Abstract
Aldo-keto reductases (AKRs) metabolize a wide range of substrates, including polycyclic aromatic hydrocarbons (PAHs), generating metabolites (o-quinones) and reactive oxygen species (ROS), which are capable of initiating and promoting carcinogenesis. Exposure to PAHs, their metabolites, and ROS further increase AKRs isoform expression that may amplify oxidative damage. Human AKR enzymes are highly polymorphic, and allelic variants may contribute to different AKRs expression in individuals. Despite the importance of AKRs in PAHs metabolism, there are no studies that evaluate, in general human populations, the effect of PAHs on AKRs expression in peripheral blood lymphocytes (PBLs). The aim of this study was to determine the effect of tobacco smoke exposure, and AKR1A1*2 and AKR1C3*2 polymorphisms, on AKR1A1 and AKR1C1–AKR1C3 messenger RNA (mRNA) levels in PBLs from smokers. In the smoker group, there is a statistically significant positive association between AKR1A1, AKR1C1, and AKR1C3 mRNA induction and urine cotinine levels in individuals with a body mass index (BMI) less than 25. However, AKR1A1*2 and AKR1C3*2 alleles did not influence AKR1A1 and AKR1C1–AKR1C3 mRNA levels. These results suggest that AKRs induction by PAHs in smokers’ PBLs is associated with BMI; therefore, the role of adipose tissue accumulation in PAHs’ effects needs further investigation.
Introduction
Xenobiotic metabolism is performed by a complex array of enzymes that transform these compounds into either reactive or nonreactive products, which are deleterious to the health of exposed individuals. In addition, xenobiotic metabolizing enzymes (XMEs) are frequently regulated by their own substrates or by intermediary products. 1 Detection of XMEs expression and activity may be useful to monitor human exposure. Therefore, XMEs expression in accessible cells, such as peripheral blood lymphocytes (PBLs), has been the focus of several studies searching for exposure biomarkers in human populations. 2 –6
Polycyclic aromatic hydrocarbons (PAHs) are environmental pollutants formed through the incomplete combustion of organic materials, including tobacco smoke. Tobacco smoke is strongly linked to several malignancies, especially lung cancer. To exert its carcinogenic effects, some tobacco constituents, like benzo[a]pyrene (BP), require metabolic activation by cytochrome P450 (CYP1A1/1B1), epoxide hydrolase, and aldo-keto reductases (AKRs). AKRs are an enzyme superfamily of soluble NAD(P)H oxidoreductases that transform a variety of substrates, including hormones, drugs, reactive aldehydes, and PAHs. 7 These enzymes catalyze the oxidation of BP-7,8-diol to form catechols, which are air sensitive, and undergo two sequential 1-electron auto-oxidation events to produce the o-semiquinone anion radical, followed by the formation of the o-quinone BP-7,8-dione. 8 Each auto-oxidation step produces reactive oxidative species (ROS), 9 promoting a pro-oxidant state that leads to DNA damage through oxidation, formation of covalent adducts, and generation of nitrogen base transversions. 10 In addition, during the formation of PAHs o-quinones, a futile redox cycle occurs in the presence of NAD(P)H, which generates ROS. Therefore, the formation of PAHs o-quinone by AKRs increases the carcinogenic potential of the parent compound. 9
Considering all that is known about AKRs, their expression may have an important impact on the biotransformation of chemical carcinogens. AKR1C1–AKR1C4 gene promoters contain several antioxidant response elements, 11 and their transcription is driven by ROS through the Nrf2 pathway. On the other hand, AKR1A1 gene promoter contains two xenobiotic response elements, suggesting that its expression is under the aryl hydrocarbon receptor control. Therefore, exposure to PAHs, their metabolites, and the formation of ROS may increase AKRs isoforms expression, thereby amplifying oxidative damage. This idea is supported by in vitro studies using human cell lines, including bronchoalveolar (H358), 12 lung (A549), 13 liver (HepG2), 14 and mouth, 15 which have shown that exposure to PAHs results in AKR1A1 and AKR1C messenger RNA (mRNA) overexpression. In addition, previous human studies in smokers have shown elevated levels of AKRs mRNA in bronchial epithelium 16 and oral mucosa 17 cells.
