Abstract
Philodryas baroni—an attractively colored snake—has become readily available through the exotic pet trade. Most people consider this species harmless; however, it has already caused human envenomation. As little is known about the venom from this South American opisthoglyphous “colubrid” snake, herein, we studied its protein composition by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), as well as its effects on the hemostatic system. Both reducing and nonreducing SDS-PAGE analysis demonstrated that the venom exhibits greatest complexity in the range of 50–80 kDa. The venom displayed proteolytic activity toward azocollagen, with a specific activity of 75.5 U mg−1, and rapidly hydrolyzed the Aα-chain of fibrinogen, exhibiting lower activity toward the Bβ- and γ-chains. The venom from P. baroni showed no platelet proaggregating activity per se, but it inhibited collagen- and thrombin-induced platelet aggregation. Prominent hemorrhage developed in mouse skin after intradermal injection of the crude venom, and its minimum hemorrhagic dose was 13.9 μg. When injected intramuscularly into the gastrocnemius of mice, the venom induced local effects such as hemorrhage, myonecrosis, edema, and leucocyte infiltration. Due to its venom toxicity shown herein, P. baroni should be considered dangerous to humans and any medically significant bite should be promptly reviewed by a qualified health professional.
Introduction
Philodryas, now considered a member of the family Dipsadidae, 1 is a genus of rear-fanged “colubrid” snakes that are found in South America, from Amazonas to Patagonia. 2, 3 However, Philodryas baroni is largely restricted to the north central Gran Chaco region of Argentina. 4 It is the largest species of the genus and is strictly arboreal, living in tropical and subtropical forests. This species is characterized by an extension of the rostral scales, being more developed in male than in female. Its body color is rather variable; there are green specimens (Figure 1) as well as blue or brown ones. For all these physical features, P. baroni snakes have become popular as pets, and they have become readily available through the exotic pet trade.

Green specimen of Philodryas baroni snake. Picture was taken by Cinthia Calamante.
Most people consider this species harmless; however, it has already caused human envenomation. Although rare and not yet reported to be lethal, human envenomation by this “colubrid” snake is characterized by pain, bleeding, swelling, heat, and ecchymotic lesions on the bitten limb, bearing a striking resemblance to local signs and symptoms of Bothrops sp.—front-fanged Viperidae snake—envenomation. 4, 5 The small number of well-documented cases of medically significant bites from P. baroni describes only mild to moderate local effects resulting from these incidents. Uncommon cases that include systemic effects (widespread ecchymoses) have occurred but are rarely documented. However, large specimens of this species may be capable of inflicting a bite resulting in significant systemic effects. 6
The properties of crude venoms from Philodryas olfersii and Philodryas patagoniensis have been studied extensively. 2, 7–18 However, little is known about the composition and biological activities of the venom from P. baroni. 19, 20 Thus, the present study deals with the biological characterization of the venom from this South American “colubrid” snake. Moreover, it provides a biochemical explanation for the hemorrhagic manifestations commonly reported to occur in patients at the site of the bite. On the whole, this study aims to contribute to a better understanding of the pathophysiology of “colubrid” snake envenomation, the consequences of which physicians and other health professionals are often poorly prepared to deal with.
Materials and methods
Reagents
Human fibrinogen, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), phenylmethylsulfonyl fluoride (PMSF), o-phenanthroline (o-Phe), azocoll, cetyltrimethylammonium bromide, hyaluronic acid, collagen, and bovine thrombin were purchased from Sigma Chemical (St Louis, Missouri, USA). Broad-range molecular mass markers were purchased from Bio-Rad Laboratories (Hercules, California, USA). Creatine kinase (CK) levels were determined using an ultraviolet kinetic kit purchased from Wiener Laboratories (Rosario, Santa Fe, Argentina). All other chemicals were of analytical grade or better.
