Abstract
The modulating effect of peroxisome proliferator-activated receptor α ligand on haemodynamic effects of phenylepherine (PE), angiotensin II (AII), endothelin 1 (ET1), acetylcholine (Ach), sodium nitroprusside (SNP) and isoproterenol (ISO) were evaluated in glycerol-induced acute kidney injury in rats. The effect of PE on fenofibrate-treated animals was a dose-dependent increase in mean arterial blood pressure (MAP). For AII and ET1, MAP was also increased for the fenofibrate group but not in a dose-dependent fashion. On the medullary blood flow (MBF), while the lower doses of PE and AII increased the perfusion unit on the fenofibrate-treated group, the higher doses decreased the perfusion unit. The ET1 increased the perfusion unit on this group but not in dose-dependent fashion. The effects of PE and AII on the cortical blood flow (CBF) of fenofibrate-treated group is similar to that of MBF for the same group but not for ET1. The effect of Ach, SNP and ISO in all the groups was the decrease in MAP. ISO caused dose-dependent increase in MBF of fenofibrate-treated group. The effect of Ach, SNP and ISO on the CBF perfusion unit was that of the increase for the fenofibrate-treated group. The study showed that fenofibrate did not attenuate increased blood pressure induced by PE, AII and ET1 but caused enhanced vasodilation by Ach, SNP and ISO.
Introduction
Glycerol-induced acute renal failure (ARF) is characterized by myoglobinuria and tubular necrosis. 1 In addition, ARF is characterized by increased vasoconstriction. 2 ARF now known as acute kidney injury (AKI) represents a significant and devastating problem in clinical medicine. 3,4 The incidence of AKI varies from 5% of hospitalized patients to 30–50% of patients in intensive care units, and there is now substantial evidence that the incidence is rising at an alarming rate. 5 Despite significant improvements in therapeutics, the mortality and morbidity associated with AKI remain high. Outstanding advances in basic research have illuminated the pathogenesis of AKI and have paved the way for successful therapeutic approaches in animal models. 3 However, translational research efforts in humans have yielded disappointing results. 6
Peroxisome proliferator-activated receptors (PPARs) are members of the nuclear hormone receptor superfamily of ligand-activated transcription factors that are related to retinoid, steroid and thyroid hormone receptors. 7 The PPAR subfamily consists of three members: PPAR-α, PPAR-β(δ) and PPAR-γ. 2 Most tissues in humans and rodents have all three receptor subtypes, although there is considerable variability in the relative expression. 7
PPAR-γ is expressed in adipose tissue, 8 endothelial cells 9 and vascular smooth muscle cells (VSMCs). 10 Activation of PPAR-γ in VSMCs inhibits proliferation and migration, the release of matrix-degrading enzyme, matrix-metalloproteinase-9 expression and oxidative stress, suggesting its role in vascular function. 1 The activation of PPAR-γ suppresses the transcription of angiotensin type-1 receptor, 11 decreases blood pressure, 12 ameliorated microalbuminuria 13 and decreases insulin resistance. 14 These observations indicate that a role exists for PPAR-γ in the regulation of renal and systemic haemodynamics and that PPAR-γ ligands can protect against renal injury.
The PPAR-α is a fatty acid-activated nuclear receptor that plays a key role in the transcriptional regulation of genes involved in cellular lipid and energy metabolism. PPAR-α is expressed in tissues with very active fatty acid metabolism such as the heart, kidney, liver and the endothelium 15 and in VSMCs, 16 suggesting that PPARα may exert direct effects on the vascular wall. 17 Fatty acids and their derivatives have been identified as natural ligands for PPAR-α, and PPAR-α is the primary target of numerous classes of synthetic ligands, including lipid-lowering fibrates and gemfibrozil. 18
Although the effect of fibrates is generally attributed mainly to their hypolipidemic and high-density lipoprotein-raising actions, they possess other actions on the vasculature such as anti-inflammatory, antiproliferative and renoprotective effects that may contribute to their beneficial effects. 18 –21 PPAR-α activation therefore reduced arterial low-density lipoprotein accumulation and endothelial layer permeability in the arterial wall in angiotensin II (AII)-induced vascular inflammation and atherosclerosis in mice. 22
PPAR-α together with PPAR-δ and PPAR-γ forms a subgroup within the nuclear receptor superfamily. 14 In contrast to PPAR-α, which is involved in the control of cellular lipid utilization, PPAR-γ has been shown to be a necessary component of the adipocyte differentiation program. 14
The biological function of PPAR-δ is unknown. A diverse group of compounds can act as activating ligands for PPAR-α including several prostaglandin derivatives, eicosanoids and long-chain unsaturated fatty acids. 1 To date, the majority of PPAR-α target genes identified are involved in cellular fatty acid oxidation. 2
