Abstract
The U.S. Congress [PL 107-188] amended the Safe Drinking Water Act and required each community water system serving more than 3,000 people to conduct vulnerability assessments. These assessments address potential circumstances that could compromise the safety and reliability of municipal water. Ferric chloride is used in coagulation and flocculation, and it is used to treat raw water with high viral loads, elevated dissolved solids or high bromide. Iron is an essential nutrient, but elevated concentrations of FeCl3 are corrosive as a result of hydrolysis to HCl. Based on a no-observed-adverse effect level (NOAEL) of 0.5% FeCl3 • 6H2O administered in drinking water to male and female F344 rats for up to 2 years, a do not consume concentration of 200 mg FeCl3 /L can be derived. Since instillation of 0.3 M (48.7 g/L) FeCl3 in saline to rodent vagina failed to elicit damage, a topical do not use concentration of 2000 mg FeCl3/L (600 mg Fe/L) can be assigned. The only FeCl3 data available to quantify ocular toxicity involved a pH 1 solution in rabbit eyes, but HCl instillation (pH 2.5) to rabbit eyes found permanent corneal ulceration after 10 min. The pH of FeCl3 in water at the do not use limit (2.4–2.6) is near the pH (2.0) considered corrosive by regulatory agencies. As direct eye contact with water at pH 4.5 or below increases complaints of ocular discomfort, emergency response plans that address FeCl3 in drinking water must account for Fe levels and the pH of the affected water.
Keywords
Introduction
Title V of the Public Health Security and Bioterrorism Preparedness and Response Act (U.S. Public Law 107–188) mandated vulnerability assessments by drinking water purveyors who service more than 3,300 connections. These assessments are intended to identify procedures that can be brought to bear in the event of a terrorist or other intentional attack on municipal water systems. 1 ,2 Emergency response plans can be valuable, given the numbers and consequences of accidents involving gross chemical contamination of drinking water supplies. 3 –10 The present analysis was conducted to define do not consume and do not use concentrations for FeCl3 that may be used to formulate emergency response plans.
The most likely scenario for an emergency during which affected water reaches the public involve short-term (possibly once-in-a-lifetime) exposure for at least one but less than 30 days. 5,8 –10 Nevertheless, the first 24 h are critical in preventing adverse health effects after which time constituent levels are reduced by dilution unless the chemical source persists. Since utility customers can be exposed as a result of ingestion and direct skin and eye contact during bathing and showering, it is important to consider oral, dermal and ocular contact with excessive concentrations of substances in drinking water.
The present evaluation of acute oral, dermal and ocular exposures to FeCl3 is intended to aid regulatory agencies in decisions to avoid or prohibit direct human contact with affected water. Carefully documented accounts of short-term (acute) human experience with reliable exposure data are the preferred key studies, but where those data are lacking, controlled studies in laboratory mammals were evaluated. A lowest acute no-observed-adverse effect level (NOAEL), an acute lowest-observed-adverse-effect-level (LOAEL) or an acute benchmark dose (where possible) are values that could be used to calculate an acute reference dose (RfD). To locate those values, the literature search strategy employed here was based on the Chemical Abstract Services Registry Number and at least one common name. The following public access databanks were searched: ChemID Plus Registry of Toxic Effects of Chemical Substances (RTECS) Hazardous Substances Data Bank (HSDB) GENE-TOX Environmental Mutagen Information Center (EMIC) Developmental and Reproductive Toxicology (DART) TOXLINE—Core and Special TRI (Toxics Release Inventory) Chemical Carcinogenesis Research Information System (CCRIS) Medline (via PubMed) Integrated Risk Information System (IRIS) Syracuse Research Corporation Online Toxic Substance Control Act Database (TSCATS)
Reports considered most valuable are detailed and published in the peer-reviewed open literature. Case reports of acute exposures usually describe symptoms and their consequences and those accounts were noted here, but nearly all case reports were limited by small numbers of patients and a near-universal absence of dose; these difficulties preclude use of case reports to establish short-term do not use and do not consume drinking water limits. Detailed reports from short-term clinical trials and summaries of (uncontrolled, but often extensive) clinical experience with short-term exposure durations are valuable. Due to the relatively few animal data for most corrosive materials (like FeCl3), all published accounts (including reviews and summary reports) that could be located were compiled. Lethality data were collected, but derivation of public warning levels based on death was not considered health protective; likewise, reports concerning mild sensory irritation and adverse taste, odor or appearance of affected water were not candidate key data. Acute studies were preferred, yet repeat dose, subchronic, reproduction and chronic duration studies can provide information on target organs and the results noted during initial exposure can be used to set or support derivation of protective short-term limits. Criteria to identify candidate key studies that document serious or irreversible adverse health consequences included relevant route of exposure, a reasonable number of subjects, concurrent controls (as available) and sufficient details to explain study design, execution and evaluation. Single-dose or short-term kinetic studies could inform interspecies dose scaling, but no controlled acute dose kinetic study in any species with FeCl3 was identified. Data from standard short-term genotoxicity and long-term bioassays for potential carcinogenicity were reviewed and where reliable studies of not more than 30 days exposure by an appropriate route demonstrate a significant genotoxic carcinogenic response in animals, the short-term theoretical excess cancer risk greater than 1:10,000 (1× 10−4) could be used in calculating the short-term exposure limits. As acute toxicity studies for most large commodity materials used in water treatment were conducted decades before the advent of standard operating procedures and adoption of defined regulatory protocols, adherence to those regulations played no part in identification of potential key studies; the limited animal and human toxicity data for FeCl3 precluded rigorous consideration of statistical parameters. Given the relatively few published studies in animals that met all these criteria, consistent results from different sources were important in identification of potential key and supporting studies. Since FeCl3 is corrosive at high concentrations, its concentration-dependent relation to water pH was integral to identification of short-term do not use and do not consume levels.
