Abstract
The present study was designed to evaluate the cardioprotective potential of lycopene (LCP) against isoproterenol (ISP)-induced myocardial infarction (MI), by assessing hemodynamic, biochemical and histopathological parameters. Wistar male albino rats were orally administered with LCP (0.5, 1.0 and 1.5 mg/kg) or with vehicle for 30 days, with concurrent subcutaneous injections of ISP (85 mg/kg) on days 28 and 29. ISP significantly (p < 0.05) decreased systolic, diastolic and mean arterial blood pressure (SAP, DAP and MAP, respectively) and heart rate (HR). ISP also decreased contractility (+LVdP/dt), relaxation (−LVdP/dt) and increased left ventricular end-diastolic pressure (LVEDP). In addition to functional impairment, ISP also caused a significant (p < 0.05) decrease in antioxidants, namely, superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSHPx), glutathione (GSH), cardiac injury marker enzymes, creatine phosphokinase-MB (CK-MB) and lactate dehydrogenase (LDH), as well as induced lipid peroxidation, malonaldialdehyde (MDA) and histopathological alterations in heart. However, pretreatment with LCP significantly (p < 0.05) attenuated ISP-induced cardiac dysfunction as evidenced by improved SAP, DAP, MAP, HR, (±)LVdP/dt and reduced LVEDP. Pretreatment with LCP also significantly (p < 0.05) prevented the depletion of antioxidants (SOD, CAT, GSHPx and GSH), myocyte injury marker enzymes (CK-MB and LDH) and inhibited lipid peroxidation and MDA formation in the heart. Furthermore, reduced necrosis, edema and infiltration of inflammatory cells on histopathological examination also depicted the protective effect of LCP against the deleterious effect of ISP. Based on the results, it is suggested that LCP possesses significant cardioprotective potential and may serve as an adjunct in treatment and prophylaxis of MI.
Introduction
Myocardial infarction (MI) continues to be an immense health problem causing mortality and morbidity across the developing and developed countries despite improved clinical care and public awareness. 1 It occurs when there is insufficient blood supply to the myocardium leading death of cardiomyocytes, a condition known as ischemia. Prolonged ischemia of the myocardium leads to necrosis, which is usually followed by many pathophysiological, functional, biochemical and electrocardiographic changes. In ischemia, free radicals and reactive oxygen species (ROS) are the central components of cellular oxidative damage, which culpably affects the myocardium. 2
Thus, a great deal of research is focused on the role of antioxidants in the prevention of many human diseases, particularly atherosclerosis, congestive heart failure and myocardial ischemia-reperfusion injury. 3,4 Numerous epidemiological, clinical and experimental studies have shown that antioxidants may slow down the progression and consequences of MI. 5 –7
In recent years, many nutrient- and non-nutrient-based natural antioxidants such as phytoestrogens in soy, epigallocatechin in green tea, anthocyanins in red berries, resveratrol in grape seeds and lycopene (LCP) in tomatoes are increasingly being recognized as potential health promoters in reducing the risk of cardiovascular diseases (CVDs). 8 LCP is one of over 600 carotenoids found in tomato, pink grapefruit, guava, papaya and watermelons, which impart a characteristic red pigmentation. Since humans are unable to synthesize carotenoids de novo, they usually acquire it from the dietary intake. 9 Chemically, LCP is a lipid soluble and highly unsaturated hydrocarbon containing 11 conjugated and 2 unconjugated double bonds, normally in the all-trans configuration (Figure 1). It is an acyclic isomer of β-carotene. 10 LCP has been shown protective against myocardial ischemic-reperfusion injury, 11 doxorubicin-induced cardiotoxicity, 12 cerebral ischemia 13 and neurodegenerative diseases. 14
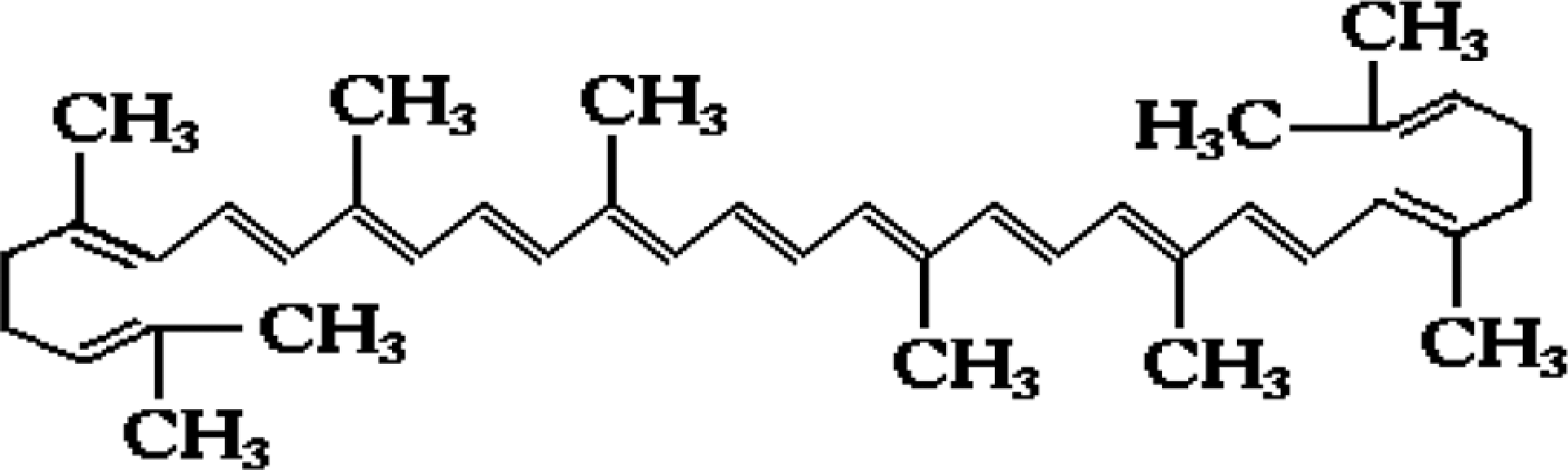
Chemical structure of lycopene.