Although the modulating effects of PPAR-α on haemodynamic effects elicited by AII under normal conditions have been studied; efforts were therefore made in this study to evaluate the haemodynamic effects of PPAR-α ligand, fenofibrate in glycerol-induced renal failure in rats. The study thus assessed the effects of a ligand of these nuclear hormone receptors on three vasoconstrictors and three vasodilators and hence its ability to ameliorate high blood pressure in ARF setting.
Materials and methods
Unless otherwise stated, all chemicals were obtained from Sigma-Aldrich (St Louis, Missouri, USA). Fenofibrate was prepared in mineral oil, while 50% glycerol was prepared in double distilled water. The vasoconstrictors phenylepherine (PE), AII and endothelin 1 (ET1) and the vasodilators acetylcholine (Ach), sodium nitroprusside (SNP) and isoproterenol (ISO) were kept on ice during the experiments.
Animal studies
The studies were carried out using adult male albino rats (age 9–12 months old; weight 350 ± 5 g) obtained from Harlan Sprague-Dawley (Houston, Texas, USA). The animals were maintained on a standard rat feed (Purina Chowo; Harlan Sprague-Dawley) and allowed ad libitum access to water and food until the day when the haemodynamic experiments will be conducted. Animals were randomly divided into the following groups: Control groups comprised rats treated with mineral oil (vehicle, 1 ml/kg) (n = 7) and double distilled water (n = 7). On the other hand, the ARF groups comprised (a) ARF rats treated with PPAR-α ligand (fenofibrate; n = 7) and (b) ARF-untreated rats or glycerol (n = 9).
Induction of ARF in rats
ARF was induced using the standard method involving administration of 50% glycerol (v/v, 4 ml/kg, intramuscularly). The 50% glycerol was obtained by dissolving glycerol in double distilled water. After 7 days of study period in which the different groups received their specific treatment orally, the required amount of glycerol was administered as a deep intramuscular injection equally distributed to both hind legs. Rats were deprived of food and water for 24 h after glycerol administration. 1 After injecting glycerol, rats were placed in individual metabolic cages for collecting 24 h urine sample used for biochemical analysis.
Biochemical analysis
Renal function of the rats was assessed to determine the extent of renal failure by measuring urinary excretion of protein. Urinary protein was measured spectrophotometrically using a kit from Sigma.
Physiological (haemodynamic) measurements
In every group, rats were anaesthetized with intraperitoneal injection of inactin (100 mg/kg; Research Biochemicals International, Natick, Massachusetts, USA) and placed on a heated platform to maintain body temperature at 37°C. A tracheostomy (PE 250; Becton Dickinson and Co., Sparks, Maryland, USA) was performed for spontaneous ventilation and a cannula (PE 250; Becton Dickinson and Co., Sparks, Maryland, USA) was placed in the right carotid artery to monitor blood pressure. A tail vein was also cannulated with a 23-G butterfly needle (Abbott Hospitals Inc., Illinois, USA) for drug administration. The left lateral side of each animal was incised so as to expose the left kidney since this was closer to the surface than its right counterpart. The mean arterial blood pressure (MAP) was measured with a pressure transducer (Statham model P231D; Statham, Oxnard, California, USA) connected to a signal manifold (Transbridge, model TBM-4; World Precision Instruments, Sarasota, Florida, USA) and recorded on a data acquisition system (Model D1720; DataQ Instruments, Akron, Ohio, USA). The cortical blood flow (CBF) was measured by a laser Doppler flow meter (Periflux System 5000, version 1.20; Perimed, Stockholm, Sweden) via a surface probe. 18 Medullary blood flow (MBF) was also measured by a laser Doppler flow meter via fibre optic that was inserted into the kidney.
Following instrumentation as detailed above, anaesthetized rats were allowed to equilibrate for 45 min, after which the effects of vasoconstrictors (PE, AII and ET1) and vasodilators (Ach, SNP and ISO) were tested for the MAP, MBF and CBF.
Data analysis
Haemodynamic responses were recorded as changes (Δ) relative to baseline (preinjection values). Data were expressed as the mean ± SEM and were subjected to analysis of variance. Significant means were separated using Duncan Multiple Range Test of SAS (1999). Bars with different superscripts a, b and ab are significantly different at p < 0.05, while bars with similar superscript are not significantly different at p < 0.05. All the groups were compared with one another.
Results
Biochemical analysis
Results of the urinary protein showed that ARF was established for the mineral oil and ARF-untreated groups, because on day 1, the urinary protein was 19 and 16 mg/ml, respectively, whereas after glycerol administration, the urinary protein concentration was 59 and 62 mg/ml, respectively. The ARF-untreated group also followed this pattern but not so for the fenofibrate group (Table 1).
Urinary protein concentration of different groups in this study (X ± SEM).

