Abstract
Cadmium (Cd) is known to cause oxidative damage in the testes of rats. The aim of this study was to investigate the protective role of rutin (RUT, 30 mg/kg) and selenium (Se, 0.15 ppm) alone or in combination against Cd (200 ppm)-induced lipid peroxidation, steroidogenesis and changes in antioxidant defence system in the rat testes. The obtained results showed that Cd increased lipid peroxidation and abnormal sperm count and decreased plasma testosterone, lactate dehydrogenase, acid phosphatase, alkaline phosphatase and testicular steroidogenic enzymes: 3β-hydroxysteroid dehydrogenase (HSD), 17β-HSD activities as well as epididymal sperm counts and motility, while RUT and Se treatment reversed this change to control values. Acute intoxication with Cd was also followed by significantly decreased activity of the antioxidant defence system (superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), glutathione reductase (GR), glutathione (GSH), and glutathione-S-transferase (GST)). Treatment with RUT and Se reversed Cd-induced alterations of antioxidant defence system and significantly prevented Cd-induced testes damage and depletion of plasma and testicular Se levels. RUT and Se appear not to have more profound effects than their separate effects against Cd-induced testicular toxicity, although Se was more potent than RUT in the recovery of testosterone levels. These results suggest that both RUT and Se do not have synergistic role against Cd-induced testicular injury.
Introduction
Cadmium (Cd) is a heavy metal, which is widely in use in batteries, dyes, plastics, electrochemistry, and paint pigment. It can be routed to soil because of the use of Cd in some insecticides, fungicides, sludge and commercial fertilizers that are used in agriculture. 1 Cd has been found to produce wide range of biochemical and physiological dysfunctions in humans and laboratory animals, 2 and there is sufficient evidence in humans to classify Cd and Cd compounds as carcinogenic substances. 2 The testis is exceedingly sensitive to Cd toxicity as reports have shown that exposure to Cd caused germ cell loss, testicular oedema, haemorrhage, necrosis and sterility in several mammalian species. 3,4 More importantly, Cd exposure may account for the recent declining fertility associated with reduced sperm count and testis function in men in developed countries. 5 However, the cellular pathway underlying the toxic effects of Cd on the testis has not yet been clearly established. 6
A variety of experiments have suggested that Cd causes testicular oxidative damage in animals. Primarily, Cd has a profound effect in reducing the weight of sex organs 7 and can also directly inhibit primary Leydig cell testosterone levels. 8 The oxidative stress and decreased testosterone level occur as a result of an increase in Cd-induced peroxidation of membrane lipids in the organs when it accumulates. 4,9 Cd also diminished the levels of several endogenous antioxidant agents in the body such as GSH and the antioxidant trace elements (Selenium (Se)) and vitamins. 10 –12 These have been shown to contribute significantly to the development of Cd-induced toxic oxidative stress. 11 Se has been reported to reduce and/or prevent both the oxidative stress and damage to the testis caused by Cd. 13,14 There are also many reports in which naturally occurring phytochemicals having antioxidant properties were used against Cd-induced testicular toxicity. 2,9,15 Similarly, dietary antioxidants such as Garlic 16 and naringenin 17 act as strong antioxidants and increase the efficacy of the chelating agent in the treatment of Cd intoxication. 18 Among these phytochemicals, rutin (RUT), a citrus flavonoid glycoside present mainly in buckwheat, onions, apples, tea and red wine, has antioxidant, anti-allergic, anti-inflammatory, anti-angiogenic and antiviral properties because of its superoxide radical scavenging nature. 11 RUT also exhibits multiple pharmacological activities including antibacterial, anti-tumour, anti-inflammatory, anti-diarrhoeal, anti-ulcer, anti-mutagenic, myocardial protecting, vasodilator, immunomodulator and hepatoprotective activities. 19 Many researchers as well as our research group have demonstrated the antioxidant properties of RUT in different experimental models. 20,21 RUT has also displayed protective effects against ethanol-induced gastric lesions, 22 against DNA damage and carcinogenesis. 23,24
The Cd-induced testicular damage as well as the protective effect of RUT has not been reported in previous studies in animal models. Similarly, a joint action of both RUT and Se against cellular oxidative damage has not yet being investigated. Therefore, this study was conducted to evaluate the potential benefit of RUT and Se concomitant treatment on Cd-induced testicular pathophysiology compared to Se or RUT treatment alone in rats exposed to Cd.