Numerous epidemiological studies and supplementation human trials have shown an inverse relationship between the consumption of carotenoid rich diet and the risk of cancer and coronary heart diseases. 15 –17 The reduction in CVD risk factors has been shown to be associated with dietary intake, serum or plasma concentrations and adipose tissue concentration of carotenoids. In a multicenter, case-control study, European Study of Antioxidants, Myocardial Infarction and Cancer of the Breast (EURAMIC), the foods rich in LCP has been found to reduce the risk of MI. 15 In another study, a reduction in the intimal wall thickness and risk of MI has been reported in persons with higher adipose tissue concentrations of LCP. 16 Despite the emerging evidences suggesting a causal–effect relationship on the possible role of LCP in the primary prevention of CVD, there is paucity of data on its role in myocardial ischemic injury. 15 –19 Also, the mechanism of its protective action to substantiate its therapeutic efficacy still lacks significant data.
Isoproterenol (ISP), a synthetic catecholamine and β-adrenergic agonist, has been known to produce MI in rat heart owing to free radical generation due to auto-oxidation and positive inotropic and chronotropic effects. 20,21 Since pathophysiological, biochemical, morphological, functional alterations and histopathological lesions following ISP administration in rats are similar to those in human MI, the ISP-induced MI serves as a well-standardized model to study the beneficial effects and mechanism of many drugs. 22,23
Therefore, the aim of present study was to evaluate the cardioprotective potential of LCP in the experimental model of myocardial ischemic injury. ISP was used to produce MI in experimental rats and LCP was administered before and at the onset of ischemia. In order to demonstrate the mechanism of cardioprotection, the hemodynamic and ventricular function parameters and biochemical markers of myocardial injury as well as antioxidants and lipid peroxidation were assessed in heart. Further, histopathological studies were also incorporated to support the biochemical and hemodynamic effects of LCP.
Materials and methods
Drugs and chemicals
The antioxidant enzyme standards and ISP hemisulfate were obtained from Sigma Chemicals Company (St Louis, Missouri, USA). LCP was the generous gift by Jagsonpal Pharmaceuticals Limited (New Delhi, India). All chemicals used in the study were of analytical grade. Double distilled water was used for the biochemical estimations. The doses of LCP in the present study have been selected based on the previous reported literature. 11
Experimental animals
Laboratory bred, 10–12 weeks old, Wistar male albino rats, weighing 150–200 g were used in the study. The study protocol was reviewed and approved by the Institutional Animal Ethics Committee and conforms to the Committee for the Purpose of Control and Supervision on Experiments on Animals. Animals were obtained from the Central Animal House Facility of All India Institute of Medical Sciences, New Delhi, India. A minimum of four rats were housed in the polypropylene cages (with stainless steel grill top) lined with husk, renewed every 24 h at standard laboratory conditions, natural light and dark cycles (approximately 14 h light/10 h dark), humidity (55 ± 10%) and an ambient temperature of 25 ± 2°C. All experiments were performed between 9.00 a.m. and 16.00 p.m. to maintain the physiological relevance of the circadian variation in measuring cardiac function. The animals were fed with standard pellet diet (Ashirwad Industries Limited, Chandigarh, India) and tap water ad libitum.