ARF: acute renal failure.
Effects of drugs on MAP
PE caused a dose-dependent increase in the MAP of fenofibrate group. This effect is similar in other groups. The effects of AII and ET1 were similar to that of PE, although less in magnitude. The effect of Ach on MAP was that of decrease but this was more pronounced in the mineral oil group than the others. The 10 ng/kg dose of SNP caused a more lowering of the blood pressure in the fenofibrate group than the others. This is similar to the 30-µg/kg dose of ISO that caused a more lowering of the MAP in the fenofibrate group than others in this category (Figures 1 to 6).

Effects of PE on MAP of rats. Note that bar with different superscripts are significantly different at p < 0.05. PE: phenylepherine; MAP: mean arterial blood pressure.

Effects of AII (ng/kg) on MAP of rats. Note that bar with different superscripts are significantly different at p < 0.05. AII: angiotensin II; MAP: mean arterial blood pressure.

Effects of ET1 (ng/kg) on MAP of rats. Note that bar with different superscripts are significantly different at p < 0.05. ET1: endothelin 1; MAP: mean arterial blood pressure.

Effects of Ach (ng/kg) on the MAP of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. Ach: acetylcholine; MAP: mean arterial blood pressure.

Effects of SNP (ng/kg) on the MAP of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. SNP: sodium nitroprusside; MAP: mean arterial blood pressure.

Effects of ISO (µg/kg) on the MAP of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. MAP: mean arterial blood pressure; ISO: isoproterenol.
Effects of drug on MBF
The effects of PE on the MBF of rats in the different groups did not follow a definite pattern. The 10-µg/kg dose of PE experienced the highest increase in perfusion unit in the glycerol group. Meanwhile, the 30-µg/kg dose also caused the least perfusion unit in the fenofibrate group. The effect of AII followed the pattern of PE to some extent, but for ET1, the 30-ng/kg dose caused the highest perfusion unit in the mineral oil group and the 100-ng/kg dose caused the least perfusion unit in the glycerol group. While the Ach and SNP caused more pronounced perfusion unit in the distilled water group, the effect of ISO on the MBF is that of dose-dependent increase in the fenofibrate group. It must be stated that 30 ng/kg dose of Ach (Figure 10) caused significant difference in perfusion unit in fenofibrate-treated animals relative to other groups and doses except that of the 30-ng/kg dose of the distilled water group (Figures 7 to 12).

Effects of PE (µg/kg) on the MBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. PE: phenylepherine; MBF: medullary blood flow.

Effects of AII (ng/kg) on the MBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. AII: angiotensin II; MBF: medullary blood flow.

Effects of ET1 (ng/kg) on the MBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. ET1: endothelin 1; MBF: medullary blood flow.
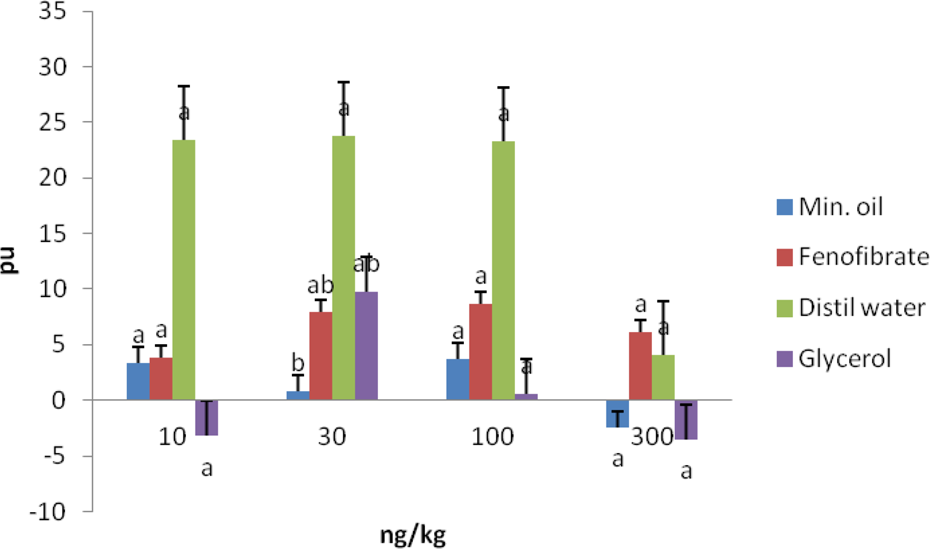
Effects of Ach (ng/kg) on the MBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. Ach: acetylcholine; MBF: medullary blood flow.

