Abstract
Several methods are used to evaluate the inflammatory changes. Measurement of edema is the most commonly used method for evaluating artificially induced inflammation in rat model. In this context, we present a method for the measurement of volume of rat paw, in which two glass columns containing fluid are connected by a glass tube. Paw is immersed in one column and the other column is placed on weighing balance. Upon immersion of paw, equal volume of fluid displaced applies a force F, which shows specific and proportional increment in the level of fluid in both the columns, which could then be detected by weighing balance, as ‘Force = Weight’ of displaced fluid, using the specific gravity of the fluid and the volume of paw, and thus the change in the volume of paw can be calculated. The rat paw immersed in column without touching the wall of the column facilitates the accurate measurement of volume of paw when measured several times. Present method is validated for its accuracy and reproducibility when internal radii of both the columns were same or different. Presented method is also cost-effective and hence can be used for the academic and the commercial animal research purpose, without compromising the precision of the evaluation.
Introduction
Inflammation is triggered by tissue injury from bacterial infection, immune activation, wounding and other source of damage. Various physiological and biochemical changes take place at the site of inflammation. Different methods are used to measure the extent of inflammation in clinical investigation. Measuring the volume of edema is the common method in experimental animal studies. Standardized method for volumetric measurement is a principal requirement for accurate and quantitative measurement of edema.
Several methods have been reported to measure the edema quantitatively. In oxazolone induced ear edema 1 and croton oil induced edema 2 the inflammation is measured by the difference in the weight of excised ear tissue. In inflammation induced by topical application of tetradecanoyl phorbol acetate 3 and arachidonic acid 4 in mice ear, the edema is measured by the changes in the thickness of ear pinna. Different methods are used for paw edema inflammation measurement. Subplantar injection of edemogens such as 0.1 mL of 0.2% carrageenan solution, 5 0.1 mL of 0.25% papaya latex, 6 0.1 mL of 2.5% brewer’s yeast powder suspension 7 have reportedly been used to induce paw edema. Various devices have been developed to quantitate the volume of edema. Measurement of dorsoventral diameter of rat hind paw pads, 8 thereafter improved by other researchers, 9 mercury plethysmography, 10 pressure transducer method, 11 sensitive method of interfacing a Mettler Delta Range top-loading balance with a microcomputer 12 and commercially available plethysmometer from Ugo Basile 13 are the devices used by many researchers.
Most preferred technique of measuring the volume of edema is by inserting the paw into a tube containing fluid and analyzing the rise in the level of fluid, where the rise in level of fluid can be observed visually or recorded with digitally aided sophisticated devices.
Present model has been designed on the basis of simple plethysmographic method and hydrostatic pressure of fluid. In this model, two open top glass columns are used, containing displacement fluid filled till the mark on column A. Glass columns of different internal radii (0.5 cm, 1.0 cm and 1.5 cm) are connected together by a glass tube of internal radius of 0.25 cm in all different possible combination of dimensions. Column B is placed on a digital weighing balance where weight of the displaced fluid is to be recorded in gram and later converted in volume (millilitre). In column A, the object will be immersed till the mark. The connecting glass tube should not touch the column B internally. The object (rat paw), upon immersion in the fluid of sampling column, is expelled by buoyant force and the resulting force displaces the specific volume of fluid from column A to column B. The volume of displaced fluid is specific to the volume of an object and is calculated with the formula
where V = volume (millilitre), W = weight recorded on digital balance, p = specific gravity of the fluid and
Carrageenan-induced hind paw edema model in rat has been used to evaluate the expediency of the measurement model.
Materials and methods
Glasswares were procured from J. Parekh & Company (Mumbai, India; authorized dealer for Borosil scientific laboratory glassware). Chemicals used were carrageenan (Sigma Chemicals, Mumbai, India) and acetylsalicylic acid (ASA; Ecosprin) manufactured by USV Ltd (Mumbai, India). Albino Wistar rats (weighing 150–200 g) were supplied by Haffkine biopharmaceutical (Parel, Mumbai, Maharashtra, India) and a digital balance having sensitivity of 10 mg was used. Experimental study procedure was approved by Institutional Animal Ethics Committee. Experimental procedure was carried out at Animal Testing Centre (Committee for Control Purpose and Supervision of Experiments on Animals (CPCSEA/315)) of Ramnarain Ruia College, Mumbai, Maharashtra, India.
