Abstract
Type I diabetes mellitus is a metabolic disease caused by the impairment of pancreatic β-cells mainly mediated through oxidative stress and related apoptosis. Islets transplantation seems a promising treatment for these patients, but during islets transplant, various types of stresses related to the isolation and transplantation procedure compromise the function and viability of islets. We recently hypothesized that the combination of cerium oxide (CeO2) and yttrium oxide (Y2O3) nanoparticles with a potential free radical scavenger behavior should be useful to make isolated islets survive until transplanted. In the present study, oxidative stress-induced apoptosis in isolated rat pancreatic islets exposed to hydrogen peroxide (H2O2) and the protective effects of CeO2 and Y2O3 nanoparticles were investigated. Exposure of islets to H2O2 (50 µm, 2 h) increased intracellular oxidant formation such as reactive oxygen species and subsequently apoptosis and decreased viability, glucose-induced adenosine triphosphate (ATP) production and glucose-stimulated insulin secretion. Pretreatment with CeO2 and/or Y2O3 nanoparticles reduced the oxidant formation and apoptosis and increased viability, glucose-induced ATP production and glucose-stimulated insulin secretion. These results suggest that this combination may protect β-cell apoptosis by improving the oxidative stress-mediated apoptotic pathway.
Introduction
Type I diabetes mellitus, also known as insulin-dependent diabetes mellitus (IDDM) is an autoimmune disease that is caused due to the death of insulin producing pancreatic β-cells. Current therapy for IDDM patients is the persistent substitutive insulin injection. However, insulin therapy cannot duplicate a normal physiological response and patients usually experience cardiomyopathy, neuropathy, and nephropathy. Therefore, there is a strong need for suitable treatments or supplements. Islets transplantation seems a promising treatment for IDDM patients and has been under attention of researchers in the recent years. During islets transplant, various types of stresses related to the isolation and transplantation procedure compromise the function and viability of islets. The main restriction is the loss of healthy islets at the end of the operation due to the inevitable-prolonged time required for islets isolation. Isolated islets are avascular and are therefore ischemic from the time of isolation through to revascularization. Prolonged ischemia makes a significant loss of islet cells. 1 As a result of ischemia and the islet isolation, oxidative stress occurs that damages some of the isolated islets. Interestingly, islets have lowest levels of intrinsic antioxidant defenses. Therefore, the free radicals easily induce their toxicity. In fact, a significant loss of islet cell mass in transplantation procedure is due to apoptosis and necrosis partially mediated by oxidative stress. 2 To overwhelm oxidative stress during islet transplantation, various antioxidants like vitamin supplements, trace elements and drugs have been widely examined. 2 –4 On the basis of evidences, we recently hypothesized that the combination of cerium oxide (CeO2) and yttrium oxide (Y2O3) nanoparticles with a potential free radical scavenger behavior should be useful to make isolated islets survive until transplanted. 5 Therefore, in the present work, we studied the protective effects of this combination in isolated islets by measuring the biomarkers involved in the survival of islets in an ex vivo model.
Materials and methods
Chemical and reagents
All chemicals were obtained from Sigma-Aldrich (Gmbh Munich, Germany) unless otherwise stated. CeO2 and Y2O3 nanoparticles were purchased from Navarrean Nanoproducts Technology (Navarra, Spain). Rat specific insulin enzyme-linked immunosorbent assay (ELISA) kit was obtained from Mercodia (Uppsala, Sweden).
Islet isolation
Rat islets were isolated from male adult Wistar rats (>12 weeks) weighting 200–250 g, according to the ethical guidelines, and the study was approved by the institute (code: TUMS-14936). Islets were isolated from the pancreas by collagenase digestion followed by repeated dextran gradient centrifugation as previously set up in our laboratory and described in detail previously. 6,7 Briefly, animals were anesthetized with intraperitoneal injection of sodium pentobarbital (60 mg/kg). After laparotomy, the common bile duct was ligated at its exit into the liver. The duct was cannulated at its exit from duodenum and then the pancreas was perfused by the injecting of 10 mL of Krebs buffer. Perfused pancreas was dissected from duodenum and the tissue surrounding the islet was digested by break down using collagenase, and after separation from fat, the tissue was washed by Krebs–N-2-Hydroxyethylpiperazine-N'-2-Ethanesulfonic Acid (HEPES) buffer three times. The extract tissue was centrifuged in Krebs buffer for two rounds of 60s and then 0.5% bovine serum albumin (BSA) was added for the completion of digestion. Finally, the islets were separated from the remaining tissue by hand picking under stereomicroscope and the purity evaluated by dithizone staining that was >95%. The islets between 100 and 150 µm in diameter were used for the experiments.
