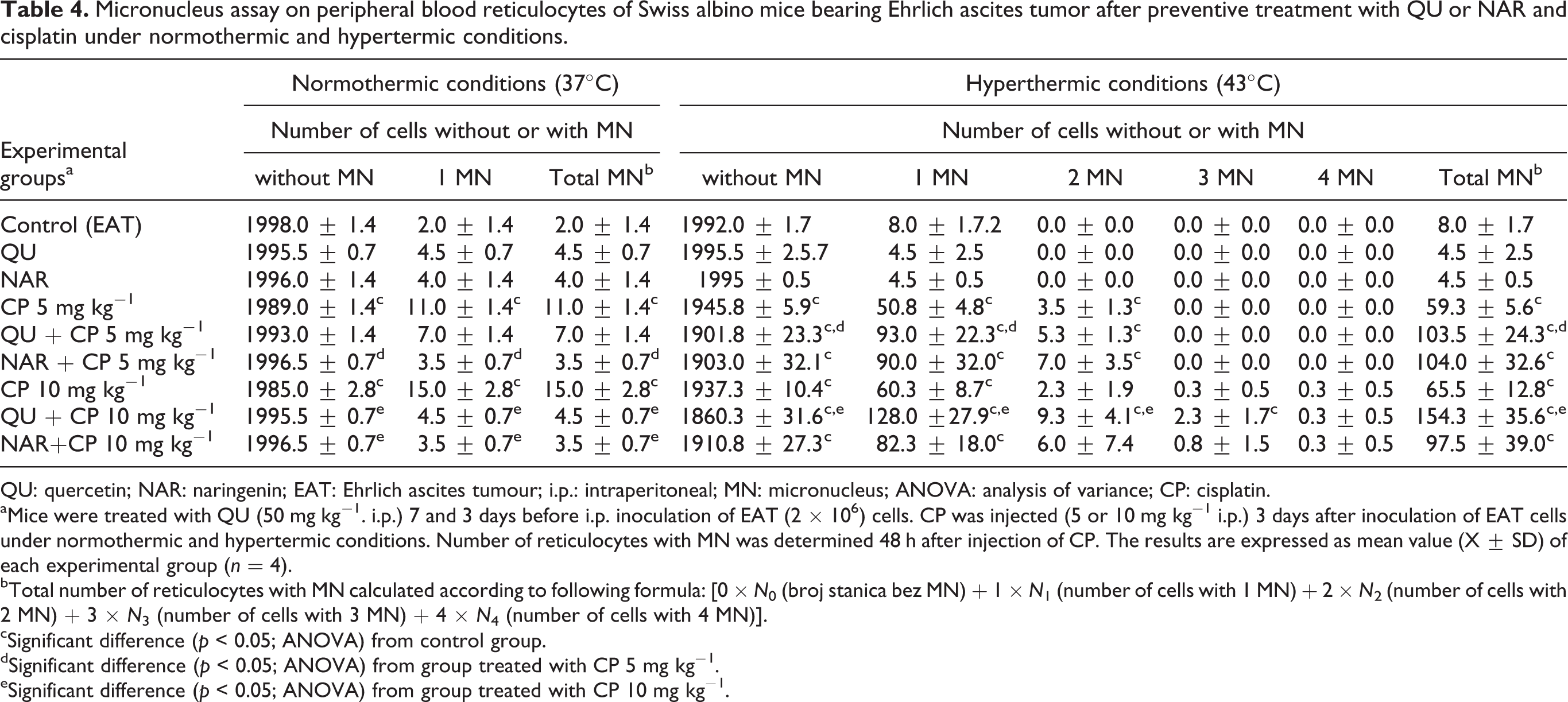
QU: quercetin; NAR: naringenin; EAT: Ehrlich ascites tumour; i.p.: intraperitoneal; MN: micronucleus; ANOVA: analysis of variance; CP: cisplatin.
aMice were treated with QU (50 mg kg−1. i.p.) 7 and 3 days before i.p. inoculation of EAT (2 × 106) cells. CP was injected (5 or 10 mg kg−1 i.p.) 3 days after inoculation of EAT cells under normothermic and hypertermic conditions. Number of reticulocytes with MN was determined 48 h after injection of CP. The results are expressed as mean value (X ± SD) of each experimental group (n = 4).