Induction of experimental MI
A standardized dose of ISP 85 mg/kg body weight was injected subcutaneously (s.c.) for two consecutive days, at 24 h interval to induce MI in rats. 19
Experimental design
The animals were acclimatized 1 week before the experiments and randomly allocated into eight experimental groups, with each consisting 12 rats. Group I (Sham): rats were administered with the vehicle orally using intragastric tube for 30 days and on 28th and 29th day administered with saline (0.5 ml s.c.) at an interval of 24 h. Group II (ISP control): rats were administered with the vehicle orally using intragastric tube for 30 days and on 28th and 29th day administered with ISP (85 mg/kg s.c.) at an interval of 24 h. Groups III–V (LCP): rats were administered with LCP orally using intragastric tube for 30 days and on 28th and 29th day administered with saline (0.5 ml s.c.) at an interval of 24 h. Groups VI–VIII (LCP + ISP): rats were administered with LCP orally using intragastric tube for 30 days and on 28th and 29th day administered with ISP (85 mg/kg s.c.) at an interval of 24 h. Changes in the body weight of each rat have been recorded weekly.
Surgical preparation and hemodynamic measurement
For all experimental groups, rats were anesthetized with pentobarbitone sodium (60 mg/kg, intraperitoneally). Atropine (0.1 mg/kg) was coadministered with the anesthetic to reduce bronchotracheal secretions and keep the heart rate (HR) elevated during surgery. Body temperature was monitored and maintained at 37°C throughout the experimental protocol. Tracheostomy was performed to ventilate rats with room air from a positive pressure ventilator (Inco, Ambala, India) using compressed air at a rate of 90 strokes/min and a tidal volume of 10 ml/kg. Ventilator setting and oxygen were adjusted to maintain arterial blood gas parameters within the physiological range.
Left jugular vein was cannulated with polyethylene tube for continuous infusion of 0.9% saline. Right carotid artery was cannulated and filled with heparinized saline and connected with CARDIOSYSCO-101 (Experimentria, Hungary) using a pressure transducer for the measurement of hemodynamic parameters, systolic, diastolic and mean arterial blood pressure (SAP, DAP and MAP, respectively) as well as HR. Left thoracotomy was performed at the fifth intercostal space and the heart was exposed for the measurement of left ventricular function. A wide bore (1.5 mm) sterile metal cannula connected to a pressure transducer (Gould Statham P23ID, USA) was inserted into the cavity of left ventricle from the posterior apical region of heart for recording left ventricular pressure dynamics; left ventricular end-diastolic pressure (LVEDP), a surrogate marker of preload, peak positive pressure development ((+)LVdP/dt: marker of myocardial contraction) and peak negative pressure development ((−)LVdP/dt: marker of myocardial relaxation) representing contractility and relaxation using Polygraph (Grass 7D, USA). After insertion of transducer, the heart was managed in its normal position in thorax. Thoracic cavity was covered with saline-soaked gauze to prevent heart from drying. Before recording the hemodynamic and ventricular function, animals were allowed to stabilize for 5 min and the hemodynamically unstable rats were excluded from the study. After recording hemodynamic and ventricular contractile function, animals were killed for biochemical and histological studies under the overdose of anesthesia.
Biochemical estimations
All biochemical parameters were estimated in heart tissues. A 10% homogenate of heart was prepared in phosphate buffer (50 mM, pH 7.4), and aliquots of 0.5 ml were used for the estimation of reduced glutathione (GSH) 24 and malondialdehyde (MDA). 25 Remaining homogenate was centrifuged at 7000 r/min for 15 min and the obtained supernatant was used for the estimation of endogenous antioxidant enzymes, such as superoxide dismutase (SOD), 26 catalase (CAT), 27 glutathione peroxidase (GSHPx) 28 and myocytes injury marker enzymes, as well as creatine phosphokinase-MB isoenzyme (CK-MB), 29 lactate dehydrogenase (LDH) 30 and protein. 31
Histological studies of myocardium
Myocardial tissue was immediately fixed in 10% formalin solution. The fixed tissues were embedded in paraffin and serial sections were cut. After hematoxylin and eosin staining, the sections were examined under light microscope (Nikon, Tokyo, Japan).
Statistical analysis
All the data were analyzed using one-way analysis of variance with post hoc analysis and Bonferroni’s multiple range test. Mean and SD were calculated for biochemical and hemodynamic variables in each group. p < 0.05 was considered statistically significant.
Results
Mortality
A total of 96 rats were included in the study, and the data obtained for 84 rats were used in this study. The hemodynamically unstable (8.5%) rats were excluded from the study. The mortality rate of 4.4% was observed in the experimental period. The reasons for mortality and hemodynamic instability were blood loss during cannulation.
Per se effect of LCP
LCP per se treatment (0.5, 1.0 and 1.5 mg/kg) did not show significant change on hemodynamic, biochemical and histopathology parameters when compared with Sham group.