Therefore, the point of departure for deriving the oral do not consume limit is designated the acute RfD. The terms acute dermal reference value (acute dermal RfV) and acute ocular reference value (acute ocular RfV) will be used in the present assessment and these values were considered as points of departure in establishing the do not use limit. The potential for acute inhalation exposure was also considered in establishing the do not use limit, although inhalation (e.g., bathing and showering) is not likely to be the determining route of exposure for a nonvolatile compound.
To derive the acute RfD and RfV, interspecies and intraspecies uncertainty factors were applied to LOAEL, NOAEL or benchmark dose values. Data from clinical studies that included susceptible individuals were preferred key observations; where data on potentially sensitive groups (e.g., extremes of age, the infirm, nutritional status, those with genetic predisposition) are not available, additional intraspecies uncertainty factors may be selected by analogy to similar chemicals but usually those factors are less than 10. The concentrations for short-term notification derived here are designed to protect nearly all consumers including “susceptible” individuals, but these emergency concentrations are such that people with idiosyncratic or unique (hypersusceptible) conditions may not be fully protected. The acute oral RfD was used to derive the drinking water do not consume limit, and the lower of the acute dermal RfV and the acute ocular RfV was used to derive the drinking water do not use limit. Following U.S. Environmental Protection Agency (EPA) convention when calculating a 1-day drinking water health advisory, the do not consume limit is defined as the acute RfD (mg/kg) multiplied by the 10-kg weight of a child assumed to drink 1 L of water per day. Due to the brief duration, a relative source contribution was not applied.
Chemical and physical characteristics
Ferric chloride (iron +3 chloride; iron trichloride; iron sesquichloride) is available commercially as the anhydrous iron perchloride and as the hydrate (FeCl3 • 6H2O; Table 1).
Physical and chemical properties of ferric chloride.

The hexahydrate is an orange-yellow crystal that decomposes in water or in moist air to produce HCl. 14 It is the HCl released from FeCl3 that accounts for the very low pH of concentrated solutions and the more concentrated the solution, the lower the pH. The pH of a 10% (100,000 mg/L) aqueous solution of FeCl3 was reported at 1.11, 18 the pH of a 0.1 M (16,200 mg/L) solution was 2.0 13 and that of a 0.01 M (1,620 mg/L) solution was 4.5. 19
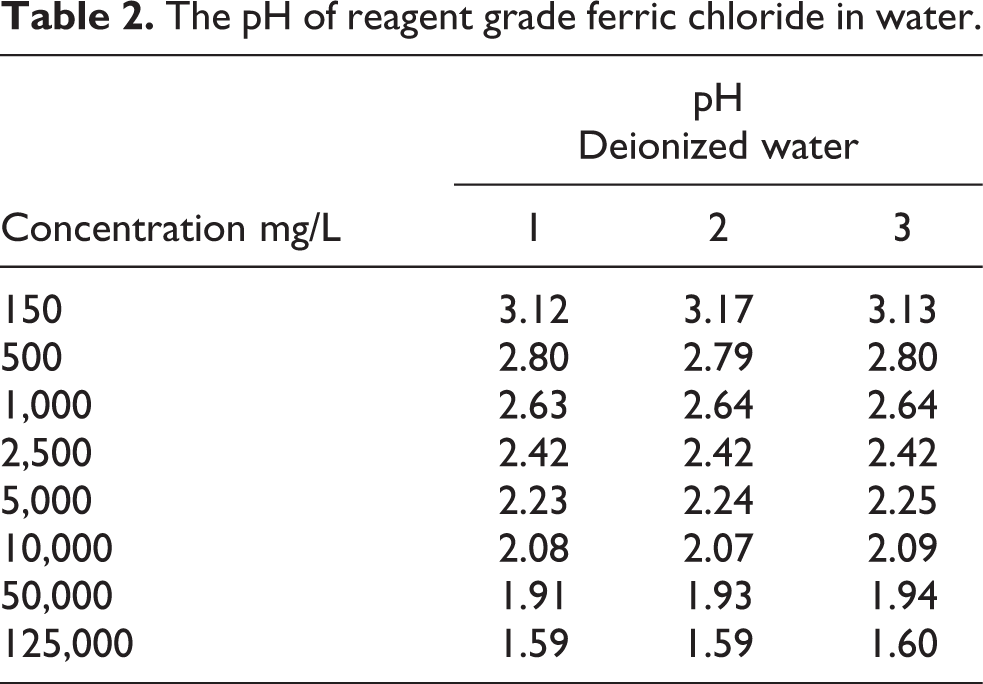
To illustrate the influence of FeCl3 on pH, measurements were taken by the NSF laboratory of reagent grade FeCl3 dissolved in distilled, deionized water at room temperature (Table 2).
The pH of reagent grade ferric chloride in water.
Ferric chloride is hygroscopic and exothermic in water. After hydrolysis, Fe reacts with water and hydroxides precipitate. In municipal drinking water and sewage treatment applications, it is the reaction of FeCl3 with hydroxide ion in slightly alkaline water that produces flocs of iron oxides
and these oxides remove suspended particulate along with H2S, PO4 −, AsO4 and microbes. 20 – 22 Due to the buffering capacity, dissolved solids, oxidizable substances and other factors, the pH of FeCl3 in deionized water (Table 2) may not be representative of FeCl3 in drinking water and the pH can differ depending on the characteristics of the particular water.
Organoleptic properties
There are no published taste or odor thresholds for FeCl3, but granular or dissolved Fe salts will stain body tissues, fabrics, porcelain fixtures and porous surfaces (e.g., concrete). Elevated concentrations of FeCl3 in water are acidic (Table 2) and consumer acceptance of drinking water with pH less than 6.5 is reduced [40 CFR 143.3]. In the presence of H2S (as in groundwater wells with elevated SO4 2−), FeCl3 will impart a blackish-brown tint to water.
