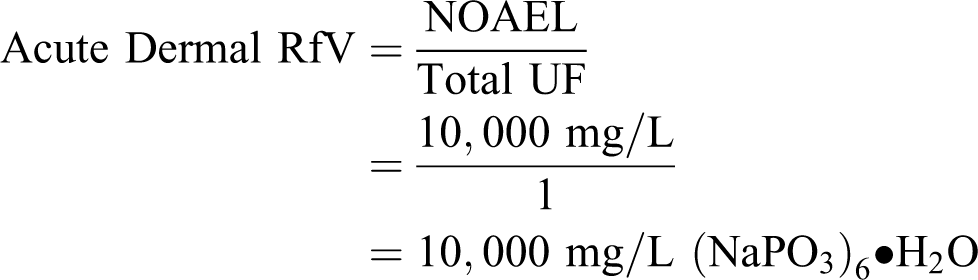Abstract
The U.S. Congress [PL 107–188] amended the Safe Drinking Water Act and required each community water system serving more than 3,000 people to conduct vulnerability assessments. These assessments address potential circumstances that could compromise the safety and reliability of municipal water. The present evaluation concerns the concentrations of the blended phosphates (also known as polyphosphates, condensed complex phosphates, polyphosphate glassy balls, and pyrophosphates) intended to aid regulatory agencies in decisions to avoid contact with affected water. Polyphosphates are direct food additives and they are used to treat municipal drinking water, but depending upon the concentration and duration of exposure these substances can induce chemical burns. Ingested polyphosphates are degraded by phosphatase enzymes to monophosphates, substances that are over-the-counter bowel purgatives. High oral doses of the monophosphates can induce transient hyperphosphatemia in older and susceptible young people, which can lead to acute phosphate nephropathy. In some patients, the condition is fatal. Based on the acute diarrhea after the ingestion of a single oral dose of monobasic (NaH2PO4) and dibasic (Na2HPO4) monophosphates in adults, a do not consume concentration of 600 mg PO4/L can be derived. Based on mild local irritation after topical application of 1.0% sodium metaphosphate [(NaPO3)6 • H2O] to intact skin of sensitive volunteers, a do not use concentration of 8,000 mg PO4/L can be assigned. Given the lack of eye irritation in rabbits after direct instillation of 0.2% (NaPO3)6 • H2O, an acute ocular contact limit of 50 mg PO4/L serves as the overall do not use level.
Keywords
Introduction
Title V of the Public Health Security and Bioterrorism Preparedness and Response Act (U.S. Public Law 107–188) mandated vulnerability assessments by large drinking water purveyors. These assessments identify procedures that can be brought to bear in the event of a terrorist or other intentional attack on municipal water systems. 1,2 Emergency response plans can be valuable, given the numbers and consequences of accidents involving gross chemical contamination of drinking water supplies. 3 –10
Blended phosphates (usually ortho and glassy polyphosphates) are used in municipal water treatment as part of scale control and corrosion control programs, as these compounds bind CaCO3, Fe, Mg and Mn. Orthophosphates, along with careful control of pH, are used to reduce Fe 11 , Pb 12 and Cu 13 concentrations in drinking water by reacting with dissolved metals to form a thin metal–phosphate coating on the inner surface of metallic pipe. The polyphosphates do not remove but sequester Fe and Mn. The polyphosphates reduce discoloration, reduce staining, reduce scaling and reduce complaints of objectionable tastes and odors present in drinking water. 14 Utilities that use phosphate inhibitors to treat water with very low alkalinity report average system lead reductions between 20% and 90% compared to those not using inhibitors. 15 Phosphate inhibitors also reduce coliform bacteria in cast iron water delivery systems by controlling corrosion; a survey of 31 North American drinking water systems found those that used phosphate inhibitors had lower coliform counts compared to systems that did not use phosphates. 16
The most likely emergency scenarios during which affected water reaches the public involve exposure for at least one but less than 30 days. 4,9 Nevertheless, planning for the first 24 h is important in preventing adverse health effects. After that time, constituent levels are generally reduced by dilution unless the chemical source persists. Therefore, the point of departure for deriving the oral do not consume limit is designated as the acute reference dose (RfD). The terms acute dermal reference value (acute dermal RfV) and acute ocular RfV will be used in the present assessment, and these values are considered as points of departure in establishing the do not use limit. The potential for acute inhalation exposure was considered in establishing the do not use limit, although inhalation is not likely to be the determining route of exposure for a nonvolatile compound.
The present evaluation of acute oral, dermal and ocular exposures to blended phosphate chemicals is intended to aid regulatory agencies in decisions to avoid or prohibit direct human contact with affected water. Carefully documented accounts of short-term (acute) human experience with reliable exposure data are the preferred key studies; but where those data are lacking, controlled studies in laboratory mammals were included. A lowest acute no-observed-adverse effect level (NOAEL), an acute lowest-observed-adverse-effect-level (LOAEL) and an acute benchmark dose (where possible) are values that could be used to calculate an acute RfD. To locate those values, the literature search strategy employed here was based on the Chemical Abstract Services Registry Number and at least one common name. The following public access data banks were searched: ChemID Plus Registry of Toxic Effects of Chemical Substances (RTECS) Hazardous Substances Data Bank (HSDB) GENE-TOX Environmental Mutagen Information Center (EMIC) Developmental and Reproductive Toxicology (DART) TOXLINE—Core and Special TRI (Toxics Release Inventory) Chemical Carcinogenesis Research Information System (CCRIS) Medline (via PubMed) Integrated Risk Information System (IRIS) Syracuse Research Corporation Online Toxic Substance Control Act Database (TSCATS)
Reports considered most valuable are detailed and published in the peer-reviewed open literature. Case reports of acute exposures usually describe symptoms and their consequences and those accounts were noted here, but nearly all case reports were limited by small numbers of patients and a near-universal absence of dose; these difficulties preclude use of case reports to establish short-term do not use and do not consume drinking water limits. Detailed reports from short-term clinical trials and summaries of (uncontrolled, but often extensive) clinical experience with short-term exposure durations are valuable. Due to the relatively few animal data for most corrosive materials (like blended phosphates), all published accounts (including reviews and summary reports) that could be located were compiled. Lethality data were collected, but derivation of public warning levels based on death was not considered health protective; likewise, reports concerning mild sensory irritation and adverse taste, odor or appearance of affected water were not candidate key data. Acute studies were preferred, yet repeat dose, subchronic, reproduction and chronic duration studies can provide information on target organs, and the results noted during initial exposure can be used to set or support derivation of protective short-term limits. Criteria to identify candidate key studies that document serious or irreversible adverse health consequences included relevant route of exposure, a reasonable number of subjects, concurrent controls (as available) and sufficient details to explain study design, execution and evaluation. Single-dose or short-term kinetic studies could inform interspecies dose scaling, but no controlled acute dose kinetic study in any species with the mixed phosphates considered here was identified. Data from standard short-term genotoxicity and long-term bioassays for potential carcinogenicity were reviewed and where reliable studies of not more than 30 days exposure by an appropriate route demonstrate a significant genotoxic carcinogenic response in animals, short-term theoretical excess cancer risk greater than 1:10,000 (1 × 10−4) could be used in calculating short-term exposure limits. As acute toxicity studies for most large commodity materials used in water treatment were conducted decades before the advent of standard operating procedures and adoption of defined regulatory protocols, adherence to those regulations played no part in identification of potential key studies; the limited animal and human toxicity data for the blended phosphates precluded rigorous consideration of statistical parameters. Given the relatively few published studies in animals that met all these criteria, consistent results from different sources were important in the identification of potential key and supporting studies. Since these phosphates are corrosive at high concentrations, the concentration-dependent relation to water pH was integral to identification of short-term do not use and do not consume levels.
