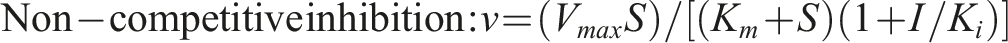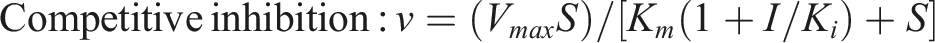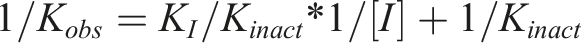Abstract
Byakangelicol, a furanocoumarin derived from Angelica dahurica, possesses anti-inflammatory and antitumor activities. Since cytochrome P450 2A6 (CYP2A6) is the major enzyme responsible for coumarin metabolism, it is important to evaluate the effect of byakangelicol on CYP enzyme activity. The purpose was to explore the effects of byakangelicol on CYPs and to provide a reference for its drug development and clinical application. The present study investigated the impact of byakangelicol on CYPs in human liver microsomes (HLMs). Byakangelicol demonstrated the capacity to suppress the activities of CYP1A2, 2A6, and 3A4 in HLMs, with IC50 values of 19.42, 10.11, and 12.80 μM, respectively. Byakangelicol exhibited competitive inhibition of CYP1A2 and 2A6 with K i values of 9.86 μM and 5.23 μM, whereas the inhibitory effect on CYP3A4 was noncompetitive with a K i value of 6.55 μM. In addition, the inhibitory effect of byakangelicol on CYP3A4 was found to be time-dependent (K inact = 0.041 min−1 and K I = 6.67 μM). This study revealed the inhibitory properties of byakangelicol on CYP1A2, 2A6, and 3A4 activity in HLMs. It suggests the potential for drug–drug interactions when byakangelicol is co-administered with drugs metabolized by these CYPs. These findings offer a foundation for investigating the interaction of byakangelicol with other drugs, which may assist in clinical prescription and drug development.
Introduction
Cytochrome P450 enzymes (CYPs) are a class of membrane-bound enzymes with a structural similarity to hemoglobin. They are primarily located in the human liver and execute a pivotal function in the biotransformation of exogenous substances and drug metabolism.1,2 CYPs are involved in the metabolism of more than 70% of clinical drugs, and induction or inhibition of CYPs is one of the mechanisms leading to drug–drug interactions.3,4 A study suggests a possible drug–drug interaction between cannabidiol and citalopram, which may be related to the former’s inhibition of CYP3A4 activity. 5 A number of herbal components were reported to suppress the activity of CYPs, including picroside II, cepharanthine, and tubeimoside I.6-8 Additionally, several studies have evaluated the potential for herb–drug interactions resulting from alterations in the activity of CYPs. 9 For example, shikonin has been reported to affect the pharmacokinetics of lapatinib in rats, leading to increased systemic exposure. 10 The investigation of the effects of the primary active ingredients in herbal medicines on CYPs can offer more precise guidance for clinical applications.
Herbs constitute a significant component of traditional Chinese medicine, with a history spanning millennia. Herbal medicines generally contain a variety of active ingredients, which enables their use in the treatment of various diseases.11,12 Byakangelicol is a prevalent component in plants belonging to the Apiaceae family, classified as a coumarin chemical compound, and it is present in high concentrations in the traditional Chinese medicine Angelica dahurica. Research demonstrates that the ethanol-soluble components of Angelica dahurica effectively suppress tumor progression and metastatic spread in murine melanoma models. 13 According to modern pharmacological studies, byakangelicol exhibits a variety of pharmacological activities, especially in anti-tumor, anti-inflammatory and analgesic, and inhibition of pathogenic microorganisms,14-16 which has attracted extensive attention from domestic and foreign researchers. For instance, network pharmacology analysis elucidates the multi-target mechanisms underlying the therapeutic potential of byakangelicol for osteosarcoma, suggesting its promise as a novel treatment candidate. 17 In addition, byakangelicol was found to suppress the proliferation of human tumor cells in a dose-dependent manner. 18 Lin et al reported that byakangelicol extracted from Angelica dahurica suppressed IL-1β-induced COX-2 expression in human lung epithelial cells. The underlying mechanism of this suppression may involve the inhibition of NF-κB activity. 19 Additionally, it was revealed that byakangelicol modulates NF-κB signaling pathway and the polarization status of macrophages in periprosthetic osteolysis, thereby effectively inhibiting the expression of pro-inflammatory factors. 20 CYP2A6, a key enzyme within the CYP family, mediates the metabolic processing of coumarins. Assessing the influence of byakangelicol on CYP enzyme activity is crucial, as it may offer important implications for the development and safe use of medications containing this furanocoumarin.
