Abstract
Knowledge of the ability of the female reproductive system to metabolize environmental chemicals is critical not only from the standpoint of toxicity but also from infertility risk assessment. Benzo(a)pyrene (BaP) is a toxicant that is released into the environment from automobile exhausts, cigarette smoke, burning of refuse, industrial emissions, and hazardous waste sites. In exposed animals, BaP becomes activated to reactive metabolites that interfere with target organ function and as a consequence cause toxicity. Studies on animal models conducted in our laboratories and those of others have shown that BaP possess endocrine disrupting properties. Thus, this chemical has the potential to cause infertility and cancers in the female genital tract. An understanding of BaP metabolism in the female reproductive system will be of importance in the diagnosis and management of female fertility as well as cancers in the reproductive tissues. Therefore, the objective of our study was to examine the metabolism of BaP by human ovarian subcellular fractions. Human ovary samples (eight individuals) were obtained from postoperative tissue removed from subjects with uterine tumors. Subcellular fractions (nuclear, cytosolic, mitochondrial, and microsomal) were prepared by differential centrifugation. BaP (1 μM and 3 μM) was individually incubated with individual subcellular fractions for 15 min and the products were analyzed by high-performance liquid chromatography. Among the different fractions tested, microsomal BaP metabolism was higher than the rest of the fractions. The BaP metabolites identified were as follows: BaP-9,10-diol, BaP-4,5-diol, BaP-7,8-diol, 9(OH) BaP, 3(OH) BaP, BaP-1,6-dione, BaP-3,6-dione, and BaP-6,12-dione. Of interest was the presence of DNA-reactive metabolites such as BaP-3,6-dione, BaP-6,12-dione, and BaP 7,8-diol, which have been implicated in the causation of infertility and cancer. Our results indicate that women who are exposed to BaP via cigarette smoke, occupational settings, and diet are more likely at a larger risk of this toxicant-induced infertility and cancer than others.
Introduction
Recently, infertility among women has become a matter of concern 1 and it is projected to increase from 4.9 × 106 in mid 1990s to about 7.7 × 106 by 2025. 2 Some infertility cases are suspected to originate from exposure/exposures to chemicals in the occupational, domestic environments, and lifestyle habits such as smoking.3,4
Some of the environmental chemicals act as reproductive endocrine disruptors by binding to the receptors of ligands that regulate reproductive system, thus contributing to impairment in sexual behavior, gamete function, and interaction and delivery of the live young. One such family of environmental chemicals that has been recognized as reproductive toxicants and endocrine disruptors is polycyclic aromatic hydrocarbons (PAH; reviewed in the study of Ramesh and Archibong 5 ). Automobile exhausts, tobacco smoke, refuse burning, industrial emissions, hazardous waste sites, and charcoal-broiled meat contribute considerable amounts of these chemicals to human exposure. 6 Studies conducted in our laboratory 7–9 and those of others 10–12 revealed that in the ovaries of exposed animals, BaP becomes activated to electrophilic metabolites that bind covalently to nucleophilic sites of DNA, form BaP-DNA adducts, 13 and may interfere with ovarian function by compromising function and viability of oocyte–granulosa complexes in follicles. 9,14
Even though an association between higher levels of PAH exposures and increased incidence of female reproductive toxicity in humans has been established, 15,16 information on the metabolism of BaP in human reproductive tissues is lacking. Therefore, the objective of this study was to assess the qualitative and quantitative patterns in ovarian subcellular metabolism of BaP, a prototypical PAH compound.
Material and methods
Sample collection
Human ovary samples (eight individuals) were obtained from postoperative tissue removed from subjects with uterine tumors, whose ovaries were ablated to suppress the progression of hormonally accelerated carcinogenic process. 17 Soon after retrieval, the samples were frozen and stored at −70°C. The investigators were not privy to subjects’ identity, race, and any other personal or medical information and the study conducted with the obtained ovarian tissues was approved by the Institutional Review Board at Meharry Medical College.