The acute oral RfD was used to derive the drinking water do not consume limit, and the lower of the acute dermal RfV and the acute ocular RfV was used to derive the drinking water do not use limit. Following U.S. Environmental Protection Agency (EPA) convention when calculating a 1-day health advisory, the do not consume limit is defined as the acute RfD (in mg/kg day) multiplied by the 10 kg weight of a child assumed to drink 1 L of water per day. Due to the brief duration, a relative source contribution (RSC) was not applied.
Chemical and physical characteristics of the blended phosphates
Inorganic phosphates are compounds that contain four phosphorous–oxygen linkages arranged in a tetrahedron. Sharing oxygen atoms between tetrahedra, the interconnected PO4 groups can form chains, rings and branched polymers. 17 The principal phosphates used to control corrosion in water distribution systems have discrete monomeric PO4 3− ions, and these include monobasic sodium phosphate (NaH2PO4), dibasic sodium phosphate (Na2HPO4) and tribasic sodium phosphate (Na3PO4). Polyphosphates are the salts of polymerized H3PO4 and these form linear P-O-P chains. Molecularly dehydrated polyphosphates include sodium tripolyphosphate ([STP] Na5P3O10) and those with cyclic rings are known as metaphosphates (e.g., tri- and tetrametaphosphoric acids, Na hexametaphosphate (NaPO3)6 • H2O). Glassy Na metaphosphate is actually a mixture of polymeric metaphosphates. 18 The crystalline Na orthophosphates are available either as food or as NSF Standard 60-certified grades and the American Water Works Association adopted purity and performance standards for the anhydrous NaH2PO4 and Na2HPO4 (AWWA Standards B505-01 and B504-05).
Proprietary linear polyphosphates are also available 19 (e.g., sodium trimeta, sodium tetrameta, sodium tripoly and sodium hexametaphosphate), but in water these materials tend to revert to orthophosphates. 12 Depending on pH, hydrolytic degradation ultimately leads to the production of H3PO4 in equilibrium with H2PO4 −, HPO4 2− and PO4 3−. 17 Table 1 lists constituents found in blended commercial phosphates.
Chemicals in phosphate blends for water treatment.

Trisodium phosphate, tetrasodium pyrophosphate ([TSPP] Na4P2O7), the complex polyphosphates and tripolyphosphate are alkaline. The pH of a 1% aqueous solution 18 of Na2HPO4 in water is 9–10;a 1% aqueous solution 18 of the tripolyphosphate has a pH of 9.7–9.8 and the pH of its slurry is 10.5. 20 The pH of an aqueous 0.1 M (12,000 mg/L) solution of NaH2PO4 ranges between 4 and 5. 18,20
The phosphate forms dissolved in municipal water are not consistent and depend on pH, the buffering capacity of the water, the dissolved inorganic carbonate concentration and the temperature of the particular system. 15 The film that is formed on the inner surface of service line pipe is usually an orthophosphate. At low levels of dissolved carbonate (<1 mg/L), a water pH of 8 is most favorable for orthophosphate film deposition; increasing the phosphate concentration reduces the optimum pH for film formation. When used to control inorganic lead (Pb) concentrations, the orthophosphates complex to form hydroxypyromorphite [Pb5(PO4)3OH] and tertiary lead orthophosphate [Pb3(PO4)2]. In systems with hard water, phosphates complex with calcium to form octacalcium orthophosphate [Ca4H(PO4)3 • H2O] which converts slowly to Ca5(PO4)3OH and that compound precipitates on the pipe surface. 12
The properties of commercial mixed phosphates used in drinking water treatment are listed in Table 2.
To illustrate the influence of blended phosphate concentration on pH, measurements were collected in the NSF laboratories using three commercial blended phosphate preparations dissolved in distilled water at room temperature (Table 3).
Physical and chemical properties of blended phosphates.

Δ Kurrol’s salt: (KPO3)n; Graham’s salt (sodium polyphosphate): (NaPO3)50; M: Na; Ca n: 220, average 7.
a Blended phosphates range from simple small molecule diphosphates to long-chain polymers with molecular weights in the millions. 17 Sodium tripolyphosphate (Na5P3O10) = 367.86.
b Depending upon the particular chemical and physical form, phosphates are available as dry powders, granular or crystalline solids and food quality phosphates are available as liquid concentrates. The physical state of the amorphous condensed phosphoric acids (polyphosphoric acids) ranges from an oil (72%–82% P2O5), to a viscous gum (82%–90% P2O5) to a mixture of amorphous glass balls and crystals (90% P2O5). 17
The pH of commercial blended phosphate solutions.

The behavior of these phosphate products, including the short-chain polyphosphates (pyro-, tripoly-, tetrapoly) and the long-chain polyphosphates, in water is complex and the hydrolysis rates depend on the chemical structure, pH and temperature. Under appropriate conditions, all P-O-P linkages can be hydrolyzed giving rise to the monomeric PO4 3− ion. At room temperature, hydrolysis of the short-chain polyphosphates at neutral pH is slow but in the presence of phosphatase enzymes, hydrolysis is 106 times more rapid than when no phosphatase enzymes are present. The pH-dependent hydrolysis rates of the short-chain polyphosphates increase as the chain length increases to 10; longer chain polyphosphate hydrolysis is more complicated than that of the shorter chain phosphates in that long-chain polyphosphates can be cleaved to produce short-chain polyphosphates and the end-group scission yields cyclic metaphosphates. At pH 4–7, hydrolysis of the end of long-chain polyphosphates favors production of trimetaphosphates and at pH 7–11, the end-group splitting favors the production of orthophosphates. Degradation rates increase with increasing temperature and higher temperatures increased the proportion of trimetaphosphate to orthophosphate. 17
Organoleptic properties
Orthophosphates and the phosphate ion per se have no established taste or odor thresholds, but aqueous solutions of phosphoric acid are used to impart a tart or sour taste to foods and beverages (e.g., root beer and cola). Elevated orthophosphate concentrations in water increase turbidity and excessive polyphosphate concentrations not only increase turbidity but increase suspended solids and impart an off-color to the water. 23
Production and use
Simple phosphate salts are prepared by reacting phosphoric acid with a base (e.g., NaOH, CaO, Al(OH)3, Na2CO3) followed by crystallization, dewatering, drying in a rotary kiln and sizing. NaH2PO4 is crystallized after evaporation on steam-heated rolls then dehydrated in an oil or gas-fired rotary dryer. Disodium phosphate (Na2HPO4) is available as either anhydrous or the commercially important dihydrate (Na2HPO4 • 2H2O), and the latter is crystallized at 95°C then centrifuged and dried. Chlorinated trisodium phosphate [(Na3PO4 • 11 H2O)4 • NaOCl] is the second-largest volume phosphate salt and it is manufactured from the reaction of hot concentrated trisodium phosphate (Na3PO4) with sodium hypochlorite.