Five AKRs isoforms are important in PAHs metabolism: AKR1A1, AKR1C1, AKR1C2, AKR1C3, and AKR1C4. AKRs polymorphism may affect its expression and activity, particularly for those individuals carrying the AKR1A1*2 (Asn52Ser) or the AKR1C3*2 (His5Gln) allelic variants. For instance, AKR1A1*2 results in a decreased enzymatic activity, 18 whereas the AKR1C3*2 polymorphism has been associated with increased lung cancer susceptibility in smokers. 10 Moreover, in vitro studies have suggested that the AKR1C3*2 variant has higher activity than the wild-type enzyme. 19 Despite the importance of AKRs in PAHs metabolism, there are no studies that evaluate its expression in PBLs of exposed human populations. Therefore, the aim of this study was to determine the effect of PAHs, as well as the AKR1A1*2 and AKR1C3*2 polymorphisms, on AKR1A1 and AKR1C1–AKR1C3 mRNA levels in PBLs obtained from smokers.
Materials and methods
Sample collection
A cross-sectional study was carried out in 140 male volunteers, students, and workers from the Universidad Autónoma de Nayarit, Mexico, from October to November 2008. Blood and urine samples were collected from 70 smokers (defined as those smoking at least three cigarettes per day) 20 and 70 nonsmokers. Information on demographic characteristics, socioeconomic status, medical history, smoking habits, and alcohol consumption was obtained from a structured questionnaire administered by the interviewers. Participants were grouped by body mass index (BMI), calculated for each subject as the body weight (kg) divided by the square of the height (m), according to the recommendations of the World Health Organization (BMI < 25: normal weight, BMI ≥ 25: overweight). 21 To be included in the study, participants had to be clinically healthy and without medication for 15 days prior to samples collection. The study was approved by the Ethical Committee for Studies in Humans (COBISH) of the Centro de Investigación y de Estudios Avanzados del IPN (Cinvestav) Mexico, and an informed consent was signed by each participant.
Urine assay for cotinine
Tobacco smoke exposure was estimated by measuring cotinine in urine samples run in duplicate with a commercial enzyme-linked immunoassay kit (DRG Instruments, Marburg, Germany), following the manufacturer’s instructions; urinary cotinine values less than 0.50 μg/mL were considered negative.
DNA extraction
Blood samples were collected by venipuncture and placed in tubes containing EDTA as an anticoagulant. DNA was isolated from whole blood using a High Pure PCR Template Preparation kit (Roche, Indianapolis, Indiana, USA) following the manufacturer’s instructions. DNA was quantified by spectrophotometry at optical density of 260 nm (OD260), and the purity was assessed by measuring the OD260/OD280 ratios. DNA integrity was evaluated by electrophoresis on 1% agarose gels stained with ethidium bromide.
Determination of polymorphisms by real-time PCR
All polymerase chain reaction (PCR) reagents and analytical equipment were purchased from Applied Biosystems (Foster City, California, USA). Real-time PCR assays of single-nucleotide polymorphisms (SNPs) were performed with primer-specific fluorescent labeled probes in a 7000 sequence detector. The probes (Table 1) were designed using Primer Express software and were labeled with FAM and VIC as the 5′-fluorescent reporters. The PCR reaction mixture contained 50 ng DNA, 1X TaqMan Universal PCR Master Mix, 200 nM of each primer, and 100 mM of the TaqMan probe. PCR reactions were performed using an ABI PRISM 7000 sequence detector system as follows: 50°C for 2 min, 95°C for 10 min, then 40 cycles of 95°C for 15 s and 60°C for 1 min. A negative control was included in each plate. Allelic discrimination was performed using the 7000 system software.
Primers and probes for the identification of the polymorphisms AKR1A1*2 and AKR1C3*2.