This study initially identified byakangelicol as an inhibitor of CYP1A2, 2A6, and 3A4 through preliminary screening. Further investigation was conducted to characterize its inhibitory kinetics against these major CYPs in vitro, thereby offering a theoretical foundation for the co-administration of byakangelicol with other drugs.
Materials and Methods
Effect of Byakangelicol on the Activity of CYPs
The CYP enzyme inhibition screening assay was performed in accordance with the U.S. Food and Drug Administration (FDA) Drug-Drug Interaction (DDI) Guidance for Industry (https://www.fda.gov/media/108130/), incorporating analytical methodologies from well-established studies.21-23 The present investigation sought to assess the inhibitory effects of byakangelicol on key CYP isoforms, namely, CYP1A2, 2A6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4. Human liver microsomes (HLMs) were provided by MedChemExpress (Monmouth Junction, NJ, USA). Specific substrates and corresponding probe reactions were referenced from previous studies22,23 (Table S1). The incorporation of representative inhibitors or byakangelicol (≥99%, CAS# 26091-79-2, MedChemExpress) (Figure 1) was employed to evaluate the activity of CYPs. The groups were as follows: negative control (without inhibitor and byakangelicol), positive control, and byakangelicol. The incubation system consisted of HLMs, a corresponding probe substrate, specific inhibitor or byakangelicol, 100 mM potassium phosphate buffer (pH 7.4), and NADPH generation system (1 mM NADP+, 1 U/mL glucose-6-phosphate dehydrogenase, 4 mM MgCl2, and 10 mM glucose-6-phosphate) in a volume of 200 μL. Before adding the NADPH generation system to start the reaction, the system was pre-incubated for 3°minutes at 37°C. The reaction was terminated by the addition of acetonitrile (10% trichloroacetic acid for CYP2A6). The supernatant was obtained by centrifugation at 12,000 r/min for 10 minutes and assayed on the Ultimate 3000 HPLC system (Thermo Fisher Scientific, Waltham, MA, USA). The conditions for HPLC analysis are listed in Table S2. All experiments were performed in triplicate (n = 3) across three independent experimental runs. Chemical structure of byakangelicol.
Inhibition Kinetic Studies
CYPs were co-incubated with 0, 2.5, 5, 10, 25, 50, and 100 μM of byakangelicol to evaluate the half-maximal inhibitory concentration (IC50). Specific probe substrates (20–100 μM of phenacetin for CYP1A2, 0.25–2 μM of coumarin for CYP2A6, and 20–100 μM of testosterone for CYP3A4) were utilized to estimate whether the inhibitory effect of byakangelicol on these CYPs was related to concentration. All experiments were performed in triplicate (n = 3) across three independent experimental runs. The fitted models are as follows:
Study of Time-Dependent Inhibition (TDI)
The time-dependent inhibitory effect of byakangelicol on CYPs was investigated and the incubation system was as described above. HLMs and byakangelicol (20 μM) were pre-incubated at 37°C for 30 minutes, after which an aliquot (20 μL) was transferred to another tube containing the probe substrate (approximately K
m
). The reaction was initiated by the NADPH generation system. Four incubation times were examined: 0, 5, 10, 15, and 30 minutes. The reaction was then terminated with the addition of acetonitrile, and the mixture was analyzed by using HPLC assay. The estimation of K
I
and K
inact
values was conducted using byakangelicol (0, 2, 5, 10, 20, and 50 μM) and substrate (approximately 4-fold K
m
) with incubation times of 0, 5, 10, 15, and 30 minutes. All experiments were performed in triplicate (n = 3) across three independent experimental runs. The formula is as follows:
K obs denotes the pseudo-first-order rate constant of inactivation at inactivated concentration [I], K inact denotes the maximum inactivation rate (theoretical value), and K I denotes the concentration of the compound at half of K inact .
Statistical Analysis
Experiments were performed in triplicate, and data were analyzed using SPSS 22.0. Lineweaver–Burk plots were generated with GraphPad Prism 8.0. One-way ANOVA was employed for the comparison of differences. P < 0.05 represents statistical differences.