Effect of LCP on hemodynamic function
A significant decrease in SAP, DAP, MAP and HR was observed in ISP-control animals as compared to Sham group (Table 1). Pretreatment with LCP at 1.0 and 1.5 mg/kg significantly restored arterial pressure such as SAP, DAP and MAP in ISP treated rats. However, LCP 0.5 mg/kg failed to improve arterial pressure. Along with restoration of arterial pressure, pretreatment with LCP at all doses (0.5, 1.0 and 1.5 mg/kg) significantly increased the HR compared with ISP control group.
Hemodynamic parameters in different experimental groups.a

LCP: lycopene; ISP: isoproterenol; SAP: systolic arterial blood pressure; DAP: diastolic arterial blood pressure; MAP: mean arterial blood pressure; HR: heart rate.
aValues are mean ± SD (n = 6).
b p < 0.05 when compared with Sham.
c p < 0.05 when compared with ISP control.
Effect of LCP on left ventricular contractile function
Figures 2 and 3 represent the alteration in left ventricular contractile function in different experimental groups. ISP-control rats showed left ventricular dysfunction as evidenced by significant decline in the values of (+)LVdP/dt and (−)LVdP/dt as compared to Sham group. However, ISP-induced decline in (+)LVdP/dt and (−)LVdP/dt was significantly prevented by pretreatment with LCP at all doses, except 0.5 mg/kg which failed to improve (+)LVdP/dt significantly in comparison with ISP control animals. In addition, a significant rise in LVEDP was also observed in ISP control animals as compared to Sham group (Figure 4). LCP pretreatment at all doses (0.5–1.5 mg/kg) significantly decreased LVEDP when compared with ISP control group.
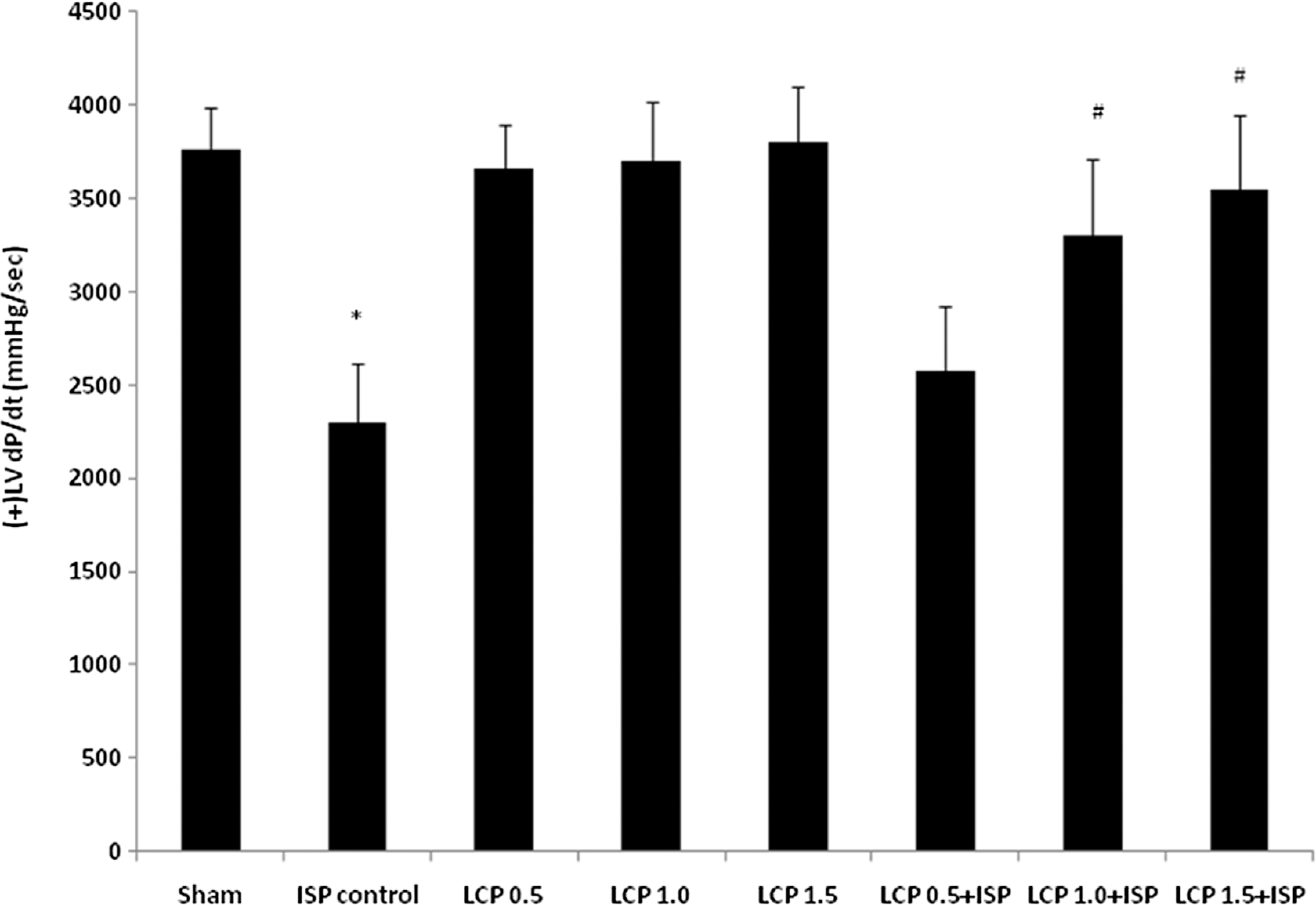
Effect of LCP on the contractility; (+)LVdP/dt in different experimental groups. Values are mean ± SD (n = 6). * p < 0.05 when compared with Sham; # p < 0.05 when compared with ISP control. LCP: lycopene; (+)LVdP/dt: marker of myocardial contraction.