Materials and methods
Animals and experimental design
Sexually mature male Wistar rats weighing approximately 139 ± 4 g were divided into six experimental groups of six animals each. The control group was not treated with Cd. The corn oil (100% pure, Vijaya Enterprises, Mumbai, India) group received 2 mL/kg body weight of corn oil. The remaining four experimental groups received 200 ppm Cd (as CdCl2, 99.99% pure, Sigma-Aldrich, Laborchemikalien GmbH, Germany), 200 ppm Cd + 30 mg/kg RUT, 200 ppm Cd + 0.15 ppm Se (as SeO2, 99.99% pure, Sigma-Aldrich), or 200 ppm Cd + 30 mg/kg RUT + 0.15 ppm Se. RUT (95% pure, Sigma-Aldrich, CHEMIE GmbH, Germany) was administered as a suspension in corn oil (2 mL/kg b.wt), while the Cd and Se experimental groups received Cd and/or Se in their drinking water simultaneously. Cd, Se doses and manner of administration were chosen on the basis of available literature data. 4,25 RUT dose was chosen on the basis of our previous studies. 21 The experiment was conducted over a period of 5 weeks. The animals were housed in individual stainless steel cages and exposed to 12-h light–dark cycle. They had access to rat pellet and drinking water ad libitum. The experimental designs and protocols for study received the approval of Institutional Animal Ethics Committee in accordance with the standard guide for the care and use of laboratory animals. Drinking water consumption and daily Cd, Se intakes were investigated according to the method described by Brzóska and Moniuszko-Jakoniuk. 26
On the last day of experimentation, the final body weight of each animal was recorded and the rats were killed by cervical dislocation. Blood was taken by cardiac puncture and was immediately collected into heparinized tubes. Plasma samples, for testosterone, Cd and Se analyses were obtained by centrifugation at 4000 rpm for 10 min at 4°C. The testes were excised, cleared of adhering connective tissues, and weighed. The left testis was divided into two halves, the first was immediately fixed in Bouin’s solution for histopathological studies and the second was kept at −20°C for Cd and Se estimation. The right testis was processed for other biochemical estimations and protein determination.
Measurement of Cd and selenium concentrations
Testicular slices destined for Se and Cd analyses were oven dried (60oC) to constant weight (0.07 ± 0.018 g). Dried tissues and plasma samples were digested with concentrated nitric acid (Merck, 65%) at 120°C. When fumes were white and the solution was completely clear, the samples were cooled to room temperature and the tubes were filled to 5 mL with ultra pure water. All samples were analyzed to determine Cd and Se using flame atomic absorption spectrometry. Samples were analyzed in triplicate and the variation in coefficient was usually less than 10%. Concentrations of Cd and Se were expressed as micrograms per gram dry weight in testis and as micrograms per liter in plasma.
Testosterone determination
Plasma testosterone was assayed using enzyme-linked immunosorbent assay (ELISA) kit (Equipar Diagnostici, SRL, Italy Code No. 74010) following manufacturer’s instruction. The absorbance was read at 405 nm within 30 min in ELISA reader (Merck). The intensity of the colour is inversely proportional to the concentration of testosterone in the sample. The sensitivity of the testosterone assay was 5 pg/mL, and the inter- and intra-run precision had a coefficient of variation of 3.9% and 6.2%, respectively
Epididymal sperm count, abnormal sperm count and motility
Epididymal sperm was obtained by mincing the cauda epididymis in 5 mL normal saline and filtered through a nylon mesh. An aliquot of the filtrate was used to count the number of spermatozoa in each sample using the Neubauer haemocytometer. For the estimation of spermatozoa motility, the epididymal sperm obtained from the cauda epididymis was placed on a slide and diluted with buffered 2.9% sodium citrate, pre-warmed to 37°C. The percentage motility of epididymal sperm was evaluated visually at 400× magnification within 2–4 min of their isolation from the cauda. Motility estimations were performed from three different fields in each sample. The mean was used as the final motility score. For morphological observation, one drop of the sperm suspension was smeared onto a glass slide, fixed in 95% ethanol and stained with 1% eosin and 5% nigrosine. At least 200 sperms from each rat were examined for abnormalities in different regions of spermatozoa according to the method described by Okamura et al. 27 The results were recorded as the percentage of abnormal sperm on each slide.