bTotal number of reticulocytes with MN calculated according to following formula: [0 × N 0 (broj stanica bez MN) + 1 × N 1 (number of cells with 1 MN) + 2 × N 2 (number of cells with 2 MN) + 3 × N 3 (number of cells with 3 MN) + 4 × N 4 (number of cells with 4 MN)].
cSignificant difference (p < 0.05; ANOVA) from control group.
dSignificant difference (p < 0.05; ANOVA) from group treated with CP 5 mg kg−1.
eSignificant difference (p < 0.05; ANOVA) from group treated with CP 10 mg kg−1.
Treatment of mice with flavonoids did not cause significant changes in the number of MNs in peripheral blood reticulocytes.
Discussion
In order to increase the effectiveness of cancer chemotherapy treatment and to reduce its side effect to normal tissues, the interest has been recently focused to the natural product that have been used in traditional medicine such as QU and NAR. The observed anti-tumour effects of these natural compounds encouraged extensive efforts to establish its usefulness in cancer prevention and cancer treatment. Recent studies suggest that using plant-derived chemopreventive agents in combination with chemotherapy can enhance the efficacy of chemotherapeutic agents and lower their toxicity to normal tissues.4,11,23,25,26,28,29 Lots of biological functions of flavonoids have been identified, but there currently exists a lack of evidence any relationship between the flavonoids and CP-induced MNs in peripheral blood reticulocytes in hyperthermal condition.
In this article, we have investigated the anti-tumour and anti-clastogenic effects of the QU or NAR alone and in combination with a chemotherapeutic agent, CP on EAT in normothermic or hyperthermic conditions.
We found that a combination of QU or NAR and CP led to a significantly reduced tumour growth and ILS of mice compared to treatment with either drug alone (Tables 2 and 3). In addition, used flavonoids sensitizes EAT cells to thermotherapy and thermochemotherapy in mice models. Thus, combined treatment of QU or NAR with HIPEC with CP (5 mg kg− 1) increased survival of mice by additional 160.33% as compared with single treatments of mice with HIPEC. These data with our and others previous data14,20,35 suggest that combination flavonoids with HIPEC might be potentially applied for clinical cancer therapy and that HIPEC with flavonoids could be used to maximally enhance the immunity of mice with EAT while potentially reducing the adverse effects of drug-heat agents (chemotherapeutic) on normal cells, paralleled with an equal or increased efficacy on tumour cells.
Today, HT is a promising approach to cancer therapy. HT potentiates the cytoxicity of several anti-tumour agents including bleomycin, 36 cyclophosphamide, 37 mitomycin C,13,38 CP,13,14,39 doxorubicin13,14,39 and l,3-bis(2-chloroethyl)-nitrosourea.40,41
In addition, HT enhanced the clastogenicity of alkylating agents. The majority of the experimental data suggest that the damage to chromosomal DNA is directly responsible for the cytotoxicity of CP.6–8 It has been conclusively demonstrated that intra- and inter-strand cross-links after CP treatment were the adducts of biological significance and responsible for cytogenetic and mutagenetic effects.6,42 A fairly good correlation between cellular sensitivity and the number of inter-strand cross-links exists. 6 In our study, hyperthermal treatment of mice with CP increased the number of MNs in the peripheral blood reticulocytes 4.36- to 5.39-folds compared to mice with normal body temperature. Given results are in concordance with Chrisman and Baumgartner, 43 who demonstrated that cyclophosphamide induces a threefold increase in micronucleated polychromatic erythrocytes. Contrary to the expected, combination of flavonoids with CP increases the number of MNs in hyperthermal conditions; number of micronucleated reticulocytes was higher 2–3 time than in group treated with CP in hyperthermal condition alone.