Volume measurement method and design of model
Two open top glass columns of 5 cm height and internal radii of 0.5 cm, 1.0 cm and 1.5 cm are connected together with a glass tube of internal radius of 0.25 cm. Column A (sampling column) is fitted to an iron stand and column B (detector column) is placed on a digital balance are connected together by the connecting glass tube. Glass tube kept from contacting the wall of column B and free end of connecting tube is immersed in the fluid of column B. Column A filled with displacement fluid till it attains height of 3.0 cm and column B will have the same level when both the columns have same internal radius, otherwise, column B shows approximately same level of fluid as of column A due to the difference in hydrostatic pressure of both the columns having different internal radii. In the present method, water and mercury were the fluids used, and in the present work, volume of edema (in millilitre) was measured as
Validation of method
Accuracy of method and calibration curve
To evaluate accuracy of the method, accurately measured volume of fluid was added to column A and the values on the digital balance were recorded. Thereafter, according to the fluid density, the expected measures were calculated and compared with the observed value using formula
The method was applied to all the possible different column combinations having internal radii of 0.5 cm, 1.0 cm and 1.5 cm. The procedure was repeated thrice. Accurately measured volume of fluid was added to column A and the accuracy of the method was checked for the calibration range of 0.5–5.0 mL in six replicates to calculate correlation coefficient.
Sensitivity
Sensitivity of the method was determined by the following formula
Reproducibility
Reproducibility was evaluated using an object of unknown volume introduced in column A with the help of a mechanical driver and its volume (V) was measured, repeated six times and the relative standard deviation (%RSD) was calculated. Accuracy, reproducibility and sensitivity were checked for different combinations of columns A and B having different internal radii.
Experimental animal test
Animal procurement and maintenance
Albino Wistar rats, weighing 180–220 g were procured from Haffkine Biopharmaceuticals (Mumbai, India). All animals were housed at Animal Testing Centre (CPCSEA/315). Procedure for animal maintenance and experimentation was carried out as per guidelines of CPCSEA, Government of India. Animals were given free access to standard laboratory food and water. Animals were maintained in polypropylene cages. Standard conditions of temperature (22 ± 5°C) and humidity (55 ± 15%) were maintained with 12 h light–dark cycles. All animals were acclimatized for 1 week before experimental procedures were carried out.
Carrageenan-induced hind paw edema model
A total of 18 female albino Wistar rats were used in the carrageenan-induced rat paw edema test. These rats were randomly divided into three groups containing six rats in each group, namely, normal control group, ASA group and positive control group. Edema was induced by subplantar injection of 0.1 mL carrageenan diluted with normal saline (1% w/v) at right hind paw of rat. Normal control group was administered with 0.1 mL normal saline as sham control. Volume of edema was evaluated by inserting the paw in column A till the marking made at tibiotarsal articulation. Animals of test group were administered with ASA dissolved in normal saline, orally (p.o.; 300 mg kg−1), 1 h prior to induction; whereas control group animals were administered with normal saline p.o. Upon induction, paw volume was measured immediately (0 h) and thereafter measured at 1 h intervals for 3 h. For the experimental animal test, water has been used as a displacement fluid and the ratio r 2 B:r 2 A = 1 was used (internal radii of columns A and B = 1 cm). Volume of edema was calculated as per method mentioned in the Accuracy of method and calibration curve section.
Statistics
The results of accuracy were compared using χ 2 test. Correlation coefficient (r) was calculated for the method when different fluids and columns of different dimension were used, reproducibility was calculated as %RSD and unpaired Student’s t tests were used to confirm anti-inflammatory activity of ASA using the present method. 15
Results
Accuracy and reproducibility
To evaluate the accuracy of the method, different volumes of displacement fluid were introduced into the column A. Table 1 shows the corresponding values read on the balance. The χ 2 test was used to compare the expected weight and observed weight. There were no significant differences observed between the expected and observed values when all the different combination of dimensions (r 2 B:r 2 A = 0.25:1 or 1:1 or 1:2.5 or 1: 0.25 or 1: 2.5 or 2.5:1 or 2.5:0.25) and when both the fluids were used. The results show high accuracy for the method. Correlation coefficient (r) was calculated for the linearity range of displacement fluid of 0.5–5.0 mL and results show that r is greater than 0.99 for all the experimental circumstances and depicts that this method has good correlation for the proposed working range and dimensions.
Comparison between the observed values in g,a following the insertion of different volumes of fluid into the column A and g for different combinations of column A and column B having different internal radii.
