Abstract
Objective:
Toluene is used as an organic solvent, and it has neurotoxic effects. Omega-3 is an essential fatty acid required for brain development. The aim of this study was to investigate the protective effects of omega-3 fatty acid against toluene-induced neurotoxicity in prefrontal cortex of rats.
Materials and methods:
A total of 21 male Wistar rats were divided into three groups with seven rats in each group. Rats in group I were the controls. Toluene was intraperitoneally injected into the rats of group II with a dose of 0.5 ml/kg. Rats in group III received omega-3 fatty acid with a dose of 0.4 g/kg/day while exposed to toluene. After 14 days, all the rats were killed by decapitation. Enzymatic activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) and the level of malondialdehyde (MDA) were spectrophotometrically studied in the prefrontal cortex of rats.
Results:
Enzymatic activities of SOD and GSH-Px were decreased, and MDA levels were significantly increased in rats treated with toluene compared with the controls. However, the increased SOD and decreased GSH-Px enzymatic activities and MDA levels were detected in the rats administered with omega-3 fatty acid while exposed to toluene.
Conclusion:
The results of this experimental study indicate that omega-3 fatty acid treatment can prevent toluene-induced neuronal damage in the prefrontal cortex of rats.
Introduction
Toluene is an aromatic hydrocarbon used in manufacturing industries of various items such as adhesives, erasers, plastics, leather, dyes, thinners, and printing pastes. People working in these fields receive too much exposure to toluene. 1,2 Furthermore, toluene is easily available and a pleasant substance. However, toluene abuse and dependence is also very common. 3 –6 Concentration of toluene in blood may induce coma. The most basic effect that may be observed by exposure to toluene is depression of the central nerve system. Additionally, toluene may cause permanent damage of the brain tissues. 2 High concentrations of toluene may demonstrate certain effects such as damage in psychomotor activities, loss of upright posture reflex, and sedation. 3,7,8 Toluene can also cause psychiatric symptoms. These symptoms include motivation failure, emotional instability, delusions, hallucinations, anxiety, and personality disorders. 9 –11 The increase in free radicals or the decrease in antioxidant enzymes may cause oxidative damage in tissues. Enzymes such as superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) are recognized as antioxidant enzymes. Furthermore, malondialdehyde (MDA) level is an indicator of an oxidative damage. Studies have indicated that the activities of SOD and GSH-Px enzymes decreased and the MDA levels increased in tissues as a result of toluene exposure. This process was reported to cause an oxidative damage in tissues. 3
Omega-3 fatty acid plays an important role in the function of the brain and nervous system. It provides the development of the brain, early speech, good coordination, and freedom from learning disabilities in child. It has also been reported that it has neuroprotective effects in brain tissues. Omega-3 fatty acid may protect against heart attack and inflammatory diseases like arthritis, lupus, and asthma. Furthermore, it has anticonvulsant, antioxidant, and anti-inflammatory properties. 12–14 Current studies showed that omega-3 fatty acid increased the activities of some antioxidant enzymes and prevented oxidative damage in tissues. 15, 16 However, although the properties of omega-3 fatty acid is demonstrated in many studies, no studies in the literature related to the effect on histopathological and biochemical changes may have occurred in the prefrontal cortex by toluene. Therefore, in this study, we investigated the preventive effects of omega-3 fatty acid against damages that may have occurred in the prefrontal cortex due to toluene exposure.
Materials and methods
Animals and treatments
Adult male Wistar rats (200–250 g) were obtained from Experimental Research Centre of Gaziantep University (Gaziantep, Turkey). The animal studies were carried out according to the guidelines of European Community Council for experimental animal care. Animals were housed individually on a 12-h light:12-h dark cycle (lights on at 8.00 a.m.), at a temperature of 21 ± 1°C, and 50% humidity. Food (i.e. standard pellet diet) and tap water were supplied ad libitum. The animals were divided into three equal groups. Rats in group I (n = 7) were the controls. Group I was given vehicle (0.1 ml/10 g/day corn oil, intraperitoneally). Toluene was intraperitoneally injected to the rats of group II (n = 7) with a dose of 500 mg/kg/day (0.1 g/ml, in corn oil). 17 Rats in group III (n = 7) received omega-3 fatty acid via intragastric gavage (0.4 g/kg/day, Marincap capsule®, Koçak Farma, Istanbul, Turkey) while exposed to toluene (500 mg/kg/day, intraperitoneally). The fatty acid composition of the Marincap capsule (500 mg) comprises eicosapentaenoic acid (18%) and docosahexaenoic acid (12%). 18 After the 14-day experimental period, all rats were killed by decapitation. The brains of the rats were removed; prefrontal cortex tissues were obtained from all of the brain specimens. Some of the prefrontal cortex tissue specimens were washed twice with cold saline solution, placed into glass bottles, labeled, and stored frozen (−30°C) for the eventual determination of SOD, GSH-Px, and MDA production. The other prefrontal cortex tissue specimens were used for light microscopic and immunohistochemical evaluations.
