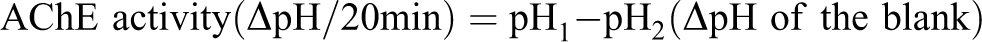Abstract
The aim of this study was to examine the effects of ubiquinone (CoQ10) on heart tissue and erythrocytes in acute organophosphate poisoning (AOP). A total of 20 rabbits were divided into three groups: sham (n = 8), pralidoxime (PAM) + atropine (n = 6), and CoQ10 + PAM + atropine (n = 6). Blood samples were taken from each test subject to measure the values of acetylcholinesterase (AChE), nitric oxide (NO), and malondialdehyde (MDA) in the plasma and erythrocyte before administration of 50 mg/kg dichlorvos by orogastric tube. Blood samples were then taken at 1, 12, and 24 h post-dichlorvos to determine plasma and erythrocyte levels of AChE, NO, and MDA. Sham group received no treatment. PAM + atropine group received 0.05 mg/kg atropine with repeated doses and PAM: first a 30-mg/kg intravenous (IV) bolus, then a 15-mg/kg IV bolus every 4 h. CoQ10 + PAM + atropine group received same dose PAM and atropine and a 50-mg bolus of IV CoQ10. Thoracotomy was performed in all the animals 24 h after poisoning and then heart tissue samples were obtained. At 12 and 24 h, erythrocyte AChE levels in the CoQ10 animals were considerably higher than those in PAM + atropine animals (p = 0.023 and 0.017, respectively). At 12 and 24 h, erythrocyte MDA and NO levels in CoQ10 animals were significantly lower than those in PAM + atropine animals (p < 0.05). Heart tissue AChE levels in CoQ10 animals were considerably higher than those of the sham and PAM + atropine animals (p = 0.001). Heart tissue MDA and NO levels of CoQ10 animals were significantly lower than those of the sham and PAM + atropine animals (p < 0.01). Treatment of AOP with CoQ10 + PAM + atropine in this animal model had a beneficial effect on both erythrocyte and heart tissue lipid peroxidation and AChE activity.
Introduction
Organophosphate (OP) pesticides are a large group of compounds that irreversibly inhibit cholinesterase, acetylcholinesterase (AChE), and neuropathy target esterase in humans and animals. Normally, AChE rapidly hydrolyzes the neurotransmitter acetylcholine (ACh) into inactive fragments of choline and acetic acid after completion of neurochemical transmission. The phosphate radicals of OP compounds covalently bind to active sites of cholinesterases, transforming them into enzymatically inert proteins, thus acting as irreversible cholinesterase inhibitors. The inhibition of ACh at synapses causes overstimulation and subsequent disruption of transmission in the central and peripheral nervous systems. 1
The toxic effects of OP pesticides are not only due to the inhibition of AChE. They may also induce in vivo and in vitro generation of reactive oxygen species (ROS) such as hydrogen peroxide (H2O2), superoxide (O2 −), and hydroxyl radicals (HO−). The extent of lipid peroxidation is determined by the balance between the production of oxidants and the removal and scavenging of those oxidants by antioxidants. Others assert that OP pesticides exert their toxic effects via lipid peroxidation. 2
Cardiovascular manifestations frequently accompany exposure to these OP pesticides. These manifestations are sinus tachycardia, sinus bradycardia, hypertension, hypotension, and impaired heart rate. Heart tissue may be sensitive to the peroxidative damage caused by OP pesticides because of its limited capacity to enzymatically detoxify hydroxyl radicals. Biochemical changes in the cardiovascular system after OP poisoning include myocardial necrosis and increased levels of creatinine kinase and lactate dehydrogenase. The following electrocardiography changes have been reported after OP poisoning: prolonged distance between Q wave and T wave in Electrocardiographi (QTc) interval, interval between S wave and T wave in Electrocardiographi (ST) segment elevation, low-amplitude T waves, and extrasystoles and prolonged distance between P wave and R wave in Electrocardiographi (PR) interval. 3 –7
Standard therapy of the overt somatic effects of OP poisoning includes anticholinergic agents (primarily atropine) and reactivators of AChE (oximes such as pralidoxime). This treatment can be life-saving in severe cases. 8
Ubiquinone (CoQ10) not only plays a central role in mitocondrial oxidative phosphorylation, but may also act as an antioxidant scavenger. CoQ10 is known to be highly concentrated in heart muscle cells due to the high energy requirements of this cell type. CoQ10 levels in the heart are five times higher than of the liver and approximately 10 times higher than that of the kidney, pancreas, and spleen. CoQ10 acts as a powerful antioxidant, scavenging lipid radicals and inhibiting lipid peroxidation as an essential component of the electron transport system (ETS) of the inner mitochondrial membrane, as well as of the membranes of the Golgi apparatus, endoplasmic reticulum, lysosomes, outer mitochondrial membrane, and plasma membrane. As a peroxyl scavenger, CoQ10 acts somewhat like vitamin E. 9,10
In this study, we aimed to determine the effects of treatment with CoQ10, in addition to atropine + pralidoxime (PAM), on erythrocyte and heart tissue lipid peroxidation, AChE activity, and nitric oxide (NO) levels in experimental acute OP poisoning (AOP).
