Abstract
The isolated and identified triterpenoid, 1-hydroxytetratriacontane-4-one (C34H68O2), obtained from the methanolic leaf extract of Leucas aspera Linn. was explored for the first time for antisnake venom activity. The plant (L. aspera Linn.) extract significantly antagonized the spectacled cobra (Naja naja naja) venom induced lethal activity in a mouse model. It was compared with commercial antiserum obtained from King Institute of Preventive Medicine (Chennai, Tamil Nadu, India). N. naja naja venom induced a significant decrease in antioxidant superoxide dismutase, glutathione (GSH) peroxidase, catalase, reduced GSH and glutathione-S-transferase activities and increased lipid peroxidase (LPO) activity in different organs such as heart, liver, kidney and lungs. The histological changes following the antivenom treatment were also evaluated in all these organs. There were significant alterations in the histology. Triterpenoid from methanol extract of L. aspera Linn. at a dose level of 75 mg per mouse significantly attenuated (neutralized) the venom-induced antioxidant status and also the LPO activity in different organs.
Introduction
Snakebite is a serious problem in tropical and subtropical countries and is responsible for morbidity and mortality. Global incidence is estimated to be about 5.4 million snakebites per year, over half of them venomous and at least 25,000 fatal. 1 Snakebite is also a major public health problem and claims a large number of lives in the Indian subcontinent. In India, approximately 15,000–20,000 people are affected every year by snake envenomation. 2 Vipera russellii, Naja naja naja and Naja kaouthia are the common snakes found throughout India, and a large number of deaths occur due to envenomation by these snakes. The four major poisonous snakes cobra, krait, Russell’s viper and saw-scaled viper are responsible for fatality due to snakebite in India. Antiserum is the only therapeutic agent available throughout the world. Antiserum sometimes does not provide enough protection against venom-induced haemorrhage, necrosis, nephrotoxicity and often produces hypersensitive reactions. 3–5 Development of antiserum in animals is time consuming, expensive and requires ideal storage condition. Over the years, many attempts have been made for the development of snake venom antagonists especially from plant sources. Many Indian medicinal plants are recommended for the treatment of snakebite, 6 but so far, no systematic analysis has been carried out. A few preliminary reports exist, which neither successfully confirmed their action nor active constituents. 7–9
The lethal effects of snake venom are largely attributed to its active ingredient phospholipase A2 (PLA2). Phospholipid hydrolysis by PLA2 enzyme releases arachidonic acid whose metabolism results in the formation of potentially toxic reactive oxygen species and lipid peroxides. 10,11 El-Asmar et al. have reported that the increase in polyunsaturated fatty acids following envenomation may lead to an increase in the rate of lipid peroxidation, which might be responsible for tissue damage. 12 PLA2 from snake venom has been implicated in multiple pathologies including neurotoxicity, 13 nephrotoxicity, 14,15 lung toxicity, 16 hepatotoxicity 17 and cardiotoxicity. 18,19 Plant extracts constitute rich sources of pharmacologically active compounds, and some of them have been reported to antagonize the activity of various crude venoms and purified toxins. 20 This activity has been attributed to different compounds such as flavonoids, coumarins and other polyphenolic metabolites widely distributed in different plant families. 21 –24 These classes of compounds are known to be powerful antioxidant agents both in hydrophilic and lypophilic environments. Antioxidants are substances that can prevent, stop or reduce oxidation damage. They have diverse pharmacological properties, including anti-inflammatory activity. 25,26
The present investigation explores the isolation and purification of an active compound from the methanolic leaf extract of Leucas aspera Linn., which was responsible for snake venom neutralization. Antagonism of cobra venom and antivenom action potentiation and antioxidant property of the active compound were studied in experimental animals.
Materials and methods
Venom and experimental animals
The lyophilized snake venom of cobra (N. naja naja) was obtained from Irula Snake Catchers Industrial Cooperative Society (Chennai, Tamil Nadu, India) with proper permission (No.WL1/7/2006 dated 13 November 2006) and preserved in a desiccator at 4°C for further use. It was dissolved in 0.85% saline and centrifuged at 2000 r min−1 for 10 min. The supernatant was collected and kept at 4°C until further use. Venom concentration was expressed in terms of dry weight. The concentration of protein in the venom of N. naja naja was determined by the Lowry’s method. 27 Swiss albino mice (weighing 18–22 g) obtained from the Institute of Veterinary Preventive Medicine (Ranipet, Tamil Nadu, India) were used for the study. They were kept in animal cages with sawdust as bedding under conditions of 12:12 h light and dark cycle and fed with standard diet. Equal numbers of male and female mice were used in each experimental group, keeping their mean weight as near as possible. All the animal studies were conducted with the prior permission of the Institutional Animal Ethics Committee, C. Abdul Hakeem College, Melvisharam, Tamil Nadu, India (No.1011/c/06/CPCSEA, dt 19 December 2008).
SDS-PAGE analysis of snake venom
The proteins of the medically important cobra (N. naja naja) snake venoms were analyzed by 12% sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE). 28 The venom samples were subjected to SDS-PAGE for protein patterns. The samples were mixed with Laemmli sample buffer (10% SDS, 10% w/v β-mercaptoethanol, 50% sucrose and 0.02% bromophenol blue), boiled for 5 min and electrophoresed at a constant current of 30 mA. After electrophoresis, the gels were stained with Coomassie brilliant blue. Molecular weight standards were co-electrophoresed.