Biochemical analyses
For biochemical analysis, the tissues were weighed and homogenized in four volumes of ice-cold Tris-HCl buffer (50 mM, pH 7.4) containing 0.50 ml/l Triton X-100 with a homogenizer (IKA Ultra-Turrax T 25 Basic) for 2 min at 13,000 r/min. All procedures were performed at +4°C. Tissue homogenates were centrifuged at 5000g for 60 min to remove debris, and the clear supernatant fluids were separated and kept at −40°C until the enzyme activity measurements were performed (about a week later).
Determination of SOD activity
Total (Cu-Zn and Mn) SOD (EC 1.15.1.1) activity was determined based on the method of Sun et al. 19 The principle of the method is based on the inhibition of nitro blue tetrazolium (NBT) reduction by the xanthine–xanthine oxidase system as a superoxide generator. Activity was assessed in the ethanol phase of the supernatant after 1 ml of an ethanol–chloroform mixture (5:3, v/v) was added to the same volume of sample and centrifuged. One unit of SOD was defined as the amount of enzyme causing 50% inhibition in the NBT reduction rate. The SOD activity was expressed as Units per gram of protein.
Determination of GSH-Px activity
GSH-Px (EC 1.6.4.2) activity was measured by the method of Paglia and Valentine. 20 The enzyme reaction in the tube containing NADPH, reduced glutathione (GSH), sodium azide, and glutathione reductase was initiated by the addition of hydrogen peroxide, and the change in absorbance at 340 nm was monitored by a spectrophotometer. Activity was expressed as Units per gram of protein.
Determination of MDA level
The tissue MDA level was determined using a method from Esterbauer and Cheeseman, 21 based on its reaction with thiobarbituric acid (TBA) at 90–100°C. In the TBA test reaction, MDA and TBA react to produce a pink pigment with an absorption maximum at 532 nm. The reaction was performed at pH 2–3 and at 90°C for 15 min. The sample was mixed with 2 volumes of cold 10% (w/v) trichloroacetic acid to precipitate the protein. The precipitate was centrifuged; an aliquot of the supernatant was reacted with an equal volume of 0.67% (w/v) TBA in a boiling water bath for 10 min. After cooling, the absorbance was read at 532 nm. Results were expressed as nanomoles per gram of wet tissue by reference to a standard curve prepared from the measurements made with a standard solution (1,1,3,3-tetramethoxypropane)
Histological examination
For light microscopic examinations, brain samples were fixed in 10% neutral buffered formalin. After dehydrating with graded alcohol series, tissues were then embedded in paraffin. Several 5-μm-thick transverse sections were obtained from brain tissue blocks and stained with hematoxylin and eosin for histological evaluation. Sections were examined and photographed with Olympus DP20 camera attached-Olympus CX41 photomicroscope for characteristic histological changes.
Statistical analysis
Data were analyzed using a commercially available statistics software package (SPSS for Windows v. 12.0, Chicago, Illinois, USA). Distribution of the groups was analyzed with one sample Kolmogrov-Smirnov test. All groups showed normal distribution, so that parametric statistical methods were used to analyze the data. One-way analysis of variance test was performed, and post hoc multiple comparisons were made using least-squares differences. Results are presented as mean ± SEM; p < 0.05 was regarded as statistically significant.
Results
Clinical findings
During the experimental study, hair loss was observed in rats on day 6 after toluene exposure. Hair loss was more excessive in group II animals when compared with animals in group III. Additionally, seizures that lasted more than half an hour occurred on day 7 after the rats were exposed to toluene. The duration of seizures continued until the end of the study in a progressive form. In addition to seizures, rats displayed aggressive behaviors.
Biochemical results
SOD, GSH-Px enzyme values, and MDA levels were determined in the prefrontal cortex of rats. Enzymatic activities of the SOD and GSH-Px were decreased, and MDA levels were significantly increased in rats treated with toluene compared with the controls (Table 1). However, the increased SOD and decreased GSH-Px enzymatic activities and decreased MDA levels were detected in the rats administered omega-3 fatty acid while exposed to toluene (Table 1).
The activities of SOD and GSH-Px enzymes and levels of MDA in prefrontal cortex in control, toluene, and toluene + omega-3 groupsa

