Abstract
Effects of different storing conditions on paracetamol concentration in biological samples of acute intoxicated rats were investigated. The stability and distribution of paracetamol was observed in postmortem serum, liver, kidney and brain tissues. The serum samples were stored for 30 days and daily changes were evaluated for paracetamol. A significant difference (
Introduction
Paracetamol (acetaminophen) is the active ingredient in most commonly used analgesic and antipyretic drugs and also an active metabolite of phenacetin, which is called soft coal analgesic. Acetaminophen is also known as
In forensic cases which are subjected to federal law in the United States, it is mandatory to analyze and to store suitable biological samples collected from deaths caused by acute poisonings. 5,6 While samples can be stored at different conditions, there are several factors that could still change the concentration of drugs over time. For this reason, there are ongoing studies to determine the stability of various substances (such as ethanol, drugs, soporifics, pharmaceuticals, pesticides, etc.) in biological samples at different storing conditions. In current literature, identification and quantitation of paracetamol in various matrices were evaluated using several analytical methods and instrumentation. 7 –14
In this study, the stability of paracetamol in serum and liver of rats was investigated for forensic and clinical purposes. Furthermore, distribution of unchanged paracetamol was quantitated from livers, brains and kidneys of the same animals using gas chromatography/mass spectroscopy (GC/MS).
The aim of this study is to investigate the distribution and stability of paracetamol in biological samples using an animal model. Future studies involving human specimens taken for clinical and forensic purposes are necessary to evaluate the distribution and stability of paracetamol in acute poisoning cases.
Materials and methods
Chemicals and reference standards
All tablets including paracetamol of pharmaceutical quality were kindly supplied by the pharmacy. Paracetamol (99% purity) and procaine (99% purity, internal standard) were obtained from Sigma (Germany). Calibrators were prepared from 200 mg/ml of paracetamol in methanol while the procaine (internal standard) was prepared using the concentration of 40 mg/ml. Calibrators and standards were all stored at 4°C prior to analyses. All solvents used were of high pressure liquid chromatography (HPLC) grade and purchased from Merck (Germany). Potassium dihydrogen phosphate (KH2PO4) was obtained from Sigma (Germany). Oasis MCX (3 cc, 60 mg) and Oasis HLB (3 cc, 60 mg) cartridges used for sample preparation were purchased from Waters (Milford, Massachusetts).
Animals and drug administration
All experiments were performed in compliance with the guidelines and the laws governing animal studies in Europe.
15
Wistar Albino, 8-week-old male rats (weighing ~250 g), were purchased from Cukurova University Experimental Medical Research Center and nine-week-old rats were used in this study. Animals were maintained in a room at a temperature and relative humidity of 24°C ± 2°C and 40–70%, respectively, and were given food and water ad libitum. Animal groups consisting of six rats were orally (gavage) administered with paracetamol at LD50 level (2404 mg/kg) for every measurement days. The experimental animals were killed 4 h after paracetamol treatment because of its half-life. Subsequently, blood, liver, kidney and brain samples were collected from each animal and sectioned and stored until used for analysis. Blood samples (
Liver, kidney and brain samples (
Gas chromatography/mass spectrometry analysis (GC/MS)
The Hewlett-Packard GC-MS system HP 6890-5973 (Agilent, Waldbronn, Germany) equipped with an autosampler, splitless injector and a DB17-MS 15 m × 0.25 mm × 0.25 µm capillary were used for the analysis. Helium (purity ≤ 99.999%) was used as the carrier gas at the flow rate of 1 ml/min. The oven temperature was programmed to increase from the initial temperature of 80°C to 280°C at 10°C/min, and the final temperature was held for 9 min. The injection port temperature was 250°C. Sample was injected in the pulsed splitless mode with an injection temperature of 250°C. The temperatures of the quadruple, ion source and mass selective detector interface were 150°C, 280°C and 280°C, respectively. Electron impact (EI) mode (70 eV) was used for ionization. For calibrating the method, six calibrators were tested: 0.5, 1, 30, 50, 100 and 150 mg/ml. The selected ion monitoring (SIM) mode was used for the distribution study; quantifying and monitored ions for the internal standard procaine were (
Sample preparation procedures column pretreatment for serum
Oasis HLB solid–liquid extraction cartridge was preconditioned with 1 ml methanol followed by 1 ml of water. One milliliter of serum was fortified with 0.1 ml internal standard (procaine) and then the sample was passed through column by gravity. Column was washed with 1 ml 5% methanol solution. Eluent was taken with 1 ml methanol and evaporated until dryness under nitrogen gas. Extract was reconstituted with 200 uL of methanol and injected into GC/MS. Fresh samples of 1 ml of serum from day 1 were immediately extracted, while the rest of samples were divided into two groups: one stored at 4°C and at −20°C. Measurements were then taken for the 1st, 2nd, 3rd, 10th, 14th, 21st and 30th days.