Determination of LD50 values of cobra venom
Toxicity of N. naja naja venom was determined in laboratory mice. In Swiss albino mice, the median lethal dose (LD50) values were calculated for four routes namely intramuscular (i.m.), intraperitoneal (i.p.), subcutaneous (s.c.) and intravenous (i.v.) by the method adopted by Meier and Theakston. 29 LD50 determinations were made using stock venom solutions of 5 mg ml−1 prepared in 0.85% saline and stored at −20°C. Portions of the stock solutions were thawed and diluted with 0.85% saline to determine lethal dose. Swiss mice of either sex weighing 18–22 g were used in the assay. Six mice were used at each dose level. The volume of dose was 0.2 ml for all the routes through which it was given. The different concentrations of venom used to determine the LD50 value for all the routes were 6.25, 7.81, 9.76, 12.20, 15.25 and 19.90 µg per animal. The experiment was carried out in triplicates (18 animals per concentration). The experiment was conducted for 24 h and mortality was recorded at 24 h after injection. LD50 value was calculated by the Spearman-Karber method. 30
Plant extracts
A total of 12 species of Indian traditional plants (Andrographis lineate, Andrographis paniculata, Leucas aspera, Mimosa pudica, Aristolochia indica, Hemidesmus indicus, Phyllanthus emblica, Phyllanthus amarus, Cynodon dactylon, Ocimum americanum, Ocimum sanctum and Musa paradisiaca) were collected in and around the region of Nambedu, Tiruvannamalai District, Tamil Nadu, India. After preliminary screening of the 12 plants, the crude extract of L. aspera Linn. was found to have strong antisnake venom activity against cobra venom. Hence, this plant was selected for further studies to purify and identify the compound responsible for antisnake venom activity. The collected plant leaves (L. aspera Linn.) were shade dried. About 100 g of powdered leaf of L. aspera Linn. was extracted in a Soxhlet apparatus first with petroleum ether (Qualigens Fine Chemicals), then with benzene (Reachem Laboratory Chemicals Pvt. Ltd, Chennai, India), diethyl ether (Nice Chemicals, Kochi, India), chloroform (Qualigens Fine Chemicals, Mumbai, India), ethyl acetate (Sisco Research Laboratories, Mumbai, India), methanol (Qualigens Fine Chemicals, England, UK), ethanol (Hayman Distillers, England, UK) and finally with distilled water. All these extracts were concentrated through distillation at a temperature of 5°C above the boiling point. The extracts were dried at 60°C for 24 h and then weighed for yield per gram of dry sample. The plant residue after each solvent extraction was completely air-dried before the next solvent extraction. 31 Such dried crude extracts were stored in a refrigerator until they were used for bioassay and lethality test to assess its antivenom activity against the venom of N. naja naja. The dry extracts (100 mg) obtained after removing the solvents were dissolved in dimethyl sulphoxide (DMSO) to obtain the desired stock solutions of the extracts for experimental purpose. The control was set-up with DMSO. Before use, it was dissolved in saline and centrifuged at 2000 r min−1 for 10 min at room temperature. The supernatant was used for further investigation and stored at 4°C. The plant extracts were expressed in terms of dry weight.
Isolation and purification of active plant constituent (triterpenoid)
Silica gel column chromatography
The methanol crude extract (40.0 g) was fractionated by column chromatography using silica gel (60–120 mesh) column (85 × 18 cm2). Elution was performed using petroleum ether–chloroform (9:1, 4:1, 2:1, 1:1 and 0:1; 500 ml for 10 fractions each) 32 to yield four fractions (A, B, C and D). Further elution of column with different proportions of choloroform–methanol (98:02, 95:05 and 90:10; 500 ml for 10 fractions each) was carried out, which yielded three fractions namely E, F and G. All the fractions collected were dried and subjected to testing at different concentrations (100, 125, 150 and 175 mg) for antisnake venom activity against N. naja naja venom in mice. Those fractions showing 100% survival of animals for a minimum of 24 h after injection alone were selected for further separation by column chromatography (Table 4). Thin layer chromatography (TLC) plates (precoated plate 0.2 mm thick, E.Merck, Germany, 60 F254) were used to determine the number of fractions in the eluted sample. The compound was spotted at 0.2 cm from the edge of the sheet and run with a mixture of chloroform and methanol at a ratio of 9:1 to a distance of 4.9 cm. It was dried at room temperature (28 ± 2°C) after which the TLC plate was visualized by pan cal D (ammonium molybdate: 21 g, ceric sulphate: 1 g and concentrated sulphuric acid: 31 ml), and the retardation factor (R f) was calculated.
Among the fractions, fraction B showed strong antisnake venom activity, and it was further rechromatographed to obtain subfractions on elution with petroleum ether–chloroform (100:0 (5 × 200 ml), 90:10 (8 × 200 ml), 80:20 (12 × 200 ml), 70:30 (9 × 200 ml), 60:40 (6 × 200 ml), 50:50 (10 × 200 ml) and 0:100 (3 × 200 ml), respectively). All the subfractions (B1, B2, B3, B4 and B5) collected were dried and subjected to testing at different concentrations (50, 75, 100 and 125 mg) for antisnake venom activity against N. naja naja venom in mice. Those subfractions showing 100% survival of animals for a minimum of 24 h after injection alone were selected for further separation by column chromatography.
Among the subfractions obtained from second column chromatography, fraction B3 showed antisnake venom activity, and it was then subjected to a subsequent column chromatography using silica gel (230–400 mesh, 20 g) to obtain various fractions on elution with petroleum ether–chloroform (100:200 (20 × 50 ml), 98:02 (22 × 50 ml), 96:04 (14 × 50 ml), 94:06 (7 × 50 ml), 92:08 (3 × 50 ml), 90:10 (1 × 50 ml) and 0:100 (1 × 50 ml). All the fractions (B3B1, B3B2, B3B3 and B3B4) collected were subjected to testing at different percentages of concentrations (25, 50, 75 and 100%) for antisnake venom activity against N. naja naja venom in mice.