Dose optimization for CeO2 and Y2O3 nanoparticles
Before starting the experiments, the purified isolated islets were cultured overnight in the culture media that contained RPMI-1640 medium, in 5% CO2 at 37°C. Optimization of dose was done by pretreating the islets with various concentrations of CeO2 nanoparticles (50, 100 and 200 µm) and Y2O3 nanoparticles (0.5, 1 and 2 µg/mL) for 3 days to reach effective doses (200 µm and 2.214 µm, respectively). Then the islets were divided into eight groups of 10 islets including: (1) Con (negative control), islets in RPMI-1640 medium alone for 3 days; (2) hydrogen peroxide (H2O2) (positive control), islets in RPMI-1640 medium alone for 3 days + H2O2 (50 µm) for 2 h; (3) Nan Cer Ox (nano cerium oxide; islets in RPMI-1640 medium + CeO2 nanoparticles (200 µm) for 3 days); (4) Nan Cer Ox + H2O2 (islets in RPMI-1640 medium + CeO2 nanoparticles (200 µm) for 3 days + H2O2 (50 µm) for 2 h); (5) Nan Ytt Ox (nano yttrium oxide; islets in RPMI-1640 medium + Y2O3 nanoparticles (2.214 µm) for 3 days); (6) Nan Ytt Ox + H2O2 (islets in RPMI-1640 medium + Y2O3 nanoparticles (2.214 µm) for 3 days + H2O2 (50 µm) for 2 h); (7) Nan Cer Ox + Nan Ytt Ox (islets in RPMI-1640 medium + CeO2 nanoparticles (200 µm) + Y2O3 nanoparticles (2.214 µm) for 3 days); (8) Nan Cer Ox + Nan Ytt Ox + H2O2 (islets in RPMI-1640 medium + CeO2 nanoparticles (200 µm) + Y2O3 nanoparticles (2.214 µm) for 3 days + H2O2 (50 µm) for 2 h).
Islets viability assay
This assay is based on the reduction of 3-4, 5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT), a yellow tetrazole to purple formazan by mitochondrial respiration in viable cells. After removing the medium, pretreated islets were washed twice by Krebs–HEPES buffer, and 20 µL of MTT solution was added and incubated for 4 h at 37°C. After washing, the formazan was resuspended in 100 µL dimethyl sulfoxide and the absorbance was measured in 570 nm by ELISA reader. The viability of the groups was shown as the percentage of controls that is assumed as 100%. 8
Measurement of reactive oxygen species
To measure reactive oxygen species (ROS) generation, a fluorometric assay using intracellular oxidation of 2,7-dichlorofluoroscein diacetate (DCFH-DA) was performed, that was set up in our laboratory and was described previously in detail. 9 Briefly, pretreated islets were washed with phosphate-buffered saline (PBS), and then incubated with 40 µM DCFH-DA for 30 min. At the end of incubation, the islets were washed with PBS, lysed with NaOH and aliquots were transferred into the blank well plate. Then, the fluorescence of dichlorofluoroscein, which is the oxidized product of DCFH-DA was measured by ELISA fluorimeter with excitation and emission wavelengths of 485 and 530 nm, respectively. The values were standardized by the amount of total protein in each well.
Glucose-stimulated insulin secretion assay
Pretreated islets were separated in tubes and washed twice by Krebs–HEPES buffer and then incubated for 30 min at 37°C at a level of 2.8 mM glucose. An amount of 1 mL of 2.8 mM glucose and 16.7 mM glucose each as basal and stimulant doses, respectively, was added to all the groups and incubated at 37°C for 30 min. Then, the supernatants were collected and stored in separate microtubes. Insulin concentration was determined by rat insulin ELISA kit according to the manufacturer’s protocol. Results are displayed as microunits insulin secreted per islet per hour. 10
Protein assay
To measure total protein concentration of islets, Bradford reagent was added to the diluted samples and the absorbance was determined after 5 min by the spectrophotometer at 595 nm. Also, the albumin was used as a standard.