Production and use
Iron normally exists in groundwater as ferrous (Fe2+) iron; but when it is exposed to oxygen, it is oxidized to the ferric (Fe3+) state, a form that accounts for the reddish-brown tint. Ferric chloride is a commodity compound that can be produced by reaction of dry chlorine gas with scrap iron or iron ore and NaCl or KCl at 500°C–700°C, but most FeCl3 is derived either from spent HCl steel pickling liquor or from recycling of etchant effluents; and it can be collected as a by-product of titanium dioxide manufacture. 23 Ferric chloride is available in several grades ranging from the anhydrous (96%) to photographic and sewage formulations. 14 As of 2001, the domestic production capacity was 465,000 short tons/year. 24
Ferric chloride is used in purification of municipal drinking water, 25 ,26 treatment of sewage and sludge 27 and in management of certain industrial wastes. 28 –30 Ferric chloride is especially valuable in treating raw water with relatively high concentrations of dissolved solids, bromides and other organics. Ferric chloride is used in coagulation flocculation treatment of wastewater 31 –34 and contaminated groundwater 35 as well as in regeneration of anion exchange resins by degradation of perchlorate ion (ClO4 −). 36 As a weak Lewis acid, it finds applications in chlorination of aromatic compounds and Friedel–Crafts reaction of aromatics. Ferric chloride is also used as an etchant in engraving, photography and in printed circuit production, 14 and it is used as a reagent to test urine for salicylates. 37
The hydroalcoholic tincture of FeCl3 has been used as a topical styptic (astringent and coagulating hemostatic) where 0.5 ml of a 35% U.S.P. solution (4.48% Fe) has been applied directly to treat acute tonsillitis. The tincture can also be diluted (Bestuscheff’s tincture), mixed with citric acid (Citro-chloridi, N.F.) or placed into gelatin capsules and consumed immediately. 38,39 It was once popular in veterinary medicine where it was used in bovine dehorning and in dewclaw and wart removal. 40
Per 21 CFR 184.1297, FeCl3 is generally recognized as safe (GRAS) when added directly to foods intended for human consumption and it is permitted as an indirect food additive (as a component of adhesives) under 21 CFR 175.105. As FeCl3 in water dissociates to hydrochloric acid, 14 HCl is also a GRAS buffer and neutralizing agent under 21 CFR 182.1057, provided it is used in accord with good manufacturing practice.
Effects on humans
Iron is an essential nutrient and Fe deficiency is the most common cause of microcytic hypochromic anemia. The pharmacology 41 ,42 and toxicology 43 ,44 of ingested Fe salts are well known. An adult male requires ~1 mg Fe/day and a female requires ~1.6 mg/day; during pregnancy, requirements increase to 5–6 mg/day and infants require about that same amount due to their rapid growth. Foods high in Fe include egg yolks, wheat germ, organ meat (liver, heart), oysters, dried beans and fruit, but it is the relative gastrointestinal bioavailability of various Fe forms in the diet that is of primary nutritional significance. Heme iron (6% of dietary Fe) represents ~30% of the Fe absorbed, but nonheme Fe is the major form ingested by economically disadvantaged people and by vegetarians where it is only poorly absorbed due to inhibition by dietary phosphate. 42
The average daily Fe supplement (usually FeSO4•H2O containing 20% Fe) used to treat Fe deficiency in adults is 200 mg (2-3 mg/kg-day). Pregnant women are given supplemental Fe at 15 to 30 mg each day (equivalent to 0.25–0.5 mg/kg day for a 60-kg adult) and small children and infants routinely receive 5 mg/kg day. The higher Fe doses used clinically are typically associated with constipation (10%), diarrhea (5%) and nausea and epigastric pain (7%). Symptoms are dose dependent with ~25% complaining at 200 mg/day (~ 3.0 mg/kg day for an adult) and ~40% complaining at 400 mg/day (~ 6.0 mg/kg day for an adult), but these effects can be mitigated to a certain extent by consuming Fe supplements with meals. 42 Patients also complain because Fe supplements color the feces black and large doses in infants can interfere with phosphorous absorption to the point that chronic ingestion can lead to iatrogenic rickets. Often, patients with enteritis or ulcerative colitis cannot tolerate oral Fe supplements. 41
Ferrous sulfate (FeO4S) is employed routinely to treat Fe-deficient anemia with doses of 150–300 mg, 3 times/day, resulting in uptake on the order of ~45 mg iron each day (1,350 mg/month). As the duration of therapy increases, the amount absorbed from that same daily dose declines (25 mg during the second and third months, to 5–10 mg thereafter as body stores plateau). Ferrous salts are used because they are about 3 times more bioavailable than their ferric counterparts; compared to the systemic uptake after ingestion of ferrous iron, the Fe uptake after ingestion of ferric salts is at least 33%–50% less. 44 ,45 There are no indications that excessive dietary Fe can cause hemosiderosis in healthy people, and continuous daily doses as high as 60 mg of FeO4S for at least 19 years are well tolerated. 44
Between 2000 and 5000 episodes of acute Fe poisoning (especially in young children) are reported each year. 46 –48 Most of the fatalities occur among infants and young children as a result of ingested FeSO4 tablets (each containing 0.3 g Fe) coated with candy or chocolate; fatality rates are on the order of 1%. 49 The actual dose is often difficult to discern due to prompt emesis; death has occurred after ingestion of as few as 15 tablets, but some patients have survived after consuming as many as 70 tablets (total dose = 20 g). 43
Wu et al. 50 described their experience with 16 cases (9 male; 7 female) of acute ingestion of aqueous FeCl3 (25% accidental, 19% occupational, 56% suicidal) in victims aged 12 to 70 yr (mean = 30.8 yr). Nine of those patients were hospitalized and one died. Of those who survived, gastritis, esophagitis with esophageal and gastric erosions were evident along with abdominal pain (37%), oral ulcers (38%), metabolic acidosis (25%), aspiration pneumonia (19%), dyspnea (12.5%), diarrhea (12.5%) and hypotension (12.5%). One of the fatalities 51 involved suicidal ingestion by a 25-year-old woman who drank 200 mL of pH 1.0 FeCl3 (57.6 g/L). Her total dose was 230 mg/kg or 80 mg/kg as elemental Fe. At 3 h after ingestion, she developed tachycardia, tachypnea, vomiting and drowsiness. Physical examination found a brown-stained tongue and gastrointestinal damage. Her serum iron at 3.2 h was 2,440 µg/dL and she subsequently expired. In a similar case described by Chen et al., 52 an adult male (24 years) ingested an unstated quantity of liquid FeCl3 in attempted suicide. He presented with emesis, epigastric pain, hemorrhagic gastritis, metabolic acidosis, elevated serum iron and evidence of renal tubular damage. He received deferoxamine chelation and he survived.