Isolation of subcellular fractions and incubation with BaP
On the day of subcellular fraction isolation, samples were removed from the freezer, thawed, and rinsed twice with ice-cold phosphate buffered saline. Subsequently, the tissues were finely diced in a small Petri dish with a surgical blade at 4°C. Cold tissue extraction lysis buffer (G-Biosciences, Maryland Heights, Missouri, USA) of 2–5 mL was added to the diced tissue and gently agitated prior to being transferred to a small Potter–Elvehjem homogenizer. Homogenization was conducted at 4°C and the homogenate was maintained at the homogenization temperature for 2 min.
Subcellular fractions (microsomes, mitochondria, cytosol, and nuclear) were isolated from the ovarian tissue samples at 4°C using standard kits. The microsomal fractions were isolated using endoplasmic reticulum isolation kit (Sigma, Catalog # ER0100) and mitochondria with FOCUS mitochondria kit (G-Biosciences, Catalog # 786-022). The cytosolic and nuclear fractions come from the intermediate centrifugation steps while isolating the microsomal and mitochondrial fractions. After isolation, the fresh subcellular fractions were subjected to incubation with BaP (CAS no. 50-32-8; 98% pure, Sigma (Sigma-Aldrich, St. Louis, MO); 1 μM and 3 μM) dissolved in dimethyl sulfoxide (DMSO). Approximately 100 μL of fractions (final protein concentration 0.5 mg/mL) were suspended in TKM buffer (sucrose 0.25 M, Tris 80 mM, potassium chloride 25 mM, magnesium chloride 5 mM, pH 7.4) and 5 mL of cocktail containing nicotinamide adenine dinucleotide phosphate (NADPH; 0.72 mM), ethylenediaminetetraacetic acid (EDTA; 0.1 mM), potassium phosphate (KPO4; 100 mM), and MgCl2·6H2O (3.75 mM). The samples were preincubated for 5 min at 37°C in a tissue shaker (Precision Scientific Instruments, Chicago, Illinois, USA). After a 15-min incubation at 37°C, the reaction was stopped with 8 mL of ethyl acetate. Benzo(a)pyrene (BaP) metabolites were extracted twice with ethyl acetate/methanol for analysis by reverse-phase high-performance liquid chromatography (HPLC) for BaP metabolites.
The purities of the subcellular fractions were assessed by the measurements of marker enzymes. Cytochrome C oxidase was measured as a marker for mitochondria. The activity of cytochrome C oxidase was assayed using reduced cytochrome C as the substrate, 18 0.5 M sodium phosphate buffer (pH 7.5), and 0.05 mg protein at 30°C. The decline in absorbance at 550 nm due to the oxidation of cytochrome C was used to calculate the specific activity. Peptidase/protease was measured as a marker for the microsomes using the Enzcheck ® peptidase/protease kit (Life Technologies Corporation, Carlsbad, California, USA). Lactate dehydrogenase was used as a marker for cytosol. This enzyme was assayed using the Lactate Dehydrogenase Kit (BioAssay Systems, Hayward, CA) according to the manufacturer’s specifications. Protein content of the subcellular preparations was determined according to the method of Bradford. 19
BaP metabolism studies
Before conducting metabolism studies, the subcellular fractions were thawed at room temperature and 120 µL of the microsomal pellet resuspended in TKM buffer (final protein concentration 0.5 mg/mL) were added to 5 mL of cocktail containing NADPH (0.72 mM), EDTA (100 mM), KPO4 (100 mM), and MgCl2·6H2O (3.75 mM). Each human ovarian sample was preincubated for 2 min at 37°C in a tissue shaker (Precision Scientific Instruments, Chicago, Illinois, USA). Treatment consisted of exposure in vitro to 1 μM or 3 μM BaP (CAS No. 50-32-8; 98% pure, Sigma) dissolved in DMSO. After a 15-min incubation at 37°C, the reaction was stopped with 8 mL of ethyl acetate containing butylated hydroxytoluene (0.2 mg/mL). BaP metabolites were extracted twice with ethyl acetate and the extracts were analyzed by reverse phase HPLC for BaP metabolites as described previously. 13 Metabolite standards were obtained from the National Cancer Institute Chemical Carcinogen Repository (Midwest Research Institute, Kansas City, Missouri, USA).