Pyrophosphates (simple linear condensed phosphates like Na2H2P2O7) are formed by condensation of two orthophosphates with the loss of one molecule of water. Some pyrophosphates are synthesized by neutralization of pyrophosphoric acid (H3P2O7) and others are made by thermal treatment (calcination) of the corresponding acid orthophosphate. Crystalline anhydrous disodium tetrapyrophosphate (NaH2P2O7) and anhydrous TSPP are made by thermal dehydration of the monosodium and disodium orthophosphates and are then crystallized from water.
Sodium salts of the tripolyphosphates are prepared by thermal dehydration of phosphate salts and calcination of NaH2PO4 and Na2HPO4. Condensed (long-chain) polyphosphates are produced by thermal dehydration of NaH2PO4. Graham’s salt (sodium polyphosphate), a water-soluble phosphate glass, is produced by heating NaH2PO4 to >620°C followed by rapid solidification to yield 90% higher molecular weight polymers and 10% mixed cyclic metaphosphates. Sodium Kurrol’s salt, (NaPO3)n, is prepared from crystallized Na2O/P2O5 at 580°C–590°C. 17
Because of its properties as a dispersant, coagulant and deflocculant, sodium metaphosphate (NaPO3)6 • H2O (also known as sodium polymetaphosphate or Graham’s salt) is the principal material used in water softeners and detergents and it is familiar with names like Micromet, Giltex, Quadrafos and Calgon. 18 Polyphosphates tend to function more efficiently in “softer” water conditions where they are used to sequester iron and manganese. The polyphosphates apparently revert to orthophosphates over time, 24 but the rate depends on structural characteristics, water temperature, bacterial count, solution characteristics of the water, and the length and duration of water travel in the distribution system. 12
The Na, K and Ca salts of various pyrophosphates are present as emulsifiers, texturing agents, sequestrants and buffers in a wide variety of foods and beverages. Food-grade NaH2PO4 is found in common baking powder. Related pyrophosphates are present in dry and cooked cereals, ice cream, nondairy creamer, canned and frozen vegetables, canned seafoods, pasta, frankfurters and other meat products. Na2HPO4 is found in detergents and buffers and it is used in tanning, dyes, enamel paints and to treat boiler water. Na3PO4 is used to remove boiler scale; it is found in detergents and water softeners and it is used to clarify sugar. 18 STP is a major commodity chemical and it is used in foods, pharmaceuticals and industrial processes; it is a “builder” in synthetic detergents (e.g., automatic dishwashing and laundry soap) and food-grade STP is used in curing bacon and ham. TSPP (Na pyrophosphate glass and Na pyrophosphate) is added to condensed milk, buttermilk, chocolate milk and instant pudding to control consistency. Sodium acid pyrophosphate (Na2H2P2O7) is used in leavening dough (particularly for donuts) and it reduces darkening of cut potatoes since it chelates iron (forming a colorless Fe pyrophosphate) and retards formation of Fe-tannin complexes. 17 The phosphate concentrations added vary with food type; for example, when used as an emulsifying agent in pasteurized processed cheese [21 CFR 133.169(7)(c)] and cheese spread [21 CFR 133.179(7)(e)] the total mixed polyphosphate concentration shall not exceed 3% by weight.
These materials are also present in common consumer products including mouthwash and as tartar control agents in toothpaste and dental floss. A case in point is the Na hexametaphosphate present in bath oils, salts and tablets, foundation makeup, eyeliner, eye makeup remover, lipstick, skin care preparations and face powder. 25
Effects on humans
Ingestion
Phosphorous (phosphate ion) is an essential nutrient. The dietary reference intake for adults of 19 years and older (including pregnant and lactating women) is at least 700 mg/day. 26 Phosphorous is normally present in plasma, intracellular fluid, cell membranes and collagen, but most of the body’s phosphate is in the skeleton where it is critical to the normal activity of osteoclasts and osteoblasts controlled by parathyroid hormone (PTH). 27 At physiologic pH (7.4), extracellular phosphate is present primarily as the Na2HPO4 and NaH2PO4 salts (4:1). Once absorbed, phosphate combines with calcium to form CaHPO4 in bones and teeth and the P content in hydroxyapatite (Ca10(OH)2(PO4)6) contributes to hardening and structural rigidity. Phosphorous is key in energy transfer reactions (e.g., ATP), nerve transmission, muscle contraction and maintenance of intracellular pH. Phosphate in extracellular fluid is primarily inorganic and the plasma concentration is inversely related to the actions of vitamin D and the rates of renal hydroxylation of 25-hydroxycholecalciferol. Phosphate is a major factor that affects tissue calcium concentrations and renal elimination of hydrogen ions. PTH and calcitonin increase phosphate elimination by blocking renal tubular reabsorption and vitamin D3 and its metabolites stimulate tubular reabsorption. 27
Free orthophosphate is the primary form by which dietary inorganic P is absorbed. Phosphate ion uptake from the gut is such that in cases where very large amounts are ingested, most is simply eliminated in the feces. Ingestion of inorganic phosphates (2-4 g) acts as a weak saline cathartic. 28 Following ingestion of lower (but elevated) doses results in most of the absorbed phosphate being eliminated promptly in the urine. Ingestion of excessive doses of phosphate salts can cause hyperphosphatemia 29 –31 , along with reductions in circulating Ca and precipitation of calcium phosphate in soft tissues including the kidney and lung where it induces metastatic calcification in the alveolar lining. 32 Ingestion of 37.6 g of NaH2PO4 and 8.6 g of Na2HPO4 as a single-dose bowel purgative can cause transient hyperphosphatemia, acute phosphate nephropathy with proteinuria and calcium phosphate deposition in the distal tubules and collecting ducts. 33 The severe hyperphosphatemia and hypocalcemia seen within a few hours to days after sodium phosphate ingestion can be followed in susceptible people by multifocal calcium phosphate deposition in the kidney, diffuse tubulointerstitial injury and acute renal failure 34 ; in some patients these changes are fatal. 35 The prognosis among survivors is such that the risk of chronic renal insufficiency is high (reaching values of 100%) with progression to end-stage renal disease in ~20% of the affected individuals. 33 The Ca-P product (CPP) is regarded as a reliable indicator of risk of calcium–phosphate precipitation in the distal tubules; the normal CPP range is 21–45.9, but after ingestion of NaHPO4 and Na2HPO4 solutions for intestinal catharsis, the mean CPP is increased to 71.28—with the highest CPP values in older patients. 36
Ingestion of a large dose of inorganic phosphate can influence serum PTH. Ittner et al. 37 found reduced PTH after oral administration of NaH2PO4 (1,500 mg P or 4,600 mg PO4 equivalent to 77 mg PO4/kg for a 60-kg woman) in some osteoporotic patients. Silverberg et al. 38 observed a 26% increase in serum P, a 50% increase in serum PTH and a 4% decline in serum Ca among eight healthy women and five men (19–36 years) given 2,000 mg oral PO4 (29 or 33 mg P/kg based on 70 or 60 kg body weight, respectively) as NaH2PO4 and KH2PO4 daily for 5 days; Silverberg et al. 38 considered those PTH values within the normal range.