SOD: superoxide dismutase; GSH-Px: glutathione peroxidase; MDA: malondialdehyde; NS: non significant.
aResults are presented as mean ± SEM.
Light microscopic results
The control group showed normal cortical morphology and neuron structure (Figure 1). In higher magnifications, neuropil appeared normal and homogen with intact vessels (Figure 2). Neuron bodies with nuclei (Figure 3) were normal.

Normal cortical morphology in control group.
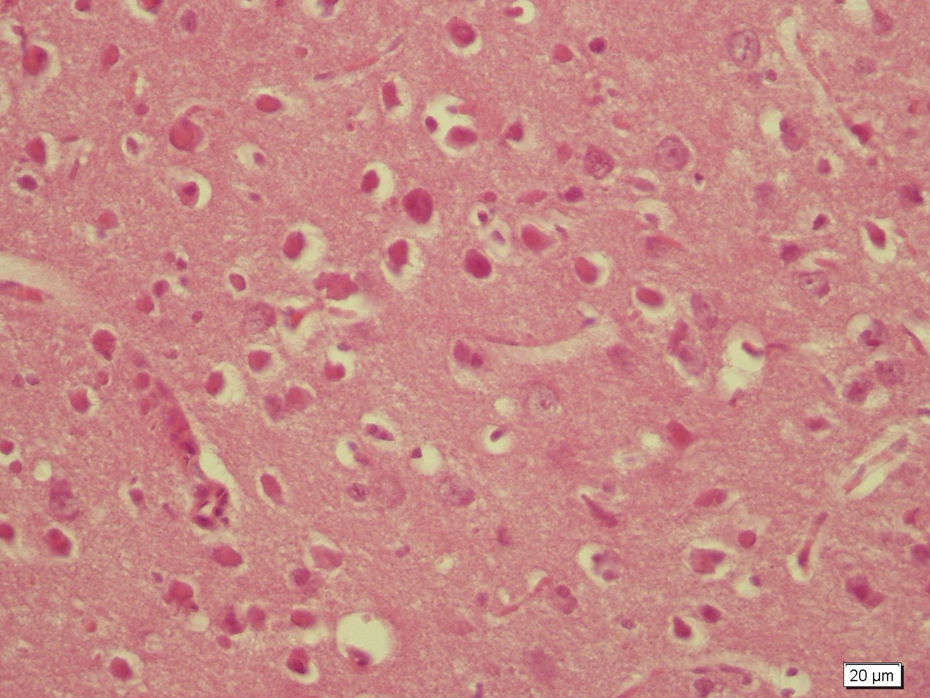
Homogen appearance of neuropil in control group.

Normal neuron bodies (→) with nuclei (*) in control group.
In the toluene group, detachments in piameter and hemorrhage were observed (Figure 4). Shrinkage in neuron bodies and degeneration (Figure 5) and vacuolization in neuropil (Figure 6) were noted.

Detachments in piameter (*) and hemorrhage (→) in toluene group.

Shrinkage in neuron bodies (→) and degeneration in toluene group.

Vacuolization in neuropil (→) in toluene group.
In toluene + omega-3 group, general structure improved (Figure 7) with no detachments in piameter and bleeding (Figure 8).

General structure improved in toluene + omega-3 group.