Statistical analysis of data
Each subcellular human ovarian tissue fractions were spread into two experimental groups and a control group. Treatment consisted of incubation of subcellular fractions in 1 µM and 3 µM BaP, while subcellular fractions in the control group were incubated in BaP vehicle (DMSO) in a 3 × 8 factorial arrangement. BaP metabolite concentrations among subcellular fractions were compared using analysis of variance with repeated measures and differences among means were tested with the Tukey–Kramer’s test.
Results
The concentrations of BaP metabolites generated by the ovarian subcellular fractions are depicted in Figure 1. The fractions also showed a BaP concentration (dose)–response relationship. The subcellular fractions incubated with 3 μM BaP generated greater concentrations of BaP metabolites than the 1 μM BaP dose and the differences were statistically significant (p < 0.05). Among the different subcellular fractions, the microsomal fraction produced greater concentrations of metabolites than the mitochondrial, nuclear, and cytoplasmic fractions and the differences were statistically significant (p < 0.05).

Comparison of the metabolism of BaP in ovarian subcellular fractions treated with 1 µM and 3 µM BaP. Values are expressed as mean concentration of total metabolites ± SE (n = 8). Data from triplicate determination (variability was <10%) of five individual samples were compared. Asterisks denote statistical significance (p < 0.05) in BaP metabolite concentrations among the various fractions and BaP concentrations used. BaP: benzo(a)pyrene.
The relative proportions of individual BaP metabolites among total metabolites are depicted in Figure 2. The different BaP metabolites resulting from the incubation of subcellular fractions were BaP 4,5-diol, BaP 7,8-diol, BaP 9,10-diol, BaP 3,6-dione, BaP 6,12-dione, 3-hydroxy, and 9-hydroxy BaP. There were some remarkable differences among subcellular fractions in the type of BaP metabolites formed. The microsomal and mitochondrial fractions produced considerably higher proportion (p < 0.05; treatment × fraction interaction) of BaP 4,5-diol and 7,8-diol than the other fractions in this study.
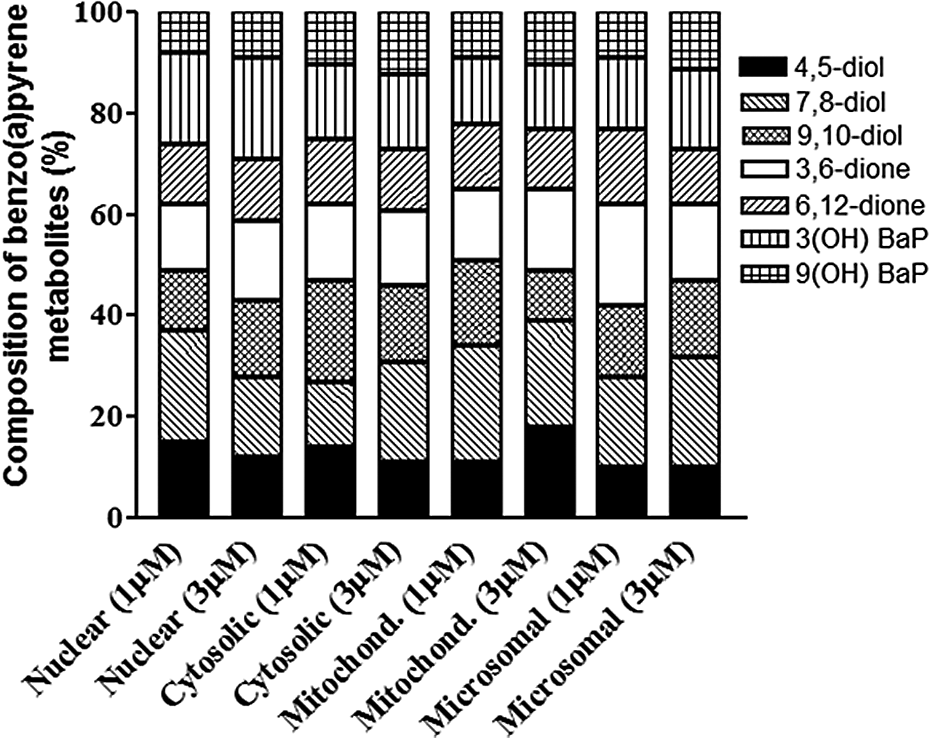
Metabolite profiles of BaP in ovarian subcellular fractions treated with 1 µM and 3 µM BaP. Values are expressed as the percentage of individual metabolite types among the total metabolites ((sum of individual concentrations of BaP 9,10-diol, BaP 4,5-diol, BaP 7,8-diol, 3,6-dione, 6,12-dione, 3-hydroxy, and 9-hydroxy BaP) formed; n = 8). BaP: benzo(a)pyrene.