Materials and methods
Experimental methods
The study was carried out in the Experimental Medicine and Research Center at our university medical center after being approved by the Board of Ethics. A total of 20 (12 male and 8 female, weighting 2500–4000 g) New Zealand rabbits were used. The subjects were divided into three groups: a sham group (n = 8), a PAM + atropine group (n = 6), and a CoQ10 + PAM + atropine group (n = 6). Subjects were anesthetized with 50 mg/kg intramuscular (IM) ketamine and 15 mg/kg IM xylazine HCl. The central ear artery and marginal ear vein were catheterized. Blood was drawn in EDTA tubes to measure baseline plasma AChE, NO, and plasma and erythrocyte malondialdehyde (MDA).
Orogastric feeding tubes were inserted and 50 mg/kg (median lethal dose (LD50) = 50 mg/kg) of dichlorvos was administered. After 1 h, when signs of poisoning (hypersalivation, bronchospasm, fasciculations, and convulsions) appeared, venous blood samples were taken again in order to measure plasma AChE, NO, and plasma and erythrocyte MDA.
No treatment was given to the sham group. Venous blood samples were taken at 12 h after OP administration to measure plasma AChE, NO, and plasma and erythrocyte MDA. In the PAM + atropine group, 0.05 mg/kg intravenous (IV) atropine was given and this dose was administered again as needed. In addition, a 30-mg/kg IV bolus of PAM was given, then 15 mg/kg of PAM was given intravenously every 4 h. In the CoQ10 + PAM + atropine group, 50 mg of CoQ10 was administered intravenously as well as PAM and atropine as in the PAM + atropine group. Blood samples were taken from the subjects in PAM + atropine and CoQ10 + PAM + atropine groups at 12 and 24 h after poisoning in order to measure plasma AChE, NO, and plasma and erythrocyte MDA.
At 24 h post-poisoning, thoracotomy was performed and heart tissue samples were taken to evaluate the levels of AChE, NO, and MDA. At the end of the study, subjects were killed by administering a high dose of ketamine.
Biochemical methods
Measurement of plasma AChE activity
Plasma was separated from erythrocytes by centrifuging for 15 min at 3000 rpm. The following 0.2 mL of plasma, 3 mL of distilled water, and 3 mL of barbital phosphate (pH 8.1) were placed into a test tube. The pH (pH1) of the mixture was measured with a glass electrode pH meter. Then, 0.1 mL of 7.5% acetylcholine iodide solution was added to the reaction mixture and incubated for 20 min at 37°C. At the end of incubation period, the pH of the reaction mixture was measured (pH2). AChE activity was calculated using the following formula:
Measurement of AChE activity in heart tissue
To measure heart AChE activity, a heart tissue sample was homogenized (at 25% of maximum speed) in barbital phosphate (pH 8.1) to weigh 3 mL/100 mg when wet. Homogenization was performed in an ice bath and the heart homogenate was preserved in ice before cholinesterase determination. For determining heart AChE activity, 0.2 mL of tissue homogenate was used. AChE activity was calculated using the same formula as shown above.
Measurement of NO in plasma and heart tissue
To measure NO in plasma and heart tissue homogenate, the Nitric Oxide Synthase Assay Kit (Colorimetric, Merck Chemicals, Darmstadt, Germany) was used.