Effects of SNP (ng/kg) on the MBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. SNP: sodium nitroprusside; MBF: medullary blood flow.

Effects of ISO (µg/kg) on the MBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. MBF: medullary blood flow; ISO: isoproterenol.
Effect of drugs on CBF
While the 3-µg/kg dose of PE caused the highest level of perfusion units of rats in the fenofibrate group, the 30-µg/kg dose also caused the least perfusion unit (−40 mmHg) in the same group. The effect of AII on the CBF is more pronounced on the mineral oils and the fenofibrate groups, whereas it caused least perfusion units in the 30 and 100 ng/kg doses. The ET1 caused increased perfusion unit in the fenofibrate group in almost a dose-dependent fashion. The effect of Ach on CBF is that of increase in a dose-dependent manner in the fenofibrate group. In the case of SNP, while the 10-ng/kg dose caused highest perfusion unit in all the groups, it was the 100-ng/kg dose that caused the least perfusion unit in the fenofibrate and distilled water groups. The effect of ISO is that of dose-dependent decrease in the perfusion unit in the fenofibrate group. In other words, the 1-µg/kg dose caused more perfusion unit than the others (Figures 13 to 18).

Effects of PE (µg/kg) on the CBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. PE: phenylepherine; CBF: cortical blood flow.

Effects of AII (ng/kg) on the CBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. AII: angiotensin II; CBF: cortical blood flow.

Effects of ET1 (ng/kg) on the CBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. ET1: endothelin 1; CBF: cortical blood flow.

Effects of Ach (ng/kg) on the CBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. Ach: acetylcholine; CBF: cortical blood flow.
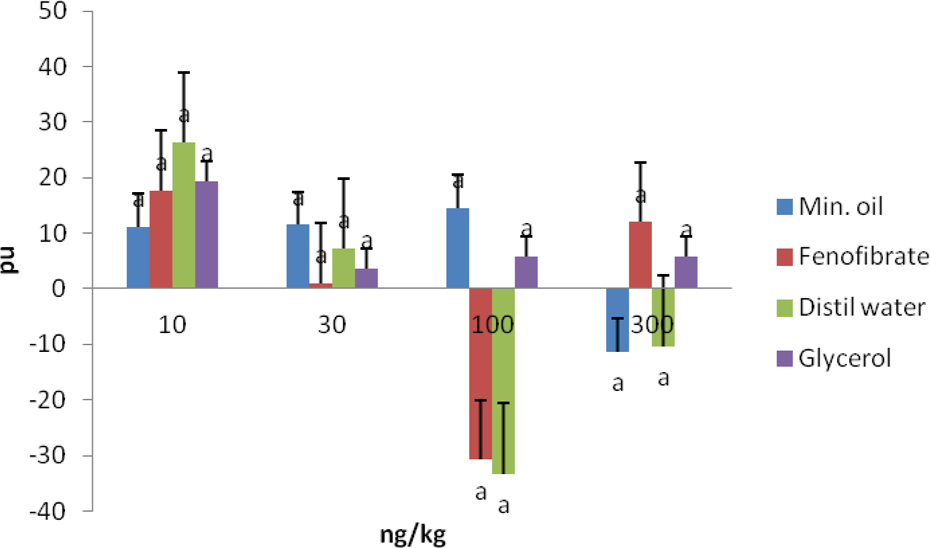
Effects of SNP (ng/kg) on the CBF of rats. Note that bar with similar superscripts are not significantly different at p < 0.05. SNP: sodium nitroprusside; CBF: cortical blood flow.