Piameter is normal; hemorrhage occurs with omega 3 treatment.
Discussion
In the present study, we investigated the toxic effects of toluene on the prefrontal cortex tissue. Additionally, we investigated the preventive effect of omega-3 fatty acid against the mentioned toxicity. Toluene is an organic solvent with neurotoxic effects. In some chronic cases, permanent damage in the central nerve system was observed due to toluene exposure. Acute exposure to toluene may induce headache, vertigo, coma, and even death. 2 Experimental studies demonstrated that toluene was responsible from disorders that may include pyramidal disorders, peripheral neuropathy, optic atrophy, neural audio loss, and impairment of cognitive functions. 3,7,22 A recent study showed that acute toluene exposure inhibited hippocampal neurogenesis in adult mice. 23 In another study, toluene exposure caused degeneration of myelins, axons, and endoneurial structures. There was deformation in some nerve fibers and vacuolization in the cytoplasm of nerve cells. 3 Kanter suggested that toluene inhalation resulted degenerative changes, shrunken cytoplasm, and extensively dark picnotic nuclei in neurons of the frontal cortex and brain stem tissues. 24 In our study, toluene exposure caused detachments in piameter, hemorrhage, degeneration, and shrinkage in neuron bodies, and vacuolization in neuropil in the prefrontal cortex. These findings are in agreement with the results of other studies on toluene-induced neurotoxicity.
In our study, toluene caused significant changes in brain tissue that were attenuated with omega-3 treatment. We observed general structure improved with no detachments in piameter and bleeding in the prefrontal cortex. Neuroprotective effects of omega-3 on brain tissue were previously shown in some studies. In a study by Zararsiz et al., 18 formaldehyde-induced injuries in prefrontal tissue were improved with omega-3 treatment. The cellular damage was also decreased in their study. In another study by Zararsiz et al., 16 omega-3 prevented formaldehyde-induced cortical damage. Moreover, Ozyurt et al. 25 conclude that omega-3 was found to be effective at reducing MK-801 (dizocilpine maleate)-induced neuronal death.
Oxidative stress can cause tissue damage. One of the parameters that can be used as an indicator of this damage is MDA. MDA is a product that occurs as a result of lipid peroxidation. 26,27 A study carried out by Coskun et al. on peripheral nerves indicated an increase in tissue MDA levels in the toluene group when compared with the control group. 3 In another study conducted on brain tissues, MDA levels in the group who received toluene containing thinner demonstrated an increase when compared with the control group. 8 Ozen et al. stated that the damage that occurred due to ischemia-reperfusion showed an improvement after administration of omega-3. In that study, the authors observed that MDA level of the prefrontal cortex tissue in rats included in the ischemia-reperfusion group showed an increase when compared with the rats in the control group. Omega-3 administration reduced the mentioned increase. 28 In another study related to formaldehyde toxicity and administration of omega-3, similar findings resembling the findings of Ozen et al. were obtained. 18 In the present study, MDA levels displayed an increase in the toluene group when compared with the control group. This finding suggests that as a result of lipid peroxidation, toluene has caused an oxidative damage in the prefrontal cortex. Furthermore, MDA levels were decreased in the group of omega-3 administration; it was assumed that omega-3 was capable to prevent the mentioned damage.
Both SOD and GSH-Px are enzymes related to oxidative damage. In a study carried out related to toluene exposure, SOD and GSH-Px enzymes were observed to decrease in the group of animals when compared with the animals in the control group. 3 Similar findings were also found in our study. These changes observed in enzymes may reflect the start of an oxidative damage. In a study related to neurotoxicity, it was determined that tissue SOD and GSH-Px enzyme values in the omega-3 group decreased when compared with the values observed in the toxic group. 25 In a study carried out by Zararsiz et al., it was emphasized that the same enzymes showed an increase in the omega-3 group when compared with the toxic group. 18 Another study conducted on the prefrontal cortex demonstrated a decrease in the SOD enzyme value in the received omega-3 group that had undergone ischemic-reperfusion group when compared with the ischemia group. 28 In the present study, SOD enzyme value demonstrated an increase in the toluene group when compared with the animals in the omega-3 group while GSH-Px enzyme value was decreased. Accordingly, we may declare that omega-3 acid may prevent the neurotoxicity caused by toluene.
Epidemiological studies have shown beneficial effects of fish consumption in the prevention of chronic diseases and specific cancers. Omega-3 fatty acid, the specific component found in fish, is responsible for this protective effect in Mediterranean populations. This protective effect is reduced in those who smoke and consume alcohol and in cardiovascular mortality. 29 Accordingly, we expect that toluene toxicity can be seen less in a community fed on fish. People who work at occupational exposure to toluene should intake omega-3 to decrease toluene toxicity. Toluene is also commonly abused (glue-sniffing). Recently, toluene abuse is an important public health problem. Omega-3 fatty acid should be applied to this people for therapy. Therefore, it has been suggested that all the people need to take omega-3 containing food for health.
In the present study, we suggested that omega-3 has neuroprotective and antioxidant properties. Furthermore, it has anticonvulsant effects. As our study is short-term, we did not use an anticonvulsant for seizure and aggressive behaviors in rats. Long-term study is needed for anticonvulsant therapy. Although, omega-3 has anticonvulsant properties, there is a literature that oral administration of omega-3 did not affect like an anticonvulsant. 30
In conclusion, the protective effect of omega-3 against toluene-induced brain injury was demonstrated in our study supported by biochemical and histological findings. However, further studies are needed to document its protective role in more detail.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