In the fatality described by Doolin and Drueck, 53 an 18-year-old man fell into a vat of saturated FeCl3 dissolved in aqueous HCl. He aspirated some of the solution into his lungs, ingested the material and absorbed Fe through his burned skin. He subsequently developed gastrointestinal hemorrhage, disseminated intravascular coagulopathy and metabolic acidosis and he died. In another case, the victim died after the only route of FeCl3 systemic uptake was via burned skin. 54 The clinical course in the latter case was similar to that described by Doolin and Drueck 53 except that sepsis, acute renal failure and respiratory arrest preceded death. No estimate of the administered or absorbed FeCl3 dose was provided in either of these reports. 53 ,54
Chronic Fe overload (hemosiderosis) results in its accumulation in cardiac muscle, reduced cardiac output and cardiac failure, 47 and it is thought that Fe-induced cardiotoxicity is related to impaired Ca2+ transport in the sarcoplasmic reticulum. It is the divalent (Fe2+) as contrast to the trivalent (Fe3+) state that is most closely associated with inhibition of the Ca2+ release channel in excitation–contraction coupling of cardiac muscle. 55 Death has been ascribed to vascular collapse; autopsy usually reveals hepatic periportal hemorrhagic necrosis and acute hepatic failure. Among survivors, pyloric stenosis with severe scarring can follow. 43
Allergic contact sensitization to Fe salts, while rare, has been confirmed with 2% FeCl3 patch testing in at least two patients. 56 –58
Effects on laboratory animals
Acute exposure
Single dose oral
Acute oral LD50 values of 28 mg/kg in rats and 500 mg/kg in mice have been reported. 59 The acute oral LD50 for anhydrous FeCl3 in mice was reported as 400 mg/kg 60 and 450 mg/kg 61 and National Institute for Occupational Safety and Health (NIOSH) 62 tabulated a rat oral LD50 value of 1,872 mg/kg. The reasons for the marked variations in the acute oral lethal dose values for rats and mice are not clear as no details of the individual protocols (e.g., whether the FeCl3 was dissolved in water or in buffered solution) were available for inspection.
Bianchini et al. 63 gave single oral intubation doses of FeCl3 • 6H2O to fed or fasted C57BL/6 J mice at 0, 10, 32.5 and 65 mg/kg (equivalent to 0, 2.0, 6.5 or 13 mg Fe/kg) and sacrificed these animals at 24 hr after exposure. No deaths were reported, but there was an increase in the numbers of nuclear aberrations in the mucosal cells of the stomach, duodenum and large bowel at the highest dose. The numbers of micronuclei in mucosal cells in the fed mice were reduced compared to the numbers of affected cells in the fasted mice.
Single dose topical
No studies assessing skin contact with FeCl3 in laboratory animals were identified.
Single dose ocular
Intracorneal injection of 0.05–0.1 mL of a concentrated (10%) aqueous solution of FeCl3 (pH 1) into rabbit eye produced a loss of the papillary response to light and reduced intraocular pressure. These changes persisted for 1–4 weeks. 64 , 65
The consequences of HCl contact with mammalian tissues depend on the initial concentration and duration of exposure. 43 , 66 In studies that compared HCl-induced damage in rabbit eye after intracorneal injection to that after a 10-min irrigation of the intact eye, Friedenwald et al. 67 found (regardless of exposure method) that as the pH declined the damage to the cornea increased. Intracorneal injection of aqueous isotonic HCl at pH 3.0 produced little or no discernible opacification, but identical treatment with pH 2.0, pH 1.5 and pH 1.0 solutions caused corneal lesions in 40%, 50% and 85% of the eyes, respectively. Corneal damage and “moderate” conjunctival ischemia began within “a few seconds” of initial irrigation with 0.1 N HCl. Dense opacification began during 10-min irrigation with isotonic (0.005 N) HCl (pH 2.5), and inspection the following day found corneal edema, mucopurulent ocular discharge and conjunctival rubor. However, serious consequences of direct contact with pH 2.5 HCl became obvious 14 days later when “most of the eyes showed gross ulceration of the cornea, with perforation resulting in about one-half the severely burned eyes.” In eyes that were less severely burned, the cornea developed a permanent “silky” opacity. 67
Repeat dose studies
Oral
Sato et al. 68 provided groups of 10 male and 10 female weanling F344 rats with drinking water in which FeCl3 • 6 H2O(98.5% purity) was dissolved at 0%, 0.12%, 0.25%, 0.5%, 1.0% or 2.0% (w/v) ad libitum for 13 weeks. Since aqueous 2% FeCl3 was stable at room temperature for at least 1 week, fresh solutions were made up twice each week in distilled water. Concurrent controls were given distilled water. Body weights and daily water consumption were measured and the mean FeCl3 intakes were calculated from the amount of the solution consumed. These concentrations were equivalent to 0, 80, 154, 277, 550, or 1,231 mg/kg day in males and 0, 88, 176, 314, 571, or 1,034 mg/kg day in the females as FeCl3. 65 The pH of drinking water with 0%, 0.25% and 0.5% FeCl3 was 6.03, 2.38 and 2.19, respectively. These animals were fed a basal CRF-1 laboratory rodent chow ad libitum throughout the study, but no food consumption data were provided.