P. baroni venom
We obtained P. baroni venom from adult wild specimens captured in northeastern Argentina and maintained at the serpentarium of Corrientes city (Argentina). Specimens were extracted by introducing a 100-μL micropipette under each fang, according to the procedure described previously by Ferlan et al. 21 Venom was lyophilized and thereafter kept frozen at −20°C until use. When required, venom was dissolved in 50 mM tris(hydroxymethyl)aminomethane–hydrochloric acid (Tris-HCl) buffer (pH 7.4). The small amount of insoluble material was removed by centrifugation, and the clear supernatant was utilized for studies. The protein content was estimated by measuring the absorbance at 280 nm, 22, 23 assuming that an absorbance of 1.183 corresponds to a protein concentration of 1 mg mL−1. 10
Animals
To determine the minimal hemorrhagic dose (MHD) and the local damage induced by P. baroni venom,
Electrophoresis
Analysis by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was used to evaluate the protein profile of P. baroni snake venom and to compare venoms from specimens of different sex and color. Venom samples of different specimens (two female brown-colored specimens, two female green-colored specimens, and two male green-colored specimens) were electrophoresed on 12% polyacrylamide slab gels following the method of Laemmli 24 and were then stained with silver or blue-silver Coomassie stain. 25, 26
Proteolytic activity
The proteolytic activity of P. baroni venom was estimated using two different substrates: collagen (the main protein of blood vessel walls) and fibrinogen (the major clotting protein in blood plasma).
Collagenolytic activity
A quantitative assay using azocoll as substrate was performed. 27 Briefly, two-fold serial dilutions (200 μL) of venom were incubated with 600 μL of 1 mg mL−1 azocoll solution in Tyrode buffer (137 mM sodium chloride (NaCl), 2.7 mM potassium chloride, 3.0 mM monosodium phosphate, 10 mM HEPES, 5.6 mM dextrose, 1 mM magnesium chloride, 2 mM calcium chloride, pH 7.4) for 1 h at 37°C, and the samples were carefully homogenized for every 5 min during incubation; the reaction was halted by placing samples on ice. After centrifugation for 3 min at 5000 g, the absorbance of the supernatant was measured at 540 nm. One unit of enzymatic activity was defined as the amount of venom that causes an increase in 0.003 units of absorbance per minute at 540 nm. Specific activity was expressed in units per milligram of lyophilized venom. For inhibition studies, P. baroni venom (51.5 μg mL−1) was incubated with the following inhibitors: o-Phe or PMSF (5 mM in both cases, final concentration) for 1 h at 37°C before incubation with azocoll or concomitantly with this substrate (without preincubation).
Fibrinogenolytic activity
Specific cleavage of fibrinogen by crude venom was determined by SDS-PAGE using 12% polyacrylamide gels, as described elsewhere. 28 A 200 µL of 2 mg mL−1 human fibrinogen dissolved in 50 mM Tris-HCl buffer (pH 7.4) was incubated at 37°C with 10 μL of venom (634 μg mL−1). At various time intervals, aliquots were withdrawn from the digestion mixture and then denatured and reduced by boiling for 7 min with denaturing solution (4% SDS, 20% glycerol, and 20% 2-mercaptoethanol) for SDS-PAGE. For inhibition studies, the reaction mixture composed of fibrinogen and P. baroni venom was incubated with the following inhibitors: o-Phe or PMSF (5 mM in both cases, final concentration) for 12 h at 37°C.
Hyaluronidase activity
A previously reported method was used to assay this activity. 29 Twofold serial dilutions (10 μL) of venom in 50 mM Tris-HCl buffer (pH 7.4) were incubated with 40 μL of hyaluronic acid solution (0.5 mg mL−1 in 0.2 M acetate buffer, pH 6.0, containing 0.15 M NaCl) at 37°C for 15 min. The reaction was stopped by adding 150 μL of 2.5% cetyltrimethylammonium bromide in 2% sodium hydroxide, and the absorbance was read at 405 nm in a Multiskan Ascent microplate photometer (Thermo Electron Co., Waltham, Massachusetts, USA). Acetate buffer was used as a blank, and a sample containing only hyaluronic acid solution was used as a measure of 100% turbidity.