On TLC, this compound produced a single spot of R f at 0.5 using benzene–ethyl acetate (70:30, v/v). The yield of the active compound was 0.2%. Chemical characterization by various spectroscopic methods namely infrared (IR) spectra, nuclear magnetic resonance (NMR) spectra and mass spectrum indicated that the active compound was triterpenoid. Those fractions showing 100% survival of animals for a minimum of 24 h after injection alone were selected for further analysis.
Neutralization of lethal effect of cobra venom
The toxicity of N. naja naja venom was assessed by injection of different concentrations of venom in 0.2 ml physiological saline through i.p. route into Swiss albino mice (20 g). 33 The cobra venom (2LD50 = 25 µg per mouse) was mixed separately with plant extract extracted with methanol at different concentrations (100, 150, 200 and 250 mg) and incubated at room temperature for 1 h. Six groups of mice (six mice per group) were used. In group I, which served as positive control, the mice were injected (i.p.) with cobra venom at a concentration of 25 µg per mouse. In groups II, III, IV and V, the mice were injected with venom mixed with plant extract at the concentration of 100, 150, 200 and 250 mg, respectively. In group VI, which served as normal control, the mice were injected with phosphate-buffered saline (PBS). The number of surviving animals was recorded at 24 h after injection and the median survival analysis was determined by Spearman-Karber method. 30 The experiment was conducted in triplicate.
I.p. administration of triterpenoid at 0, 1, 2, 4, 6 and 8 h after injection of cobra venom
Two sets of experiments were conducted to study the efficacy of triterpenoid from methanol extract and commercial antiserum to neutralize cobra venom in mice. Each set consisted of eight groups of mice (six per group). In the first set, the mice of group I were given i.p. injection with a lethal dose (2LD50 = 25 µg per animal) of N. naja naja venom and served as positive control. In group II, the mice were administered with i.p. injection of N. naja naja and triterpenoid from methanol extract of plant (75 mg permouse) simultaneously. In groups III, IV, V, VI and VII, the mice were given i.p. injection of N. naja naja venom (25 µg per mouse) and then the mice in group III were administered triterpenoid from methanol extract of plant 1 h after venom injection, after 2 h in group IV, 4 h in group V, 6 h in group VI and 8 h in group VII. The mice of group VIII were given PBS alone and served as a negative control. The number of surviving mice was recorded 24 h after injection of triterpenoid from methanol extract of plant. The experiment was conducted in triplicates. In the second set of experiments, the commercial antiserum obtained from the King Institute of Preventive Medicine (Chennai, India) was used to neutralize the cobra venom and compare its efficacy with triterpenoid from methanol extract.
Antioxidant analysis in envenomated mice treated with triterpenoid from methanol extract of L. aspera Linn
Four groups of mice were used in this experiment. In group I (six mice), the mice were injected with N. naja naja venom at a concentration of 25 µg per mouse by i.p. route. Blood and tissue samples were collected at the moribund stage (about 24 h after venom injection) for analysis. In group II (12 mice), the mice were given i.p. injection with the mixture of venom and triterpenoid from plant extract (75 mg per mouse), and the tissue samples were collected at 24 and 48 h after venom injection (2LD50 = 25 µg per mouse). In group III (12 mice), the mice were injected with the mixture of venom and commercial antiserum (400 µl per animal), and the tissue samples were collected at 24 and 48 h after venom injection (2LD50 = 25 µg per animal). In group IV (12 mice), which served as normal control, the mice were injected with the normal saline, and the tissue samples were collected at 24 h after injection of normal saline. After 24 and 48 h, the animals which survived and the moribund animals were dissected and the organs such as heart, liver, lung and kidney were removed. These organs were homogenized individually with Tris buffer (pH 7.4) using mortar and pestle. All tissue homogenates and plasma were stored at −20°C and used for the evaluation of the activity of the antioxidants such as superoxide dismutase (SOD), 34 glutathione-S-transferase (GST), 35 glutathione peroxidase (GPX) 36 and catalase (CAT) 37 present along with total reduced glutathione (GSH) 38 and lipid peroxidation. 39 The experiments were conducted in triplicates.
Histopathological analysis
For histological investigation, organs such as heart, liver, lungs, kidney, spleen and brain were dissected out from N. naja naja venom-injected mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. After processing the tissue and deparaffinising it in xylene, the sections were hydrated through graded series of alcohol up to 70% and stained with Harris alum haematoxylin, counterstained with 1% alcoholic eosin 40 and then mounted with glass cover slip in DPX mount through xylene and observed under the light microscope.
Statistical analysis
Data are expressed as mean ± standard error (SE). The Mann–Whitney nonparametric and parametric Student’s t tests were used for checking the significance of differences between groups. A p < 0.05 was considered significant. Statistical calculations were performed using SPSS (version 9, IBM, US) software.
Results
SDS-PAGE analysis of cobra (N. naja naja) venom
The concentration of protein in the venom of spectacled cobra was 62.56 ± 0.59 µg µl−1. The protein profile of cobra venom was studied using SDS-PAGE. Figure 1 shows the protein pattern of cobra venom. Eight protein bands (six major bands and two minor bands) were observed on SDS-PAGE in reducing condition after staining with Coomassie brilliant blue. The approximate molecular weight of protein bands I, II, III, IV, V, VI, VII and VIII calculated based on the standard protein markers were 14, 24, 29, 45, 48, 65, 72 and 99 kDa, respectively (Figure 1).