Interestingly, used flavonoids in normothermic condition are able to protect mouse bone marrow erytropoesis against the CP-induced clastogenicity. Moreover, the current study demonstrates that QU or NAR was not clastogenic at the doses tested. These results are in concordance with earlier studies, where administration of QU or nar did not cause DNA damage in the bone marrow cells.4,11,23,26 In vivo, QU displays rather a protective activity against DNA damage. In vitro experiments indicated that QU used at high concentrations may be mutagenic, but when given in low concentrations, it provided protection against the DNA damage induced by hydrogen peroxide (H2O2). 44 The lack of clastogenicity of QU in vivo can be explained by its low bioavailability and by degradation of its aglicone form caused by intestinal bacterial flora or O-methylation, glucuronidation and sulphatation in the gastrointestinal tract. 45
According our data, QU or NAR are effective in reducing clastogenesis induced by CP in bone marrow cells and may possibly decrease the risk of secondary tumours in cells that were not originally neoplastic in normothermic condition but in hyperthermic condition used flavonoids may increased clastogenicity of CP.
The protective effect of these flavonoids could be possibly ascribed to its radical scavenger effect that modulated the changes induced by CP. Based on the data presented here, strategies can be developed to decrease the deleterious effects of CP in normal cells using QU or NAR in normothermic condition.
Contrary, HT is known to cause conformational changes in proteins affecting the spindle apparatus. In addition, HT after drug treatment could also have an effect on the post-replication repair of CP-induced DNA lesions. Thus, the individual effect of the two agents and their interaction resulted in the higher enhancement of cytogenetic damage expression as seen in the significant increase of MN frequency on day 2 after CP + HT. Several studies suggest that high doses of CP could induce nephrotoxicity, hepatotoxicity, neurotoxicity, myelosuppression and genotoxicity, which are closely associated with the increased generation of ROS, such as hydroxyl radical and superoxide anion. ROS are noxious to cells, leading to oxidation of essential cellular components, such as proteins and lipids, and to eventual genomic instability events resulting from DNA, which can be seen as DNA fragmentation or MN formation. 46 Several reports have also indicated that a host of phenolic phytochemicals, including etoposide and flavonoids, can alter intracellular ROS levels. 47
QU, probably in the presence of Fe2+ and increased levels of ROS caused by CP, increases CP toxicity on reticulocytes. It is possible that auto-oxidative activity of QU may be result of high concentration of QU and its inhibition of mitochondrial respiration with concomitant production of the superoxide anion and H2O2. H2O2 causes DNA strand breakage by generation of hydroxyl radicals close to the DNA molecule, by the Fenton reaction. We speculated that the synergism was attributable to a decreased ability of the cell to repair that damage in hyperthermal condition.
It seems that high-dose CP with hyperthermal conditions induce irreversible lesions or inefficient post replication repair, which leads to increased chromosomal instability and formation of MN. It is possible that increased oxidative stress and lipid peroxidation of erythroid cells in bone marrow can lead to the release of iron and prooxidant effects of flavonoids by simultaneously increasing mitochondrial ROS, resulting in increased MN in combined treatment of mice bearing EAT. A strong hypothesis is that these flavonoids inhibit repair processes of DNA by inhibiting poly(ADP-ribose) polymerase 48 which participates in DNA repair of normal or tumour cells after being damaged by alkylation.
Conclusion
This study demonstrates that HIPEC is superior to tumour growth control, tissue-drug retention and survival compared to equivalent chemotherapy doses given in normothermic condition. QU or NAR, two plant-derived chemopreventive agents, markedly enhances hyperthermic cytotoxicity to tumour cells when administered in combination with CP. These flavonoids are effective in reducing clastogenesis induced by CP in bone marrow erytropoesis in normothermic condition but they increased clastogenicity of CP in hyperthermic condition. We believe that it is possible to achieve increased effectiveness of CP, and better therapy results with fewer side effects, if together with flavonoids, CP is administered in non-toxic doses and if HP is applied either by using the same doses of CP or even by decreasing them.
Footnotes
Authors’ contribution
N.O.: designed the study and coordination, performed the experiments, participated in the data acquisition, analysed samples, gathered data, and wrote the manuscript, read and approved the final manuscript. D.S.: participated in the data acquisition and participated in its design, read and approved the final manuscript. M.K.: analysed samples, read and approved the final manuscript. N.C.: analysed samples and performed statistical analysis, read and approved the final manuscript.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the Ministry of Sciences, Education and Sports of the Republic of Croatia project No. 119-0000000-1255.