Evaluation of energy as ATP/ADP ratio in islets
Pretreated groups of 100 islets were sonicated in 250 µL of trichloroacetic acid (TCA) (6%) and then centrifuged at 12,000
Caspase-3 and -9 activity assays in islets
Caspase-3 and -9 activities were measured by colorimetric assays based on the identity of specific amino acid sequences by these caspases. The tetrapeptide substrates were labeled with the chromophore ρ-nitroaniline (ρNA). ρNA is released from the substrate upon cleavage by caspase and produces a yellow color that is monitored by an ELISA reader at 405 nm. The amount of caspase activity present in the sample is proportional to the amount of yellow color produced upon cleavage. 11 Briefly, the pretreated islets were lysed in the supplied lysis buffer and were incubated on ice for 10 min. The whole cell lysates were incubated in caspase buffer (100 mM HEPES, pH 7.4, 20% glycerol, 0.5 mM EDTA, 5 mM dithiothreitol) containing 100 µM of caspase-3 and -9 specific substrate (N-acetyl-Asp-Glu-Val-Asp-p-nitroanilide (Ac-DEVD-pNA:), N-acetyl-Leu-Glu-His-Asp-p-nitroanilide (Ac-LEHD-pNA), respectively) for 4 h at 37°C. Then, absorbance was measured at 405 nm. Caspase activity was defined as nanomoles per ρNA released per hour per milligram of protein (nanomoles per hours milligrams of protein) by a ρNA calibration curve.
Detection of apoptotic cells
Apoptosis was investigated by staining the islets with Hoechst 33342. The islets were washed twice with PBS and fixed in PBS containing 10% formaldehyde at room temperature for 4 h. Fixed islets were washed with PBS and stained with Hoechst 33342 for 30 min at room temperature. After that cells were evaluated under fluorescence microscope for nuclei showing apoptotic features such as chromatin condensation and fragmentation. Apoptotic cells were counted in a blinded fashion in two slides per condition and per experiment. 11
Results
Islet viability
The cell viability decreased to approximately50% when cells were treated with 50 µM of H2O2 for 2 h. Then, we investigated the effects of Nan Cer Ox or/and Nan Ytt Ox on oxidative stress-induced apoptosis. As seen in Figure 1, MTT assay shows that the pretreatment of islets with Nan Cer Ox or Nan Ytt Ox significantly increases the oxidative stress-reduced cell viability (

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on the viability of isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 hours, and then cell viability was measured. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
ROS production
As shown in Figure 2, exposure of islets to H2O2 significantly increased the production of ROS when compared with controls (
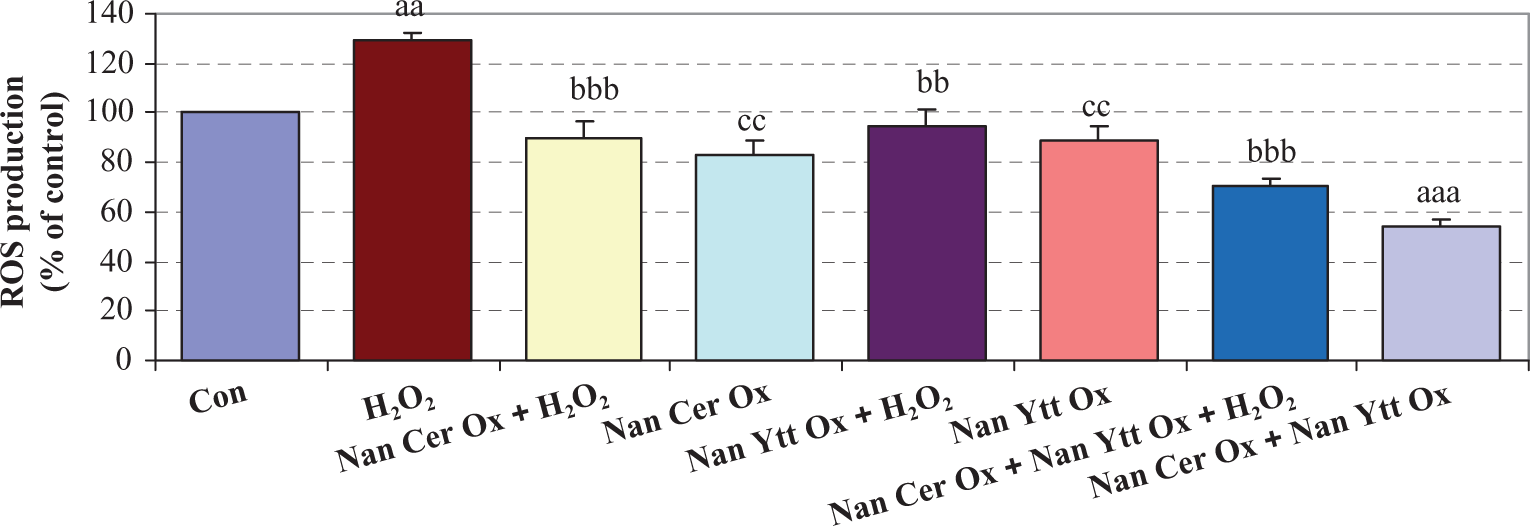
Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on ROS production in isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, and then ROS production was measured. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
Insulin secretion
Islet cells in the presence of 2.8 mM glucose did not exhibit a significant change in insulin secretion. As shown in Figure 3, high glucose caused a fourfold increase in insulin secretion compared with low glucose. Incubation with H2O2 significantly decreased glucose-induced insulin secretion (

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on insulin secretion in isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, and then insulin secretion was measured. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
Glucose-induced ATP generation