SDS-PAGE showing the protein pattern of Indian spectacled cobra (Naja naja naja) venom. Lane M: marker; lanes 1–12: N. naja naja venom. SDS: sodium dodecyl sulphate; PAGE: polyacrylamide gel electrophoresis.
Determination of LD50
The LD50 value of cobra venom was determined in the mice model by different routes and the results are shown in Table 1. For i.v. route, the LD50 was found to be 8.4 µg per mouse and the LD50 values for i.p. route, i.m. route and s.c. route were 13.73, 13.37 and 13.72 µg per mouse, respectively. The upper and lower limits for these LD50 values are given in Table 1.
LD50 values of cobra venom given to mice by different route of injection at 24 h post injection.

LD50: median lethal dose.
Neutralization of cobra venom by methanol extract
Among the different solvent extracts, the methanol extract was found to be effective in neutralizing the cobra venom. The methanol extract at the concentration of 200 mg per mouse and above neutralized the lethal activity of cobra venom, whereas other solvent extracts failed to neutralize the cobra venom (Table 2). The median effective dose (ED50) value for methanol extract was determined as 103.5 mg per mouse (Table 3).
Percentage survival of mice injected with the mixture of venom and extract of Leucas aspera Linn. extracted with different solvents after 24 h post injection.

PBS: phosphate-buffered saline.
a Naja naja naja venom (25 µg per mouse) mixed with plant extract and incubated at room temperature for 30 min followed by injection into mice to test the inhibition of lethality of venom. (n = 18). Values are mean ± SE.
Neutralization potency of the aqueous and methanol extracts and fractions of methanol extract of Leucas aspera Linn. against lethal effect of cobra venom in mice.a
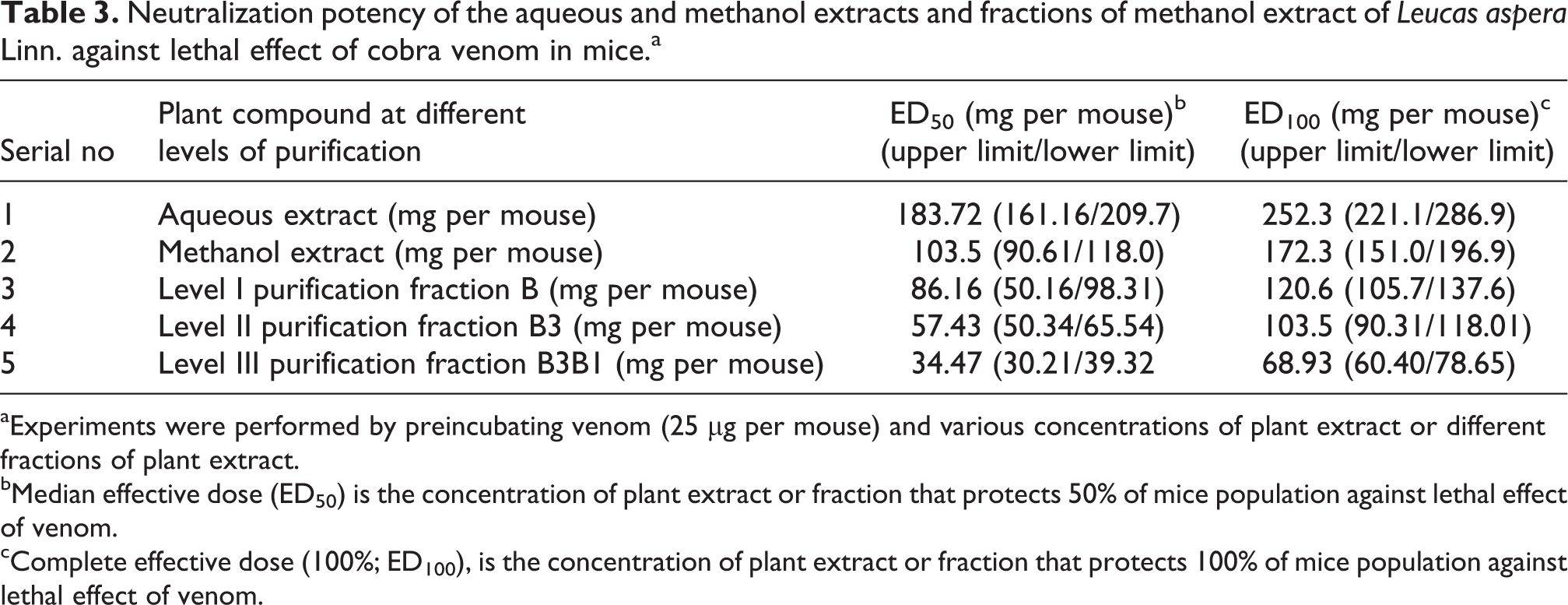
aExperiments were performed by preincubating venom (25 µg per mouse) and various concentrations of plant extract or different fractions of plant extract.
bMedian effective dose (ED50) is the concentration of plant extract or fraction that protects 50% of mice population against lethal effect of venom.
cComplete effective dose (100%; ED100), is the concentration of plant extract or fraction that protects 100% of mice population against lethal effect of venom.
Since the methanol extract of L. aspera Linn. was found to have strong antivenom activity, it was selected for detailed study on isolation, purification and characterization of the active compound responsible for neutralizing the cobra venom. The methanol crude extract was fractionated by column chromatography using silica gel column and seven fractions (A, B, C, D, E, F and G) were collected. All the seven fractions were tested to check the inhibitory activity of cobra venom and the results are given in Table 4. Among these seven fractions, the fraction B was found to have strong antivenom activity against cobra venom. The fraction B at the concentration of 125 mg per mouse and above protected envenomated mouse. All the envenomated mice treated with fraction B at the concentration of 150 mg per mouse and above survived. Fraction B neutralized the lethal activity of cobra venom at the concentration of 125 mg per mouse and above, whereas other fractions failed to neutralize the cobra venom. The ED50 value for fraction B was found to be 86.16 mg per mouse (Table 3). The active fraction B was rechromatographed and five subfractions (B1, B2, B3, B4 and B5) were collected. All the subfractions were tested for their inhibitory activity against cobra venom in mice, and the results are shown in Table 5. The subfraction B3 was found to be very active and neutralized the cobra venom completely at the concentration of 100 mg per mouse and above. All other subfractions failed to neutralize the cobra venom. All the mice injected with venom and subfraction B3 at the concentration of 100 mg per mouse and above survived. The ED50 value for subfraction B3 was determined as 57.43 mg per mouse (Table 3). The active subfraction B3 was chromatographed again and four subfractions (B3B1, B3B2, B3B3 and B3B4) were collected. All the subfractions were tested for their inhibitory activity against cobra venom in mice, and the results are shown in Table 6. The subfraction B3B1 was found to be very active and showed very strong inhibitory activity against the cobra venom at the concentration of 75 mg per mouse and above. All other subfractions failed to neutralize the cobra venom. All the mice injected with venom and subfraction B3B1 at the concentration of 75 mg per mouse and above survived. The ED50 value for subfraction B3B1 was found to be 34.47 mg per mouse (Table 3).
Percentage survival of mice injected with the mixture of venom and different fractions of level I purification of methanol extract of Leucas aspera Linn. after 24 h post injection.

PBS: phosphate-buffered saline.
a Naja naja naja venom (25 µg per mouse) mixed with different fractions of level I purification of methanol extract of L. aspera Linn. and incubated at room temperature for 30 min followed by injection into mice to test the inhibition of lethality of venom (n = 18). Values are mean ± SE.
Percentage survival of mice injected with the mixture of venom and different fractions of level II purification of methanol extract of Leucas aspera Linn. after 24 h post injection

PBS: phosphate-buffered saline.
a Naja naja naja venom (25 µg per mouse) mixed with different fractions of level II purification of methanol extract of L. aspera Linn. and incubated at room temperature for 30 min followed by injection into mice to test the inhibition of lethality of venom. (n = 18). Values are mean ± SE.
Percentage survival of mice injected with the mixture of venom and different fractions of level III purification of methanol extract of Leucas aspera Linn. after 24 h post injection.