Results
Byakangelicol Suppressed the Activity of CYP1A2, 2A6, and 3A4
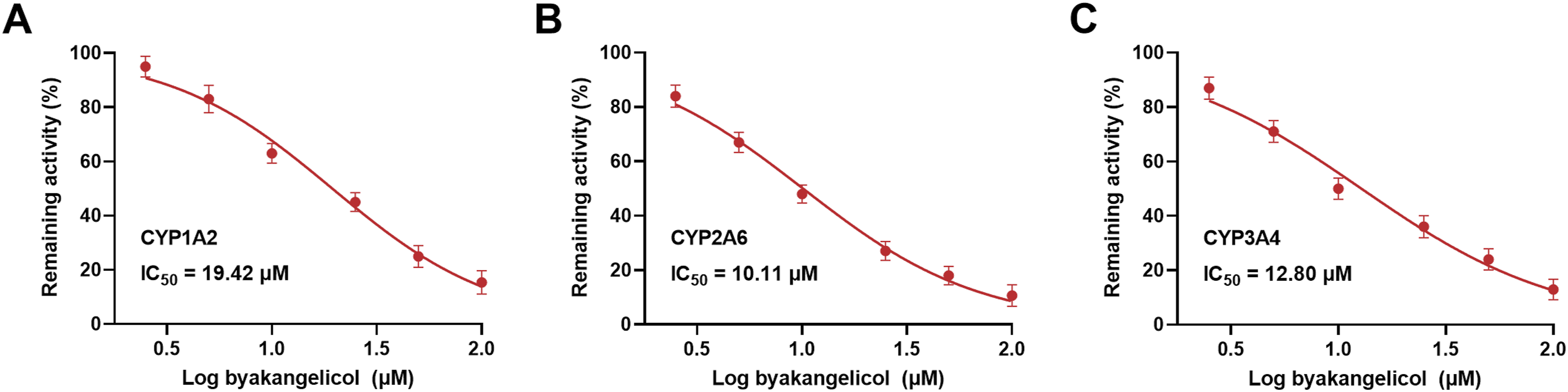
As demonstrated, CYP activities were significantly inhibited by specific positive inhibitors. Significant inhibition of CYP1A2, 2A6, and 3A4 was detected following the treatment of byakangelicol (Figure 2). The inhibitory effects of byakangelicol on CYP1A2, 2A6, and 3A4 were found to be concentration-dependent. The IC50 values for the suppression of CYP1A2 (Figure 3(A)), 2A6 (Figure 3(B)), and 3A4 (Figure 3(C)) by byakangelicol were determined to be 19.42, 10.11, and 12.80 μM, respectively. Effect of byakangelicol on common CYPs. Positive control: specific concentration of inhibitor, byakangelicol: byakangelicol (100 μM), negative control: without inhibitor. ***P < 0.001. The concentration-dependent inhibition of CYP1A2 (A), 2A6 (B), and 3A4 (C) by byakangelicol was observed with IC50 values of 19.42, 10.11, and 12.80 μM, respectively.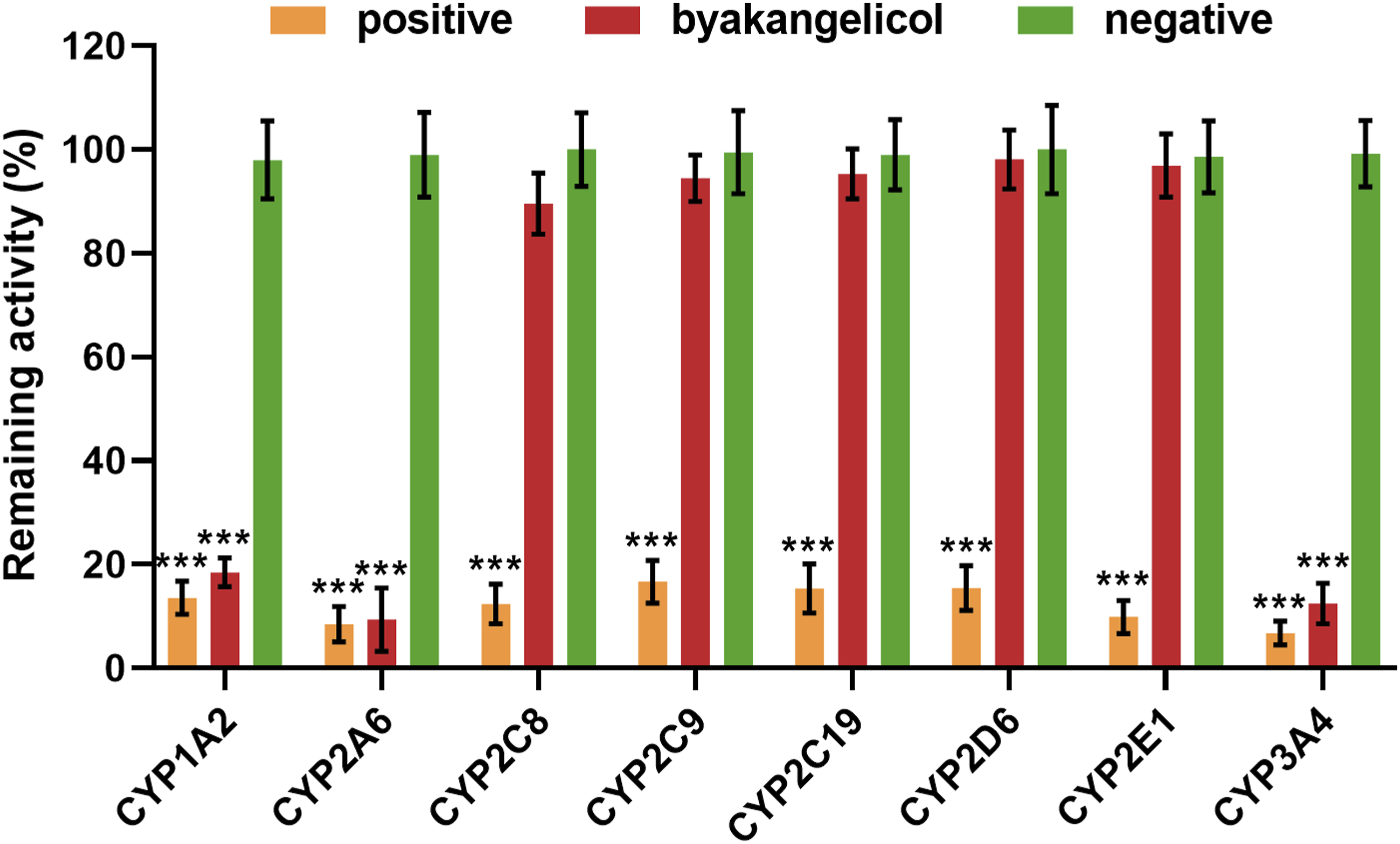
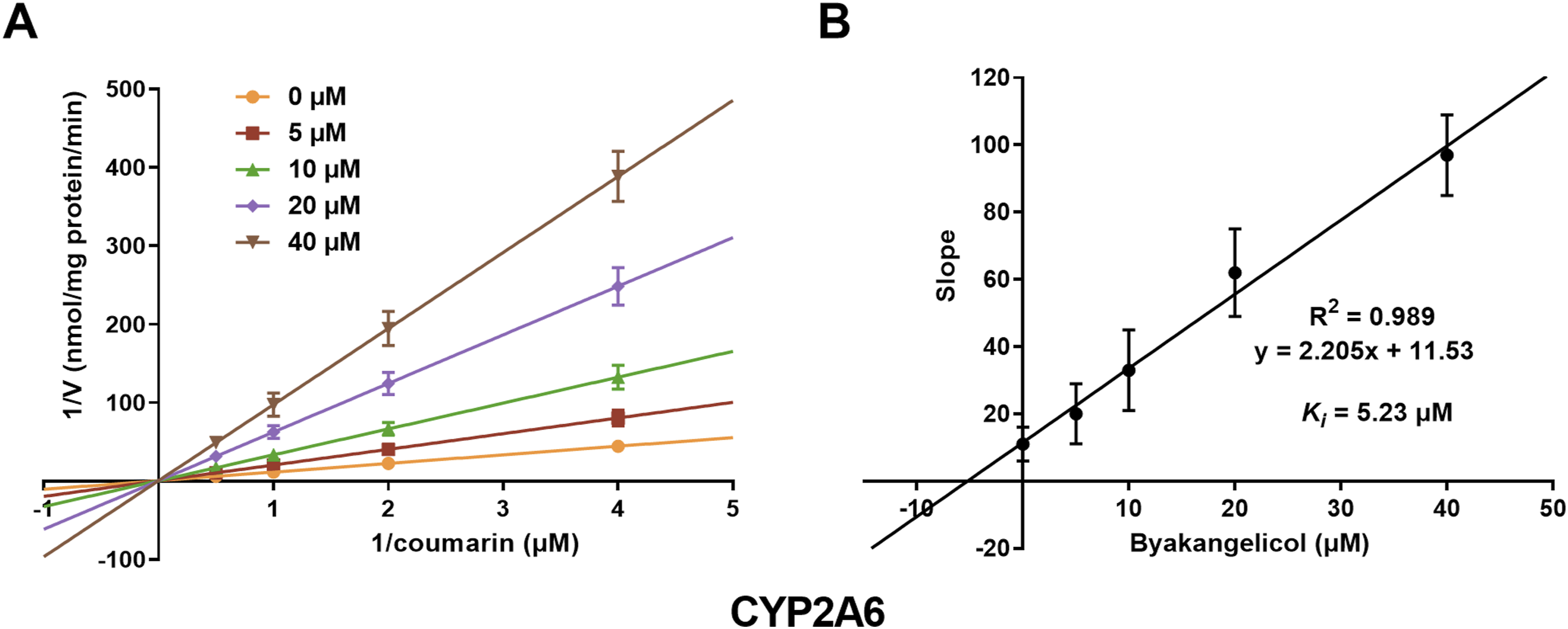
Inhibitory Properties of Byakangelicol on CYP1A2 and 2A6