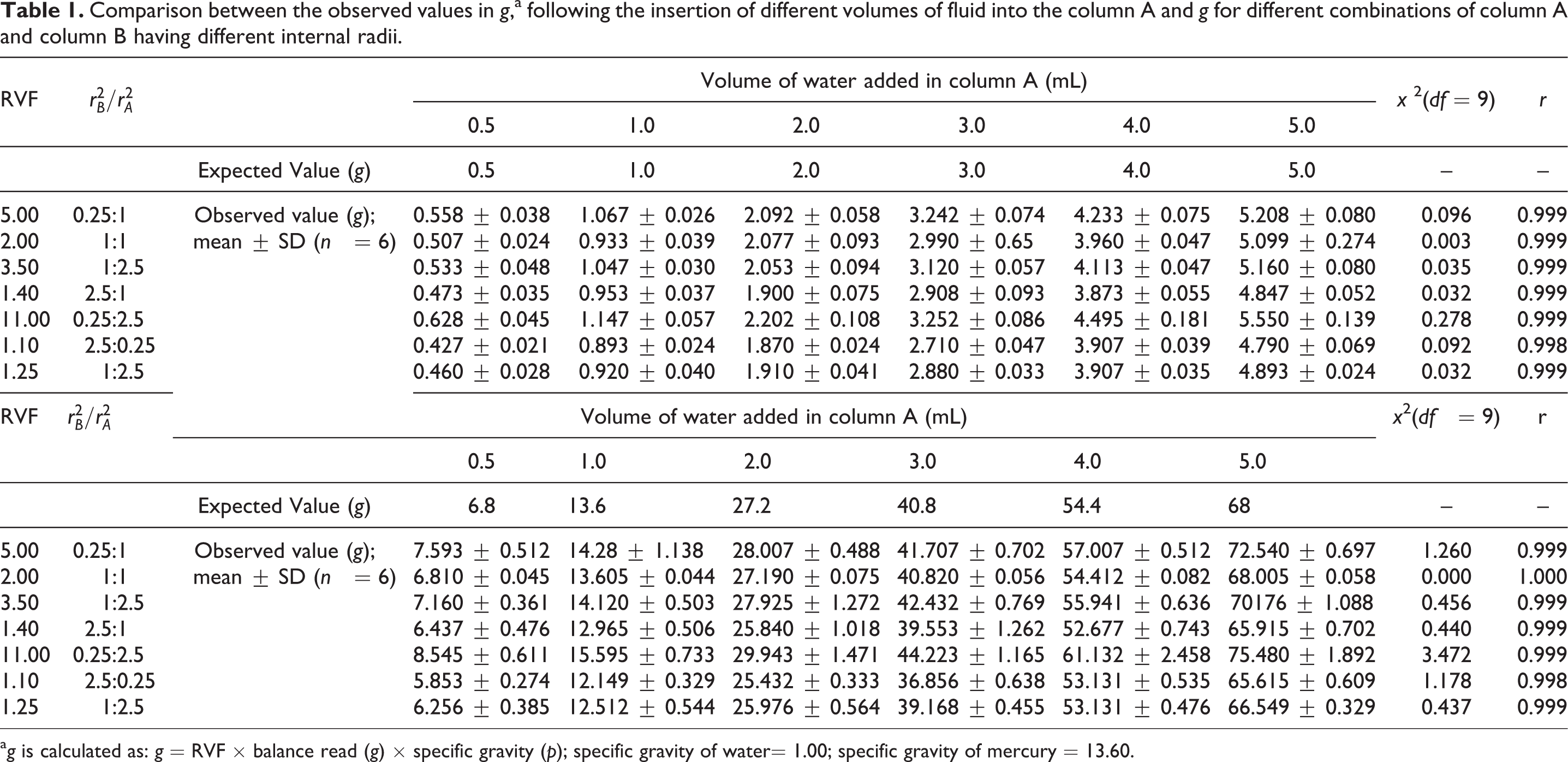
a g is calculated as: g = RVF × balance read (g) × specific gravity (p); specific gravity of water= 1.00; specific gravity of mercury = 13.60.
Table 2 shows reproducibility calculated as %RSD. Results show that the method is reproducible for all the experimental circumstances presented here.
Volumea of bizarre-shaped glass object calculatedb when different ratios of dimensions and different fluids were used.