PBS: phosphate-buffered saline.
a Naja naja venom (25 µg per mouse) mixed with different fractions of level III purification of methanol extract of L. aspera Linn. and incubated at room temperature for 30 min followed by injection into mice to test the inhibition of lethality of venom. (n = 18). Values are mean ± SE.
Identification of different compounds of L. aspera Linn
The results of TLC were shown in Figure 2. Seven bands were observed in fraction B and the R f values for these bands were 0.16, 0.40, 049, 0.59, 0.65, 0.71 and 0.82 cm. Five bands with R f values of 0.27, 0.69, 0.73, 0.30 and 0.86 cm were observed in fraction B3. A single band with R f value of 0.5 cm using benzene–ethyl acetate (70:30, v/v) was observed in the fraction B3B1.

Thin layer chromatography plates showing different fractions isolated from Leucas aspera Linn. Plate 1: fraction B; Plate 2: fraction B3; Plate 3: fraction B3B1.
Confirmation of antivenom activity of isolated pure compound
The isolated pure compound was tested for its antivenom activity against the cobra venom, and the results are given in Table 6. All the mice injected with venom and pure compound (B3B1) at the concentration of 50 mg per mouse and above survived. The ED50 and complete effective dose (ED100) values for the pure compound were found to be 34.47 and 68.93 mg per mouse, respectively (Table 3).
Spectral analysis of B3B1 compound from L. aspera Linn
Fraction B3B1 was obtained as a brown solid at the end of level III purification. The TLC pattern showed one spot indicating that it is a pure compound. The identification and structural characterization of isolated pure compound were carried out by analyses of various spectral data (ultraviolet (UV), IR, proton ( 1 H) and carbon ( 13 C) NMR and mass spectrum) and the results are given below (Figure 3). 13 C NMR of the mixture gave 10 signals, which corresponds to the δ values given in the results. Interestingly, the mass spectrum of the sample was interpreted based on the proposed structures. Hydroxyl group was confirmed by IR band observed at 3434 cm−1, carbonyl functionality at 1644 cm−1, 842 at long aliphatic chain methyl and methylene at 1.08, C=C at 4.08 Based on the results obtained in the present study, this compound was identified as 1-hydroxytetratriacontan-4-one.

Gas chromatography–mass spectroscopy of B3B1 fraction of compound from Leucas aspera Linn.
The identification and structural characterization of isolated pure compound were carried out by analyses of various spectral data (UV, IR, 1 H and 13 C NMR and mass spectrum) and the results are given as follows (Figure 3).
M f C34H68O2 Mass m/z; 508, 433, 407, 379, 295, 267, 194, 152, 135, 106, 89, 79, 67 (Figure 3).
UV λmax (MeOH): 508 nm.
IRν; cm−1 3434 (OH), 1644(α, β-COOH), 842, 819 (–C–C–C–) (Long chain aliphatic)
1 H NMR (deuterated chloroform (CDCl3), 400 MHz, δ ppm) values: 0.8–1.08 (–CH3), 2.08 (–CH2), 3.38 (OH), 4.08 (–CH=CH–).
13 C NMR (CDCl3, 400 MHz, δ ppm) values: 28(–CH3), 29 (CH2), 205(–C=O).
I.p. administration of triterpenoid from plant extract at 0, 1, 2, 4, 6 and 8 h after injection of cobra venom
The results of envenomated mice administered with triterpenoid from methanol extract or commercial antiserum at 0, 1, 2, 4, 6, and 8 h after injection of cobra venom revealed 100% survival in envenomated mice treated with triterpenoid from methanol extract at the dosage of 75 mg per mouse at 0, 1, 2, 4 and 6 h after injection of cobra venom and 83.32% survival in mice administered with triterpenoid at 8 h after venom injection (Table 7).
Percentage survival of envenomated mice administered with triterpenoid from methanol extract of Leucas aspera Linn. or commercial antiserum at different time intervals (in hours) after injection of cobra venom (n = 18).a

PBS: phosphate-buffered saline.
aValues are means ± SE.
bAdministration of triterbenoid from plant extract or commercial antiserum at 0, 1, 2, 4, 6 and 8 h after injection of cobra venom.
Antioxidant status
The levels of antioxidant enzymes such as SOD, GPX, CAT, reduced GSH and GST and LPO were measured in different organs (heart, liver, kidney and lungs) of normal mice, cobra venom injected mice and envenomated mice treated with triterpenoid from the plant extract and commercial antiserum separately at different time intervals, and the results are presented in Tables 8 to 15. The levels of antioxidant enzymes and LPO were found to be significantly different between normal mice and envenomated mice, and these values became normal in envenomated mice treated with plant extract and commercial antiserum separately after 48 h of treatment
The activity of antioxidant enzymes and lipid peroxidation in the heart tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 24 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal).
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the heart tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 48 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bSignificantly different (p < 0.001) from normal mice.
cMice injected with venom (25 µg per animal).
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the liver tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 24 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal)
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the liver tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 48 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal)
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the kidney tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 24 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal)
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the kidney tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 48 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal).
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the lung tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 24 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal)
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
The activity of antioxidant enzymes and lipid peroxidation in the lung tissue of normal mice, envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum after 48 h of treatment (n = 18).a