Isolation of PBLs, total RNA extraction, and cDNA synthesis
Lymphocytes were isolated from 5 mL fresh blood using Ficoll-Paque PLUS (GE Healthcare Bio-Sciences, Uppsala, Sweden) according to the manufacturer’s instructions. Lymphocytes were washed twice with phosphate-buffered saline prior to RNA isolation. Total RNA was isolated using the Trizol reagent according to the manufacturer’s instructions (Invitrogen Life Technologies, Carlsbad, California, USA). RNA was quantified spectrophotometrically at OD260, and the purity was assessed by measuring the OD260/OD280 ratio. The integrity of the RNA was evaluated by electrophoresing RNA samples on 1% agarose gels. Complementary DNA (cDNA) was prepared from 5 µg total RNA using the kit SuperScript III First-Strand Synthesis System for RT-PCR (Invitrogen Life Technologies, Carlsbad, California, USA) and an oligo dT primer.
Evaluation of mRNA levels by quantitative PCR
Quantitative PCR was carried out using cDNA prepared from PBLs’ mRNA. Probe sequences are shown in Table 2. mRNA levels were normalized to the housekeeping gene GAPDH. The PCR reaction mixture contained 1 μl of cDNA, 1X TaqMan Universal PCR Master Mix, and 0.9 μM and 0.25 μM of primers and probe, respectively. PCR reactions were performed using an ABI PRISM 7000 sequence detector system, with the following cycling protocol: 50°C for 2 min and 95°C for 10 min, then 40 cycles of 95°C for 15s and 60°C for 1 min. Each sample was run in triplicate. Results were analyzed using the comparative threshold cycle (Ct) method as described previously by Livak and Schmittgen. 22 Results are expressed as fold differences in gene expression relative to the control, and calculated as fold change = 2−ΔΔCt.
Primers and probes used for evaluation of AKRs mRNA levels.

AKR: aldo-keto reductase; mRNA: messenger RNA.
Statistical analysis
StatXact-8 (v. 8.0.0; Cytel, Cambridge, Massachusetts, USA) software was used for the Fisher–Freeman–Halton test and Bonferroni’s correction method, conducted to analyze the difference between genotypes and allele frequencies among human samples. For mRNA expression, Stata version 10.1 software (StataCorp LP, College Station, Texas, USA) was used for statistical analysis. The statistical analysis was carried out using descriptive statistics of variables such as age, alcohol consumption, BMI, relative AKRs levels, and AKRs ΔCt, which were evaluated using the Mann–Whitney test. In addition, the probability of multiple comparisons in AKRs ΔCt was adjusted using the Bonferroni’s correction method. Results from the human studies are provided as geometric mean (GM). A multiple regression analysis of the AKRs ΔCt variation in participants grouped by BMI was carried out with variables such as age, BMI, SNP, alcohol consumption, number of cigarettes smoked, cotinine levels, and exposure to other PAHs sources. An interaction analysis model was developed to evaluate interaction between normal weight and overweight participants with cotinine level, for each gene analyzed. Statistical significance was set at p < 0.05.
Results
Demographic data are shown in Table 3. The GM of the participants’ age was 22.7 years, while the GM of their BMI was 24.5 kg/m2. There were no differences in age or BMI between smokers and nonsmokers, but 35.71% of the population were overweight (BMI ≥ 25). The majority of participants (91.4%) regularly consumed alcohol, with a significant higher number for smokers (84.29% of nonsmokers vs. 98.57% of smokers, p = 0.002). These data are in agreement with the behavioral link between cigarette smoking and alcohol consumption. All participants had clinical chemistry results within the normal ranges and were diagnosed as clinically healthy.
Demographic characteristics of study participants.

BMI: body mass index; GM: geometric mean; CI: confidence interval.
aThe p value was determined using the Mann–Whitney test.