The effects of H2O2 and Nan Cer Ox and/or Nan Ytt Ox on ATP have been examined in isolated rat islets by calculating the ratio between ATP and ADP (Figure 4). This ratio is more important for glucose-stimulated insulin secretion than the ATP level. At low-glucose levels (2.8 mM), neither H2O2 nor pretreatment with Nan Cer Ox and/or Nan Ytt Ox resulted in changes of ATP/ADP ratio. But high levels of glucose (16.7 mM) resulted in a twofold increase in the ATP/ADP ratio, and H2O2 treatment significantly inhibited the increase in this ratio (

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on ATP/ADP rations in isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, and then ATP/ADP rations was measured. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
Caspase-3 and -9 activities
As shown in Figure 5, H2O2-induced apoptosis significantly increased the activity of caspase-3 and -9 when compared with controls (

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on caspase-3 and -9 activities in isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, and then cell caspases-3 and -9 activities were measured. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
Apoptosis of Islet cells
We evaluated the apoptotic cells after Nan Cer Ox or/and Nan Ytt Ox pretreatment under oxidative stress. As seen in Figure 6, apoptosis was assessed morphologically by Hoechst staining where arrows indicate Hoechst-stained cells with apoptotic bodies. Hoechst staining also revealed a significant increase in apoptotic cell in islets exposed to H2O2 when compared with controls (

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on morphological assessment of apoptosis in isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, washed, fixed and stained with Hoechst 33342 5 µg/mL for 30 min. The arrows point to apoptotic cell nuclei. Quantification of two independent experiments performed in triplicates. (a) Control cells and (b) H2O2-exposed cells. Con: control; Nan Cer Ox: nano cerium oxide; Nan Ytt Ox: nano yttrium oxide.

Effects of Nan Cer Ox and/or Nan Ytt Ox pretreatments and H2O2 treatment on apoptosis of isolated rat islets. Islets were pretreated with Nan Cer Ox (200 µm) and/or Nan Ytt Ox (0.5 µg/mL) for 3 days and after that were exposed to H2O2 (50 µm) for 2 h, and then the apoptotic cells were counted. Results are mean ± SEM of three independent experiments performed in duplicate. Difference between control and other groups is significant at
Discussion
The prerequisite for successful islet transplantation is possessing a large number of viable and functional pancreatic islets after isolation. Regarding the production of oxidative stress during islet isolation and transplantation in one hand, and the lowest levels of intrinsic antioxidant defenses in islets on the other hand, a major challenge in the preparation of islet happens. ROS levels were found to correlate with viability and apoptosis. 12 Apoptosis is implemented by the activation of a family of caspases. Caspases-8 and -9, as initiators, are activated through self-processing and then cleave downstream procaspase-3 to the active form of caspace-3, which acts as the executor of apoptosis. 11 In the present study, we observed a significant increase in ROS and a decrease in the cell viability of islets exposed to H2O2. Also, we found that H2O2-induced apoptosis increases the activity of caspases-9 and -3, whereas pretreatment of CeO2 or/and Y2O3 nanoparticles in these conditions improved all of these defects.