Biochemical assay
Testicular slices were homogenized in cold sodium phosphate buffer (pH 7.4). The homogenates were then centrifuged at 4000 rpm for 15 min at 4°C. The supernatants were separated and used for enzyme assays and protein determination. The activity of superoxide dismutase (SOD) using epinephrine as substrate was assayed according to the method of Misra and Fridovich. 28 Catalase (CAT) activity was assayed by the reduction of dichromate in acetic acid to chromic acetate when heated in the presence of hydrogen peroxide (H2O2). The chromic acetate produced was measured colorimetrically at 610 nm. 29 Glutathione-S-transferase (GST) activity was assayed by the method of Habig et al. 30 The specific activity was expressed as μmol GSH-CDNB (1-chloro-2,4-dinitrobenzene) conjugate formed/min/mg protein using an extinction coefficient of 9.6 mM−1cm−1. GSH was determined by the method of Jollow et al. 31 where the colour developed was read at 412 nm. Glutathione peroxidase (GSH-Px) was assayed based on the reaction between glutathione (GSH) remaining after the reaction of GSH-Px and 5,5-dithiobis-(2-nitrobenzoic acid) to give a compound that absorbs light at 412 nm. 32 Glutathione reductase (GR), which utilizes NADPH to convert oxidized glutathione (GSSG) to the reduced form (GSH), was assayed according to the method described in Staal et al. 33 Acid phosphatase (ACP) and alkaline phosphatase (ALP) activities were assayed according to the method described in Ketaki et al. 34 and was based on the hydrolysis of p-nitrophenyl phosphate. Liberated p-nitrophenyl was spectrophotometrically quantified at 420 nm. Lactate dehydrogenase (LDH) activity was determined by the method of Cabaud and Wroblewski 35 and was based on the interconversion of pyruvate and lactate. The activity level of testicular 3β-hydroxysteroid dehydrogenase (HSD) was measured based on the conversion of NAD to NADH while that of 17β-HSD was measured based on the conversion of NADPH to NADP. 36 The absorbance at 340 nm was measured at 30 s intervals for 5 min in a spectrophotometer against blank controls. The enzyme activities were expressed in mmol of NAD converted to NADH/mg protein/min (3β-HSD) and mmol of NADPH converted to NADP/mg protein/ min (17β-HSD). One unit of enzyme activity is the amount causing a change in absorbance of 0.001/min at 340 nm. Lipid peroxidation was determined by the method of Ohkawa et al. 37 This method was based on the reaction of malondialdehyde (MDA) and thiobarbituric acid to generate a coloured product with an absorbance at 532 nm. A standard curve using serially diluted 1,1,3,3-tetramethoxypropane was prepared and MDA concentrations in the experimental samples were extrapolated from the standard curve. Lipid peroxidation was expressed as mmol MDA/mg protein. Protein content in the tissue samples was determined by the method of Lowry et al. 38
Statistical analysis
All data were presented as the mean ± SD. Data were subjected to statistical analysis via one-way ANOVA. Student’s t test was utilized for comparison of data between control and experimental groups. The difference was considered to be statistically significant at p < 0.05.
Results
Effect of Se and RUT on Body and Testicular weights
As shown in Table 1, significant decreases in body and testes weights were observed as a result of Cd administration in Cd group compared to control group. Se and RUT administration alone or in combination significantly attenuated Cd-induced changes in body weight and testes weights. No significant change in body and testes weights were found in rats treated with corn oil only in comparison with the control group.
Changes in body weight, testis weight, plasma testosterone level and sperm characteristics in rats after Cd treatment and simultaneous supplementation with Se and RUT alone or in combination.a
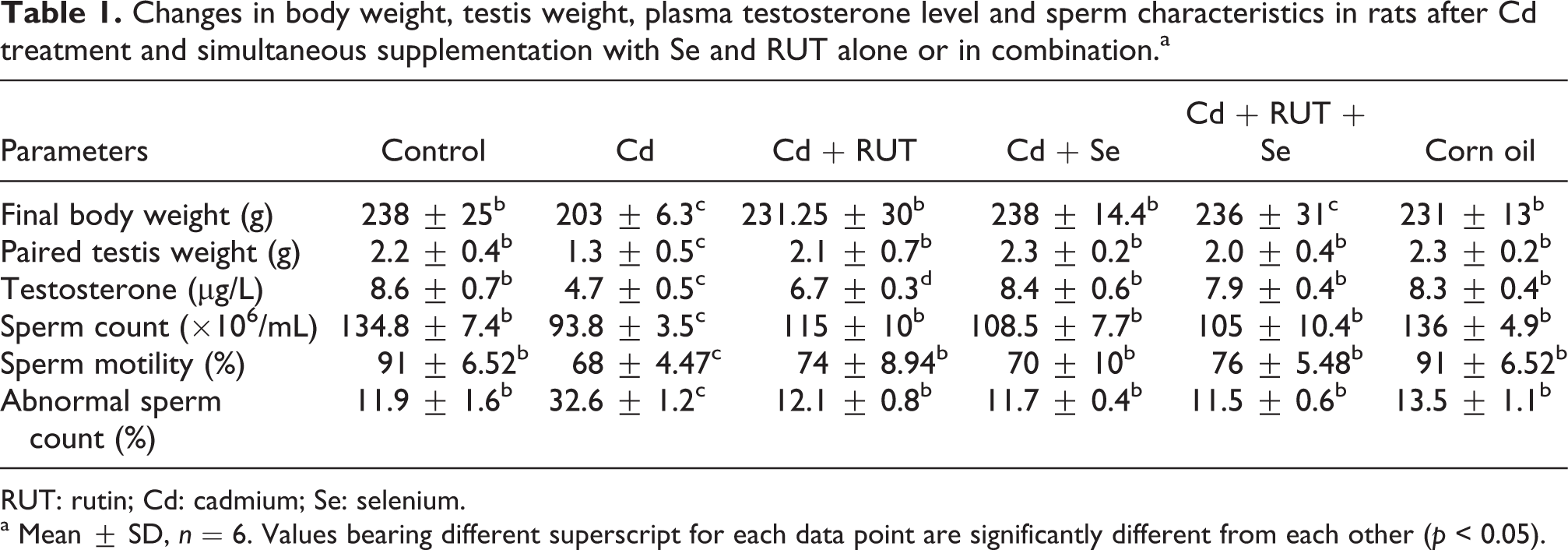
RUT: rutin; Cd: cadmium; Se: selenium.
a Mean ± SD, n = 6. Values bearing different superscript for each data point are significantly different from each other (p < 0.05).