In order to evaluate the exposure to cigarette smoking, urine cotinine levels were determined. The urinary cotinine concentration GM in smokers and nonsmokers was 3.083 μg/mL and 0.239 μg/mL, respectively (Table 4). A total of 65 smokers (92.86%) had urinary cotinine concentration above 0.50 μg/mL, while only 5 (7.14%) had cotinine values below 0.5 μg/mL; they reported to have smoked their last cigarette around 17 h before taking the urine sample. In contrast, only one participant (1.4%) in the nonsmoker group had a value above 0.50 μg/mL.
Urinary cotinine concentration.

GM: geometric mean; CI: confidence interval.
aThe p value was determined using the Mann–Whitney test.
We then determined the AKR1A1 and AKR1C1–AKR1C3 mRNA levels in both nonsmokers and smokers (AKR1C4 was not measured because it is expressed in the liver only). No differences were observed on AKR1A1 and AKR1C1–AKR1C3 mRNA levels between groups (data not shown). Similar results were observed when a multiple regression analysis was carried out with variables such as age, alcohol consumption, number of cigarettes smoked, cotinine levels, and exposure to other PAHs sources. However, the BMI of the participants had an impact on the expression levels of the isoforms AKR1A1, AKR1C2, and AKR1C3. Smokers with BMI < 25 had higher levels of mRNA expression compared to those with a BMI ≥ 25 (Table 5). An association between the urine cotinine levels and the ΔCt values for AKR1A1, AKR1C1, and AKR1C3 in smokers with BMI < 25 was also observed. In contrast, this correlation was not present in smokers with BMI ≥ 25 (Figure 1). An interaction in AKR1A1 and AKR1C3 ΔCt values between smokers with BMI < 25 and BMI ≥ 25 was also observed (p = 0.018 and p = 0.002, respectively), indicating differential behavior between these two subgroups.
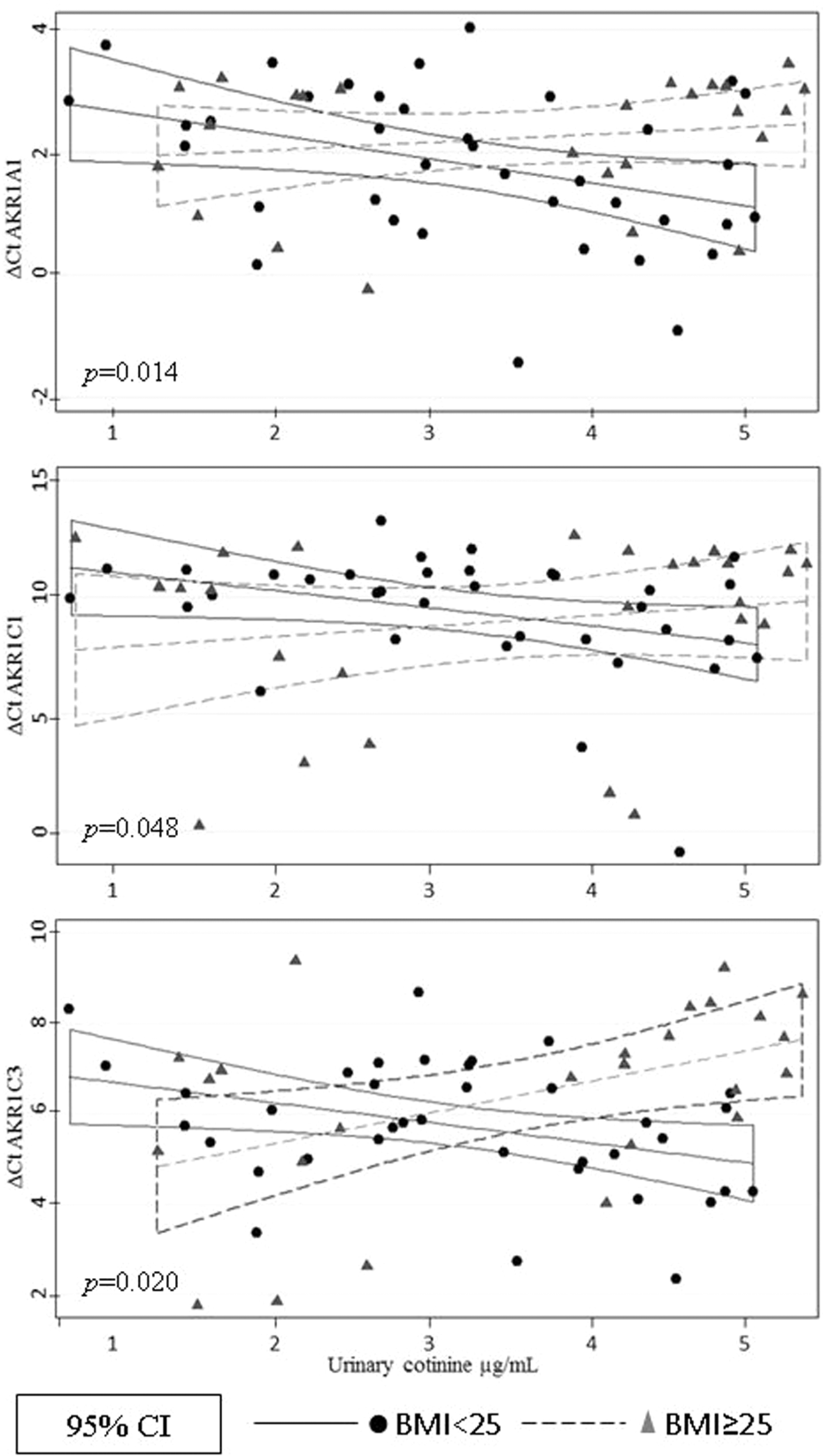
Interaction analysis to evaluate the difference in AKR1A1, AKR1C1, and AKR1C3 ΔCt values between normal weight and overweight smokers. Data were normalized to GAPDH. The p value represents the significance of the interaction between cotinine, BMI, and the ΔCt for each isoform. Based on relative levels of mRNA, the levels of ΔCt in participants with normal weight correspond to higher levels of expression than in overweight participants. BMI: body mass index; mRNA: messenger RNA.