Dermal and other routes
When present at dilute solutions in water, sodium hexametaphosphate [(NaPO3)6 • H2O] is alkaline and direct contact can be irritating. 39 Based on unpublished data generated by the Dutch Contact Dermatitis Group tabulated by deGroot 40 and referenced in Lanigan, 25 application of 1% (NaPO3)6 • H2O to intact skin of 1–20 people with suspected or verified contact allergy to cosmetic products failed to elicit any signs of local irritation. The 1% concentration was the highest recommended for dermal testing. 40 Based on a mean irritation score of 1.8 out of 4.0, a primary irritation index of 0.9 (“slight”) and tissue vesiculation/destruction at abraded but not intact skin of one of six volunteers, a 50% solution of STP was judged as having negligible irritant potential. 41
Effects on laboratory animals
The toxicology and metabolic control of mono-, di- and tribasic sodium and calcium phosphates, sodium polyphosphate (Graham’s salt), sodium hexametaphosphate, sodium potassium polyphosphate (Tammann’s salt), potassium pyrophosphate and the ammonium, calcium and potassium pyrophosphates have been reviewed. 20,25,28,42 –45
Acute exposure
Oral
Acute oral LD50 values of the monophosphates, diphosphates and the tri- and tertapolyphosphates in mice, rats, hamsters and rabbits range from >100 to more than 4,000 mg/kg (tabulated in World Health Organization [WHO] 28 and Weiner et al. 46 ). For example, the acute oral LD50 of NaH2PO4 in guinea pigs was >2,000 mg/kg, 47 that of technical grade STP [Na5P3O10] in rats was 6,500 mg (4,030–10,480 mg)/kg 48 and that for blended polyphosphates in rats was 10,000 mg/kg. 21
Dermal
In unpublished reports from Stauffer Chemical Company reviewed in Lanigan 25 direct application of 0.2% aqueous sodium metaphosphate under occlusion to abraded rabbit skin for 24 h was “mildly irritating,” whereas similar treatment with concentrated sodium hexametaphosphate caused severe skin irritation. Skin contact with powdered sodium hexametaphosphate was considered a “mild” irritant (reviewed in Lanigan 25 ).
Direct contact with neat NaH2PO4 was not irritating to rabbit skin after 24 h, but mild-to-moderate irritation was observed with Na2HPO4. No to slight local irritation was seen after topical application of trisodium phosphate and 15%–19% solutions of trisodium phosphate were slightly irritating when applied to abraded rabbit skin. 46 Similar studies 44 –46 with STP found little or no evidence for skin irritation. STP and other condensed polyphosphates were nonirritating to moderately irritating, but concentrated polyphosphoric acid (116%) was corrosive on rabbit skin.
Ocular
Direct instillation of a concentrated (unstated) solution of aqueous sodium hexametaphosphate into the eyes of New Zealand rabbits induced severe and irreparable ocular damage; identical treatment with 0.2% was said to be “nonirritating” in rabbit eye (reviewed in Lanigan 25 ).
When placed into rabbit eyes, the solid NaH2PO4 and Na2HPO4 caused minimal ocular irritation. 46 Four studies with STP concluded there was no or little evidence for ocular irritation, 44,45 but solid trisodium phosphate was considered slightly-to-moderately irritating and a 15% aqueous solution of trisodium phosphate was a mild ocular irritant. 46 Studies with solid condensed phosphates, including Na2H2P2O7 or STP, found that marked irritation was induced if the eyes were not washed within 24 h after initial contact compared to the minimal-to-mild irritation seen when the eyes were rinsed. 46 A 10% solution of TSPP was irritating to rabbit eyes even when the eyes were rinsed 24 h after exposure (reviewed by Weiner et al.). 46
Repeat dose studies
Feeding elevated dietary phosphate to rats can lead to nephrocalcinosis, 49 but the response depends on dose, frequency and duration of exposure. Weanling rats (5/group) were fed 0% or 5.0% Na2HPO4 in the diet for 1 month. 50 Calcium was added to each diet to ensure a “reasonably balanced” ratio of calcium to phosphorus. Although the body or kidney weights were not affected, renal tubular necrosis was seen in the treated rats. Assuming a 0.35-kg rat consumes 18 g food/day, the 5% level was equivalent to 2,571 mg/kg day. In a similar study carried out in the same laboratory, weanling male rats (5/group) were fed 0%, 0.2%, 2.0% or 10.0% sodium hexametaphosphate, STP or sodium trimetaphosphate in the diet for 1 month. The highest level of all phosphates reduced body weight, increased relative kidney weights and induced renal tubular necrosis. Some rats fed 2% exhibited acute renal pelvic inflammation or tubular lesions. The dietary NOEL of 0.2% for all three phosphates was equivalent to 103 mg/kg day, assuming a 0.35-kg rat consumes 18 g food/day.
Twenty rats per sex and dose were fed diets containing 0%, 1.0%, 2.5% or 5.0% Na2H2P2O7 or 5.0% Na2HPO4 for 100 days. 51 The basal diet contained 0.6% calcium and 0.5% phosphorus. All of the treated groups developed renal histopathology, decreased renal function and increased kidney weights except among those fed 1% Na2H2P2O7. Assuming these rats consumed 18 g food/day, the LOELs for 1% of Na2H2P2O7 and 5% of Na2HPO4 were 450 and 2,571 mg/kg day, respectively.
In 1989, Ritskes-Hoitinga and associates 52 fed diets with either 0.4% or 0.6% total P as NaH2PO4 ad libitum to juvenile female Wistar (RIV:TOX) rats for 28 days. Rats fed the higher P dose had twice the P in urine as those fed the lower dose; renal weights were increased significantly (25%) as was the incidence of nephrocalcinosis (as evidenced by mineral deposits at the corticomedullry junction) in rats consuming the higher P feed. In the more severely affected rats, interstitial fibrosis and signs of focal epithelial regeneration in the renal tubules were seen. Feeding the higher P diet reduced plasma Ca and increased kidney Ca concentrations.
Similar results were seen after feeding diets high (1.5%) in P (be it as monophosphate or tripolyphosphate Na or K salts) to male rats for as short as 13 days; the high P dose was associated with increased urinary albumin. 53 Feeding high P diets (as NaH2PO4, KH2PO4, Na5P3O10 or K5P3O10) for 21 days to rats increased kidney Ca and P concentrations, produced nephrocalcinosis and reduced renal function. The severity of nephrocalcinosis and the concentrations of urinary albumin were greater in rats fed the tripolyphosphates than in rats fed the monophosphates. 54,55
Male rats fed 8% Na orthophosphate in their diet for 7 months (or until the animal died) developed nephrocalcinosis and degenerative changes in the parathyroids and bone. 49 Rats fed 10% sodium trimetaphosphate for 1 month developed nephrocalcinosis and transient tubular necrosis; rats fed 10% sodium metaphosphate experienced reduced body weights and rats fed 10% sodium hexametaphosphate developed pale and “swollen” kidneys (reviewed in Lanigan 25 ). However, when adequate calcium (0.47%-0.56%) was incorporated into diets containing 0.43%-1.3% total P (as K2HPO4), which were fed to rats for 50–150 days, there was no indication of adverse gross or histologic changes in those animals. 50
The Joint Food and Agricultural Organization of the United Nations (FAO)/WHO Expert Committee on Food Additives 56 identified at least five dietary studies conducted in rats given total phosphate at 0.47% to 0.90% that failed to induce histologic evidence of renal damage. Weiner et al. 46 summarized feeding studies (28-100 days) with inorganic phosphates in rats, dogs and sheep and concluded that after high phosphate loads, excess phosphate increased bone demineralization and increased Ca release as part of a physiological regulatory mechanism. As a group, subchronic NOAELs for inorganic phosphates are greater or equal to 103 mg/kg day (based on the data for sodium triphophate, sodium trimetaphophate, and sodium hexametaphosphate). 46 Nonetheless, the WHO 56 noted the difficulty in determination of the dietary phosphate levels that fail to induce nephrocalcinosis in rats since areas of focal renal calcification were observed in the control animals and other factors—notably the Ca and vitamin D content and the acid–base balance of the laboratory chow—were important determinants of study outcome.