Plasma testosterone levels, abnormal sperm count, epididymal sperm count and motility
The Cd-treated rats showed significantly lower plasma testosterone level as compared to the control animals. However, the level of plasma testosterone was reversed by the administration of Se and RUT alone or in combination. The testosterone level in the Cd + RUT + Se group was not significantly different from the Cd + Se group but was significantly increased when compared with the Cd + RUT group, indicating that Se was more effective than RUT in restoring testosterone level to near control value (Table 1). In fact, Se administration increased the plasma testosterone level by 79%, whereas RUT administration increased testosterone level by 43% when compared with Cd-treated animals. Cd treatment decreased epididymal sperm concentration and motility and increased abnormal sperm morphology rate compared with control rats. The treatment of Cd-exposed rats with Se and RUT separately or in combination prevented the Cd-induced decrease in sperm concentration, motility and increase in abnormal sperm count (Table 1). No significant change in plasma testosterone level, abnormal sperm count, epididymal sperm concentration and motility in animals treated with corn oil in comparison to the control animals was observed.
Cd and Se concentrations in plasma and testis
Our results showed that Cd administration significantly increased Cd accumulation in the plasma and testes and simultaneously decreased Se concentrations in testicular tissue and plasma in comparison with control rats. The treatment of Cd-exposed rats with Se alone had no effect on the Cd-induced increase in the plasma and testicular concentrations of Cd, whereas it reversed the Cd-induced depletion of Se concentrations in testis and plasma. The combination of RUT + Cd alone decreased the plasma and testicular Cd concentrations compared with Cd-exposed animals. This combination also prevented Cd-induced depletion of Se concentrations in the plasma and testis. The co-administration of Se + RUT in Cd-treated rats was also effective as it resulted in a significant increase in plasma and testicular Se concentrations in comparison with the Cd-treated rats and reversed the Cd-induced increase in the plasma and testicular concentrations of Cd. There were no significant differences in plasma and testicular Se and Cd concentrations between control and corn oil groups (Tables 2 and 3).
Se concentration in plasma and testes of rats after Cd treatment and simultaneous supplementation with Se and RUT alone or in combination.a

RUT: rutin; Cd: cadmium; Se: selenium.
a Mean ± SD, n = 6. Values bearing different superscript for each data point are significantly different from each other (p < 0.05).
Cd concentration in plasma and testes of rats after Cd treatment and simultaneous supplementation with Se and RUT alone or in combinationa.

RUT: rutin; Cd: cadmium; Se: selenium.
a Mean ± SD, n = 6 for each treatment group. Values bearing different superscript for each data point are significantly different from each other (p < 0.05).
Activities of testicular steroidogenic enzymes
Testicular 3β-HSD and 17β-HSD enzymes play a pivotal role in testicular steroidogenesis. Figure 1 shows that activities of these enzymes were decreased in Cd-exposed group of rats compared with control rats. The decreased testicular 3β-HSD and 17β-HSD activities resulting from Cd treatment were reversed in animal groups treated with Se or RUT separately or in combination compared with Cd-exposed group of rats. However, we noticed that co-exposure to Cd and Se completely reversed these changes, whereas RUT supplementation only partially reversed the Cd-induced decrease in 3β-HSD and 17β-HSD activities. The activities of these enzymes were also restored completely to the control values with the combined Se and RUT treatment. No significant change in testicular 3β-HSD and 17β-HSD enzymes activities in animals treated with corn oil in comparison to the control animals were observed (Figure 1).

Effects of selenium (Se) and/or rutin (RUT) on testicular steroidogenic enzyme activities namely, 17β-HSD (a) and Δ 5 3β-HSD (b) in rats after Cd treatment. (Mean ± SD, n = 6). Bars bearing different superscripts are significantly different from each other (p < 0.05).
Antioxidant defence system and lipid peroxidation in testes
Cd exposure significantly decreased the antioxidant defence system, GR, GSH-Px, GSH, GST, SOD, CAT and increased MDA concentration as compared with control rats (Control and Corn oil groups). The treatment of Cd-exposed rats with Se or RUT alone or in combination significantly increased the level of the antioxidant defence system and decreased the MDA concentration as compared with Cd-exposed rats (Figures 2 and 3). We did not observe any significant change in the antioxidant defence system and lipid peroxidation in testes of rats treated with corn oil in comparison to the control animals.