Platelet aggregation assay
The effect of P. baroni venom on the aggregation of washed human platelets was tested as described previously. 30 Platelet count was ascertained to be 300 × 109 L−1 and platelet aggregation was performed in a lumiaggregometer (model 560VS; Chronolog Corporation, Havertown, Pensylvania, USA) by the method of Born. 31 Collagen (2.5 µg mL−1, final concentration) or bovine thrombin (0.2 U mL−1, final concentration) were used as agonists of platelet aggregation.
Hemorrhagic activity
This activity was evaluated by a method described previously by Peichoto et al. 28 Different doses of the venom (2–127 μg) were injected intradermally in mice, using three to five animals per dose. Two hours after injection, animals were killed, and the skin was removed. Hemorrhagic haloes were immediately cut, fragmented, and added to tubes containing 4 mL of Drabkin’s reagent. The tissue fragments were immediately homogenized, and the reaction mixtures were incubated in the absence of light at room temperature for 24 h. Thereafter, tubes were centrifuged at 5000 g for 5 min. The absorbance of the supernatant at 540 nm was determined in a spectrophotometer (Boeco S-22, Hamburg, Germany) and then hemoglobin concentration was calculated. 32 The MHD was defined as the minimal concentration of venom able to induce a three-fold increase in hemoglobin concentration in relation to that of a control tissue injected only with vehicle. 28
Local tissue damage induced by P. baroni venom on the mouse gastrocnemius muscle
Groups of four mice were injected in the right gastrocnemius muscle with 40 μg of P. baroni venom dissolved in 100 μL of 50 mM Tris-HCl buffer (pH 7.4). Control mice were injected only with vehicle under identical conditions. After 1, 3, 6, 9, 12, 15, 18, 21, and 24 h of injection, mice were anesthetized with chloral hydrate intraperitoneally (300 mg kg−1), and blood samples were collected from the abdominal portion of the aorta. Serum was obtained for the enzymatic determination of CK levels using an ultraviolet kinetic kit. For histological analysis, small samples of muscle were collected and fixed in Bouin’s solution for 24–48 h. Thereafter, the samples were dehydrated in an ethanol ascending series, cleared in xylol, and embedded in liquid paraffin. Sections of 4 µm thickness were cut and stained with hematoxylin and eosin and examined under a light microscope.
Statistical analysis
Where appropriate, the results were expressed as mean ± SD. The differences between groups were compared using one-way analysis of variance followed by Dunnett’s test. Statistical analyses were performed using the software GraphPad InStat, version 3.01 (GraphPad Software, San Diego, California, USA.) A value of p < 0.05 indicated statistical significance.
Results and discussion
Although the rear-fanged snakes in general are not regarded as clinically important due to their less efficient (to humans) venom delivery apparatus, a growing number of accidents caused by these snakes have been reported in epidemiological studies 33 and reviewed by Weinstein et al. 6 However, the vast majority of “colubrid” venoms are still unstudied, mainly because of the exceedingly small quantities of raw material obtained upon extraction. 34 Herein, we characterized the venom of the argentine long-nosed tree snake—P. baroni— a striking rear-fanged snake gaining increasing use as an exotic pet in the last years, but potentially capable of causing envenomation in humans. 4, 5
Both reducing and non-reducing SDS-PAGE analysis were in accordance with recent results from our group, 20 showing that the “colubrid” venom studied in this work contains numerous protein bands, mainly in the range of 50–80 kDa (Figure 2). These protein bands may represent metalloproteinases of the P-III class, which are the major components of P. olfersii venom and the main ones responsible for its most toxic effects. 12 As in many other “colubrid” venoms, 35 a prominent band at approximately 25 kDa was also observed. It may correspond to a cysteine-rich secretory protein that is a common component of most reptile venoms. 34, 36, 37

SDS-PAGE in 12% gel of Philodryas baroni venom (4 μg). Lane 1: venom obtained from a female brown-colored specimen. Lane 2: venom obtained from a female green-colored specimen. Lane 3: venom obtained from a male green-colored specimen. Lane 4: venom obtained from a female green-colored specimen. MM: molecular mass standards. Gel was blue-silver Coomassie stained. SDS: sodium dodecyl sulfate; PAGE: polyacrylamide gel electrophoresis.