A series of concentrations of byakangelicol were utilized to evaluate its inhibitory effects on CYP1A2 and 2A6. Lineweaver–Burk plots demonstrated that the inhibition of CYP1A2 (Figure 4(A)) and 2A6 (Figure 5(A)) by byakangelicol was most accurately described by a competitive model with a constant Vmax. Subsequent analysis indicated that byakangelicol exhibited inhibitory activity on CYP1A2 (Figure 4(B)) and 2A6 (Figure 5(B)), with K
i
values of 9.86 and 5.23 μM, respectively. Characteristics of the inhibition of CYP1A2 by byakangelicol. (A) The inhibition of CYP1A2 by byakangelicol was consistent with a competitive model. (B) The secondary plot yields a K
i
value of 9.86 μM. Characteristics of the inhibition of CYP2A6 by byakangelicol. (A) The inhibition of CYP2A6 by byakangelicol was consistent with a competitive model. (B) The secondary plot yields a K
i
value of 5.23 μM.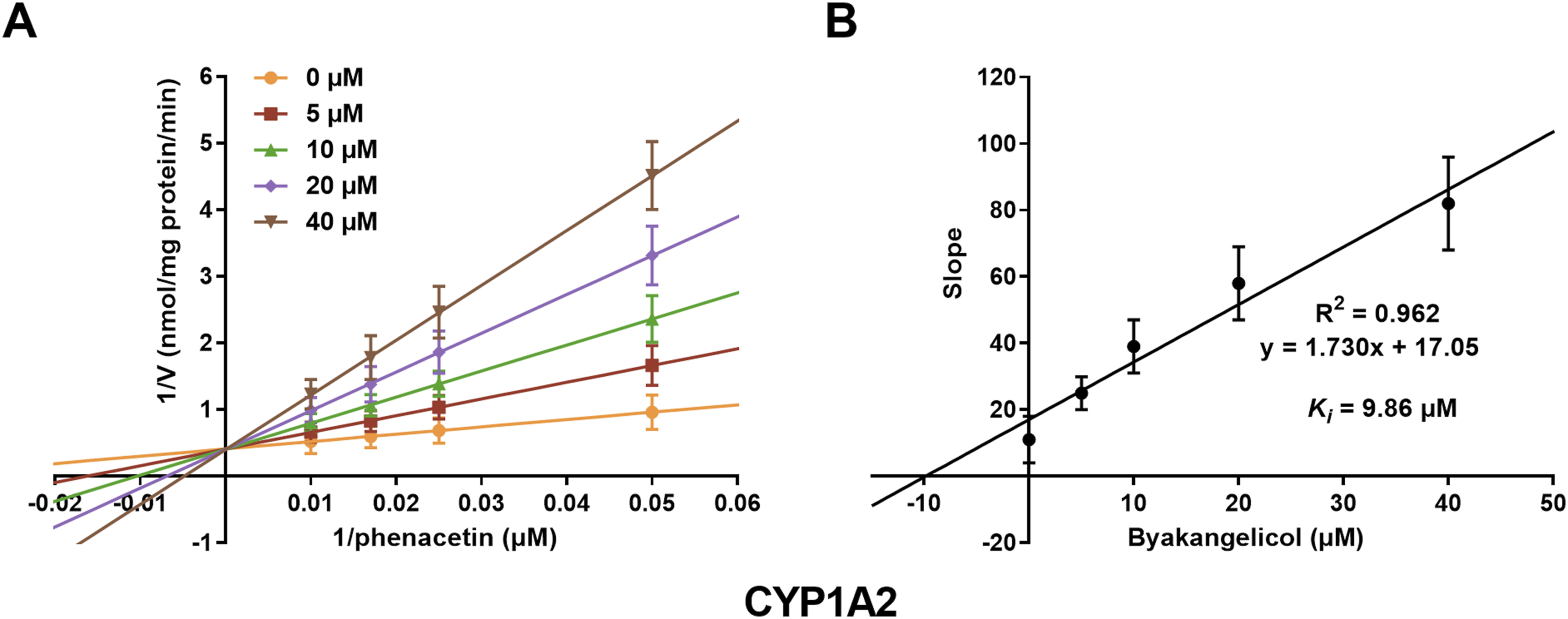
Suppression of CYP3A4 by Byakangelicol
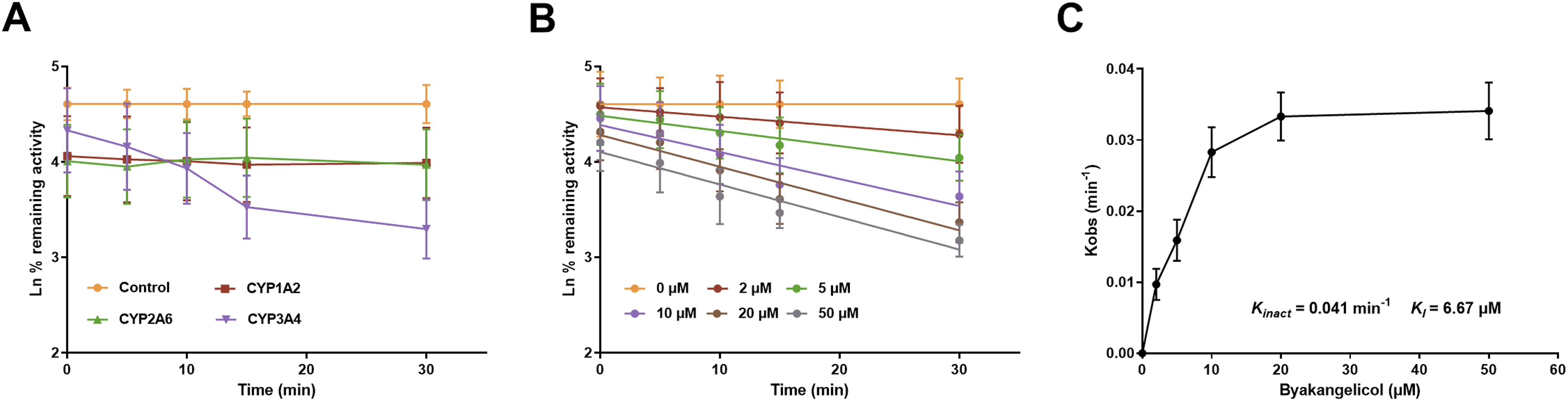
The suppression of CYP3A4 by byakangelicol best fitted to the non-competitive inhibition model, with a K
i
value of 6.55 μM (Figure 6), and the Vmax declined with increasing byakangelicol concentration. Additionally, the association between pre-incubation time and the inhibitory effect of byakangelicol was investigated. The results demonstrated a decline in