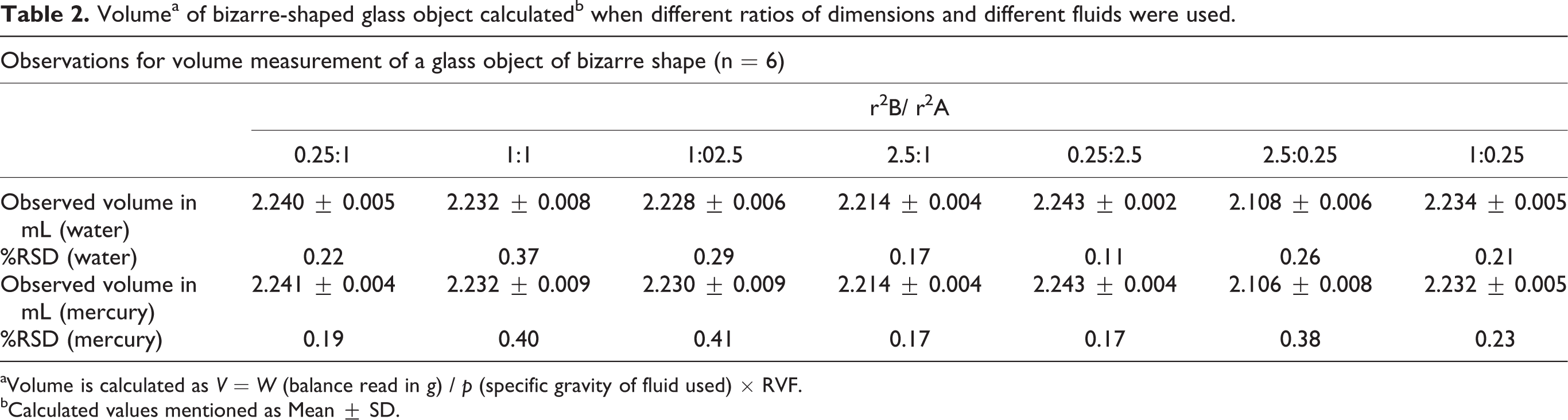
aVolume is calculated as V = W (balance read in g) / p (specific gravity of fluid used) × RVF.
bCalculated values mentioned as Mean ± SD.
Table 3 shows mean ± SEM of edema volume induced by carrageenan in the presence and absence of ASA (300 mg kg−1). Edema volume of control rat and treated rat paw were compared by unpaired Student’s t test (p ≤ 0.01). Significant difference in edema volume was observed at 2 h and 3 h and clearly confirms the anti-inflammatory activity of ASA with this model.
Effect of acetylsalicylic acid on carrageenan induced paw edema in rats.

aVolume of edema (mL): mean ± SEM. b p ≤ 0.01 evaluated by Student’s t test.

Plethysmometer coupled with digital balance.
Discussion
Present method is based on the Archimedes’ principle of buoyancy and hydrostatic pressure
14
; briefly, when an object immersed into the fluid displaces the volume of fluid exactly equal to the volume of the object immersed. By force (F), the displacement of fluid results an opposing force that pushes against the object to expel it. Physically, F = weight of displaced volume of fluid that in turn is equal to the immersed volume of object in fluid column and thus balance shows this force (F = W). At equilibrium,
Same principle has been used to measure the volume of an unknown object, 16 simply by inserting the object in the fluid filled column placed on balance. This method is sensitive but lacking reproducibility and accuracy, as it is difficult to hold the object like rat paw exactly at different time intervals, without touching the wall of fluid column. An improvised plethysmometer method has been used, 17 where water expelled upon insertion of rat paw is to be collected and measured in the insulin syringe, results error in the measurement due to air space. Dorsoventral measurement of paw pad is not reliable for quantitative measurement. 8 Present model overcomes this drawback and is dominant over simple hydroplethysmometry methods for its accuracy, sensitivity and reproducibility. Sensitivity of presently reported model is high, when water as a fluid is used (0.02 mL) and much high for mercury (0.002 mL), enough to measure the volume of an object like a rat or mice paw, for experimental animal studies to evaluate toxicology and pharmacology of chemicals. Sensitivity could further be enhanced using more sensitive balance.
Present model has advantage that the object to be inserted in sampling column need not to be kept from touching the wall of column, facilitates investigator to hold rat paw accurately at desired height and minimizes investigator error. Water being nontoxic and user-friendly can be preferred as a displacement fluid and the method can be calibrated prior to each sampling by just setting balance read zero. This troubleshooting fluid evaporation error usually occurs in hydroplethysmometry. This method can be used with all the different ratios of
Reported method is simple and cost-effective and hence would be useful for the academic and the commercial laboratory animal research purpose, without compromising the precision of the measurement.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.