Phosphorous deficiency can increase its loss from maternal bone during pregnancy and the osteopenia and reduced mineral mass seen in the bones of premature infants is characterized by Ca and P deficiency, 57 but feeding monovalent or divalent inorganic phosphates failed to elicit signs of developmental toxicity in laboratory animals. 46 Dietary P deficiency reduced neonatal survival in rats 58 ; however, there were no adverse effects on mammalian reproduction after feeding monovalent inorganic phosphates. 46
Long-term studies
Chronic feeding studies with sodium metaphosphate, sodium trimetaphosphate and sodium hexametaphosphate found reduced body weight gain, calcium deposition in the kidney, bone decalcification, parathyroid hypertrophy and hyperplasia, focal hepatic necrosis and elevated phosphate in the urine. 25,46
The Joint FAO/WHO Expert Committee on Food Additives 56 stated, “A more important and more sensitive criterion for the deleterious action of phosphate overdosage is the appearance of metatastic calcification in soft tissues especially in the kidney, stomach and aorta. Kidney calcification may be observed in a few weeks or months, depending on the dose level.” The Committee 56 summarized the factors that influence the response to excessive dietary phosphates including total phosphorous content, the presence of calcium and other minerals and the degree of hydrolysis of polyphosphates to short-chain phosphates and orthophosphates. The Committee 56 reviewed the chronic studies in rats and concluded the data demonstrated a repeat dose no adverse effect level of 0.75% total phosphate (equivalent to 375 mg/kg-day).
Absorption, distribution, metabolism and elimination
Ingested diphosphates are converted to monophosphates and are excreted as such in the urine. 59 The higher polyphosphates are not absorbed intact to any appreciable extent from the gut but are hydrolyzed to orthophosphates 60 ; the larger the polyphosphate, the slower the hydrolysis. 61 The metaphosphates are first hydrolyzed to tripolyphosphate then to the corresponding orthophosphate (reviewed in Lanigan 25 ). After oral administration of either the hexametaphosphate 62 or sodium potassium polyphosphate, 63 there were no more than trace amounts of phosphate in the urine and blood. Calcium tetany occurred in at least one instance after ingestion of a phosphate detergent, a condition that was attributed to absorption of the intact polymers 20 ; following a very large oral bolus, sodium hexametaphosphate can apparently give rise to free H3PO4 that can induce metabolic acidosis. 39,60,64
After ingestion of the short polyphosphates (e.g., (KPO3)n, Na polyphosphate), a small amount of the parent compound was detected in urine, but up to 30% was recovered as the corresponding monophosphate. 65 After oral administration to rats, the tripolyphosphates were not absorbed from the gut intact and at least half of an administered (KPO3)n dose was eliminated intact in the feces. Bacterial hydrolysis in the intestine results in the slow liberation of the mono- and diphosphates. 66 The pyrophosphates are either absorbed from the gut intact or are slowly hydrolyzed (t ½ = 400 h) in stomach acid to the corresponding orthophosphate. 67
Phosphate uptake is mediated by the activity of sodium–phosphate (NaPi) cotransporters present in the enterocyte apical membrane and phosphate balance is maintained by coordinated action between the small intestine, skeleton, parathyroid gland and kidney. The kidney maintains serum phosphate in that phosphate filtered by the kidney is reabsorbed as a result of NaPi-2a activity. PTH decreases renal tubular phosphate transport; increased PTH leads to internalization of the renal proximal tubular cell NaPi cotransporters, reduces their activity and a higher proportion of tubular phosphate is eliminated. 43
Bone-derived fibroblast growth factor (FGF)-23 regulates vitamin D metabolism, regulates urinary phosphate elimination and maintains phosphate homeostasis via downregulation of NaPi-2a and NaPi-2c. Transgenic mice with excessive FGF-23 have increased urinary phosphate excretion and they develop hypophosphatemic rickets as a result of suppressed NaPi-2a and NaPi-2c just as in humans with autosomal hypophosphatemic rickets. Alternatively, missense mutations in FGF-23 lead to hyperphosphatemia. GALNT-3 (UDP-N-acetyl-α-D-galactosamine: polypeptide-N-acetyl-galactosaminyl transferase 3) is a protein that controls proteolytic processing of FGF-23 and GALNT-3 knockout mice have reduced O-glycosylation of FGF-23. As a result, functional levels of FGF-23 are reduced and the mice develop hyperphosphatemia. 43
Chronic kidney disease in humans is characterized by increased FGF-23 and as the condition becomes more severe, FGF-23 levels increase; despite the higher FGF-23, serum phosphate can continue to increase and can lead to compensatory hyperparathyroidism.
FGF-23 interaction with its receptors is facilitated by Klotho, a 550-amino-acid protein that controls FGF-23 receptor downstream signaling. Mouse Klotho is the product of the mouse Kl gene and it is expressed in the renal distal convoluted tubules, parathyroid gland, and choroid plexus. Mouse Klotho has 94% homology with rat and 80% homology with human Klotho. Mice with reduced or inactive Klotho develop hyperphosphatemia, exhibit signs of hypervitaminosis D, become infertile, develop generalized tissue atrophy and they die prematurely. 43
Hazard assessment
Phosphorous is an essential element. Depending upon the particular phosphate, dilute concentrations are used in baking, pharmaceuticals, foods, buffers and as direct additives to municipal drinking water systems. Free orthophosphate is the primary form by which ingested phosphate is absorbed. Ingestion of a large dose of the monophosphate salts produces prompt diarrhea and in some people, these doses induce a transient hyperphosphatemia that can lead to kidney damage of sufficient severity as to require permanent hemodialysis. The diphosphates, the triphosphates and the common small and large polyphosphates are degraded in the gut to the orthophosphates.
Direct eye or skin contact with neat or concentrated (alkaline) phosphates can cause local irritation and severe chemical burns (reviewed in Lanigan 25 ).
Cancer characterization
There are no data to suggest that common mono-, hexa-, pyro- or polyphosphate salts are genotoxic in standard short-term tests 28,46 or that ingestion of phosphorous, PO4 or metaphosphate salts presents an increased carcinogenic risk. 25
Susceptible populations
Ordinarily, the phosphate present in food is sufficient to meet metabolic requirements. However, genetic (X-linked familiar hypophosphatemia) and medical conditions (primary and secondary hyperparathyroidism) can result in low-normal to reduced circulating phosphate. Patients with marked hypophosphatemia may progress to osteitis fibrosa cystica and develop hypophosphatemic rickets. In these cases, 80% of the patients are diagnosed with adenoma of the parathyroid and 15% are diagnosed with hyperplastic parathyroid glands. 27 Thus, in some patients, it is a metabolic phosphate deficiency rather than phosphate excess that elicits medical complications.