Body weights were measured once per week for the first 13 weeks and once every 4 weeks thereafter. None of the rats died, but the terminal body weight gain was reduced at least 10% in treated compared to control animals. The high-dose groups consumed significantly less water compared to the concurrent control. Necropsy found the esophageal mucosa was stained brown in those given ≥0.25% FeCl3 and the lamina propria of the large intestine accumulated brown stains in rats given 2.0% FeCl3. The hepatic Kupffer cells, the cartilage of the trachea and bronchii, the tongue, forestomach, the mucosa of the small intestine, the renal tubular epithelium and the white pulp of the spleen were all stained brown in rats that drank water with ≥0.25% FeCl3. Sato et al. 68 identified 0.5% as a drinking water maximum tolerated FeCl3 concentration for both genders.
Topical
No signs of general siderosis were seen in adult female Wistar rats and NMRI mice after 10–40 direct vaginal instillations of 0.15 or 0.3 M FeCl3 in saline (24.3 or 48.7 g FeCl3/L). 69 Mice received twice daily instillations at 0.1 ml and the rats received twice daily instillations at 0.2–0.25 ml. Rats were given a total of 30 instillations over three 5-day periods and killed at 15 h after the last dose or identical rats were given 40 installations over four 5-day periods with termination either at 15 h or at 7 days after the final dose. Mice received 10 instillations over 5 consecutive days with sacrifice 15 h after the last installation, or they were given 20 installations over two 5-day periods with sacrifice 15 or 60 h after the final treatment. No signs of local inflammation, degeneration or other lesions were observed in any of these animals.
Ocular
No studies assessing repeat ocular contact with FeCl3 in laboratory animals were identified.
Long-term studies
Groups of 50 weanling (5 week old) male and 50 female F344 rats were fed a basal CRF-1 diet and provided with 0%, 0.25% or 0.5% FeCl3•6 H2O (98.5%) ad libitum in their drinking water for 2 years. 68 Mean daily FeCl3•6 H2O intakes in the 0.25% and 0.5% groups were 169.7 and 319.7 mg/kg day in the males and 187.9 and 336 mg/kg day in the females, respectively. The rats were given free access to a stock chow, but no food consumption data were reported. At 104 weeks, all of the animals were given distilled water for an additional 8 weeks. Body weight gains were reduced significantly in both groups of treated male and female rats compared to the concurrent controls, and cumulative mortality among rats given 0.5% FeCl3•6 H2O was reduced significantly compared to the control. There were no significant differences in the overall tumor incidence between control and treated groups of either gender; none of the treated groups experienced an increase in the incidence of any specific tumor type compared to the control. Other than brown stains in the gastrointestinal tract (as noted during the subchronic study), there were no histologic changes found that were considered specific to FeCl3 ingestion.
Topical or ocular
No data assessing long-term topical or repeated ocular exposure to FeCl3 in laboratory animals were identified.
Absorption, distribution and elimination
No empirical data concerning Fe uptake after acute FeCl3 exposure were located, but the absorption and distribution characteristics of Fe salts after single or repeated oral doses are well known. In the only repeat exposure study with FeCl3, F344 male and female F344 rats had total serum Fe concentrations of 102 ± 5, 107 ± 5, 109 ± 8, 129 ± 22, 139 ± 10 and 156 µg/dL after consuming water for 13 weeks with 0%, 0.12%, 0.25%, 0.5%, 1.0% and 2.0% FeCl3, respectively. 68
Absorption of dietary Fe is controlled by the small intestine, but the normal physiologic control mechanism can be overwhelmed after ingestion of large amounts. 41 Iron is absorbed most readily in the ferrous (Fe2+) state and increasing the amount ingested increases the amount absorbed, but it also reduces the fraction of the administered dose that is absorbed. Ingested ferrous (Fe2+) iron is transported through the intestinal mucosa where it is oxidized to ferric iron (Fe3+) and the systemic dose is present as ferric iron. After a single oral dose, initial rapid absorption is followed by slower uptake into the systemic circulation that is rate limiting. Systemic Fe uptake is greatest from the duodenum. 70,71 Transferrin (siderophilin) mediates transport of Fe after it has been absorbed; plasma Fe varies with disease states and physiologic conditions, including infectious disease, anemia and nutritional status. The Fe bound to transferrin accounts for only ~0.1% of total body iron; ~66% is found in hemoglobin, 3% in myoglobin and 25% is present as hemosiderin and ferritin. The primary routes of Fe elimination include exfoliation of epithelial cells from the skin and gastrointestinal tract, via menstruation and via body sweat.