Relative levels of AKRs mRNA in smokers, according to BMI.

AKR: aldo-keto reductase; BMI: body mass index; mRNA: messenger RNA; CI: confidence interval; GM: geometric media compared to GM (1.0) for nonsmoker.
aThe p value was determined using the Mann–Whitney test.
We then determined the AKR1A1*2 (Asn52Ser) and AKR1C3*2 (Gln5His) allelic variants. The genotype distribution for AKR1A1*2 was 90% for *1/*1 and 10% for *1/*2, whereas the allelic frequencies were 95% for *1 and 5% for *2 (Table 6). On the other hand, the genotype distributions for AKR1C3*2 were 10% for *1/*1, 46% for *1/*2, and 44% for *2/*2, and the allelic frequencies were 33% for *1 and 67% for *2 (Table 7). All genetic variants determined in the present study were in Hardy–Weinberg equilibrium (data not shown). In order to determine whether the AKR1A1*2 and AKR1C3*2 genotype influences the AKR1A1 and AKR1C1–AKR1C3 mRNA levels, a correlation analysis was done. However, no association was observed between AKRs mRNA levels and the SNP determined in this study.
AKR1A1*2 polymorphism frequency in human samples.

MC: Omnibus test Monte Carlo p value (upper 95% CI); TSE: all test versus Mexicans. Two-sided “exact” p value. aBonferroni adjusted a criterion = p£ 0.0125, equivalent to unadjusted a criterion of 0.05, according to 4 test: 1 globaland, 3 sample populations compared with Mexicans.
Finally, the genotype and allelic frequencies in the present study were compared with other populations (Tables 6 and 7). The AKR1A1*2 allele is not present in Asian and Sub-Saharan African populations, while the European population showed an allelic frequency similar to the Mexican population. In contrast, the Mexican population had a higher AKR1C3*2 allelic frequency than European and Sub-Saharan African populations, but lower than the Chinese population.
AKR1C3*2 polymorphism frequency in human samples.