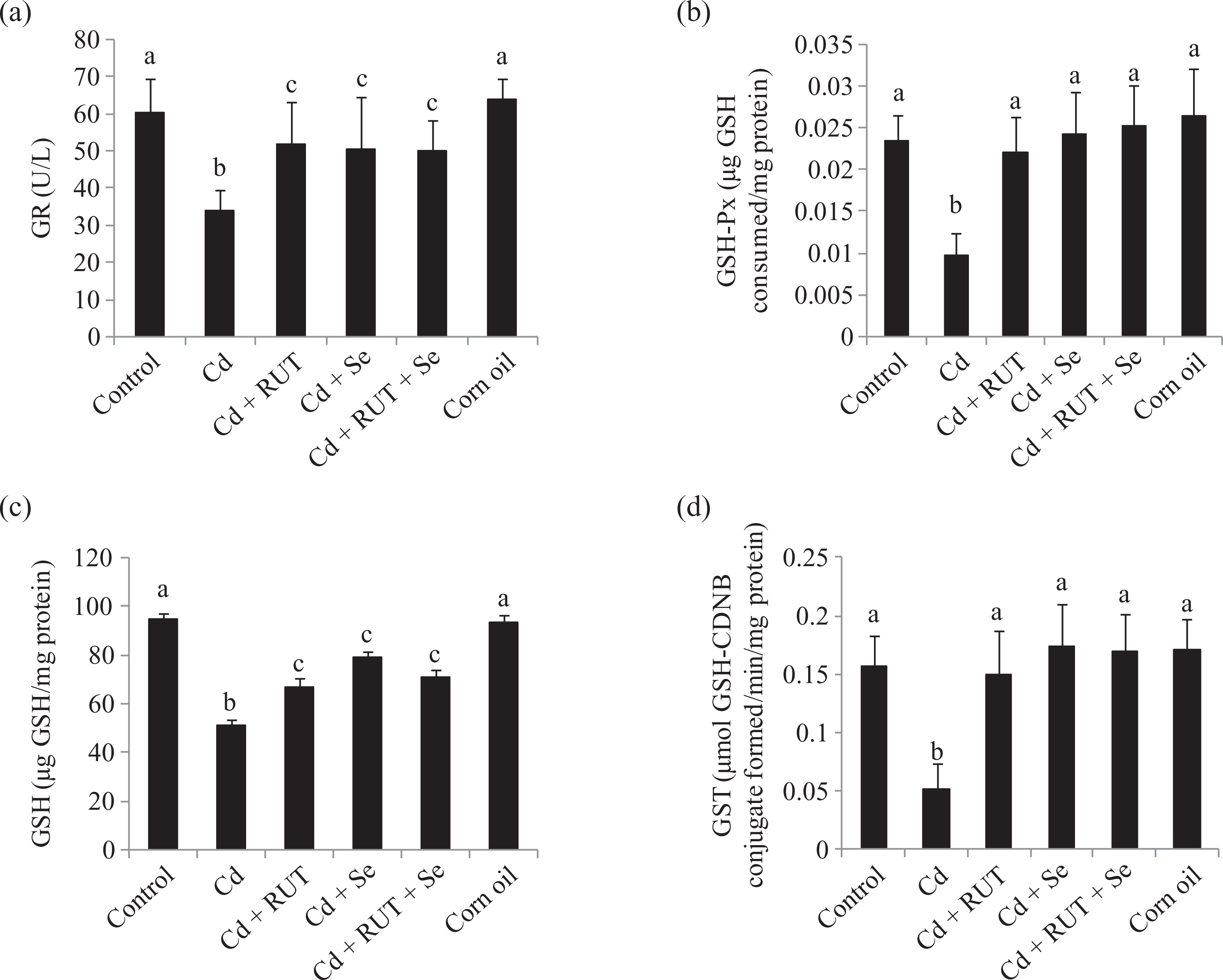
Effects of selenium (Se) and/or rutin (RUT) on testicular glutathione reductase (GR) activity (a) glutathione peroxidase (GSH-Px) activity (b) reduced glutathione (GSH) level (c) glutathione-S-transferase (GST) activity (d) in rats after Cd treatment. Results are expressed as mean ± SD. n = 6 for each treatment group. Bars bearing different superscripts are significantly different from each other (p < 0.05).

Effects of selenium (Se) and/or rutin (RUT) on testicular superoxide dismutase (SOD) activity (a), catalase (CAT) activity (b) and malondialdehyde (MDA) level (c) in rats after Cd treatment. Results are expressed as mean ± SD. n = 6 for each treatment group. Bars bearing different superscripts are significantly different from each other (p < 0.05).