Measurement of MDA in heart tissue
A heart tissue sample (0.5 g) was taken and preserved at −80°C. Using a cold 150 mM KCl solution, the tissue sample was homogenized to make a 10% homogenate and was then centrifuged at 10,000 r/min for 10 min. The following substances were mixed: 0.1 mL of the homogenate, 0.2 mL of 8.1% sodium dodecyl sulfate solution, 1.5 mL of 20% acetic acid solution (sodium hydroxide was added to this mixture to adjust pH to >3), and 1.5 mL of 0.8% thiobarbituric acid liquid; this mixture was then stirred with a vortex. The mixture was then boiled in distilled water at 95°C for 60 min. Then it was cooled, and the following were added to the mixture: 1 mL of distilled water, 5 mL of n-butanol and pyridine (15:l, v/v), and then the mixture was rinsed. The resulting mixture was spun at 4000 r/min for 10 min. A sample from the upper layer of the mixture was taken, and absorbance at 532 nm was measured spectrophotometrically. MDA concentrations were then derived with the following formula:
Measurement of MDA in erythrocytes
Blood was centrifuged and the plasma was separated. After being washed with normal saline solution once, 1.5 mL was taken from the erythrocyte plug and 1.5 mL of the buffered sodium azide was added. From this hemolysate, 50 mL was taken and 12.5 mL of Drabkin solution was added and the hemoglobin (Hb) was measured. From this mixture, 5 mL was taken and 5 mL of 35% H2O2 was added and this mixture was incubated for 2 h at 37°C with tubes open. After this was cooled, 3 mL was taken and 2 mL of trichloroacetic acid–arsenide solution was added and the mixture was then centrifuged at 2500 r/min. From this supernatant, 3 mL was taken and 1 mL of thiobarbituric acid was added and then the mixture was boiled for 15 min. After it was cooled, absorbance at 532 nm was measured spectrophotometrically and the results were calculated for each gram of Hb.
Statistical methods
Statistical analyses were performed using SPSS for Windows® version 13.0 (SPSS, Inc., Chicago, USA). Between groups comparisons were made by repeated measurements with analysis of variance (ANOVA). For significant values, Bonferroni’s one-way ANOVA as a post hoc test and then the Tukey honestly significant difference (HSD) test was applied. Comparisons with a p < 0.05 were regarded as statistically significant. When comparing intra-group repeated measurements, the student t test was used. Means of each group’s values were calculated and reported in figures. To compare tissue AChE and tissue MDA values, one-way ANOVA and then Tukey HSD tests were performed.
Results
Half of the subjects in the sham group died before 12 h post-poisoning and the other half died before 24 h; therefore, no blood samples were collected from those subjects at 24 h.
One hour after poisoning, no significant differences between groups in plasma AchE, MDA, or NO levels were found (p > 0.05). At 12 h after poisoning, the mean erythrocyte AChE level of the CoQ10 + PAM + atropine group was significantly higher than that of both the PAM + atropine and sham groups (p = 0.023 and p = 0.016, respectively). At 24 h after poisoning, the mean erythrocyte AChE level in the CoQ10 + PAM + atropine group was significantly higher (p = 0.007) than that of the PAM + atropine group (Figure 1).

Mean erythrocyte AChE levels (U/L) at various times after intoxication with dichlorvos in the three treatment groups. AChE: acetylcholinesterase.
The mean NO levels at 12 and 24 h post-poisoning in the CoQ10 + PAM + atropine group were significantly lower than that of the PAM + atropine group (p = 0.0063 and p = 0.023, respectively; Figure 2).

Mean erythrocyte NO levels (mmol/g Hb) at various times after intoxication with dichlorvos in the three treatment groups. NO: nitric oxide.
At 12 hours, the mean erythrocyte MDA level in the CoQ10 + PAM + atropine group was lower than that of both the sham and PAM + atropine groups (p = 0.001 and p = 0.012, respectively). At 24 h, the mean erythrocyte MDA level in the CoQ10 + PAM + atropine group was significantly lower than that of the PAM + atropine group (p = 0.002; Figure 3).

Mean erythrocyte MDA levels (nmol/mL)) at various times after intoxication with dichlorvos in the three treatment groups. MDA: malondialdehyde.
The mean heart tissue AChE level in the CoQ10 + PAM + atropine group was significantly higher than that of the sham and PAM + atropine groups (p = 0.001 and p = 0.001, respectively; Figure 4). The mean heart tissue NO level in the CoQ10 + PAM + atropine group was significantly lower than that of the sham and PAM + atropine groups (p < 0.001; Figure 5). The mean heart tissue MDA level in the CoQ10 + PAM + atropine group was significantly lower than that of the sham and PAM + atropine groups (p < 0.001; Figure 6).