Effect of LCP on the relaxation; (−)LVdP/dt in different experimental groups. Values are mean ± SD (n = 6). * p < 0.05 when compared with Sham; # p < 0.05 when compared with ISP control. LCP: lycopene; (−)LVdP/dt: marker of myocardial relaxation.

Effect of LCP on the LVEDP in different experimental groups. Values are mean ± SD (n = 6). * p < 0.05 when compared with Sham; # p < 0.05 when compared with ISP control. LCP: lycopene; LVEDP: left ventricular end diastolic pressure; ISP: isoproterenol.
Effect of LCP on myocardial injury markers
ISP administration showed a significant decrease in the myocyte injury markers, CK-MB isoenzyme and LDH enzyme as compared to Sham group (Table 2). However, LCP pretreatment at all doses (0.5, 1.0 and 1.5 mg/kg) significantly prevented the depletion of CK-MB and LDH enzymes from heart in comparison with ISP control group.
Myocytes injury marker enzymes in myocardium of different experimental groups.a
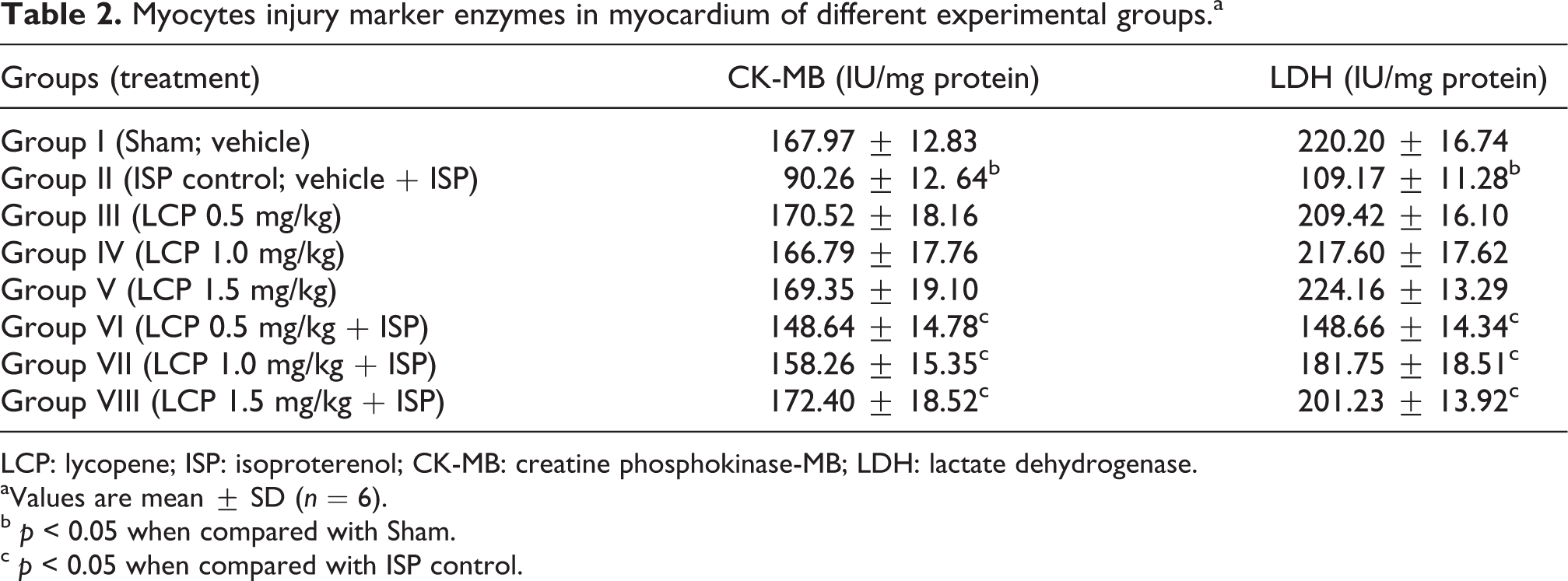
LCP: lycopene; ISP: isoproterenol; CK-MB: creatine phosphokinase-MB; LDH: lactate dehydrogenase.
aValues are mean ± SD (n = 6).
b p < 0.05 when compared with Sham.
c p < 0.05 when compared with ISP control.