CYP3A4 activity with prolonged duration (Figure 7(A)). The inhibition of byakangelicol on CYP3A4 over time was identified by incubating CYP3A4 with different concentrations of byakangelicol for 0–30 minutes (Figure 7(B)). Subsequently, the values of K
inact
and K
I
were calculated as 0.041 min−1 and 6.67 μM, respectively (Figure 7(C)). Characteristics of the inhibition of CYP3A4 by byakangelicol. (A) The inhibition of CYP3A4 by byakangelicol was consistent with a noncompetitive model. (B) The secondary plot yields a K
i
value of 6.55 μM. Effect of time on the inhibition of CYPs by byakangelicol. (A) The suppressive effect of byakangelicol on CYP3A4 was time-dependent. (B) The inhibition of CYP3A4 by different concentrations of byakangelicol was time-dependent. (C) The K
inact
and K
I
values were determined to be 0.041 min−1 and 6.67 μM, respectively.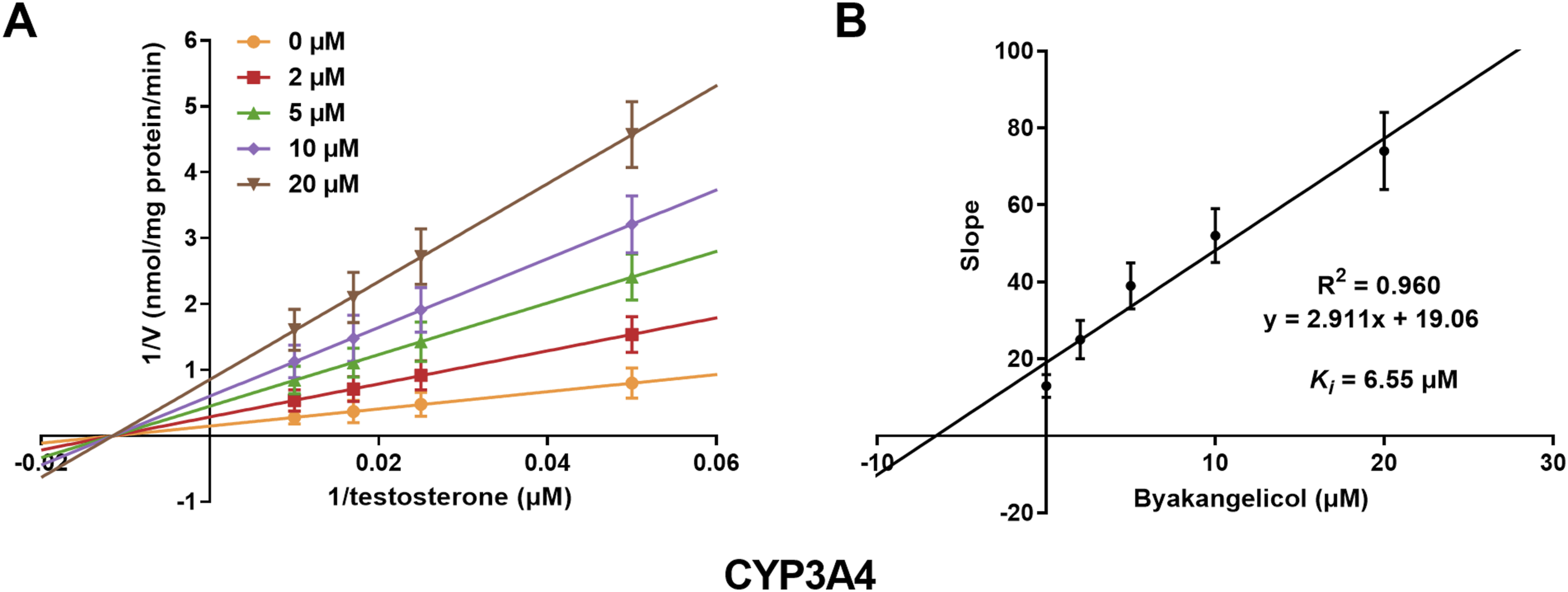
Discussion
In recent years, the combination of Chinese and Western medicine has become a common strategy for treating diseases, especially chronic diseases such as hypertension and diabetes mellitus. These diseases are often treated with Western medicine in conjunction with proprietary Chinese medicines.24,25 The inhibition of hepatic