Risk characterization
While several groups have been identified as particularly susceptible to chronic Fe accumulation, the major health problem associated with Fe is nutritional deficiency. It is not known whether excessive Fe in the diet or drinking water may increase the incidence of preclinical hemochromatosis in the general population. 44
Hazard assessment
Direct contact with FeCl3 involves its immediate corrosive properties together with the danger of systemic Fe poisoning. 51 Acute intoxication after ingestion of excessive amounts of soluble inorganic Fe (be it as tablets, syrup or elixirs) presents in several distinct phases: tissue corrosion at the site of initial contact, vomiting, hematemesis and diarrhea followed either by recovery (with delayed pyloric stenosis) or (depending on the dose) by profound metabolic acidosis, shock, pneumonitis and death. 43 Because of the immediate pain and pharyngeal spasms associated with its prompt dissociation to free HCl, 14 the quantity of FeCl3 consumed is limited except in accidents 51 or among highly motivated individuals. 50 –52 Ingestion of concentrated aqueous FeCl3 caused rapid corrosion in the mouth and throat, glottic edema, esophagitis, gastrointestinal injury, metabolic acidosis, coagulopathy, progressive dyspnea and shock. Based on a single oral lethal dose of 230 mg/kg, 51 FeCl3 is considered “very toxic” and the probable lethal dose for a 70-kg person is between 1 teaspoon and 1 ounce. 43 Just as with HCl, 43 ,66,67 it is the normality of the FeCl3 solution and the rate of contact that dictates outcome as evidenced by the fact rats survived 13 weeks of daily FeCl3 doses in drinking water 68 at upward of 1,230 mg/kg day, 65 yet a single oral bolus in rats of 28 mg/kg was lethal. 59
Cancer characterization
There are no data to suggest that ingestion of either inorganic Fe salts or the chloride ion presents an increased carcinogenic risk. Chronic studies conducted in rats with FeCl3 in drinking water failed to show increased tumor incidence at any site. 68
Susceptible populations
Several groups have been identified as inclined to chronic Fe accumulation, but the major problem for most otherwise healthy people is nutritional deficiency. It is not known whether excessive Fe in the diet or drinking water could increase the incidence of preclinical hemochromatosis in the general population. 44
Idiopathic hemochromatosis is a condition that affects ~1 in 10,000 people, and it is characterized by chronic slow Fe accumulation in peripheral tissues. Males are more often affected than females and clinical signs are usually delayed until 40 to 60 years of age; metabolic defects in mucosal and reticuloendothelial regulation of Fe stores are thought to be responsible for the abnormal Fe accumulation. 44 People with congenital atransferrinemia develop excess Fe deposits in all organs except the bone marrow; others with congenital errors in iron metabolism accumulate Fe in the liver. 41 ,72 The World Health Organization (WHO) 44 identified those with metabolic defects in regulation of Fe absorption as at risk for Fe accumulation and earlier onset of symptoms. People homozygous for β-thalassemia major (a genetic condition found in 1 of 300 people of Italian, Greek, Middle Eastern or Asian ancestry that is expressed in impaired hemoglobin synthesis and anemia) also have increased Fe uptake to the point of overload and they experience an increased risk of cardiac disease. 73 –75
Exposure assessment
Iron is an essential element and the bulk of one’s daily requirement is met by the Fe present in foods and dietary supplements. The daily Fe dose used in the treatment of dietary deficiency is 200 mg/day or ~3 mg/kg day for an adult woman, and pregnant women and infants require 5-6 mg/day. 41 ,42
Do not consume level derivation
Key study and critical effect
While the total FeCl3 dose is not known in most cases of accidental or suicidal poisoning, a sufficiently high bolus dose (> 200 mg/kg) of ingested FeCl3 has been responsible for overt intoxication and fatalities. 50 ,51 Some of the rodent acute oral LD50 values 16,60 are consistent with the human lethal dose, but the overall range of reported lethal dose values in rodents is uncharacteristically large and no experimental details for any of the values are available that might provide reasons for the marked variability.
Due to the absence of details from the information in humans, health risk assessment of acute oral exposure to FeCl3 must rely on toxicity data collected in laboratory animals. As with other mineral acids, it is the rate of FeCl3 contact and the normality of the solution that dictates the outcome of exposure. Rats survived daily oral doses of FeCl3•6 H2O at up to 336 mg/kg day drinking water without effects other than the facts the esophageal mucosa was stained brown in those given ≥0.25% FeCl3 and the lamina propria of the large intestine was stained brown in those given 2.0% FeCl3. The hepatic Kupffer cells, the cartilage of the trachea and bronchii, the tongue, forestomach, the mucosa of the small intestine, the renal tubular spithelium and the white pulp of the spleen all accumulated brown stains. Rats given FeCl3 in their drinking water at pH 2.19 responded by reducing their water consumption and they displayed dose-dependent reductions in body weight. Sato et al. 68 identified a rat subchronic maximum tolerated dose of 0.5% FeCl3•6 H2O in drinking water based on decrements in body weight gain. The NOAEL values from the subchronic study (227 and 314 mg/kg day for male and female rats, respectively) 65 were essentially the same as the 0.5% NOAEL (equivalent to 319 and 336 mg/kg day) from the chronic study. It is noteworthy that the NOAEL is based on reduced body weight as a consequence of avoidance of the acidic (pH 2) drinking water in contrast to hemosiderosis or signs of chronic Fe intoxication.
The key study used to determine the do not consume limit was the drinking water study in F344 rats. 68 The reasons for the reductions in cumulative mortality seen in the 2-year bioassay were not articulated by the authors, but reduced drinking water consumption was associated with reduced body weight gains. Although no food consumption data were reported, 68 it is well known that caloric restriction can reduce body weight, increase animal (including rodent) life span and decrease the incidence of spontaneous tumors. 76 –79 The most likely explanation for the reductions in body weight and cumulative mortality seen after chronic ingestion of FeCl3 68 rests not with supplemental Fe per se, but rather with the significant reductions in water consumption (likely a result of reduced palatability of pH 2 drinking water) together with concomitant caloric restriction. 80 ,81
Dose response
Clinical experience demonstrates that as the concentration of ingested FeCl3 increases, the consequences of exposure increase. Iron deficiency is a chronic problem and the daily nutritional requirements often cannot be met by the Fe content of the typical Western diet and FeCl3 has been added as a source of dietary Fe to fortify flour and milk powders. The minimum daily Fe requirements for newborns (10 mg), 0.5- to 3.0-year-old infants and young children (15 mg) are somewhat less than those for adolescent males and females (18 mg), but in pregnant and lactating women existing body stores and dietary sources are such that daily supplements of 30–60 mg (0.5–1 mg/kg day) are necessary. 44 The daily Fe supplement used to treat anemia is 3.0 mg/kg day, but 25% of those who consume that level experience nausea, epigastric pain, diarrhea and/or constipation. At 6.0 mg/kg day, 40% of patients complain of gastrointestinal distress 41 and ingestion of concentrated FeCl3 solutions has been held responsible for systemic intoxication and death. 50 –54
Comparing the published rat acute oral LD50 values (400–450 mg/kg) 60 ,61 to the doses tolerated by rats for their lifetimes (320–336 mg/kg day) 68 suggests that a large oral bolus of FeCl3 delivers a high localized concentration of HCl to the upper alimentary tract. The HCl liberated from FeCl3 in aqueous solution 14 is corrosive and upon reaching the pyloric sphincter and antrum, the acid can be entrapped causing full-thickness necrosis that can progress to perforation of the stomach wall. 43 No such damage was observed in rats given equivalent but divided doses of FeCl3 in their drinking water 68 ; therefore, it is the maximum concentration not the total dose of FeCl3 that most clearly accounts for different results with variable dosing protocols.