Discussion
The BaP concentrations used in this study were relevant to dietary, environmental, occupational, and smoking exposures. Dietary exposures of humans to BaP range from 8.4 µg to 17 µg/person/day. 20 An additional intake of 0.1 µg/day is expected for individuals who smoke a pack of cigarettes per day, as mainstream smoke yield of BaP per cigarette amounts to 10 ng/cigarette 21 and 215–375 ng/m3 of PAHs through secondhand cigarette smoke in pubs and tavern patrons. 22 Exposures from other occupational hazards (industrial workers—1.4–25 ng/m3, 23 and restaurant cooks—6.9 ng/m3 24 ) contribute to additional BaP exposure. Furthermore, people living in the vicinity of hazardous waste sites (soil PAH levels ranging from 600 µg/kg to 10,000 µg/kg20) and in unvented homes that use biomass for cooking and home heating (15–162 µg/m2 25 ) are at an increased risk of getting exposed to additional amounts of BaP. Because of the increasing environmental contamination by BaP, in laboratory studies allowance has to be made for scenarios where exposure levels that approximate or higher than those mentioned above. Taken together, the BaP concentrations used in this study could be achieved in human body under the above-mentioned exposure situations.
BaP is metabolized by CYP1A1, 1A2, 1B1, and epoxide hydrolase (EH) in human tissues. 26–28 Even though constitutive expression of CYP1B1 was reported to be very low in normal human tissues, 28 Muskhelishvili et al. 29 demonstrated CYP1B1 messenger RNA (mRNA) and protein expression in human ovary using in situ hybridization and immunohistochemistry, respectively. The levels of estrogen resident in steroidogenic tissues such as uterus and ovary may have a bearing on the functioning of cytochromes P450 (CYP) enzymes. 30 CYP1A1 activity has been reported in endometrium. 28 The drug-metabolizing enzymes (DME) in steroid-rich tissues such as ovaries of these subjects may have been induced to a greater extent as increased expression of CYPs in tumors and adjacent tissues has been reported. 31 Therefore, the biotransformation of BaP by subcellular fractions could have been the result of either CYP1A1 or 1B1 or both. Due to the issues arising from integrity of mRNA obtained from these limited number of samples, molecular studies could not be conducted to examine the protein and mRNA expression. The debate regarding the specific DME responsible for BaP metabolism in human ovary notwithstanding, there is anecdotal evidence to indicate that both these isozymes (CYP1A1 and 1B1) are involved in the metabolic activation of BaP to DNA reactive species. 32
Our studies have demonstrated the ability of human ovary to metabolically process BaP. Therefore, questions arise as to the functional significance of the identified metabolites. Matikainen et al. 15 grafted human ovarian cortical biopsies into non-obese diabetic severe combined immunodeficiency (NOD-SCID) mice. When these mice were exposed to PAHs, the oocytes were found to undergo apoptosis mediated by the Bax gene. In an attempt to unravel whether PAH parent compound or its biologically active intermediates contribute to oocyte loss, neonatal mice ovaries were cultured ex vivo in the presence of 7,12-dimethylbenz(a)anthracene (DMBA) and its metabolite DMBA-3,4-dihydrodiol. The metabolite appeared to be more potent than the parent compound in initiating germ cell destruction. These findings highlight the consequences of PAH exposure and the resultant metabolite accumulation contributing to premature ovarian failure. Additionally, these findings were supported by the studies of Kee et al., 33 who also reported that the DMBA metabolite affects the development of human primordial germ cells. As metabolites have been implicated in causation of apoptosis 34 leading to ovarian follicular atresia, 35,36 information on the composition of metabolites that are generated from BaP in the presence of subcellular fractions is relevant in the context of toxicity.