Effect of LCP on reduced GSH and lipid peroxidation
In ISP-control group, a significant decrease in GSH content was observed in comparison with Sham group. Subsequent to the depletion of GSH, an increase in lipid peroxidation product, MDA was observed as compared to Sham group (Table 3). LCP pretreatment at 1.0 and 1.5 mg/kg significantly prevented the depletion of GSH from heart and LCP at all doses (0.5, 1.0 and 1.5 mg/kg) inhibited lipid peroxidation as evidenced by reduced MDA levels, concomitant to increased GSH when compared with ISP-control animals. However, LCP 0.5 mg/kg did not show significant effect on the GSH.
Glutathione and lipid peroxidation in myocardium of different experimental groups.a
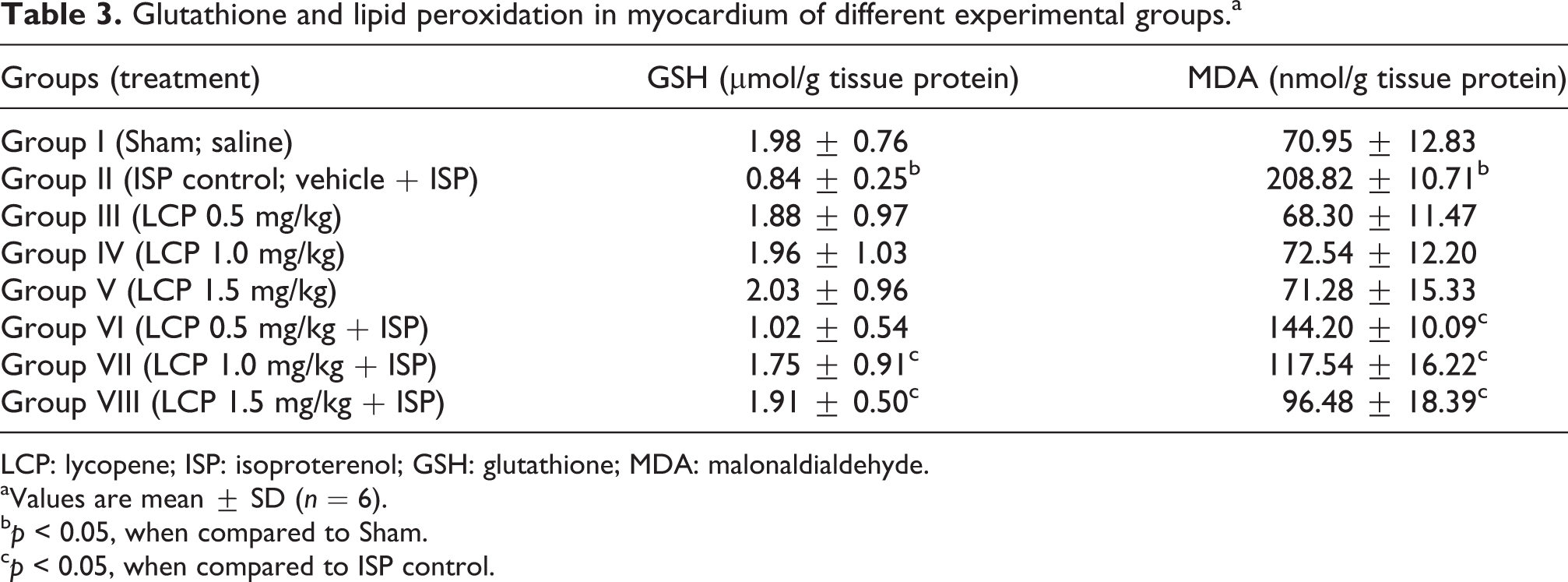
LCP: lycopene; ISP: isoproterenol; GSH: glutathione; MDA: malonaldialdehyde.
aValues are mean ± SD (n = 6).
b p < 0.05, when compared to Sham.
c p < 0.05, when compared to ISP control.
Effect of LCP on the activities of SOD, CAT and GSHPx enzymes
In ISP control group, a significant decrease in the values of SOD, CAT and GSHPx enzymes was observed as compared to Sham group (Table 4). Pretreatment with LCP at all doses (0.5, 1.0 and 1.5 mg/kg) significantly prevented the decline of antioxidant enzymes in a dose-dependent manner when compared with ISP control group.