Systemic poisoning with the salts of phosphoric acid is uncommon except in the elderly or in those with bowel obstructions, those with renal insufficiency or those who have cardiovascular disease. 68 People with autosomal mutations in FGF-23 and GALNT-3 (familial tumor calcinosis) develop hyperphosphatemia and ectopic calcification of the extensor surfaces of the large joints due to impaired phosphate handling by the kidney. 43
Acute renal failure and death can follow ingestion 31 –35,69,70 or rectal administration 71 –73 of sufficient quantities of phosphate salts, and people aged 70 years or more develop higher circulating phosphate concentrations after a single large bolus than do younger people. 33,36 Both children 74 and older people 33,36 are particularly susceptible to phosphate-induced nephrocalcinosis as are those with preexisting cardiac or renal conditions, advanced liver disease, ascites or bowel obstruction. Risk of impaired kidney function after consumption of phosphate-based laxatives is increased among those with diabetes mellitus or treatment with angiotensin-converting enzyme inhibitors (ACE-Is), angiotensin receptor blockers (ARBs) and nonsteroidal anti-inflammatories. 75 Acute nephropathy after consuming phosphate-based cathartics occurs in fewer than 1 in 1,000 patients and patients with smaller body weights are at increased risk. 75
The hazards of direct skin or eye contact with ortho- or polyphosphates are expected to be similar between genders, across all age groups and ethnic backgrounds. No group was identified that is uniquely sensitive to the irritant or corrosive actions of direct topical or ocular contact with ortho- or polyphosphates.
Exposure assessment
Phosphorous is an essential element and the daily requirement is at least 700 mg/day and for most people, the phosphate content of a regular diet provides all of the P required. 26 Dairy, meat and beans are good sources and cow’s milk contains 1 mg of elemental P/liter. 66 Phosphates and phosphoric acid are general purpose additives in foods, drugs and related products for humans [21 CFR 182.1073] and animals [21 CFR 582.1073], and these are Generally Recognized as Safe (GRAS) provided they are used in accord with good manufacturing or feeding practices. No published exposure estimates for the ortho- or polyphosphates were located, but the U.S. Food and Drug Administration ([FDA] summarized in Weiner et al. 46 ) reported that the average per capita daily P intake due to inorganic phosphates added to food was approximately 300 mg.
Do not consume level derivation
Key study and critical effect
The similar chemistry and toxicity of inorganic phosphates allow data from one compound to represent related compounds in the same class. 46 The robust toxicological profile shows clearly that inorganic phosphates within a particular class exhibit similar toxicities with similar magnitudes. Further, the larger orthophosphates and the short- and long-chain polyphosphates are absorbed intact only to a very limited extent, if at all, and the larger molecules are hydrolyzed by phosphatases present in the gut to the corresponding highly bioavailable and pharmacologically active monophosphates. Thus, the toxicity of the monphosphate metabolites can be utilized to derive an acute oral RfD for the parent ortho- and polyphosphates.
Aqueous solutions of NaH2PO4 and Na2HPO4 are used as cathartics in preparation of the large bowel for endoscopy. 42 Intentional evacuation of the large bowel is brought on by ingestion of a single dose of a prescribed standard bolus of sodium phosphate solution. This cathartic regimen is a reliable method for prompt “bowel cleansing prior to medical procedures or relief of occasional constipation” (Package Insert OSPS; Phospho-soda, CB Fleet, Lynchburg, Virginia, USA) and it will “produce a semifluid or watery evacuation in 3 to 6 h or less” 76,77 Hypertonic saline cathartics result in water being extracted from the circulation and being evacuated in the feces. An essential aspect of that regimen is continuous ingestion of copious quantities of water to prevent dehydration. Since the fraction of ingested phosphate that gains access to the systemic circulation is eliminated primarily in the urine, it is important to maintain sufficient hydration so as to not concentrate the filtrate and precipitate calcium phosphate in renal tubules. Saline cathartics are contraindicated in people with kidney disease and if the healthy patient is noncompliant with instructions on proper hydration, renal phosphates will accumulate and these can lead to acute phosphate nephropathy. This is precisely what occurred among the 21 patients (from a total biopsy survey of 7,349 individuals or 0.29%) described by Markowitz et al. 33 Nephrocalcinosis with abundant distal tubular calcium phosphate deposits was confirmed upon renal biopsy of these patients and this condition developed after a single oral dose of 37.6 g NaH2PO4 and 8.6 g Na2HPO4. Among these 21 people, there were 17 men and 4 women (mean age 64 years; range: 39–82 years); most were caucasian (81%) and the majority (16 or 76.2%) had a history of hypertension; 7 of those were on ACE-Is and 7 were on ARBs; 4 had diabetes, 4 had degenerative joint disease and 3 had coronary artery disease. Not even 1 of the 21 patients had a history of hypercalcemia or any medical condition associated with hypercalcemia (neoplastic disease, sarcoidosis, vitamin D intoxication, milk alkali syndrome). The onset of renal failure was rapid (≤2 weeks) for 8 (38%) and it was discovered somewhat later (≤4 weeks) in 12 (57%); overall, the median time to discovery of acute phosphate nephropathy after colonscopy was 4 weeks. Four of the affected people went on to develop end-stage renal disease that required dialysis, and one of those underwent renal transplant. Follow-up at a mean of 16.7 months found 17 whose renal function had improved, but renal function never returned to baseline among any of the affected individuals. The ACE-I and ARB drugs are associated with diuresis, a condition that with dehydration can increase susceptibility to phosphate-induced complications and increase the risk of nephrocalcinosis. 42,77
Dose response
The toxicity of ingested orthophosphates depends upon both the phosphate content and the acid/base properties of the specific material. 78 Phosphate dose–response relationships range from its minimum dietary requirement of 380 mg/day for children 1–3 years old to 1,055 mg/day for adolescents including during pregnancy 79 to ingestion of sufficiently concentrated H3PO4 that can cause death, 80 yet dilute H3PO4 has been used safely for decades to adjust the tart taste of carbonated beverages 81,82 and inorganic phosphates are common in foods.
Consumption of NaH2PO4 and Na2HPO4 at 2–4 g is a weak saline cathartic. 28,42 Dose–response relations cannot be identified from Markowitz et al. 33 due to the single-dose nature of the standard laxative regimen; however, an acute oral RfD can be derived from the single oral NaH2PO4/Na2HPO4 total phosphate dose (as HPO4) of 36 g (600 mg/kg day for a standard medium-frame woman) and that value can be considered an acute LOAEL for notable gastrointestinal distress 76 and induction of acute phosphate nephropathy among susceptible people. 33
Uncertainty factor selection criteria
The following uncertainty factors were applied to the 600 mg/kg day LOAEL for acute phosphate–induced diarrhea. 33 Nephrocalcinosis and renal failure occur in a subset of patients that receive oral phosphates as bowel purgatives and the onset of renal injury can be complicated by underlying cardiovascular disease and intervention with diuretic-like drugs, both of which increase susceptibility to phosphate-induced kidney damage. 42
Nevertheless, the customary high-dose standard phosphate laxative regimen is considered safe for well-hydrated patients with normal kidney function.