Activities of testicular maker enzymes (marker of spermatogenesis)
Compared with the control and corn oil groups, testicular LDH, ACP and ALP, activities showed a significant decrease in the Cd-treated group. Co-treatment with Se or RUT separately or in combination effectively increased the activities of LDH, ACP and ALP toward normalcy (Table 4). There was no significant change in the activities of the testicular marker enzymes (LDH, ACP and ALP) in animals treated with corn oil in comparison to the control animals were observed.
Effect of Se and/or RUT on testicular LDH, ACP, and ALP activities in rats after Cd treatment.a

RUT: rutin; Cd: cadmium; Se: selenium; LDH: lactate dehydrogenase; ACP: acid phosphatase; ALP: alkaline phosphatase.
a Mean ± SD, n = 6 for each treatment group. Values bearing different superscripts are significantly different from each other (p < 0.05).
Histopathological study of testis
Histological study as shown in Figure 4 demonstrates normal seminiferous tubular diameter in control and corn oil-treated animals. On the other hand, shrinkage of tubular diameter was observed in Cd-treated testis. Se and RUT supplementation alone or in combination restored the shrinkage in tubular diameter.
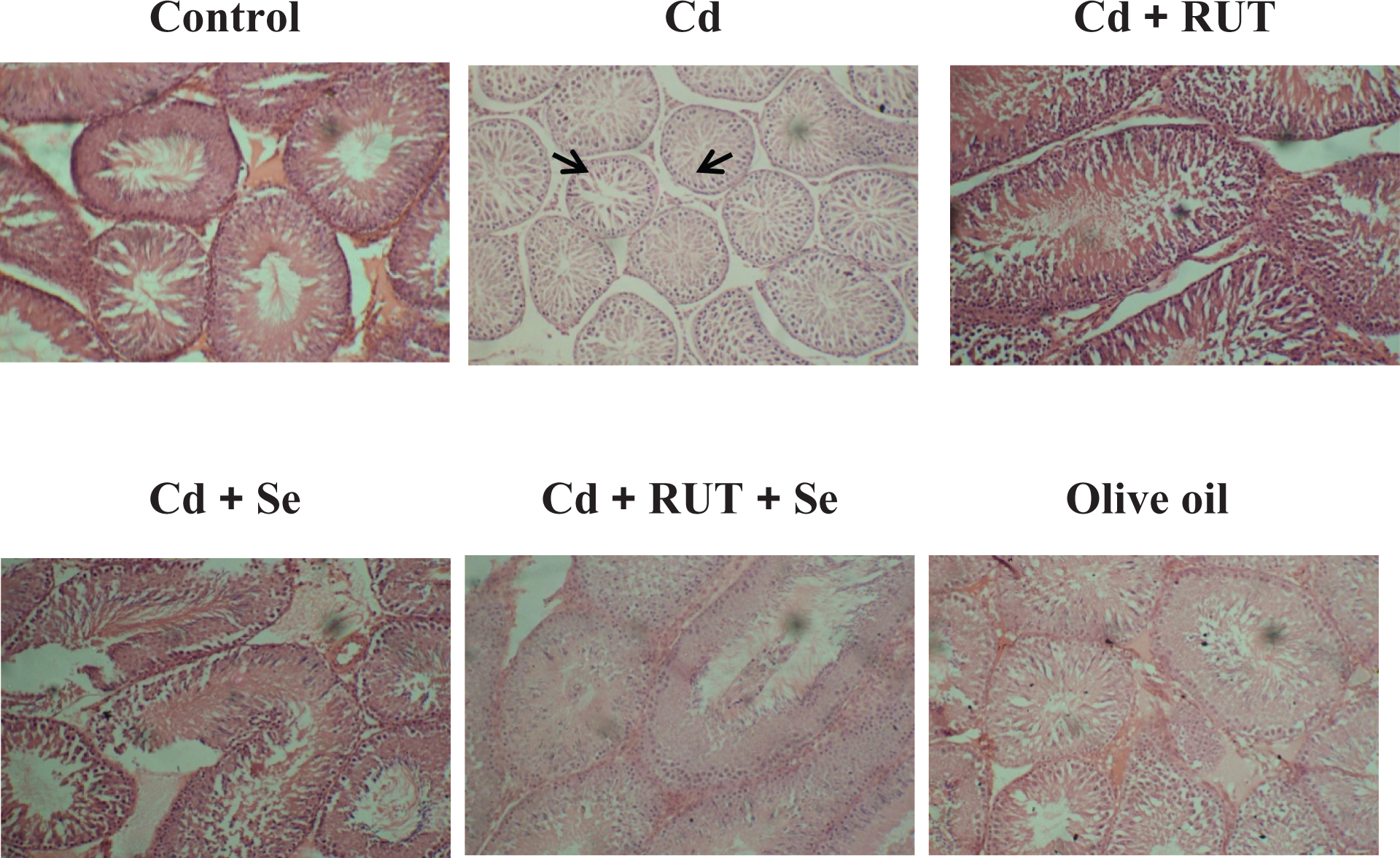
Representative photomicrograph of haematoxylin and eosin stained sections of testis of control and experimental rats (400×).