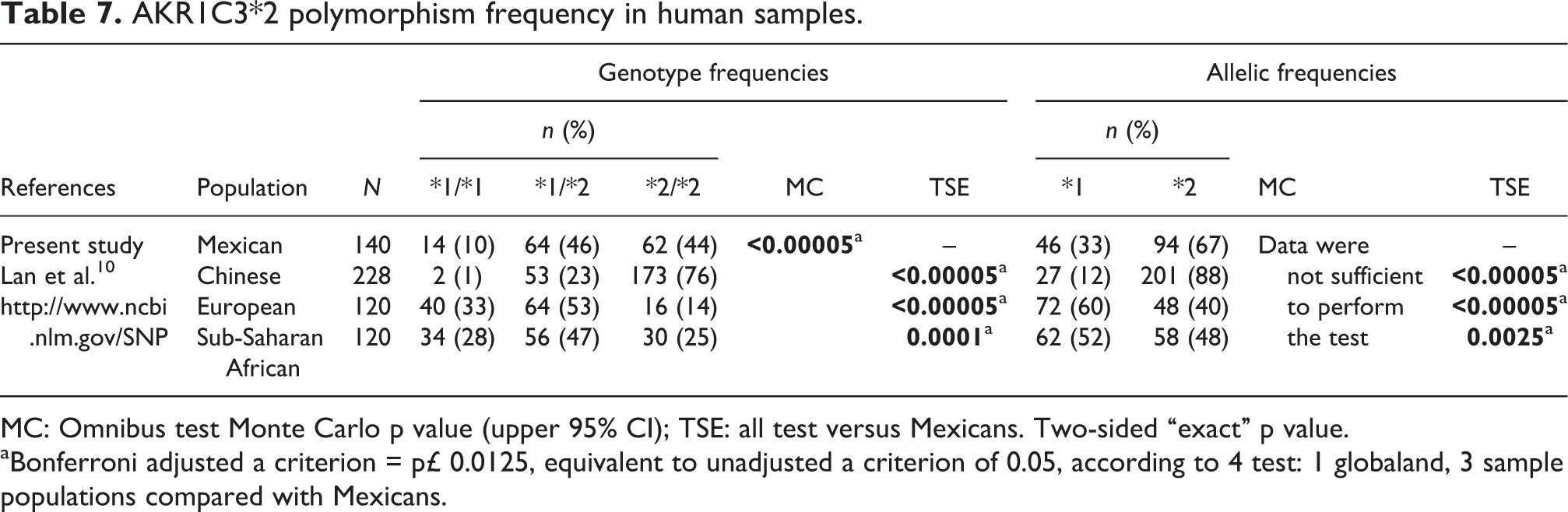
MC: Omnibus test Monte Carlo p value (upper 95% CI); TSE: all test versus Mexicans. Two-sided “exact” p value. aBonferroni adjusted a criterion = p£ 0.0125, equivalent to unadjusted a criterion of 0.05, according to 4 test: 1 globaland, 3 sample populations compared with Mexicans.