Initial oxidation of BaP catalyzed by CYP450 family of enzymes (CYP1A1, CYP1A2, and CYP1B1) yield arene oxides (9-OH-BaP, 7-OH-BaP, 6-OH-BaP, 3-OH-BaP, and 1-OH-BaP). These arene oxides rearrange to phenols or undergo hydration catalyzed by EH generating BaP-9,10-diol, BaP-7,8-diol, and BaP-4,5-diol. 37,38
The generation of 3,6- and 6,12-dione metabolites is of importance as they are capable of inducing cellular damage through the production of reactive oxygen species and oxidatively damaging bases of DNA. 39 Among the diol metabolites, the subcellular fractions preferentially formed BaP-7,8-dihydrodiol, a precursor of the DNA reactive BaP-7,8-dihydrodiol epoxide (BPDE). Our findings are indicative of marked differences in BaP metabolite profiles among the subjects and therefore may contribute to differential susceptibilities of individuals exposed to BaP. Additionally, the preponderance of BaP-7,8-dihydrodiol in these samples is interesting, given the fact that this precursor of BPDE has been linked to BaP-induced cancer 38 and infertility. 13
Taken together, our results show that exposure of humans to PAHs and the likely consequences of such exposure to the reproductive health of women deserve attention. As part of the National Health and Nutrition Examination Survey, urine samples were collected from 2400 nonoccupationally exposed individuals and PAH levels were measured. 40 These studies revealed most of the PAH compounds and their monohydroxy metabolites in urine of sampled individuals, which indicates the inevitable exposure of general population to PAHs. Additionally, consumption of red meat and diet rich in saturated fat, 20,41,42 occupational exposure, 43 and cigarette smoke 22,44 may elevate the levels of PAHs in the human body and consequently trigger disease. Also of relevance in this regard are gender-related differences in DME gene expression in humans. This places females at an increased risk of susceptibility to PAH-induced toxicity 45 and cancer 46 compared to males.
As mentioned above, the samples were originated from women suffering from uterine cancer. Clinicopathological details such as prior exposure of the subjects to chemicals at workplace, 43 their medication, 47 nutritional status (dietary factors), 45 or smoking habits 22 are not known. All these factors are capable of inducing CYP enzymes in addition to gonadal cancer-inducing steroids. Additionally, we are beset by the lack of knowledge of the phases of menstrual cycle of the subjects at which the ovarian tissues were retrieved. If the ovaries were retrieved at different phases of the menstrual cycle due to exigency of medical condition, it is conceivable that variations in the ovarian steroid levels may also have contributed to the variations in metabolism of BaP by subcellular fractions inasmuch as metabolism of PAHs in mammalian ovary is hormonally regulated. 48 Nonetheless, our findings raise awareness for the possible role of BaP in ovarian failure, a toxicant to which women are exposed to in the environment, cigarette smoke, and at occupational settings.
Conclusion
Our data indicate that human body has the capability of metabolizing BaP, the products of which if accumulated in target organ tissues such as ovary and uterus can cause reproductive toxicity and cancer. Additionally, our findings advocate the need for adopting preventive measures to curb PAH including BaP exposure to women at occupational settings.
Footnotes
Funding
This research was supported by the National Institutes of Health (NIH) grants G12 RR03032, 5R01CA142845-04, and 1RO3CA130112-01.
Conflict of interest
The authors declared no conflicts of interest.