Discussion
The combined treatments of antioxidant in the prevention of Cd-induced testicular pathophysiology have been previously reported in rats. 4,14,18 In this study, we combined two potent antioxidants, a micro-nutrient, Se, and a dietary antioxidant flavonoid, RUT, to investigate their protective effects separately and then in combination against Cd-induced testicular pathophysiology in rats. In the present study, the administration of Cd significantly decreased the body weight, testis weight and the seminiferous tubular diameter. The observed weight loss of the testis may be due to the adverse effect of Cd on the number of germ cells and elongated spermatids in the testis. 39 The decrease in the weight of the testis and the reduced gain in body weight observed in this study are consistent with the report of others. 40 –42 Co-administration of Se + RUT to Cd-treated rats showed results approximating those in the control group with respect to testes weight, seminiferous tubular diameter and gain in body weight. The antioxidant effects of Se and RUT have been demonstrated previously in rats. 4,20,21,43 It is also reported that RUT could suppress lipid peroxidation and scavenge free radicals such as superoxide and hydroxyl radicals produced during Cd intoxication 11,44 Consistent with our results, supplementation of Se + RUT normalized the altered activities of the enzymatic and non-enzymatic antioxidant defence systems (CAT, SOD, GSH-Px, GR, GST, and GSH) in testicular tissues of Cd-treated rats. The combination of Se + RUT prevented the reduction of testicular weights in the Cd-treated rats when compared with those in Cd-alone-treated animals. The protective effects of Se + RUT can be explained by the prevention of cellular damage occurring because of oxidative stress in testicular tissues. A significantly increased accumulation of Cd in testis observed in animals treated with Cd is in accordance with previous studies. 4,25
In this study, it was observed that Cd treatment significantly decreased epididymal sperm count, motility and significantly increased abnormal sperm morphology in comparison with the control group. These results correlate with previous findings of Cd treatment in animals. 4,6,15,41 The reduction in sperm count, motility and increased abnormal sperm morphology in Cd-treated rats may be caused by lipid peroxidation of unsaturated fatty acids in the sperm plasma membrane, resulting in a loss of its fluidity and function. 40 Furthermore, overproduction of reactive oxygen species (ROS) or their accumulation due to dysfunction of the antioxidant defence system following Cd treatment leads to lipid peroxidation. 18,45 Cd is also known to bind directly to the active site of the enzyme containing –SH groups or could displace the metal cofactors from the active sites resulting in their inactivation. 46 Lipid peroxidation is a significant manifestation of oxidative damage and has been found to play an important role in the toxicity of many xenobiotics. 18
Co-administration of Se + RUT alone or in combination to the Cd-treated group prevented the adverse effect of Cd on sperm characteristics, and values were close to those in the control group regarding sperm count, motility and abnormal sperm count. The obtained results correlate with the previous findings 4,21 and could be explained on the basis of the antioxidant properties of Se and RUT.
In the present study, LDH, ACP, and ALP activities showed a significant decrease after Cd administration. Since the activities of these enzymes are closely associated with spermatogenesis, the decreased activity of these enzymes in Cd-administered animals represents a defect in spermatogenesis and testicular maturation. Similar patterns of testicular LDH, ACP and ALP activities have been reported with a number of chemicals causing testicular toxicity. 2,21,47 Furthermore, the activities of ALP and ACP have also been reported to correlate with testicular steroidogenesis, suggesting that the decreased steroidogenesis was responsible for the reduction in the number of spermatids. 2 Administration of Cd-treated rats with Se + RUT revealed significant increase in testicular LDH, ACP and ALP when compared with the Cd-alone treated group. The obtained results agree with the previous findings, 2,21,47 suggesting that Se and RUT protected testicular function from the adverse effects of Cd.