In addition, electrophoretic similarity was observed between the venoms of brown and green specimens (Figure 2), which suggests that there may be no variation in venom sample composition related to body color of the snake. However, male and female venoms under non-reducing conditions showed minor variation, characterized by the presence of a higher amount of high molecular mass components in the male venom (Figure 2). Therefore, a sex-based difference in the protein composition of P. baroni venom might exist, as it was previously reported for the venom of Bothrops jararaca. 38 Unfortunately, the low amount of venom produced by this “colubrid” snake and the fact that only a few male specimens were kept in captivity for the production of venom limited the possibilities of studies. Further experiments were carried out with venom from three female specimens, all performed in duplicate.
P. baroni venom showed proteolytic activity on both the substrates tested in this study, that is, collagen and fibrinogen. The venom displayed dose-dependent proteolytic activity toward azocollagen, with a specific activity of 75.5 U mg−1, which is 25 times greater than that reported for adult B. jararaca venom. 39 It is in accordance with a previous report 7 showing that P. patagoniensis venom is much more proteolytic than the venom of Bothrops alternatus. Similarly, Rocha et al. 17 demonstrated that P. olfersii and P. patagoniensis venoms exhibit higher proteolytic activity than B. jararaca venom. 1,10-Phenanthroline (5 mM, final concentration) completely abolished the collagenolytic activity of the venom (about 98% inhibition), but 5 mM PMSF had only a partial inhibitory effect that was significant (about 70% inhibition) only when the venom was pre-incubated with the inhibitor. This suggests the predominance of proteases exhibiting a divalent cation-dependent behavior in the venom of P. baroni, which is in agreement with a recent work from our group 20 showing that this venom displays low levels of catalytic activity toward synthetic substrates for serine proteinases. Rocha et al. 17 suggested that metalloproteinases present in the venoms of P. olfersii and P. patagoniensis are the components responsible for their major toxic effects.
Some “colubrid” snake venoms, including those of P. olfersii and P. patagoniensis, have been shown to degrade fibrinogen, interfering with blood coagulation via hypofibrinogenemia. 2, 3, 10, 28 Similarly, in this work, P. baroni venom rapidly hydrolyzed the Aα-chain of fibrinogen, exhibiting lower activity toward the Bβ-chain, and even lower toward the γ-chain (Figure 3(a)). It is worth noting that most “colubrid” venoms tested for this activity before this work did not affect the γ-subunit of fibrinogen, 2, 10, 35 but in all those cases, the incubation time did not exceed 2 h.

Fibrinogen degradation by Philodryas baroni venom, analyzed by SDS-PAGE (12% gel), under reducing conditions. (a) Time dependence of the proteolytic activity on human fibrinogen of the venom. (b) Effect of inhibitors of metalloproteinases (o-Phe) or serine proteinases (PMSF; 5 mM in both cases, final concentration) on the digestion of fibrinogen by the venom at 37°C for 12 h. Lane 1: fibrinogen control. Lane 2: fibrinogen incubated with venom. Lane 3: fibrinogen incubated with venom + o-Phe. Lane 4: fibrinogen incubated with venom + o-Phe + PMSF. Lane 5: fibrinogen incubated with venom + PMSF. Gels were silver stained. o-Phe: o-phenanthroline; PMSF: phenylmethylsulfonyl fluoride.
Fibrinogenolytic activity was completely abolished when the venom was simultaneously incubated with 1,10-phenanthroline and PMSF for 12 h at 37°C (Figure 3(b)). These results show that some of the fibrinogenases present in P. baroni venom are metalloproteinases capable of degrading Aα-, Bβ- and γ-chains, and others are serine proteinases exhibiting ability of degrading only Aα-chain throughout the incubation period examined. Similar results were obtained by our group, 10 when testing the fibrinogenolytic activity of P. patagoniensis venom.