Tissue pretreatment
One gram of tissue samples (liver, kidney and brain) was homogenized with 0.1 M, pH 4.4 phosphate buffer and fortified with 0.1 ml of internal standard. Homogenate samples were vortexed for 2 min and centrifuged at 3500 rpm for 15 min. Samples were filtered through 0.45 µ hypodermic syringe filter. The filtered homogenate was extracted using Oasis MCX solid–liquid extraction cartridge. Oasis MCX solid–liquid extraction cartridge was conditioned with 2 ml methanol and followed with 1 ml phosphate buffer. The filtered homogenate was passed through the column by gravity. Column was washed sequentially with 2 ml water, 2 ml 0.1 N HCl, 2 ml 5% methanol and 2 ml acetone/methanol (70:30, v/v) solutions. Column was eluted with 2 ml 5% ammonium hydroxide and the eluate was dried under nitrogen gas. Eluate was reconstituted with 200 uL of methanol and injected into GC/MS.
Method validation and determination
The GC/MS toxicological methods were developed and validated in compliance with international standards to detect and/or to quantify paracetamol in rat samples under the operating conditions described above. Blank rat plasma and tissue samples collected from male laboratory Wistar Albino rats were used for calibration and validation purposes. The internal standard method was used for quantification purposes. The calibration curves were based on linear regression analysis using the ratio of the analyte peak area related to the internal standard.
The GC/MS method validation parameters were as follows: blank serum and tissue samples fortified with calibrators and quality controls were tested for selectivity and no interferences were found. For serum, the linearity was fulfilled in the concentration range of 0.5–150 mg/ml with regression coefficients greater than 0.999 for paracetamol. For tissues, the linearity was established to be within 0.5–10 mg/g with regression coefficients greater than 0.992 for paracetamol. The detection limits (LOD, S/N >3) in plasma and tissues were 10 ng/ml (ng/g) and the lower limits of quantification (LLOQ, S/N > 8) were 20 ng/ml (ng/g).
Statistical analysis
SPSS 16.0 packet program was used in the statistical analysis of data. Categorical measurements were summarized as numeric and percentage constant measurements were summarized as mean and standard deviation (median and minimum–maximum when necessary). Mann–Whitney test was used for comparisons of two situations (the storing conditions, the distribution of paracetamol in organs, paracetamol concentrations changing by days), Kruskall–Wallis test was used for statistical analysis of the results obtained in toxicological analysis for more than two situations (comparing the values of different days, etc.) and Mann–Whitney test with Bonferroni correction for the
Results
Serum samples collected immediately after drug administration were used as the baseline values for measuring the stability and distribution of paracetamol. When the concentrations of paracetamol in serum were quantitated, it was observed that the percentage change among the groups of samples from 0 to 10 days were well within 20% at both storing temperatures, that is 4°C and −20°C with significant
Changing profile of serum paracetamol concentration according to the storing conditions and days

a Abbreviation: S.D., standard deviation

Changing of serum paracetamol concentration according to days and storing conditions (box plot).
When the percentage change in paracetamol concentrations in liver were measured and evaluated from day 1 through day 30, the results showed a statistically significant decrease in the overall concentration (Table 2, Figure 2). Although the percentage change in paracetamol concentration between days was statistically meaningful, the percentage change concentration between days 14 and 30 was not statistically meaningful. When it is evaluated as percentage change, paracetamol concentration change in first day liver sample is 30.26%, and it was 94.97% in 30th day liver sample.