PBS: phosphate-buffered saline; GST: glutathione-S-transferase; GSH: glutathione; GPX: glutathione peroxidase; CAT: catalase; SOD: superoxide dismutase; LPO: lipid peroxidase.
aValues are mean ± SE.
bMice injected with venom (25 µg per animal)
cSignificantly different (p < 0.001) from normal mice.
dMice injected with mixture of triterpenoid from plant extract (75 mg per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
eMice injected with mixture of commercial antiserum (400 µl per animal) and venom (25 µg per animal) after incubating for 1 h at 37°C.
Histological investigation
Significant histological changes were observed in kidney tissue of envenomated mice at moribund stage were glomerular congestions, vacuolar degeneration of tubular cells and haemorrhagic regions (Figure 4(b)). The disturbances and circulation of protein dystrophy in hepatocytes were observed in liver tissue of envenomated mice. Degenerated hepatocytes were also observed in different regions of liver tissue (Figure 5(b)). The lung tissue of envenomated mice showed infiltration of erythrocytes within bronchus alveoli and thickening of partitions between alveoli (Figure 6(b)) were observed. Interalveolar partitions were torn in some places in the lung tissue. Haemorrhagic areas were also observed in the lung tissue of envenomated mice. In the heart, a disruption in the striations of the muscle with haemorrhagic areas were seen (Figure 7(b)). Several blood vessels were found to be congested and capillaries were filled with erythrocytes. Histological observation of spleen of envenomated mice revealed the congestion of red pulp (Figure 8(b)). Cells with autophagy and polynucleated cells were observed in the spleen tissue of envenomated mice at moribund stage. In the brain tissue of envenomated mice, no significant changes in the cells except some marked infiltration of leukocytes (Figure 9(b)) were seen. The envenomated-treated mice with methanol extract or commercial antiserum did not show any observable changes in kidney (Figure 4(c) and (d)), spleen (Figure 5(c) and (d)), lung (Figure 6(c) and (d)), heart (Figure 7(c) and (d)), liver (Figure 8(c) and (d)) and brain (Figure 9(c) and (d)) after 48 h of treatment.
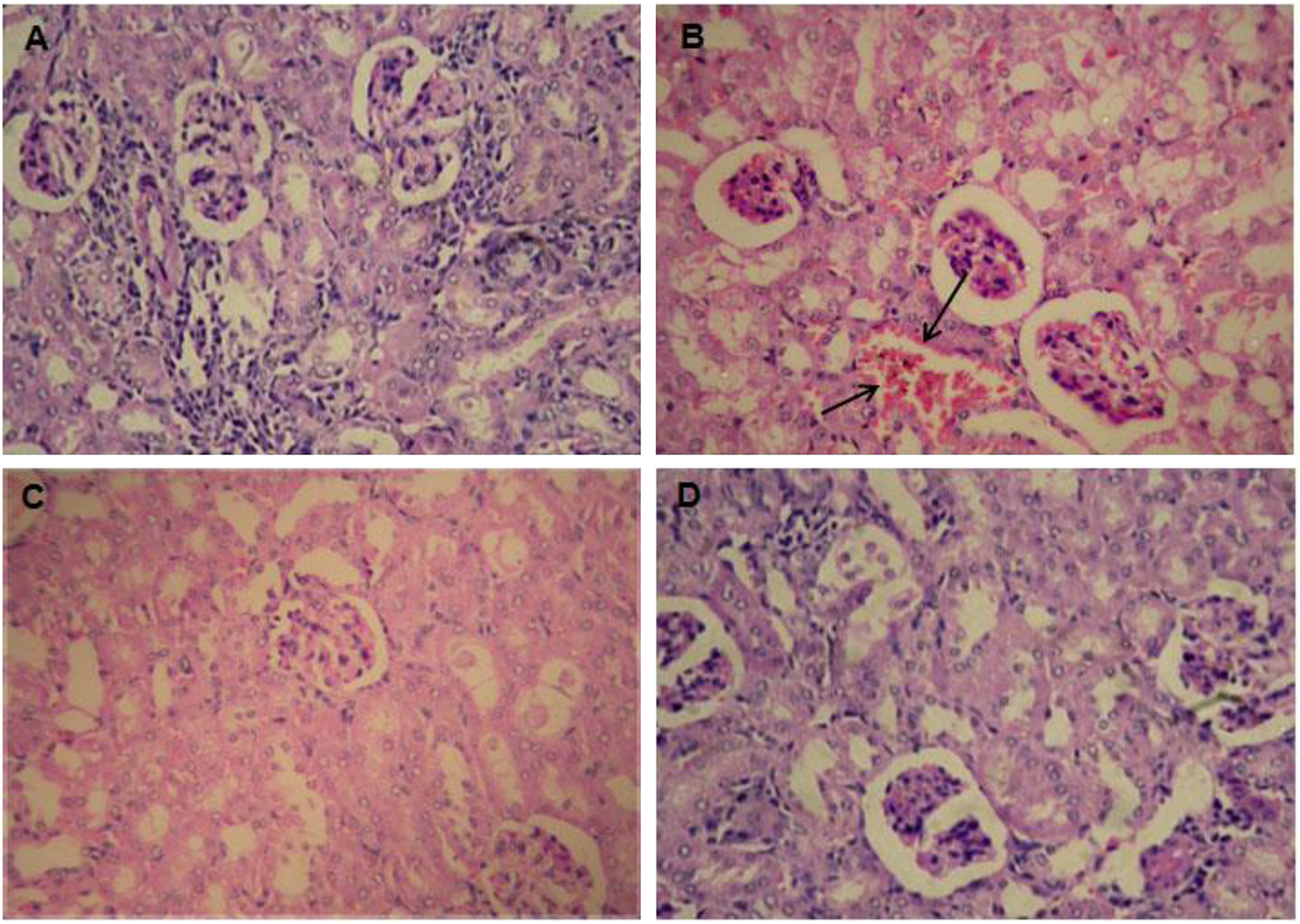
Histological changes in the kidney of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a) Normal mice; (b) envenomated mice, the glomerular congestion and haemorrhagic regions are denoted by arrows; (c) envenomated mice treated with triterpenoid from plant extract; (d) envenomated mice treated with commercial antiserum.

Histological changes in the liver tissue of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a)Normal mice; (b) envenomated mice, the congestion of blood vessels is denoted by arrows; (c) envenomated mice treated with triterpenoid from plant extract; (d) envenomated mice treated with commercial antiserum. No changes were observed in (c) and (d).

Histological changes in the lung tissue of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a)Normal mice; (b) envenomated mice, the infiltration of erythrocytes and thickening of partitions between alveoli are denoted by arrows; (c) envenomated mice treated with triterpenoid from plant extract; (d) envenomated mice treated with treated with commercial antiserum with infiltration of erythrocytes.

Histological changes in the heart tissue of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a) Normal mice; (b) envenomated mice, the disruption in muscle fibres with hemorrhagic regions is shown by arrows; (c) envenomated mice treated with plant extract; (d) envenomated mice treated with commercial antiserum. No changes were observed in (c) and (d). Normal texture was observed in the heart tissue in treated envenomated mice.

Histological changes in the spleen of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a) Normal mice; (b) envenomated mice, the congestion of red pulp is shown by arrows; (c) envenomated mice treated with triterpenoid from plant extract with normal structure; (d) envenomated mice treated with commercial antiserum and normal feature was observed in spleen.