When added to washed human platelet suspensions, P. baroni venom showed no platelet pro-aggregating activity per se up to a final concentration of 84.1 μg mL−1, but it partially inhibited platelet aggregation induced by 0.2 U mL−1 thrombin (Figure 4). At a final concentration of 14.9 μg mL−1, the venom completely inhibited the aggregation of washed human platelets induced by 2.5 μg mL−1 collagen. Similarly, Peichoto 40 demonstrated that the venom of P. patagoniensis inhibits the aggregation of washed human platelets induced by either thrombin or collagen.

Inhibitory effect of Philodryas baroni venom on platelet aggregation from human washed platelets (300 × 109 L−1) stimulated by thrombin or collagen. Final venom concentrations in the suspension of washed platelets are shown.
Prominent hemorrhage developed in mouse skin after intradermal injection of the crude venom (Figure 5), and its MHD was 13.9 μg, which is lower than that reported for adult B. jararaca venom. 39 We can assume that hemorrhagic phenomena caused by P. baroni venom in experimental animals, and likely also in human victims, is the result of the synergistic action of metalloproteinases that degrade the extracellular matrix surrounding the blood vessels, fibrinogenolytic enzymes that interfere with the blood clot cascade, and components that inhibit platelet aggregation and thrombus formation. Moreover, it is possible that the metalloproteinases that possess hemorrhagic activity are, at least in part, the very same enzymes that are responsible for the cleavage of fibrinogen and/or the platelet aggregation inhibition, as it was already demonstrated for P. patagoniensis venom. 28

Local hemorrhage induced by different amount of Philodryas baroni venom in mouse skin.
After intramuscular (i.m.) injection, P. baroni venom (40 μg per animal) induced an increase in serum CK activity (Figure 6), which is a specific marker for muscle damage. Macroscopically, the appearance of the venom-treated muscle was clearly different from the control sample at all periods of time (data not shown). Histological analysis of the muscle samples obtained after 1, 3, 6, 9, 12, 15, 18, 21, and 24 h of injection corroborated the myotoxicity of the venom. The control sample retained a normal appearance, whereas those samples of gastrocnemius muscle injected with 40 μg of P. baroni venom showed prominent damage characterized by hemorrhage, necrosis, inflammatory infiltrate, and edema (Table 1 and Figure 7). Our group previously reported similar CK changes and morphological alterations induced by the venoms of P. olfersii and P. patagoniensis, when injected into the gastrocnemius muscle of mice. 8, 9 Moreover, the very same local lesions observed herein were also observed when studying the venom of the viper B. alternatus, 41, 42 which is one of the main species responsible for snakebites in Argentina.
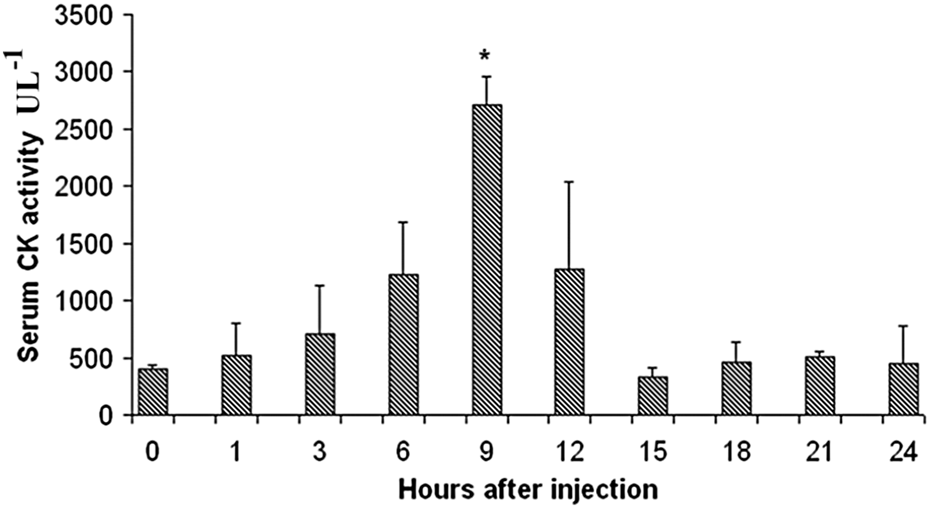
Changes in mouse serum CK levels after intramuscular injection of 40 μg Philodryas baroni venom. Bars represent the mean ± SD of four individual experiments. *p < 0.05 indicates statistically significant differences with the untreated control (first bar). CK: creatine kinase.