The mean heart tissue AChE levels of groups. AChE: acetylcholinesterase.
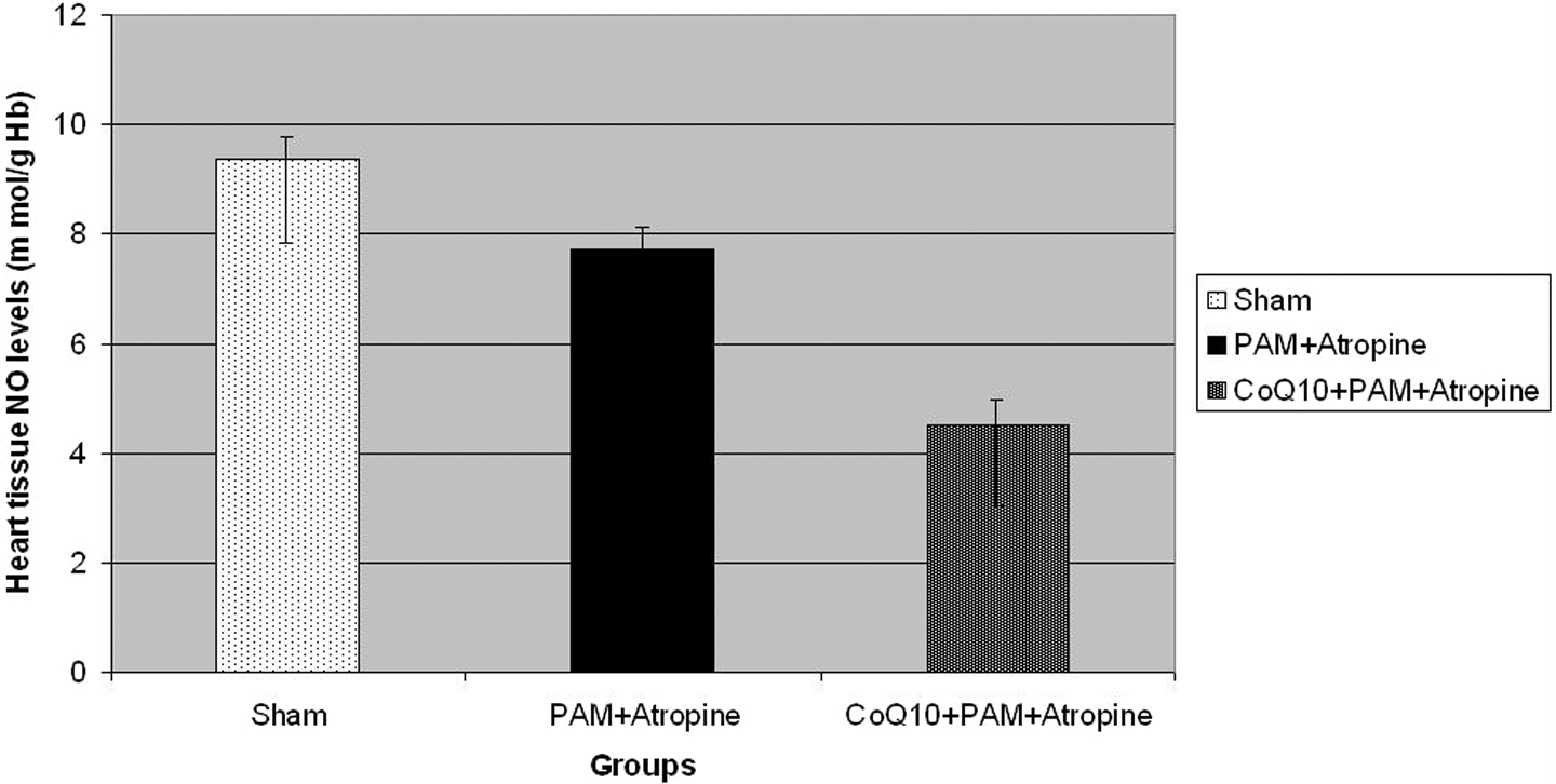
The mean heart tissue NO levels of groups. NO: nitric oxide.

The heart tissue MDA levels of groups. MDA: malondialdehyde.