Antioxidant enzymes in myocardium of different experimental groups.a

LCP: lycopene; ISP: isoproterenol; SOD: superoxide dismutase; CAT: catalase; GSHPx: glutathione peroxidase.
aValues are mean ± SD (n = 6).
b p < 0.05 when compared with Sham.
c p < 0.05 when compared with ISP control.
Effect of LCP on histopathological changes of rat myocardium
Figure 5(a) to (e) shows the light micrograph of myocardium of rats in different experimental group. Histopathological changes were evaluated by a pathologist, All India Institute of Medical Sciences, New Delhi, who was masked to the treatments and biochemical results. The observations were graded and summarized in Table 5. Sham group showed a normal myocardial histoarchitecture (Figure 5(a)) and ISP-control group showed confluent necrosis of muscle fibers, edema and increased connective tissue among myocardial fibers along with extravasations of RBCs and infiltration of the inflammatory cells (Figure 5(b)). LCP (0.5, 1.0 and 1.5 mg/kg) per se did not produce histopathological alterations in the myocardium. Rats pretreated with LCP 0.5 mg/kg, showed a mild degree of myocardial damage (Figure 5(c)). However, rats pretreated with LCP 1.0 and 1.5 mg/kg exhibited a remarkable protection from deleterious effects of ISP as evidenced by reduced edema and absence of myocardial necrosis resembling to the normal myocardial histoarchitecture (Figure 5(d) and (e)).

Light micrograph (hematoxylin and eosin; ×100) of the myocardium of the rat of (a) Sham group showing normal architecture of myocardium, (b) ISP-control group showing confluent focal necrosis of myofibrils and edema with infiltration of inflammatory cells and extravasations of red blood cells, (c) LCP 0.5 mg/kg + ISP showing slight myocardial necrosis and edema with inflammatory cells, (d) LCP 1.0 mg/kg + ISP showing lesser edema and myocardial necrosis, (e) LCP 1.5 mg/kg + ISP showing normal myocardial architecture with lessened necrosis and edema. LCP: lycopene; ISP: isoproterenol.
Histopathological changes in myocardium of different experimental group.

LCP: lycopene; ISP: isoproterenol; –: nil; +: mild; ++: moderate; +++: severe.
Discussion
The results of present study demonstrated the preventive effect of LCP in the animal model of ISP-induced MI. LCP exhibited its cardioprotective effects by improving cardiac function, reducing lipid peroxidation and strengthening the endogenous defense system of rat myocardium. Also, LCP pretreatment salvaged myocardium against ISP-induced myocardial necrosis as observed in histopathological examination.
Catecholamines are important regulators of myocardial metabolism and function. Excessive level of catecholamines have been known to cause cellular damage as observed in clinical conditions like angina, acute coronary insufficiency and MI. 32 Generation of oxygen free radicals (OFRs) and ROS through auto-oxidation of catecholamines has been recognized as one of the important causative factors. 21,22 Numerous studies have demonstrated that ISP-induced oxidative ischemic injury of myocardium is associated with deterioration of hemodynamic and left ventricular contractile function. 21,32,33 LCP pretreatment in present study have significantly prevented ISP-induced systolic and diastolic dysfunction as evidenced by improved SAP and DAP and subsequent increase in MAP, a marker of after load. LCP pretreatment also attenuated left ventricular dysfunction as evidenced by improved inotropic ((+)LVdP/dt) and lusitropic ((–)LVdP/dt) states of the heart, which was decreased after ISP administration. Furthermore, attenuation of ISP-induced rise in LVEDP, a surrogate marker of preload also demonstrates the recovery of ventricular function from ISP-induced myocardial damage. Favorable improvement in hemodynamic and left ventricular contractile function with LCP treatment indicates improved cardiac performance and recovery from ISP-induced MI.
Apart from hemodynamic and ventricular contractile function, several cardiospecific diagnostic marker enzymes like CK-MB isoenzyme and LDH are also present in myocardium which is used to predict pathological changes in the myocardium. 2 These marker enzymes are leaked out into the extracellular fluids after myocardial injury due to disintegration of the cellular and subcellular compartments. 23 Following administration of ISP, a significant decrease in myocardial CK-MB and LDH enzymes indicates their depletion from heart and delineates myocardial injury. However, rats pretreated with LCP showed a significant rise in myocardial enzymes; CK-MB isoenzyme and LDH demonstrate that LCP renders the cardiomyocytes less leaky due to membrane stabilization following reduced membrane lipid peroxidation.