On the other hand, as in all aerobic cells, the mitochondrial respiratory chains represent a major source of ROS in beta-cells and also are a primary target for ROS attacks. 13 One of the first events occurring after the addition of H2O2 is an intensive decrease in the concentration of reducing equivalents but it is unknown if this is secondary to the depolarization of mitochondrial (mitochondrial membrane potential) ΔΨ. These changes in ΔΨ reduce the production of ATP. It seems that there is a close link even for mild oxidative stress to the KATP current and beta-cell function. 13 Low levels of ROS facilitate the regulation of insulin gene expression and beta-cell function. In contrast, excessive concentrations of ROS decreases the insulin gene expression and thus insulin secretion that led to the damage of islets. 14 Three factors, glucose metabolism, ROS generation and ATP production (and thus KATP channel activity) are closely coupled. Increasing the exogenous H2O2 impairs ATP production and opens KATP channels that result in hyperpolarizing the beta-cells to limit Ca2+ influx and glucose-induced insulin secretion. 15 The ROS accumulation in mitochondria, ATP depletion and depolarization of ΔΨ are closely coupled to cell metabolism and also apoptosis. 13 Our findings confirm these subjects by an increase in ROS and apoptosis, and decrease in ATP levels, viability and insulin secretion in islets after exposure to H2O2. Interestingly, we observed a significant improvement in these factors via the pretreatment of islets by CeO2 or/and Y2O3nanoparticles.
Reducing the deleterious oxidative stress on islets function, viability and ROS-induced cell death by enhancing antioxidant capacity or decreasing ROS generation are the promising strategies to prevent the oxidative damage of pancreatic islets for transplantation. For this, we now confirm a novel approach for the improvement of islet transplantation procedures using a combination of CeO2 and Y2O3 nanoparticles. 5 CeO2 nanoparticles are known as a biological free-radical scavenger or antioxidant having oxygen vacancies for dual oxidation states that is created by the loss of oxygen and the reduction in Ce+4 to Ce+3. Moreover, these nanoparticles have several advantages over other antioxidants: first, CeO2 nanoparticles act as catalysts to mimetic superoxide dismutase activity. 16,17 Second, the antioxidant activity of these nanoparticles is mediated at oxygen vacancies on the surface. Therefore, one CeO2 nanoparticles offer many sites for catalysis, despite the fact that enzymes or pharmacological agents have only one active site per molecule. Finally, these nanoparticles remain resident and active in a living cell for an extended period of time. 18 Y2O3 is notable because the free energy of oxide formation from elemental yttrium is among the highest known. Y2O3 nanoparticles have also shown potential scavenger behavior similar to ceria. 19 Most recently, CeO2 nanoparticles have been tested for the ability to serve as free-radical scavengers or antioxidant. 20–23 Also, similar properties were found with Y2O3 nanoparticles. 19
In summary, with no doubt, we need a strong antioxidant mixture to protect islets during transplantation 24,25 and to overcome diabetes. 26 Nanoparticles composed of CeO2 and Y2O3 can have antioxidant properties that promote cell survival under the conditions of oxidative stress. Our study suggests that these nanoparticles may be a therapeutic regenerative material that will scavenge ROS that are responsible for ROS-induced death of islets and the accompanying loss of function. This study demonstrates that the combination of CeO2 and Y2O3 nanoparticles may protect the β-cells by improving the oxidative stress-mediated apoptotic pathway and have the potential to become a ‘theranostic agent’.
Footnotes
Funding
This work was supported by Tehran University of Medical Sciences and Iran National Science Foundation (INSF).