Effects of ISO (µg/kg) on the CBF of rats. Note that bar with different superscripts are significantly different at p < 0.05. CBF: cortical blood flow; ISO: isoproterenol.
Discussion
In this study, we have demonstrated that the administration of 50% glycerol could actually lead to the development of AKI as evidenced by high urinary protein. Glycerol-induced ARF is characterized by myoglobinuria and tubular necrosis. 1 Glycerol-induced ARF is a phenomenon widely recognized experimental model of myoglobinuric nephropathy, and the renal vasoconstriction seen in the initial phase of the injury is considered as an important pathogenic event. 2 Increased renal production of AII and thromboxane A2 (TxA2) have been reported suggesting a role for AII and TxA2 as mediators of the vasoconstriction and the decrease in renal function seen after glycerol administration. 2,23 However, it is interesting to note that 24 h after glycerol administration to the fenofibrate group, the urinary protein actually experienced a decrease in its level when compared with day 7 of drug administration. This observation would give impression that ARF was not developed. However, one would like to state that fenofibrate like other PPAR activators have anti-inflammatory activities. 18 Studies showed that PPAR activation confers cardioprotective effects that seem to involve modulation of the nitric oxide (NO)/NO synthase system. 24 –26 In this regard, clinical observations showing that PPAR-α activators lower the progression of atherosclerotic lesions 25,26 was ascribed to increased NO, which is known to be antiatherogenic and anti-inflammatory. 27 This observation is consistent with the fact that PPAR-α activators improved endothelial-mediated NO vasodilation. 19 In other words, PPAR-α prevented the development of hypertension and improved myocardial inflammation and collagen deposition in angiotensin-infused rats. 20 Therefore, this suggested that a role exists for PPAR-α as a target for prevention and treatment of myocardial disease. The focus of this study, however, is to actually evaluate the renoprotective effect of fenofibrate, a PPAR-α activated ligand.
The involvement of several vasoconstrictors, including ET1 28 and AII, 29 in haemodynamic changes has been identified. Namely, it has been reported that renal injury induces synthesis of endogenous ET1, which is then able to ensure continuation of its own production, after the initial injury ends. In addition, ET1 has greater vasoconstrictor activity in the renal vasculature than in any other vascular bed and it has direct glomerular and tubular effects. 30 At high concentrations, ET1 induces severe reduction in renal blood flow and glomerular filtration rate (GFR), a fall in the urinary flow rate and a decrease in urinary sodium excretion. ET1 is also able to contract mesangial cells. Renal effects and mesangial cell contraction induced by ET 31 are mediated, at least in part, by platelet-activating factor. AII is also a potent intrarenal vasoconstrictor. It modulates the GFR by directly affecting the tone of both efferent and afferent arterioles, but it also affects mesangial cell function. 29 It was, therefore, in the context of these haemodynamic changes, we set out to evaluate the effect of fenofibrate on AII- and ET1-induced high blood pressure and hence changes in the medullary and CBF.
In this study, after 7 days of fenofibrate administration and thereafter glycerol-induced acute kidney failure, fenofibrate did not blunt high blood pressure in rats when PE, AII and ET1 were administered at different times. As a matter of fact, the MAP was highest in the fenofibrate group when PE, AII and ET1 were administered. This may corroborate the reports of Iglarz et al. 32 that in deoxycorticosterone acetate (DOCA)-salt hypertensive rats, blood pressure increase and endothelial dysfunction were prevented in part by the PPAR-γ activator, rosiglitazone, but not by the PPAR-α activator fenofibrate. Both PPAR activators, however, prevented the enhanced production of reactive oxygen species and the hypertrophic remodeling typically found in blood vessels of DOCA-salt hypertensive rats, consistent with their anti-inflammatory effects.
All the vasodilators used in this study were able to achieve enhanced vasodilation in the presence of fenofibrate. This further corroborates the cardioprotective effect of this agent. With respect to MBF changes, fenofibrate in the presence of PE and AII caused a mixed reaction in that at lower doses, there is increase in pulsation unit and vice-versa. The effect of ET1 and the three vasodilators was similar. Since MBF is a region of low oxygen supply, increasing pulsation may further lead to hypoxia and hence could lead to ischaemia. It is good to note, however, that at higher doses of the vasoconstrictors, the pulsation unit decreased. The effect of fenofibrate on the CBF in the presence of the vasoconstrictors (PE and AII) is similar to that of MBF. In the case of ET1 and the three vasodilators, the pulsation unit experienced increased level and this may to some extent bring about vasodilation and hence renoprotective effect. It should be noted that the cortex constitutes the major portion of the kidney and receives a disproportionately higher percentage (90%) of blood flow compared with the medulla (about 6-10%) or papilla (1–2%). Thus, when a blood-borne toxicant is delivered to the kidney, a high percentage of the material will be delivered to the cortex and will have greater opportunity to influence cortical rather than medullary or papillary functions. 33
It was expected that fenofibrate will give a pronounced renoprotective effect; however, the short duration of 7 days as against the usual duration of 25 weeks may have affected this. 18 Again, although PPAR-α is expressed in vascular tissues, it is lacking in renal vasculature. 34 These and others may have accounted for seemingly ineffective haemodynamic changes seen in this study.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