Discussion
AKRs have an important role in the bioactivation of PAHs leading to oxidative DNA damage. 23 Therefore, it is very important to characterize not only the expression of these oxidoreductases in easily accessible human tissues but also the factors that might modify their expression and/or activity. This is the first study that determines the mRNA levels of the main AKRs isoforms (AKR1A1 and AKR1C1–1C3) involved in PAHs transformation in PBLs obtained from the general human population. Increased AKR1A1, AKR1C1, and AKR1C3 mRNAs levels (1.52-, 1.70-, and 1.50-fold induction, respectively) were observed for smokers compared with nonsmokers (all with a BMI < 25). Although these differences were not statistically significant, probably due to the size of the sample; AKRs induction has been observed in human oral cell lines exposed to tobacco smoke. 24 In contrast, statistical analysis indicated that, for smokers with BMI < 25, there is an association between AKRs mRNA levels (ΔCt) and urine cotinine levels, suggesting that excessive adipose tissue may affect the expression of these enzymes. Since AKRs expression is mediated by Nrf2 in response to oxidative stress, and at the same time they produce ROS, 25 it is plausible that AKRs functional polymorphisms affect their own expression. However, the present results did not find any association between AKR1A1*2 and AKR1C3*2 polymorphisms and mRNA levels. Additionally, this study confirms that PBLs are a useful and convenient surrogate model for other less accessible tissues to study gene regulation affected by contaminants, like in this study or, for diagnostic purposes in order to detect early changes in gene expression. 26,27
PAHs are highly liposoluble and, therefore, stored in body fat. 28 Ahotupa and Mantyla 29 demonstrated that the adipose tissue content of mice modified the tissue distribution of liposoluble polychlorinated biphenyls (PCBs), which altered the activity of several drug metabolizing enzymes, including epoxide hydrolase. Therefore, individuals with excessive adipose tissue may have more PAHs storage capacity than normal weight individuals. Being overweight decreases PAHs availability, and results in reduced biological effects such as AKRs mRNA induction. These data also suggest that overweight smokers, when they lose weight, may release stored PAHs into their bodies that may modify the levels of AKRs and other drug metabolizing enzymes. Carlson 30 showed that starvation causes PCB mobilization and increased xenobiotic metabolism activity.
The AKR1A1*2 variant frequency observed in the present study (5%) was similar to that previously reported in Europeans (4%), but differed from that of Asians (0%) and Sub-Saharan Africans (0%). In contrast, in the present population, the AKR1C3*2 allele was observed at a frequency of 67%, which is lower than that previously found in a Chinese population (88%), 10 but higher than either a European (40%) or a Sub-Saharan African population (48%). Carrying any of these allelic variants may have different outcomes depending upon the type of xenobiotic. AKR1A1 has been implicated in the metabolism of anthracycline antibiotics such as doxorubicin and daunorubicin. Both drugs are used for the treatment of breast cancer, childhood solid tumors, Hodgkin’s disease, and acute lymphoblastic leukemias. 31,32 However, the use of these drugs results in cardiotoxic susceptibility. 33 In particular, the AKR1A1*2 allelic variant has reduced catalytic activity when compared with the wild-type allele, and has been associated with anthracycline toxicity. 18 On the other hand, due to the fact that AKR1A1 is involved in PAHs activation, 34 individuals carrying AKR1A1*2 variants may have a decreased capacity to catalyze the oxidation of BP-7,8-diol to form catechol, decreasing ROS production. However, the formation of BP-7,8 dihydrodiol-9,10-epoxide from BP-7,8-diol might result in more toxicity than BP-o-quinone and ROS. 35 This fact suggests a protective role for AKRs in response to PAHs exposure. On the other hand, the AKR1C3*2 allele has been associated with a susceptibility to bladder, lung, and prostate cancer. 10,36,37 This allelic variant might also have a role in alcohol dependence. 38
In summary, the present study showed that AKR1A1, AKR1C1, and AKR1C3 mRNA are induced in smokers PBLs and correlate with urine cotinine, but this effect is influenced by BMI. Such AKRs induction is observed in individuals with low BMI (<25); the toxicological relevance of this fact to potential damage to or protection for human health should be studied further. In addition, toxicokinetic studies on individuals with different BMI are necessary in order to investigate PAHs accumulation in body fat stores. It is pointed out that the relatively small size of sample for a genetic study is a limitation of this study. Finally, an extensive analysis of SNP of AKRs is needed to understand the impact of genetic polymorphisms on protein expression in human populations.
Footnotes
Funding
This work was supported by SEMARNAT-CONACYT (SEMARNAT-2002-CO1-293) and Conacyt (60463) awarded to AA. BSBV received a scholar grant from Conacytwith number 6544.
Acknowledgments
The authors are grateful to Patricia Vázquez-Vázquez, Emmanuel González Barbosa, Miriam Jackeline Loera Rosales, Marisol Fuentes Reyes and Cyndia Azucena González Arias, for their technical assistance and/or data collection.