Changing profile of liver paracetamol concentration according to the days
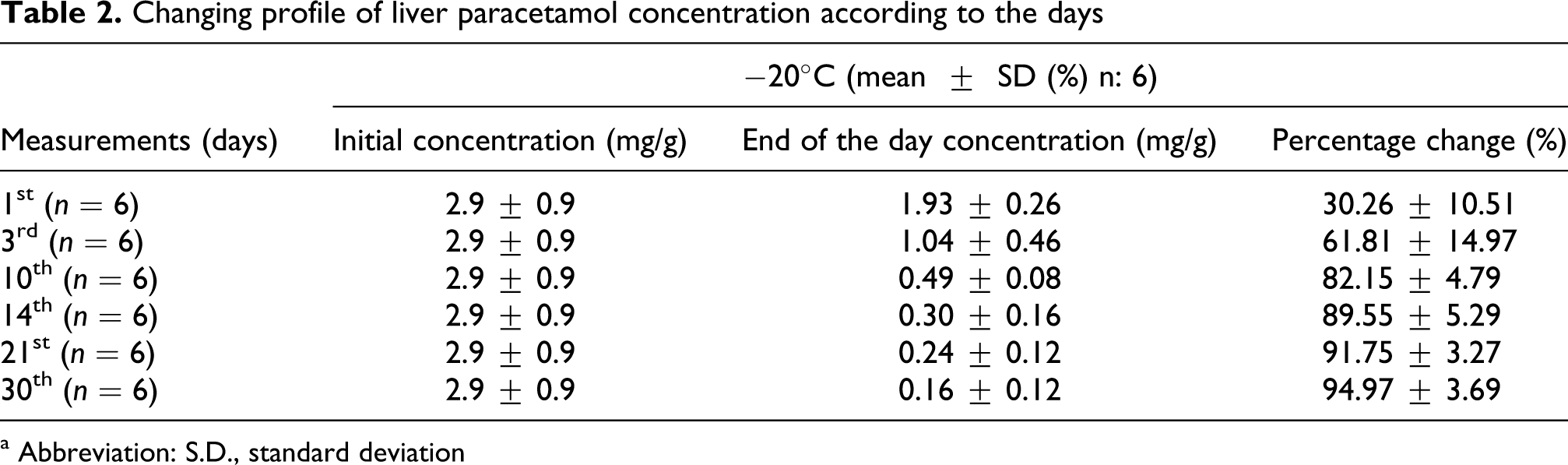
a Abbreviation: S.D., standard deviation

Changing of liver paracetamol concentration according to days (box plot graphic).
The quantitative distribution of unchanged paracetamol amounts in liver, brain and kidney was provided in Table 3. In this evaluation, the difference between the measurements of liver, kidney and brain paracetamol concentrations was determined to be statistically significant. Paracetamol concentration was the highest in liver and the lowest in brain. There was no significant statistical difference between the measured concentration of paracetamol from kidney and brain.
Paracetamol distribution of organs in case of acute intoxication

a Arithmetic mean ± standard deviation.
Discussion and conclusions
In postmortem forensic toxicological analysis, paracetamol ingestion and/or administration can be detected from liver, kidney and blood samples. In case of paracetamol if taken more than the recommended dose, the drug concentration quantitated from serum samples depicts a clearer picture of its level of toxicity. Therefore, for the quantitation of paracetamol in serum, a sensitive and reliable analytical method is used. Although acute paracetamol intoxication cases occur frequently, scientific literatures on evaluating the stability of paracetamol from biological samples are very few.
A review of 80 cases by Hawton et al. has shown that 50% of the patients who have used paracetamol in their suicidal attempts believed that the drug is easy to obtain. While 29% perceived that the drug is dangerous and only 4% indicated that it is inexpensive. 16
In the study performed by Frank Peters, it is indicated that the stability of the substances in biological samples taken for clinical and forensic purposes generally degrade with factors like oxidation, reduction and esterification and recommended that samples should be stored in refrigerator or at −20°C to minimize degradation. In addition, it is stated that there may be degradation to some degree even if the samples were stored in suitable conditions for a long time. 17
In our study, it was observed that the percentage change in paracetamol concentrations in serum measured after the 10th day was similar in both storing conditions: 4°C and −20°C. However, at 4°C, as time passed, after the 14th day, it was observed that the level of paracetamol concentration in serum significantly decreased but at the same period the change at −20°C is not statistically significant. In addition, statistical difference was observed for serum samples stored in neither storing conditions between the measurements of the first 10th day and after the 14th day. When the percentage change in the effects of storing conditions on paracetamol concentration was studied, samples stored at −20°C manifested minimal change in the drug stability compared to samples stored at 4°C. This was further demonstrated in samples from the 30th day with the percentage change of 33.78% and 66.30% for −20 oC and 4°C, respectively.
In different storing conditions for serum samples, although mean average values were different from each group it was observed that they decreased in a statistically similar way. Only the 30th day, measurement was determined to be different among the storing conditions according to days.
In the study of Speed et al., it is stated that paracetamol concentration did not show any significant change in blood samples when preservative and anticoagulant were added to the samples and were stored at 4°C . 18 The results of our study related to serum samples correlate with the study of Speed and his friends although they are not identical in the purpose of the studies. In our study, it was observed that the amount of paracetamol in liver stored at −20°C decreased with each passing day.