83
Interspecies extrapolation (1×). An interspecies uncertainty factor of 1× was appropriate as the key study was conducted in humans. Intraspecies extrapolation (3×). An intraspecies uncertainty factor of 3× was selected based on the fact that children and older adults are apparently prone to acute phosphate nephropathy.
33,74 In addition to underlying cardiovascular disease and treatment with diuretics, the predisposition of some older women to phosphate-induced kidney damage may be related to their smaller body mass.
33
People with autosomal mutations of the NaPi cotransporters and their intracellular processing (familial tumor calcinosis) are uniquely susceptible to hyperphosphatemia as a result of impaired renal phosphate handling.
43
Since the key study
33
included susceptible individuals and the do not consume level refers to an acute (possibly once-in-a-lifetime) drinking water exposure, a total uncertainty factor of 3× was assigned. LOAEL to NOAEL extrapolation (3×). A 3× adjustment from the LOAEL to an acute NOAEL can be made since diarrhea is minimally adverse and reversible. With respect to phosphate-induced renal failure associated with advanced age, gender and medical complications, those factors were addressed using a 3-fold intraspecies uncertainty factor.
Thus, the total uncertainty factor for acute ingestion of NaH2PO4 and Na2HPO4 as representative of the higher ortho- and polyphosphates is 10×.
Acute oral RfD derivation
Ingestion of the standard two 45-mL doses of NaH2PO4 and Na2HPO4 in preparation for bowel endoscopy 42 results in prompt evacuation of the large intestine, 76 abdominal cramping, nausea, vomiting, bloating, anal and perianal irritation along with dizziness, weakness, thirst, chest pains, chills, headache and fecal incontinence. 68 Electrolyte disturbances including hyperphosphatemia and hypocalcemia can be severe particularly in the elderly and in those with bowel disorders. 42 If left uncorrected, metabolic acidosis and Ca deposition in peripheral soft tissues can precipitate acute nephropathy that can cause death or result in permanent renal insufficiency among survivors. 33,35,69,70
An oral RfD can be based on the prompt diarrhea
76
and the potential for phosphate-induced nephropathy
33
after ingestion of 18.8 g NaH2PO4 and 4.3 g Na2HPO4 as two doses in 45-mL water (38 g NaH2PO4 and 8.6 g Na2HPO4). Given the molecular weights of anhydrous NaH2PO4 (119 g/mole) and anhydrous Na2HPO4 (142 g/mole) (PO4 equivalent to 87 and 67% of the total, respectively), the acute bolus HPO4 dose used as an over-the-counter purgative is 30.5 and 5.8 g, respectively. For a 60-kg woman the total oral HPO4 dose of 36 g or 600 mg/kg day can be identified as an acute LOAEL. Therefore, an acute RfD is derived according to the following:
By way of comparison, ingestion of NaH2PO4 and Na2HPO4 as a maintenance nutritional supplement (Neutra-Phos®) provides 765 mg PO4 (250 mg P) per 2½ fluid ounce serving—equivalent to 11 mg PO4/kg for the 70 kg adult. The usual dose range for an adult (75–600 mL) is taken in divided doses 4 times per day, a daily dose that provides 250 mg to 2 g total P. Under this regimen, the highest recommended daily total PO4 dose is 44 mg/kg day. The recommended daily dose for infants and children under 4 years of age is 60 mL (200 mg P), and it is consumed after meals or at bedtime and taken 4 times per day (800 mg P or 80 mg P/kg day for a 10-kg child). The total ingested PO4 as phosphorous is generally no greater than 250 mg/day (~ 4 mg/kg day) when consumed as a dietary supplement. At these dosages, the package insert instructions indicate “some individuals may experience a mild laxative effect for the first day or two.”
Do not consume limit
The do not consume limit is based on the oral RfD and a default 10 kg body weight and 1 L/day water consumption of a child. This procedure is consistent with methods used by U.S. EPA for calculation of 1-day health advisories.
The do not consume level for phosphate in drinking water derived above is rather similar to that present in cola drinks (432–521 mg PO4/L). 81,82
Do not use level derivation: dermal
There are very few published data upon which to base a direct skin contact value for the structurally diverse orthophosphates and polyphosphates. Other than the results with rabbits and humans treated with Na metaphosphate [(NaPO3)6 • H2O] described by Lanigan, 25 no published studies of controlled contact with polyphosphates were located.
Key study and critical effect
Based on the absence of irritation with patch testing of patients who had a history of allergic contact dermatitis (deGroot 40 as reviewed by Lanigan 25 ), a 1% (10,000 mg/L) concentration of (NaPO3)6 • H2O can be identified as a point-of-departure for calculation of a dermal do not use level. The pH of a 1% solution of (NaPO3)6 • H2O is 6.8 to 7.2. 25
Dose response
There are few published accounts upon which to assess dose–response relationships of direct skin contact with orthophosphates or polyphosphates. Unpublished studies referenced by deGroot 40 and cited by Lanigan 25 found that concentrations of sodium hexametaphosphate in water at up to 1% failed to elicit skin irritation in people with a history of skin contact allergy. Although concentrated sodium hexametaphosphate is a severe skin irritant in rabbits, a 0.2% solution “was only mildly irritating.” In light of the absence of adverse effects on the skin of patients with a history of allergic contact dermatitis with cosmetics, a 1% concentration (10,000 mg/L) of (NaPO3)6 • H2O can be used as a NOAEL to derive the acute dermal RfV.
Uncertainty factor selection
The following uncertainty factors were applied to the 1% (10,000 mg/L) (NaPO3)6 • H2O concentration causing no adverse effects on the intact skin of patients with a history of allergic contact dermatitis.
25
Interspecies extrapolation (1×). Qualitative data in humans are summarized in Lanigan
25
who examined direct skin contact with aqueous 1.0% (NaPO3)6 • H2O. As there was no evidence that a single contact produced any sign of local irritation, an uncertainty factor of unity was assigned. Intraspecies extrapolation (1×). Thresholds for irritant responses can vary between individuals, but the volunteers described by deGroot
40
(as cited in Lanigan
25
) had a history of suspected or verified allergic contact dermatitis associated with cosmetics. Since these subjects represent a potentially sensitive population, an intraspecies uncertainty factor of 1 was assigned. LOAEL to NOAEL extrapolation (1×). Because no signs of local skin irritation were seen after topical application of 1% (NaPO3)6 • H2O,
40
no adjustment from the NOAEL is necessary here.
Thus, the total uncertainty factor for acute skin contact with polyphosphates is 1×.
Acute dermal RfV calculation
An acute dermal RfV based on avoidance of skin irritation can be derived from results in sensitive volunteers who received a single topical application of 1% aqueous
Considering that (NaPO3)6 • H2O has a molecular weight of 630, 10,000 mg/L (NaPO3)6 • H2O is approximately equivalent to:
Do not use level: dermal
The acute dermal RfV can be used directly as the skin contact do not use level.

Do not use level derivation: ocular
There are very few published data upon which to base a direct eye contact NOAEL or LOAEL value for the structurally diverse orthophosphates and polyphosphates. Other than the results with rabbits and humans exposed to Na metaphosphate [(NaPO3)6 • H2O] described by Lanigan, 25 no published studies of controlled contact with the complex polyphosphates were located.