CYPs by traditional Chinese medicines or their components directly affects the pharmacokinetics of the substrates of the enzymes, thereby influencing the therapeutic efficacy of the drugs.26-28 The in vitro metabolism study of HLMs has been demonstrated to be a valuable tool in investigating the effects of traditional Chinese medicine or its components on related metabolizing enzymes. This approach facilitates the elucidation of the potential mechanisms underlying drug-induced adverse reactions.29,30 In the present study, byakangelicol was revealed to exhibit relatively strong inhibition of CYP1A2, 2A6, and 3A4 in HLMs, with no significant inhibition of other CYP isoforms.
In the human liver, CYP1A2 constitutes approximately 13% of the total CYPs. 31 CYP1A2 mRNA expression has been documented to demonstrate variability of up to 40-fold among individuals. The enzymatic activity of CYP1A2 and the drug metabolism mediated by the enzyme also exhibit considerable individual variability and can be influenced by factors such as drugs, smoking, and genes.32,33 CYP1A2 exerts a pivotal function in the metabolic processing of drugs including clozapine, caffeine, lidocaine, and theophylline. 34 Research has demonstrated that the active constituents of several herbs, including echinacoside, total saikosaponins, and suberosin, exhibit the capacity to impede the activity of CYP1A2.23,35,36 CYP2A6, an isoform of the CYP2A subfamily, is predominantly expressed in the human liver and is responsible for the transformation of procarcinogens, including nicotine, coumarin, and nitrosamines. Moreover, CYP2A6 is involved in the metabolism of some clinical drugs (eg, clomethiazole, fadrozole, and valproic acid).37,38 The research by Lim et al demonstrated that cathine suppresses CYP2A6 activity and may affect the pharmacokinetics of drugs metabolized by CYP2A6. 39 Here, the suppressive effect of byakangelicol on CYP1A2 and CYP2A6 was enhanced with increasing concentration. Moreover, the competitive inhibition of CYP1A2 and 2A6 activity by byakangelicol was analyzed, and this inhibition may be related to the structure of individual CYP isoforms. In particular, byakangelicol is a coumarin-like compound that exhibits a chemical structure analogous to that of coumarin, which is a substrate of CYP2A6. Future experiments could be designed to further identify the key sites of the action of byakangelicol with CYP1A2 and 2A6 by using in silico analysis.