In the postmortem study Hilberg et al. performed with rats, the rate of ante mortem and postmortem serum paracetamol concentrations was found to be approximately 1. In addition, postmortem paracetamol concentration in liver, kidney and lung were determined to be low. This is partly due to the absorption rate of paracetamol from the stomach and its low distribution volume and nonionic structure. When serum and organ samples used in our study were evaluated after acute paracetamol poisoning, it was observed that paracetamol was lower in brain as in the study of Hilberg et al. Accordingly, the paracetamol concentration measured in 16 rats had mean concentrations of 2.68, 1.11 and 0.68 mg/g in liver, kidney and brain, respectively. 19
The most important organ in the detoxification process is liver. In most studies, about 10% of the patients who could not be cured of paracetamol poisoning, liver damage was observed and 10%–20% of them died of hepatic failure. In our study, the organ distribution of paracetamol was determined to be the highest in liver in accordance with other studies in literature. Kidney is also one of the target organs in paracetamol poisoning. In the study of 51 cases by Mahadevan et al., 13.5% of cases presented with nephrotoxicity along with hepatotoxicity. 20 In the study of Blakely et al. published by American Blood Renal Diseases Center, it is stated that 2% of total acute renal damages was because of paracetamol poisoning. In the same study, it is stated that paracetamol may damage kidneys in the case of glutathione shortage or a drug-inducing cytochrome P450 microsomal enzymes in therapeutic doses. 21 In our study, when organ distributions were studied, it was observed that brain accumulated the least amount of the drug. In the study of Hendrickson et al. on rat brain, it is stated that paracetamol affected the central nervous system by consuming glutathione. Although this study is accepted as a pre-study, it is not certain how the central nervous system is affected by acute paracetamol poisoning. Similarly, there are pre-studies about the depression caused by overdosed paracetamol ingestion due to the increasing metabolic acidosis. 2
The chemical and physical denaturation of the biological sample before analysis could be attributed to the absence of the preservative effect of drugs bound to plasma proteins. This could also be due to bacterial contamination within the sample and the poor storing conditions. In forensic studies and investigations, stability of drugs in stored biological samples is very important. If there is a claim on analysis or evaluation of samples subsequently datas must ensure that they are kept viable. Factors such as temperature change, prolonged storage and lack of preservative can affect the stability of the drug.
In the study by Holmgren et al., stability of 46 drugs including benzodiazepines, antidepressants, analgesics, hypnotics and paracetamol in postmortem blood for 1 year is evaluated using preservatives in −20°C. In this 1-year study, no statistically significant change was observed in femoral vein blood paracetamol concentration at −20°C using preservatives. However no statistically significant change was observed in femoral vein blood paracetamol concentration at −20°C using preservatives a meaningful difference was observed between the first and second measurements of 7 drugs (ethanol, ketobemidon, desmetilmianserin, 7-amino-nitrazepam, THC, thioridazie, zolpidem). 22
In stability studies, the difference between the two analytical results may be related not only to the stability of the drug in the sample but also to the reliability and accuracy of the method. In the study Drummer et al., it is stated that bioanalytical procedures in postmortem toxicological analysis must be standardized and this must be started by choosing the suitable sample for the analysis. 23 In our study, as biological samples, liver, kidney and brain were used as well as serum which is the most related biological sample in paracetamol poisonings. For validation of the method used in the analysis of these samples, recovery, limit of detection and limit of linearity studies were performed. As a result, it is important to know the stability of drugs in biological samples both in clinical and in forensic studies. Paracetamol stability in serum until the 30th day after collection using preservative (sodium fluoride) did not show any statistically significant decrease in concentration. Although postmortem liver samples stored at −20°C were not suitable for future reevaluation of paracetamol concentration. This condition should be taken into consideration while reevaluating the samples stored for greater than 30 days.
In conclusion, stability of the paracetamol from biological samples are interdependent not only on the source of the sample but also on the storing conditions. For clinical purpose, organ distribution of paracetamol is limited to the dose and the time of ingestion. Detection and reevaluation of paracetamol concentration in the field of forensic studies is limited to the storing conditions, time and the matrix of the biological samples. Considering these given factors of storing the biological samples for a certain time, similar longer studies should be performed to determine the stability of xenobiotics.
Footnotes
Funding
This study is financed by Cukurova University Scientific Research Fund with the project number of TF 2007 D5.