Key study and critical effect
Based on the observation of “mild” irritation in rabbit eye following direct instillation of a 0.2% solution of (NaPO3)6 • H2O, 2,000 mg/L can be identified as a point-of-departure for calculation of an ocular do not use level. Rabbits have been used for decades to evaluate acute responses in the eye and the results in rabbits are generally indicative of the potential for an irritant response in humans. 84
Dose response
The only dose–response relationships for ocular contact with the ortho- or polyphosphates were those described by Lanigan. 25 A 0.01% solution of sodium hexametaphosphate was said to be “injurious” to human eyes. Although concentrated sodium hexametaphosphate was a severe eye irritant in rabbits, a 0.2% solution was “mildly irritating.”
Uncertainty factor selection
The following uncertainty factors were applied to the 0.2% sodium metaphosphate minimal LOAEL in rabbit eye described by Lanigan.
25
Interspecies extrapolation (3×). Thresholds for corrosive response in the eye likely vary little between species and animal studies designed to measure potential corrosive and reactive chemicals in the eye are considered reasonably accurate indicators of the irritant potential in humans. Rabbits have been used for many years to evaluate acute responses in the eye and the results in rabbits are generally indicative of the potential for an irritant response in humans.
84,85
An interspecies kinetic uncertainty factor of 1 is appropriate since the end point represents a point-of-contact response that involves neither absorption nor metabolism; dynamic factors may contribute to species differences. Since the key study was conducted in rabbits, a total interspecies uncertainty factor of 3 was assigned. Intraspecies extrapolation (3×). Complex sodium phosphates have been used at 10%–24% in household and laundry detergents for decades
17,20
and consumer contact with these products is commonplace. When these products are splashed or otherwise enter the eye, transient stinging, tearing and hyperemia occur. While thresholds for corrosion in the eye likely vary little, the range of individual irritant response after ocular contact with complex phosphates is not known; therefore, a total intraspecies uncertainty factor of 3× was assigned to address the lack of empirical data that address the response in human eyes after acute phosphate exposure. LOAEL to NOAEL extrapolation (3×). Concentrations of 0.1% and 5% sodium hexametaphosphate were said to be injurious to human eyes (reviewed in Lanigan
25
). Because no or only possible slight irritation with no permanent damage was observed in rabbit eyes after a single direct instillation of 0.2% sodium hexametaphosphate and the response was consistent with the transient irritation experienced after a phosphate soap contacts the eye, these changes are considered minimal. Therefore, a 3× adjustment from a mild LOAEL to a NOAEL was made.
Thus, the total uncertainty factor for direct eye contact with polyphosphates is 30×.
Acute ocular RfV calculation
An acute ocular RfV based on avoidance of excessive eye irritation can be based on the report of little or no ocular irritation in rabbits after direct instillation of 0.2% (NaPO3)6 • H2O.
25
Since sodium hexametaphosphate is present in common dishwashing and laundry soaps and the response in rabbit eye was consistent with the hyperemia experienced after a dilute solution of common phosphate soap contacts the human eye, an ocular RfV can be derived as:
Considering that (NaPO3)6 • H2O has a molecular weight of 630, a concentration of 67 mg/L (NaPO3) • H2O is approximately equivalent to:
Do not use level: ocular
The acute ocular RfV can be used directly as the ocular do not use level.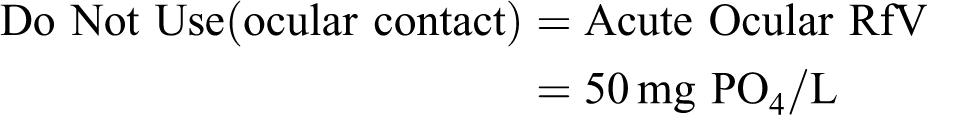
Since the ocular do not use level is lower than the topical do not use level, it serves as the basis of the overall do not use level.
Other standards and guidelines
The basis for the oral RfD and the drinking water levels derived above can be compared to health advisory values and regulations promulgated by federal and international regulatory agencies. The U.S. EPA has not established primary or secondary maximum contaminant levels for phosphates in drinking water. The U.S. EPA has not published a tap water health advisory or other guidance for phosphates.
The National Research Council 26 adopted a dietary reference intake of 380 mg P/day for children ages 1–3 years and 405 mg P/day for children ages 4–8 years. The highest daily required amounts were assigned to adolescent males and females and to pregnant and lactating women (1,055 mg P/day). An acceptable daily intake (ADI) of 63 mg P/kg-day proposed by Weiner et al. 46 was based on a dietary NOAEL of 5% for STP (equivalent to 57 mg P/kg-day) or sodium hexametaphosphate (equivalent to ~68 mg/kg-day); the composite uncertainty for interspecies and intraspecies extrapolation was 1× and the average of the two NOAEL values was used as the point of departure. The U.S. FDA [21 CFR Part 182] granted GRAS status to the orthophosphates and the polyphosphoric acids and to their calcium, potassium, sodium and ammonium salts.
In 1970, the Joint Food and Agricultural Organization of the United Nations and the WHO Committee on Food Additives 56 assigned an “unconditional acceptance” level for dietary phosphate of 0–30 mg/kg day and a “conditional acceptance” level of 30–70 mg/kg day. The distinction between the two levels depended on levels of dietary calcium. The unconditional acceptance value was regarded as suitable for communities with low calcium intake and the conditional acceptance value was applied for those with a high calcium level in a normal human diet. The daily phosphate dose (Neutra-Phos) used in long-term maintenance and treatment of chronic phosphate deficiency and hypophosphatemic rickets is 44 mg/day and phosphate-deficient humans tolerate chronic daily supplements of up to 14–28 mg PO4/kg day. 86
The Cosmetic Ingredient Review Panel 25 expressed concern over reports of severe dermal and ocular damage in rabbits after contact with concentrated sodium hexametaphosphate, but Lanigan 25 noted that dilute concentrations were “nonirritating to mildly irritating” and concluded that these ingredients could be used safely in cosmetics at the levels and under the conditions of normal use.
Conclusion
Consumption of 2 L of water at the do not use limit corresponds to ingestion of 390 mg P/day and that value is equivalent to 55% of the adult RDA or 30% of the phosphorous RDA for boys and girls aged 9–18 years. 26 Based on the long history with complex phosphates used as emulisifiers and texturizers in foods at concentrations up to 3% and their use in cosmetics at ≤0.1% to 50%, 25 taken together with the results of controlled studies conducted in human volunteers and laboratory animals, the short-term do not consume and do not use concentrations derived here can be considered protective of the public health.
Footnotes
Authors’ notes
Mention of trade names, proprietary products, or specific equipment does not constitute an endorsement, neither does it imply that other products may not be equally suitable. The drinking water do not consume/do not use limits derived here are based on the examination of the toxicity of specific materials in mammalian systems. These limits are designed to protect the public from short-term adverse health effects associated with either ingestion or other direct contact (skin and eye) during bathing and showering. As such, beneficial uses of water intended for other than domestic use (e.g., firefighting, landscape or agricultural irrigation, sanitary or storm sewers, industrial process waters, hemodialysis) are not considered here and the do not use/do not consume limits should not be applied to exposure situations other than potable water or to species other than mammals (including livestock and domestic animals).
Funding
This study was supported by the State of Pennsylvania Department of Environmental Protection. The authors are indebted to the volunteer members of the NSF Health Advisory Board for critical review of the draft document.