Discussion
The mechanisms by which OP induces cardiotoxicity are still uncertain, but are thought to involve several systems. These include sympathetic over-activity, hypoxemia, acidosis, electrolyte derangements, and direct toxic effects of the compounds on the myocardium. 11
Recent findings indicate that at least some toxic manifestations of organophosphates are caused by enhanced production of ROS. 12 Superoxide anions, hydroxyl radicals, and H2O2 enhance the oxidative process and induce lipid peroxidative damage in cell membranes. Heart tissue may be susceptible to oxidative damage due to the presence of polyunsaturated fatty acids and oxygen, which may produce oxidative changes in myocytes. 13 MDA, the end product of lipid peroxidation, has been used as a measure of cardiotoxicity induced by free radicals and lipid peroxidation. 2
Atropine blocks the development of OP-induced cardiac lesions in a dose-dependent fashion. The clinical severity of OP poisoning and appearance of cardiac lesions are well correlated. Atropine has been found to diminish myocardial necrosis associated with OP toxicity. 14,15 In addition, a previous study found that atropine + PAM treatment reduced mortality. 16
Although all of the animals in the sham group died by 24 h post-poisoning, none of the animals in the PAM + atropine or PAM + atropine + CoQ10 groups died.
In an OP toxicity study in rats, heart tissues of animals receiving vitamins C and E had significantly lower MDA levels than the nontreated animals. 2 They reported that vitamins C and E altered antioxidant enzyme activity in heart tissue and decreased lipid peroxidation. In a study of subacute OP toxicity in rats, vitamin E decreased ultrastructural changes in heart tissue and MDA levels but could not completely protect against toxic effects. 17
In our study, we used CoQ10, a strong antioxidant, which can prevent myocardial destruction and decrease lipid peroxidation in heart tissue. In our study animals, both erythrocyte and heart tissue AChE levels were higher and the MDA levels are lower in the group treated with CoQ10 added to PAM + atropine compared with PAM + atropine treatment. This indicates that CoQ10 decreased lipid peroxidation in heart tissue and enhanced the protective effect of atropine against cardiac toxicity linked to OP poisoning.
In a rat model of OP toxicity, the cardioprotective antioxidants vitamin E and selenium resulted in increased heart tissue levels of AChE and decreased levels of MDA. 18 Thus, these antioxidants are promising agents against cardiotoxicity linked to OP poisoning. Similar results were also found in a subchronic OP toxicity study in rats that were given vitamins C and E; heart tissue MDA levels were significantly lower when compared with nontreated group. 19 They summarized that the vitamins C and E combination decreased heart damage and lipid peroxidation caused by organophosphates.
AChE enzyme reactivators (oximes) are used as an antidote in the standard treatment of OP poisoning along with atropine, especially in those with signs of cardiotoxicity. In our study, erythrocyte and AChE levels in heart tissue were higher in the PAM + atropine-treated group than in the nontreated sham group, and the levels in the group with CoQ10 added to this regimen were even higher. Besides, CoQ10 treatment could have increased PAM’s effect on erythrocyte and heart tissue. Altuntaş et al. 20 found that a single dose of OP significantly decreased both in vivo and in vitro AChE activity. While the combination of vitamins C and E did not have significant effect on AChE activity in vitro, they significantly increased in vivo AChE activity. They did not evaluate the effects of PAM or atropine in their study.
In a study of malathion toxicity in rats, magnesium magnetic isotope nanoCarrier (PMC16) was added to PAM + atropine treatment as an additional antioxidant. 21 PMC16 maintained higher levels of AChE activity and decreased ROS production more than PAM + atropine. However, they did not measure heart tissue erythrocyte or AChE activity or lipid peroxidation. They reported that cardiac toxic effects could be minimized by adding antioxidants to standard treatment for OP poisoning.
In our study, CoQ10 had beneficial effects when added to standard PAM + atropine treatment in this model of OP poisoning, as NO levels in erythrocytes and heart tissue were higher and MDA levels were lower in the group given with PAM + atropine when compared with those given with CoQ10 along with PAM + atropine. CoQ10 has been found to decrease lipid peroxidation, one of the most important pathophysiological mechanisms leading to tissue damage in OP poisoning.
The limitation of this study was the small number of subjects. However, our board of ethics did not allow a greater number of subjects to be used.
In this animal model of AOP, CoQ10 added to PAM + atropine was superior to PAM + atropine treatment alone in terms of oxidative effects in erythrocytes and heart tissue. In future studies in larger animals and humans, CoQ10 added to standard PAM + atropine treatment may prove to be a beneficial agent to minimize cardiac damage, morbidity, and mortality in severe OP poisoning.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.