Histological changes in the brain of envenomated mice and envenomated mice treated with triterpenoid from plant extract or commercial antiserum. (a) Normal mice; (b) envenomated mice; (c) envenomated mice treated with triterpenoid from plant extract; (d) envenomated mice treated with commercial antiserum. No significant changes were observed in envenomated mice and treated envenomated mice.
Discussion
The four major poisonous snakes cobra, krait, Russell’s viper, and saw-scaled viper are responsible for fatality due to snakebite in India. The venoms of cobra and krait, belonging to the Elapidae family, are neurotoxic in nature and usually result in heart failure. Immunotherapy using polyvalent antivenom is the only effective treatment against snake venom poisoning. Antisnake venom therapy may cause various side effects such as anaphylactic shock, pyrogen reaction and serum sickness. 33,41,42 These side effects may be due to the presence of high concentrations of non-immunoglobulin proteins in many commercially available antivenoms. Moreover, antivenom production in animals is time consuming, expensive and requires ideal storage conditions. In view of the above facts, an alternate technology for the production of antivenom has to be developed or a suitable snake venom neutralizing agent has to be found. The present study was carried out to explore the cobra venom neutralizing activity of triterpenoid from methanol extract of Indian medicinal plant L. aspera Linn.
Eight protein bands with molecular weights of 14, 24, 29, 45, 48, 65, 72 and 99 kDa were observed on SDS-PAGE in the venom of N. naja Naja. Similar type of protein banding pattern was observed by many workers. 43 –46 Cobra venom subjected to SDS-PAGE analysis indicated the presence of prominent protein components with molecular weights of 10, 20, 24, 55, 105 and 110 kDa. 45
In the present study, the toxicity of cobra whole venom was tested in vivo using mice. The LD50 values of cobra venom in mice weighing 20–22 g for different routes were determined as 13.73, 8.44, 13.37 and 13.73 µg per mouse for i.p., i.v., i.m. and s.c. routes, respectively. The LD50 values of cobra venom through different routes in the present study coincided with values reported by many workers. 2,47 –52 . The lethality of cobra venom was determined by i.p. injection into 20 g mice and the LD50 value was calculated as 7.3 µg per mouse. 47 Shashidharamurthy et al. (2002) conducted toxicity experiments with cobra venom collected from different regions of India in mice through i.p. route and calculated the LD50 value as 14 µg per mouse for eastern, 24 µg per mouse for western and 40 µg per mouse for southern venoms of cobra.44
India is very rich in herbal plants and traditional herbal medicine is readily available in rural areas for the treatment of snakebite. Application of the plant or its sap onto the bite area, chewing leaves and bark or drinking plant extracts or decoctions are some procedures intended to counteract snake venom activity. In the rural areas (e.g. Nambedu village, Thiruvannamalai District, Tamil Nadu, India), the leaves of L. aspera are chewed as an antidote by victims of snakebite particularly cobra bite. The results of preliminary screening of 12 plants showed that the aqueous extract of L. aspera Linn. protected all the envenomated mice treated with 300 mg per mouse of aqueous plant extract. Based on the traditional use of L. aspera for snakebite and the results of preliminary screening, this plant was selected for further studies. The observation of the present study on antivenom activity against cobra venom was supported by Kannappa Reddy et al. who observed antivenom activity against viper venom but detailed studies particularly on isolation and identification of the compound responsible for neutralization have not been carried out. 53 Similar type of antivenom activity against different snake venoms was also observed by different workers in different medicinal plants such as Hemidesmus indicus, 54 Mimosa pudica, 55 Vitex negundo and Emblica officinalis, 56 Acalypha indica, 57 Annona senegalensis, 58 Crinum jagus, 59 Musa paradisiaca, 60 Casearia sylvestris 61 and Parkia biglobosa. 62 Perumal Samy et al. conducted a survey covering 72 medicinal plants belonging to 53 families used for snakebite treatment in the traditional way, and the results showed that the fractions of six plants showed potent neutralizing effect against the venom. 63
In the present study, various Indian medicinal plants were screened for their ability to neutralize the lethality of cobra venom. These plants were selected based on previous works. 9,55,64 –67 The results obtained from preliminary investigations on the use of various previously reported plants with antivenom activity against cobra venom provide evidence of the importance of some plants to neutralize cobra venom in experimental studies using mice. Complete (100%) neutralization was fixed as the bench mark to define an extract with antivenom activity. Of the 12 plants tested, the aqueous extract of the leaf of the herb L. aspera Linn. was found to be the most effective against cobra venom; the same herb was reported for its antivenom activity against viper venom. 55,65 This plant was selected for the present study since not much work has so far been carried out especially with regard to its ability to neutralize venom and the compound responsible for neutralization.
The aqueous extract of L. aspera Linn. showed strong antivenom activity against cobra venom. Various solvents were used to obtain the plant extracts to test their antivenom activity in mice. The methanolic extract of L. aspera Linn. leaves showed 100% survival in the envenomated mice treated with methanolic extract of L. aspera Linn. at the concentration of 200 mg per mouse, while extracts with ethanol, petroleum ether, chloroform, ethyl acetate, benzene and diethyl ether could not protect the envenomated mice. Our observation agrees with the studies carried out by many researchers 56,59,68 –70 on antivenom activity of methanloic root extract of Pluchea indica. Alam et al. reported that the methanolic root extract of P. indica could neutralize the lethality of viper venom. 68 The compounds, such as β-sitosterol and stigmasterol, responsible for neutralizing activity were isolated from the methanolic extracts of the root of P. indica and were found to neutralize viper venom-induced lethality, haemorrhage and oedema in experimental animals. 70 The results of the present study on testing the different solvent extracts of L. aspera Linn. showed significant antivenom activity in the methanol extract as observed by many researchers. 59,68 –70
Fraction B3B1 was obtained as a brown solid at the end of level III purification. The TLC pattern showed one spot indicating that it is a pure compound.