The acute oral lethal dose in humans of aqueous FeCl3 is on the order of 230 mg/kg. 51 The acute oral lethal FeCl3 dose in humans compared to the daily FeCl3 ingested dose tolerated by rats for their entire life (336 mg/kg day) might be taken to suggest a species difference. Although circulating Fe is increased after a massive oral FeCl3 bolus, in each case of clinical FeCl3 acute intoxication 50 –52 the victims vomited. The difference between the single oral lethal dose in rats (400–450 mg/kg) 60 ,61 and that in humans 51 may not be necessarily attributed to the absorbed dose of Fe per se, but to human tracheobronchial aspiration of ingested acid 14 ,53 and vomitus, asphyxia due to glottic and laryngeal edema compounded by corrosive esophagitis, hemorrhage and shock. The rodent oral LD50 values were generated using gastric intubation, and rats are physiologically incapable of emesis. Thus, it is the oral exposure regimens taken together with the physiological differences between rats and humans that account for the differences in the rodent and human lethal dose values. From these observations, it is evident that the rodent oral acute lethality data have little relation to repeated consumption of FeCl3 in drinking water.
Uncertainty factor selection criteria
The following uncertainty factors were applied to the FeCl3•6 H2O concentration (0.5%) in drinking water given to F344 rats and considered an NOAEL of 300 mg/kg day.
68
Interspecies extrapolation (3×). While empirical data specifically quantifying interspecies toxicokinetic differences after oral exposure to FeCl3 were not identified, no appreciable interspecies kinetic differences are anticipated as a result of brief (possibly once in a lifetime) exposure to elevated Fe in drinking water. An interspecies uncertainty factor of 3× was applied to account for potential toxicodynamic differences in response between humans and rats in FeCl3 ingestion. Intraspecies extrapolation (3×). Selection of an intraspecies (human variability) factor is based on the availability of data that identify unique or sensitive subpopulations. The toxicokinetic portion of the intraspecies uncertainty factor was considered unity since no appreciable kinetic-based differences are anticipated as a result of acute (possibly once in a lifetime) FeCl3 ingestion. The toxicodynamic portion of the intraspecies uncertainty factor was 3× since several genetic conditions exist that limit the ability to handle and eliminate ingested Fe.
44
,72
–75
Therefore, the total intraspecies uncertainty factor was 3×. LOAEL to NOAEL extrapolation (1×). Since the reduced body weight gain seen in the Sato et al.
68
lifetime drinking water study in rats was likely secondary to refusal of pH 2 drinking water rather than a consequence of systemic Fe intoxication, a LOAEL to NOAEL uncertainty factor of unity was applied.
The total uncertainty factor for acute ingestion of FeCl3 is, therefore, 10×.
Acute oral RfD derivation
An acute oral RfD for FeCl3•6 H2O based on NOAELs of ~300 mg/kg day in rats
68
can be derived as follows:
Adjusting this value by the molecular weight of FeCl3/FeCl3•6 H2O or 162.2/270.3 gives the equivalent as FeCl3:
Adjusting the acute RfD of 30 mg/kg-day by the atomic/molecular weight ratio of Fe/FeCl3•6 H2O (56/270.3) gives the equivalent as Fe:
Do not consume limit
The do not consume limit can be calculated by adjusting the acute oral RfD for the 10 kg weight of a child drinking 1 L/day of water.
Adjusted for the atomic or molecular ratios as for the acute oral RfD, this concentration is equivalent to:

At this concentration and assuming an adult drank 2 L of water/day from a single source, daily Fe contributed from affected water would be not more than 120 mg. The total daily Fe contribution at the do not consume limit is less than the average 200 mg daily Fe dose used to treat deficiency. 44 ,45
Do not use level derivation: dermal
Key study and critical effect
No standardized skin irritation studies with FeCl3 were located. The key study for derivation of a dermal do not use limit is that of Loewit et al. 69 in which no signs of general siderosis, Fe-dependent lesions, inflammation, or degeneration were seen in the vaginal epithelium of adult female rats or mice after 10–40 direct instillations of 0.15 or 0.3 M FeCl3 in saline (24.3 or 48.7 g/L FeCl3; 8.4 or 16.8 g/L Fe).
Dose response
Skin contact with concentrated FeCl3 solutions apparently contributed to acute systemic intoxication and death, 53 ,54 yet tinctures of FeCl3 have been used as a topical astringents and hemostatics with no evidence of systemic poisoning. 39 ,40 However, no controlled evaluation other than the rat and mouse studies with FeCl3 69 was available.