Light micrographs showing the histopathological changes induced by 40 μg Philodryas baroni venom in mouse gastrocnemius muscle. In the figure, extravasation of erythrocytes, inflammatory infiltrate of polymorphonuclear leukocytes, and cell necrosis induced by the venom are shown. *coagulation-type necrosis. #myolysis. Sections were stained with hematoxylin–eosin.
Histological analysis of samples of gastrocnemius muscle injected with 40 μg of Philodryas baroni venom.

−: no lesion detected; +: mild lesion; ++: moderate lesion; +++: severe lesion.
In most cases, snakebite results in subcutaneous/i.m. injection of venom into the prey or human victims leading to complicated pathology. The tissue destruction and hemorrhage is primarily due to the degradation of structural and adhesive components such as collagen, elastin, fibronectin, and glycosaminoglycans of the extracellular matrix of connective tissue, surrounding the blood vessels and capillaries, by matrix-degrading venom hyaluronidases and metalloproteinases. These two groups of hydrolytic enzymes appear to be the principal agents responsible for tissue degradation at the bite site. 43 However, these degradative enzymes vary in relative abundance in different venoms. 44 Rear-fanged snake venoms are rich in hemorrhagic metalloproteases, 3, 28, 34, 45–48 but hyaluronidase activity has been detected only in a few “colubrid” venoms. 34 In this work, P. baroni venom did not exhibit hyaluronidase activity, even at a concentration of 4 mg mL−1. This is in accordance with a previous report 45 showing that none of the venoms from 12 species of “colubrid” snakes exhibit hyaluronidase activity, which is typically found in front-fanged snake venoms. Maybe, because of the absence of this activity, systemic toxicity induced by P. baroni venom has not been observed to date. But the absence of this toxicity could more likely be due to small amounts of or not very active neurotoxins since Prado-Franceschi et al. 18 and Carreiro da Costa et al. 13 have demonstrated that the venoms of P. olfersii and P. patagoniensis contain components that can affect neuromuscular function.
In conclusion, this work shows for the first time the biological characterization of the venom from P. baroni, an attractively colored snake, and thus commonly kept as an exotic pet. In addition, it provides a biochemical explanation for the hemorrhagic manifestations commonly reported to occur in patients bitten by P. baroni. Due to the toxicity exhibited by the venom of this snake, herein, it is advisable to consider this species dangerous to humans—especially infants and young children for whom its fangs are large enough to penetrate their skin—and special care must be taken while manipulating this snake. It is also advisable to take into account that any medically significant bites are promptly reviewed by a qualified health professional, who must know very well this kind of accident in order to provide the correct treatment to victims. Finally, the data presented in this article give insight into future directions for research on the venom of P. baroni.
Footnotes
Acknowledgment
We thank Marcelo L Santoro (Instituto Butantan, Brazil) for helping with the platelet aggregation assays.
Conflict of interest
The authors declared no conflict of interest.
Funding
MNS and AT are recipients of a fellowship from Secretaría General de Ciencia y Técnica (SGCyT), Universidad Nacional del Nordeste (UNNE), Argentina. Author MNS was granted with a research travel fellowship from the SGCyT-UNNE (Res. N° 5114/2011 C.S.) . Additional financial support was provided by SGCyT-UNNE (PI B013-2010), CONICET (PIP 114-200801-00088), Agencia Nacional de Promoción Científica y Tecnológica (PICT-2010-1908) from Argentina, and the international cooperation project CONICET/FAPESP (2010/52559-3).