Based on the results obtained in the present study, this compound was identified as 1-hydroxytetratriacontan-4-one as reported by Misra et al. who identified different compounds including 1-hydroxytetratriacontan-4-one in L. aspera Linn. 71 They isolated two long-chain compounds, 1-hydroxytetratriacontan-4-one and 32-methyltetratriacontan-8-ol, from the leaves and shoots of L. aspera Linn. 71 –73 The compound isolated from L. aspera Linn. responsible for its antivenom activity against cobra venom was identified as 1-hydroxytetratriacontan-4-one as reported by Misra et al. 71 This plant is being used for the treatment or as a supplementary treatment before immunotherapy for cobra snakebite in the rural areas. Among the compounds reported in L. aspera Linn., α-sitosterol, β-sitosterol and β2-sitosterol were found to have strong antivenom activities. 71,74,75 In the present study, strong antivenom activity against cobra venom was found in 1-hydroxytetratriacontan-4-one isolated from L. aspera Linn. As observed in the present study, Gomes et al. reported the neutralization of viper and cobra venom by β-sitosterol and stigmasterol isolated from the root extract of P. indica, and these authors also suggested that these compounds may play an important role, along with antiserum, in neutralizing snake venom-induced actions. 70 Based on the results of the present study, we also recommend the use of our plant compound along with antiserum to enhance the potency of antiserum to neutralize the snake venom after carrying out clinical studies.
The antivenom activity of pure compound against cobra venom was tested in vivo using mice and the pure active compound showed strong antivenom activity against cobra venom at the concentration of 69.50 mg per animal with 100% survival, while lesser concentrations showed different mortality rates. This indicates that the pure compound was found to be dose dependent against cobra venom.
Since it has strong antivenom activity against cobra venom, further works on isolation and characterization of active compound of the plant responsible for its antivenom activity have been carried out to explore the possibility of developing an alternative treatment to serum therapy.
Analyses of antioxidant enzymes and histological investigations were carried out in mice injected with cobra venom, and the results were compared with that of mice injected with mixture of cobra venom and plant extract or commercial antiserum.
Significant decrease in the levels of antioxidant enzymes was found in heart, liver, kidney and lung of envenomated mice but no alteration was found in the envenomated mice treated with plant extract or commercial antiserum. All organisms are known to contain high levels of polyunsaturated fatty acids, which are the substrates for lipid peroxidation, and the presence of antioxidants elevates the resistance of their cell membranes to oxidative stress. 76 The present study has shown that the level of lipid peroxidation was significantly increased in cobra venom-injected tissues (heart, liver, kidney and lung) of mice when compared with control animals. The increased level of lipid peroxidation might have led to the production of malondialdehyde enhancing the formation of free radicals on polyunsaturated fatty acids in the cell membranes. In addition, the extensive lipid peroxidation especially of the highly unsaturated fatty acids, altering the ratio of polyunsaturated to other fatty acids could have resulted in decreased membrane fluidity and membrane disorganization. 54,63,77
SOD is one of the main antioxidant defence enzymes generated in response to oxidative stress. It converts the highly toxic superoxide anions into hydrogen peroxide (H2O2). 78 The activity of SOD was significantly lowered in heart, liver, kidney and lung of cobra venom-injected mice in the present study as observed by Perumal Samy et al.. 63 No change in the activity of SOD was observed in the cobra venom-injected mice treated with the plant extract or commercial antiserum. H2O2 is toxic to cells, and CAT is a major primary antioxidant defence component that catalyses the decomposition of H2O2, which is produced by the action of SOD or GPX to water. Although both the enzymes (SOD, GPX) detoxify H2O2, organic peroxides are the preferred substrate for GPX. At high H2O2 concentrations, organic peroxides are metabolized by CAT. 79 The present study showed that the activity of CAT and GPX was significantly decreased in heart, liver, kidney and lung tissues of cobra venom-injected mice, while no change was found in envenomated mice treated with plant extract or commercial antiserum. 80 Bannister et al. 86 (1986) have reported that GPX is inactivated with increased levels of H2O2, which in turn is due to the lower activity of CAT. 57,81 In tissues, GSH is maintained predominantly in the reduced form, where it acts as major source of reducing power and functions in the reduction of oxidized tissue components. 82 GSH is regenerated from oxidized GSH by nicotinamide adenine dinucleotide phosphate, requiring GSH reductase. In the present study, the activity of GST was also significantly decreased in different tissues of envenomated mice, while there was no significant alteration in envonamted mice treated with plant extract or commercial antiserum. The decreased activity of GST in cobra venom-injected mice might be due to the decreased availability of reduced GSH. 57 In conclusion, phytochemicals from the medicinal plant L. aspera Linn. herbs possess antioxidative properties, which are associated with their direct free radical scavenging abilities.
Significant histological changes such as infiltration of erythrocytes and congestion of blood vessels were observed in the lung tissue as observed by Ali et al. 83 and Aznaurian and Amiryan. 84 Ali et al. have concluded that the mechanism for lung injury was not due to the direct action of the venom or its components, but probably due to factors related to renal dysfunction due to envenomation. Light microscopic examination of the heart tissue sections of envenomated mice revealed a disruption in the striations of the muscles as observed by many workers. 81,82 This change may cause irregularities in contraction of atria and ventricles of the heart and rhythmical beating of heart, which in turn affects the circulation of blood in the body. Histological observation of liver tissue of envenomated mice revealed the congestion of blood vessels and capillaries. The obstruction of blood vessels may cause liver dysfunction.
In conclusion, our study confirms that L. aspera Linn. extract is a possible way forward for effectively antagonizing the cobra venom toxicity of different parameters in replacing the commercially available antiserum. Triterpenoid from the methanol extract of L. aspera Linn. could effectively counteract and neutralize the toxicity of cobra venom with reference to the antioxidant and histological derangements in different vital organs of mice. Furthermore, this efficacy of L. aspera Linn. must be confirmed both with in vivo and in vitro cell models. This needs to be further elucidated using higher mammals before proceeding to preliminary clinical trails.
Footnotes
Acknowledgements
The first author is recipient of a Young Scientist award from the Department of Science and Technology, Government of India. The authors thank the management of C. Abdul Hakeem College for providing facilities to carry out this work and the secretary, Irula Snake Catchers Industrial Cooperative Society, Chennai, Tamil Nadu, India, for providing the snake venoms for this study. The authors are grateful to Mr P. Thomas John, Former Head, Department of Zoology, C. Abdul Hakeem College, Melvisharam, Vellore, Tamil Nadu. The authors are also thankful to the King Institute of Preventive Medicine for providing snake venom antiserum.
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