Uncertainty factor selection
The following uncertainty factors were applied to the NOAEL of 0.3 M (16.8 g/L) FeCl3 after repeated direct application to intact rat and mouse vaginal epithelium: Interspecies extrapolation (3×). Case reports have suggest that Fe uptake through damaged skin can contribute to acute poisoning by FeCl3
53
,54
, but there are no data to suggest changes in total body burden after topical application of FeCl3 tinctures or other products to intact skin. No data were located that evaluated controlled exposure of intact human or animals skin to FeCl3 in water and an interspecies uncertainty factor of 3× is warranted.” Intraspecies extrapolation (10×). Thresholds for mineral acid-induced digestion of the stratum corneum, the underlying epidermis and mucous membranes (etching) likely vary little between individuals. There are at least two reported cases of FeCl3 sensitization,
56
–58
and it is possible that topical Fe may be absorbed to a greater extent by those with eczematous or atopic dermatitis, psoriasis or other conditions that impair the normal barrier properties of intact skin. Those factors taken together with the fact thresholds for skin irritation can vary depending on individual complexion and other factors
82
warrant an intraspecies uncertainty factor of 10×. LOAEL to NOAEL Extrapolation (1×). As no adverse effects were observed in rodent vaginal epithelium after repeated application of 24.3 or 48.7 g/L FeCl3,
69
a LOAEL to NOAEL extrapolation factor was not warranted.
The total uncertainty factor assigned for acute FeCl3 topical contact is, therefore, 30×.
Acute dermal RfV calculation
An acute dermal RfV based on avoidance of skin and mucous membrane irritation can be due to the lack of local or systemic damage after aqueous FeCl3 was applied repeatedly to the vaginal epithelium of mice and rats.
69
Application of a 30-fold uncertainty factor to the highest NOAEL to derive an acute dermal RfV is as follows:
Do not use limit: ocular
Key study and critical effect
As with HCl and other mineral acids, FeCl3 can be irritating to the eyes and direct contact with concentrated solutions carries with it risk of serious ocular damage. The only study with FeCl3 examined a pH 1 solution in rabbit eyes and that study was carried out using intraocular injection. 64 ,65 Due to the fact that only one, highly corrosive concentration was studied and that intraocular administration may not be directly applicable to FeCl3 present in drinking water, a specific ocular do not use limit expressed as Fe was not derived.
The pH of FeCl3 in distilled water ranges from 2.6 at 1,000 mg/L to 2.8 at 500 mg/L (Table 2). Irrigation of rabbit eyes with pH 2.5 HCl produced marked corneal damage that began within 10 min 67 and solutions with pH less than 2.0 are corrosive on tissues. 83 ,84 The risk manager should note the pH of water at the do not consume limit (Table 2) is consistent with the pH of HCl that can cause corneal damage in rabbit eye after 10 min of direct contact. As complaints of ocular discomfort increase after contact with pH ≤ 4.5, 85 an ocular do not use concentration for FeCl3 in drinking water could be based on pH.
Other standards and guidelines
The basis for the oral RfD and the drinking water levels derived above can be compared to health advisory levels and regulations promulgated by federal and international agencies.
The U.S. EPA promulgated a secondary maximum contaminant level (SMCL) of 0.3 mg/L for Fe in drinking water, based on aesthetic effects including a rusty color in the water and reddish or orange staining of plumbing fixtures, as well as increased sediment and metallic taste [40 CFR 143.3].
The WHO 86 referenced a Joint FAO/WHO expert committee on food additives (JECFA) 87 provisional maximum tolerable daily intake (PMTDI) of 0.8 mg/kg iron from all sources except “iron oxides used as coloring agents, supplemental iron taken during pregnancy and lactation, and supplemental iron for specific clinical requirements.” Applying a 10% relative source contribution for drinking water to the PMTDI, the resulting value of ~2 mg/L based on a 60-kg adult drinking 2 L/day was considered to present no health hazard. 86
The National Research Council 88 established dietary Fe reference intakes. The daily adequate intake for infants 9–6 months of age is 0.27 mg/day. Recommended dietary allowances (RDAs) range from 7 mg Fe/day for children ages 1–3 years to 27 mg Fe/day during pregnancy. Assuming a 70-kg adult consumes 2 L of drinking water (from a single source) at the do not consume concentration (60 mg Fe/L) each day, the total dose would be 120 mg or 1.7 mg/kg day or ~50% of the average daily Fe dietary supplement used to treat nutritional iron deficiency. Assuming a 10-kg child consumes 1 L of drinking water at the do not consume level, the total dose would be 6 mg/kg day or approximately equal to the childhood and infant daily dose used to treat Fe deficiency and very near the 7–10 mg Fe/day RDA for children 1–8 years, respectively. 88
To control Fe concentrations in infant formula, the American Academy of Pediatricians Task Force on Infant Nutrition recommended a lowest concentration of 0.26 mg Fe/L and an upper limit of 4.35 mg Fe/L. 44 The WHO 44 established a provisional maximum tolerable intake for dietary Fe of 0.8 mg/kg day.
The U.S. EPA promulgated an SMCL of 250 mg/L for chloride in drinking water, based on a salty taste above that level [40 CFR 143.3]. The WHO 89 reviewed chloride in drinking water and, while noting that there is a detectable taste to water with chloride concentrations >250 mg/L, declined to set a health-based guideline because chloride toxicity has been observed only on very rare occasions in humans.
Footnotes
Authors’ Notes
Mention of trade names, proprietary products, or specific equipment does not constitute an endorsement, neither does it imply that other products may not be equally suitable. The drinking water do not consume/do not use limits derived here are based on examination of the toxicity of specific materials in mammalian systems. These limits are designed to protect the public from short-term adverse health effects associated with either ingestion or other direct contact (skin and eye) during bathing and showering. As such, beneficial uses of water intended for other than domestic use (e.g., firefighting, landscape or agricultural irrigation, sanitary or storm sewers, industrial process waters, hemodialysis) are not considered here and the do not use/do not consume limits should not be applied to exposure situations other than potable water or to species other than mammals (including livestock and other domestic animals).
Funding
This study was supported by the State of Pennsylvania Department of Environmental Protection. The authors are indebted to the volunteer members of the NSF Health Advisory Board for critical review of the draft document.