Plasma testosterone level and testicular steroidogenic enzyme (3β-HSD and 17β-HSD) activities were significantly decreased in Cd-treated rats compared with those in the control. Our findings are in agreement with many previous studies and was thought to be related to the decreased level of gonadotrophins. 48 –50 The decrease in plasma testosterone level in Cd-treated rats may be due to the inhibition of these testicular androgenic enzyme activities, because these enzymes are responsible for the regulation of testosterone biosynthesis. RUT and Se prevented the deleterious effects of Cd on steroidogenesis as manifested by restoring the activities of the two key enzymes and plasma testosterone. The observed significant increase in plasma testosterone level in the group treated with Cd + Se is consistent with the previous reports, 4,51 confirming that Se is required for testosterone biosynthesis. The increased testosterone level in the groups treated with Cd + Se has been attributed to the antioxidant action of Se 4,51 as ROS is known to inhibit steroidogenesis. 52 However, this is the first time we are reporting reversal of testosterone level to normalcy by RUT in Cd-induced testicular injury. The RUT-reversed Cd-induced reduction of steroidogenesis might be through scavenging of Cd-induced ROS, which can affect steroidogenic enzyme activities at protein and transcription levels. 52 Other flavonoids with antioxidant properties as RUT have also been reported to prevent the decrease in testosterone levels in other models of testicular toxicity. 53,54 However, Se was more potent than RUT in reversing to normalcy the adverse effects of Cd on steroidogenesis. This might be due to the increased supply of Se to the testis in this group of animals. It has been reported that severely Se-deficient rats and men have lowered serum testosterone concentrations, suggesting that Se is essential for male fertility and normal development of spermatozoa. 55,56 Our observations that plasma and testicular Se concentrations were higher in Se-treated rats confirm the relevance of Se for testosterone biosynthesis.
Testicular oxidative stress is known to play an important role in male fertility. It is now known that lipid peroxidation is one of the toxic manifestations of Cd administration in testis. In the present study, the treatment of male rats with Cd resulted in a significant increase in testicular tissue levels of MDA (malondialdehyde), a by-product of lipid peroxidation, whereas GSH levels, CAT, SOD, GSH-Px, GR and GST were significantly decreased. These results correlate with previous findings of Cd treatment in animals. 4,6 The treatment of Cd-treated rats with Se + RUT revealed significant reduction in testicular tissue MDA as compared with the Cd-treated group. Se + RUT administration to Cd-treated animals significantly elevated testicular tissue GSH level and CAT, SOD, GSH-Px, GR and GST when compared with values in the Cd-alone-treated animals. The obtained results agree with the findings of others. 20,21,51,57 The reduction in lipid peroxidation in testicular tissue of Cd-treated animals because of the administration of Se + RUT alone or in combination apparently indicated that Se and RUT potently scavenged free radicals and suppressed oxidative damage.
From our study, the combination of RUT and Se did not exert stronger effect than when each was administered separately against Cd-induced testicular pathophysiology, suggesting that there was no cooperative protective action between RUT and Se against Cd-induced testicular toxicity. Therefore the combined treatments of flavonol-based phytochemicals such as RUT with antioxidant trace elements such as Se do not have greater benefits than the administration of the individual molecules during conditions of Cd-intoxication. This protective effect of RUT and Se is attributed to their antioxidant 21,51 and metal-chelating properties of RUT. 44 The capacity for Se to increase steroidogenesis better than RUT could be attributed to the increased supply of testicular Se which is required for testosterone biosynthesis. It is thus likely that testicular Se concentration could be maintained to prevent the testicular disorder caused by Cd. Supplementing with Se may improve testicular function as Se is an essential cofactor in GSH enzymatic function 58 as well as a component of selenoproteins such as glutathione peroxidase, thioredoxin/glutathione reductase which play important roles in the redox regulation of normal spermatogenesis. 4 Furthermore, Se might also be the cornerstone of the antioxidant defence in Cd intoxication because the Cd-treated rats showed low levels of Se in the testes as well as in the plasma which were directly correlated with defects in spermatogenesis and steroidogenesis.
When the present study was initiated, the hypothesis was that the provision of a combination of endogenous antioxidant micronutrient and a dietary antioxidant (i.e. a multimodal approach) during Cd-intoxication would provide a stronger effect. For instance, Se and antioxidant vitamins such as vitamin E and vitamin C function synergistically to regenerate both water- and fat-soluble antioxidants. 59 Our results do not confirm the hypothesis that a combination strategy of antioxidant supplementation is better than single Se delivery. The reason for this could be that flavonoid–flavonoid or Se-vitamin interactions are more feasible than Se-flavonoid antioxidant interaction possibly due to a different mechanism of action.
In summary, Cd treatment increased lipid peroxidation and altered the antioxidant defence systems, resulting in oxidative damage to rat testes and decreased steroidogenesis and sperm quality. Treatment with RUT and Se alone or in combination showed protective effects against Cd-induced lipid peroxidation and changes in enzymatic and nonenzymatic (SOD, CAT, GSH-Px, GR, GST and GSH) antioxidant defence systems, steroidogenesis and sperm quality of rats. RUT and Se act as potent antioxidants alone or in combination which may be due to their free radical scavenging and metal-chelating properties. Although, RUT and Se alone and in combination did not function cooperatively in reducing the harmful effects of Cd, Se was more potent than RUT in the prevention of Cd-induced decrease in steroidogenesis. Both antioxidants can protect testicular function from Cd-induced oxidative stress.
Footnotes
Acknowledgements
The technical expertise of Mr Omoko Ejiro of the Department of Veterinary Reproduction, University of Ibadan, Oyo State on spermatozoa analysis is gratefully appreciated.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