As mentioned earlier, ISP-induced MI leads to the gradual loss of endogenous oxidant/antioxidant balance. The endogenous defense network constitutes enzymatic and nonenzymatic antioxidants; SOD, CAT, GSHPx and GSH which neutralizes OFRs and ROS in ischemic tissue. 2 Decrease in the values of SOD and CAT following ISP administration indicate overwhelming of free radicals, which ensues oxidative damage to the myocardium. In previous studies, LCP has been reported as the most efficient biological carotenoid singlet oxygen quencher and shown to participate in first line of defense, maintained by SOD and CAT. 34,35 LCP in the present study prevented the decline of SOD and CAT in myocardium that is suggestive of its antioxidant and free radical scavenging activity in consonance to previous studies. 36
In addition, the present study also showed a significant decrease in myocardial GSHPx enzyme and GSH, an endogenous antioxidant substrate following ISP administration. GSH has a direct antioxidant function by reacting with superoxide radicals, peroxy radicals and singlet oxygen followed by the formation of oxidized GSH and other disulfides and well known to replenish GSHPx, being its substrate. 3 Depletion of GSH from heart following ISP administration may have exceeded the ability of antioxidant enzymes to dismutate the radicals, resulting in oxidative damage of the myocardium. Reduction in the activities of GSH-dependent antioxidant enzyme, GSHPx following ISP administration might be corroborated to the decreased availability of its substrate, GSH. Reduced GSHPx activity causes accumulation of lipid peroxides and other oxidants which renders the cellular and subcellular membranes more susceptible to the oxidative damage. Pretreatment with LCP-prevented depletion of GSH and restored the GSHPx activity in myocardium of ISP-treated rats indicates its antiperoxidative activity in consonance to the previous study, which demonstrate its antiperoxidative activity against adriamycin-induced cardiotoxicity. 36
By impairing antioxidant defense mechanisms, ISP renders the myocardium more susceptible to lipid peroxidation, an important oxidative degenerative pathogenic event in MI. 34 In the present study, after ISP administration, the enhanced lipid peroxidation represented by MDA level might be due to compromised antioxidant defense, thus the free radical were not effectively neutralized and causes the myocardial damage. LCP pretreatment decreased the level of MDA, which might be due to enhanced activities of myocardial antioxidants and quenching of ROS and free radicals. Among naturally occurring carotenoids, LCP has the strongest ability to scavenge free radicals, being 10-fold, 47-fold and 100-fold more effective in quenching singlet oxygen than α-tocopherol, β-carotene and vitamin E, respectively. 34 Among the various defense mechanisms, LCP is most likely to be involved in the scavenging of two of the ROS, singlet molecular oxygen and peroxy radicals, even at low-oxygen tension. 37 In the present study, ISP-induced myocardial ischemia invoked a condition of low oxygen availability consequent to reduced blood supply, where this unique property of LCP to quench free radicals at low oxygen tension might contribute to the afforded cardioprotection. 37,38
The protective effects of LCP on myocardial histoarchitecture were further confirmed by light microscopic examination. Several studies suggested that ISP produces histopathological lesions similar to those of coagulative myocytolysis or myofibrillar degeneration, which is one of the signs of acute MI in man. 18 Subsequent to ISP administration, significant myonecrosis, edema and infiltration of inflammatory cells were observed in the myocardium of ISP control rats. However, rats pretreated with LCP have shown resistance toward myocardial damage by reduced necrosis and edema. LCP-treated groups show preservation of structural integrity with predominantly normal cardiomyocytes morphology at all three doses and with no sign of leukocyte infiltration. Increased CK-MB and LDH in heart further supports this observation suggesting the preservation of membrane integrity. Improved antioxidant status and inhibition of lipid peroxidation might have attributed to the myocardial salvage and morphological recoup of the myocardium against ISP-induced myocardial necrosis. LCP per se treated rats showed normal cardiac myofibers without pathological changes, therefore indicating that LCP is safe to cardiomyocytes and does not render any adverse effects on myocardium.
Furthermore, the protective effect of LCP against the myocardial membrane damage can also be explained based upon its lipophilic nature. 39 It is presumed that LCP similar to other lipophilic agents such as vitamin E, antipyrine, nifedipine and β-blocking drugs intercalate into the lipid matrix and impart stabilization to myocardial cell membranes pertinent to the degree of their lipophilicity. 40 Thus, it is possible that LCP may also render resistance and impart stabilization to the myocardial cell membranes by extending the viability from necrotic damage.
To conclude, present study findings shows that LCP treatment maintained the antioxidant levels, preserved histology of myocardium and improved cardiac performance following ISP administration. The results of this study suggest that LCP may serve as an adjunct in prophylaxis and treatment in delaying the progression of MI.
Footnotes
Acknowledgments
The authors thank Mr BM Sharma for his technical assistance in the course of study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