CYP3A4 is one of the most abundant isoforms of drug-metabolizing CYPs in HLMs and functions in the metabolism and excretion of drugs in the body. A significant number of prevalent drugs, including antibiotics, antihistamines, antifungals, and antivirals, are metabolized by CYP3A4. 40 This suggests that the active state of CYP3A4 can directly impact the rate of metabolism and the effects of these drugs. A study has indicated that the altered pharmacokinetics of clozapine in vivo may be attributable to TDI of CYP3A4 by lemborexant. 41 In addition, the suppression of CYP3A4 activity by herbal medicines has been documented. 42 For instance, Wang et al reported that polyphyllin H significantly suppressed CYP3A4 activity. 29 In rats, bergapten has been demonstrated to result in elevated serum levels of macitentan, a phenomenon that is concomitant with its inhibitory effect on CYP3A4. 43 In this study, byakangelicol suppressed CYP3A4 activity in a noncompetitive manner. Subsequent research disclosed that the inhibition of CYP3A4 by byakangelicol intensified over time. Consequently, attention needs to be paid to the dosing interval and the risk of potential adverse reactions when byakangelicol-containing proprietary Chinese medicines are co-administered with CYP3A4-metabolized drugs. Furthermore, the results demonstrate that byakangelicol exhibits TDI of CYP3A4, suggesting potential mechanism-based inactivation (MBI). As a furanocoumarin compound, byakangelicol shares structural characteristics with established MBI inhibitors that undergo metabolic activation by CYPs.44-46 This process typically involves enzyme-catalyzed generation of reactive intermediates (eg, furan ring epoxides) that form covalent adducts with the enzyme, leading to irreversible inactivation. The observed TDI provides functional evidence for the formation of such reactive metabolites. The MBI behavior indicates a risk of persistent drug–drug interactions in vivo, as recovery of enzymatic activity relies on de novo protein synthesis rather than inhibitor clearance. 47 Further mechanistic studies are needed to identify the specific reactive metabolites and elucidate the molecular basis of byakangelicol-mediated CYP inhibition.
This study provides preliminary evidence of the inhibitory potential of byakangelicol in vitro, highlighting the need for further attention and investigation. However, an accurate assessment of its actual risk profile requires measuring systemic exposure levels in vivo and comprehensively evaluating potential risks of clinical drug–drug interactions. As indicated in regulatory guidelines such as ICH M12, the translation of in vitro inhibition parameters (eg, Ki and IC50) into clinically relevant risk assessments requires a comparison with systemic exposure levels, particularly the unbound fraction. To date, no animal or human studies have reported plasma concentrations (Cmax) of byakangelicol following the administration of Angelica dahurica or its extracts, and comprehensive pharmacokinetic data are currently lacking. Interpreting the inhibitory efficacy of byakangelicol observed in vitro within a clinical context remains a significant challenge and a key limitation of this study. Coumarin and its derivatives (eg, 7-hydroxycoumarin) are primarily metabolized via glucuronidation catalyzed by uridine diphosphate glucuronosyltransferases (UGTs). As byakangelicol is a furanocoumarin compound, it may undergo similar metabolic processing. However, this study did not directly examine UGT activity or the formation of glucuronide metabolites. Future studies should examine the impact of byakangelicol on phase II enzymes such as UGTs and identify its glucuronide conjugates to better understand its clearance mechanisms. Given the potential of byakangelicol for TDI, its effects on glutathione S-transferases (GSTs) are closely associated with cellular stress responses and detoxification pathways that may be triggered by this compound. GSTs, key phase II enzymes, facilitate detoxification by conjugating reduced glutathione (GSH) to electrophilic compounds—including reactive metabolites that may arise from CYP-mediated metabolism. Further research is warranted to determine whether byakangelicol influences GST activity or intracellular GSH levels.
In summary, the findings of this study characterize the suppressive effect of byakangelicol on CYP1A2, 2A6, and 3A4 activity in HLMs, implying potential interactions between byakangelicol and drugs that are metabolized by CYP1A2, 2A6, and 3A4. These findings may provide theoretical guidance for the safe use and development of drugs.
Supplemental Material
Supplemental Material - Inhibitory Effects and Characteristics of Byakangelicol on the Activity of Multiple Cytochrome P450 Isoforms
Supplemental Material for Inhibitory Effects and Characteristics of Byakangelicol on the Activity of Multiple Cytochrome P450 Isoforms by Ting Lin, Xiao He, Pingfa Lin, and Ying Chang in International Journal of Toxicology
Footnotes
Author Contributions
All authors designed this study. T L, Y C, PF L and X H conducted the experiment and analyzed the data. T L wrote the manuscript. Y C revised the manuscript. All authors reviewed and approved for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.